Abstract
Fish skin mucus is the first line of defense that provides physical and chemical barriers against pathogens and toxins. The mucus is produced continuously and sloughed off regularly from the skin to defend against infections through the skin. However, the molecular properties of the mucus content that prevent pathogen invasion are yet to be fully understood. In this study, a proteomic approach using liquid chromatography–mass spectrometry (LCMS) was applied to explore the changes in the mucus protein content of resistant and susceptible groupers in response to Vibrio alginolyticus. The Vibrio-resistant groupers showed no observable clinical sign of infection after the immersion challenge, while the Vibrio-susceptible groupers presented either hemorrhagic- or non-hemorrhagic ulceration of the skin. A comparative proteome analysis on the mucus samples yielded 1488 identified proteins. The immune-related proteins, namely Cystatin B, Complement Component C6, Complement factor 1, Allograft inflammatory factor 1, Deleted in malignant brain tumors protein, MHC class 1 and Annexin A1, that were significantly abundant in the resistant group responded to V. alginolyticus infection. Interestingly, there was an expression of immune-related proteins that possibly could be the non-invasive biomarkers, namely 3-hydroxybutyrate dehydrogenase type 2 and L-rhamnose-binding lectin SML.
1. Introduction
The world aquaculture production in 2018 surpassed capture fisheries with a record of 114.5 million tons of production. Fish protein primarily derived from aquaculture is expected to comprise 59% of human consumption by 2030 [1]. Grouper is one of the most abundant fishery species due to its significant commercial value that has been established in Singapore, Malaysia, Hong Kong, Thailand and Taiwan since the 1970s. Among the most common grouper species are the Epinephelus fuscoguttatus (Brown Marble grouper), Epinephelus coiodes (Orange Spotted grouper), Epinephelus lanceolatus (Giant grouper), Epimetheus tauvina (Greasy grouper), Mycteroperca tigris (Tiger grouper) and Cromileptes altivelis (Mouse grouper). However, throughout the years, the transition of the aquaculture technique from extensive to semi-intensive and intensive farming has led to the emergence and outbreak of infectious diseases. In order to mitigate the outbreak, several approaches have been taken to improve the quality of fish seeds produced in the hatchery. Hybridization, an approach to crossbreeding between two species, has been practiced in aquaculture to improve the quality of the fish seeds. The hybrid offspring inherited the desirable traits of the parental species, such as a superior growth rate, robustness and disease resistance [2,3,4]. The hybrid of E. fuscoguttatus × E. lanceolatus represents 70% of the total cultured grouper population in Indonesia, and this hybrid has become the second target species for aquaculture production in Hong Kong [5,6,7,8].
Although the novel hybrid of E. fuscoguttatus × E. lanceolatus yielded a significant and superior growth performance over its parental species [9], the inheritance of the desirable disease-resistant trait requires further evaluation [9,10,11]. Notwithstanding the reported improved immunological parameters in this hybrid grouper [9], the aquaculture of the hybrid species is still being affected by the prevailing infectious diseases, such as grouper iridovirus [12], vibriosis [13] and marine leech infestation [14]. Vibriosis is among the most common infectious diseases that affect grouper aquaculture. This disease is caused by the Gram-negative Vibrio sp., which includes V. alginolyticus, V. vulnificus, V. harveyi and V. parahaemolyticus [15]. Dark skin, pale gills, excessive mucus production, hemorrhage and ulceration of the affected areas, such as body, mouth and fins, are the common clinical signs of infection. The outbreak of vibriosis commonly resulted in a high mortality rate of the affected farms, causing an inevitable slump in marine aquaculture production [16,17].
Proteomics has been widely applied in studies related to infectious diseases, which include drug discovery, biomarker identification, disease pathogenesis, vaccine development and disease diagnosis, as well as in fundamental research to comprehend the host’s physiological and immunological responses against pathogens [18,19]. The characterization of the immunological responses against pathogen invasion using the proteomics approach facilitates the identification of novel immune-related proteins and potential biomarkers for disease diagnosis and therapy [20,21]. This approach has successfully identified differentially expressed proteins in different immune organs of the disease-resistant/tolerant and the susceptible fish [22]. The fish skin mucus is rich in biochemicals, especially proteins and metabolites [23,24]; hence, a comparative analysis of the mucus has become of great interest in understanding the immunological responses of skin- and mucosal-associated lymphoid tissues. The functional skin mucus proteome of disease-resistant grouper is hypothesized to provide new insights into the disease-resistant/tolerant phenotype, which could assist in the screening of broodstock/fish seed quality. This study aimed to identify the proteome changes in the grouper skin mucus in response to Vibrio alginolyticus infection and the potential protein biomarkers that correspond to the disease-resistant phenotype.
2. Materials and Methods
2.1. Fish and Bacterial Infection, Mucus Sample Collection and Preparation
All animal handling and experimental protocol were performed in accordance with the ethics guidelines approved by Universiti Putra Malaysia Animal Ethics Committee (Approval number: UPM/IACUC/AUP-R054/2022). Hybrid grouper fingerlings (Epinephelus fuscoguttatus ♀ × Epinephelus lanceolatus ♂) in 9.6 ± 0.4 cm size were obtained in April 2021 from a local hatchery in Kuala Selangor, Malaysia, and transported to the laboratory using plastic bags containing oxygen. The fingerlings were screened for health issues, from which only healthy fish were subjected to acclimatization under laboratory conditions for 2 weeks in 25 ppt saltwater with continuous aeration and fed with commercial feed twice daily on an ad libitum basis. The water temperature was maintained at 27 to 34 °C throughout the day.
Vibrio alginolyticus isolate was obtained from the Laboratory of Fish Health and Disease, Department of Aquaculture, Universiti Putra Malaysia. The isolate was cultured on marine agar for 24 h at 16 °C. A single colony was inoculated in 10 mL marine broth, and then the culture was up-scaled to 1 L. Bacterial cells were then collected through centrifugation at 11,000× g for 15 min. Bacterial cell suspension of 9.4 × 107 CFU/mL was prepared in 25 ppt saltwater for subsequent experimental infection in grouper fingerlings.
Eighty healthy grouper fingerlings with a mean total body length of 9.6 ± 0.4 cm were randomly selected. For experimental infection, 60 fingerlings were exposed to V. alginolyticus at a dose of 9.4 × 107 CFU/mL by immersion for 4 h, whereas the control group consisted of 20 fingerlings were not exposed to V. alginolyticus. The fingerlings were then transferred to clean saltwater with continuous aeration for observation, and the control group was kept in a separated aquarium. On day 7 post-infection, fingerlings were categorized into Vibrio-susceptible group according to the presence of clinical signs of infection that include red spots, hemorrhagic and/or non-hemorrhagic ulceration on the body, while the Vibrio-resistant fingerlings presented no observable clinical sign of infection. The fish were fasted 12 h before harvesting.
In the pre-harvesting process, all (46 samples) of the fish mucus from resistant and susceptible groups, including those from the dead fish, were spread individually on marine agar and thiosulfate citrate bile salts sucrose agar (TCBS: selective media for Vibrio sp.) to confirm the presence of Vibrio. The bacteria from the dead fish spleen were also detected by plating on the designated agar. All the subjected samples showed a positive Vibrio growth on the TCBS agar and thus confirmed the presence of Vibrio through the immersion method.
Fish were anesthetized using tricaine methanesulfonate (MS222) before skin mucus sampling. Skin mucus (n = 5) was collected by gently scraping the body using a sterile glass slide avoiding the collection of blood and the anal area. The 5 mucus samples of fish from the same group were pooled to represent one biological replicate due to the minute amount of mucus from each fish. All samples were stored at −80 °C until further use. The mucus samples were reconstituted in 1× phosphate-buffered saline and sample buffer (7 M Urea, 2 M Thiourea, 2 mM PMSF). The samples were mixed well and sonicated on ice at 20% amplitude for 3 min (10 s pulses on, 15 s pulses off). The samples were then centrifuged at 15,000× g for 15 min to collect supernatant containing skin mucus total protein which concentration was quantified using Bradford assay (Solarbio, Beijing, China) before subsequent analysis. The total protein in 9 µg from each sample was electrophoretically separated on 12% SDS polyacrylamide gel and double stained with Coomassie-silver.
2.2. Liquid Chromatography–Mass Spectrometry (Hybrid Quadrupole-Orbitrap Technology)
Nine samples of 3 biological replicates from each group were submitted to proteomic analysis using LCMS. Prior to LCMS, protein samples of 75 µg were loaded into 12% SDS polyacrylamide gel. Protein bands were then excised and in-gel digested using trypsin gold (Promega, Madison, WI, USA). Subsequently, clean-up of the digested samples was performed using Ziptip (Millipore, Burlington, MA, USA). LCMS analysis of the peptide samples was conducted according to the previous report [25]. In brief, peptide samples were reconstituted in 30 μL of 0.1% formic acid and 5% acetonitrile, before 2 μL of the samples were loaded onto an Acclaim PepMap 100 C18 column (2 μm, 0.075 × 150 mm) (Thermo Scientific, Waltham, MA, USA). The reverse phase column was equilibrated with 0.1% aqueous formic acid (mobile phase A) and 80% acetonitrile containing 0.1% formic acid (mobile phase B). Elution of gradient 5–35% mobile phase B was performed at a flow rate of 0.3 mL/min for 75 min using EASY-nano liquid chromatography (EASY-nLC) 1200 System (Thermo Scientific, MA, USA). An online Q Exactive Plus Hybrid Quadrupole-Orbitrap mass spectrometer system (Thermo Scientific, MA, USA) was used to generate the peptide ions with a spray voltage of 1900 V in positive mode. A precursor ion scan was conducted with a resolution of 70,000 and a mass range of m/z 310–1800. Precursors containing charge states from 2+ to 8+ were further fragmented via collision induced and high-energy collision induced (CID and HCD) at normalized energy of 28%. Precursor mass was scanned at the range of 110–1800 m/z.
2.3. Data Processing and Protein Annotation
Mass spectra of the peptides were acquired using Tune (Ver. 2.11 QF1 Build 3006; Thermo Scientific, Waltham, MA, USA) and deconvoluted with Proteome Discoverer Ver. 2.4 (Thermo Scientific, Waltham, MA, USA) to create the peptide mass list. SEQUEST HT search engine, incorporated in the Proteome Discoverer, was used to match the generated mass list against Epinephelus FASTA sequences downloaded from NCBI. Mass tolerance for the peptides and their fragments was fixed at 10 ppm and 0.02 Da, respectively. Trypsin was indicated as the digestion enzyme with up to two miscleavages allowed during the search. Carbamidomethylation (CAM) modification on cysteine residues was set as a static modification, while variable amino acid modifications were set as deamidation for asparagine and glutamine residues, and oxidation for methionine residues. The mass list was also searched against a decoy database generated from the randomized protein sequences. The identified proteins were accepted with at least a Rank 1 peptide and a false discovery rate of 1%. Spectra that matched the sequences were further validated with the Percolator algorithm (Ver. 2.04) where q-value at 1% false discovery rate (FDR).
2.4. Statistics and Protein Enrichment
The statistical analysis was performed using Perseus [26] to identify differentially expressed proteins. One-way ANOVA was applied and the comparison between groups was analyzed by applying a two-tailed Student’s t-test (p ≤ 0.05). Proteins with fold change ≥2 were identified to be differentially expressed [27], and the differentially expressed proteins identified were mapped to Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) for pathway analyses. Fisher’s exact test was applied to perform enrichment analysis (p-value ≤ 0.05).
3. Results
3.1. Experimental Infection of Hybrid Grouper with Vibrio alginolyticus
The hybrid grouper fingerlings were exposed to V. alginolyticus by immersion in the saltwater of 25 ppt containing approximately 9.4 × 107 CFU/mL of V. alginolyticus for 4 h. On day 7 post-infection, 10 mortalities out of 60 infected fingerlings were recorded (16.7% of cumulative mortality), 28 fingerlings (46.7%) were resistant to V. alginolyticus and 18 fingerlings (30%) were susceptible to the infection, exhibiting clinical signs of infection where ulceration and red spots were observed on the mouth, body and fins (Figure 1). The resistant fingerlings recorded no observable clinical signs of infection.

Figure 1.
Development of ulcer and lesions on the body (a), mouth (b), anal fin (c), dorsal fin (d) and caudal fin (e) of hybrid grouper that were susceptible to Vibrio alginolyticus experimental infection, comparable to the resistant hybrid grouper where no observable ulcer and lesion were recorded.
The infection of V. alginolyticus on the selected healthy hybrid grouper was confirmed by applying the bacteria plating method. The fish mucus from resistant and susceptible groups was spread individually on marine agar and TCBS agar to confirm the presence of Vibrio in the fish. All the subjected samples, including the spleen of the dead fish, showed positive for Vibrio infection on the TCBS agar (Appendix A Figure A1). However, the bacterial load was not quantitatively analyzed. The immersion in the same concentration of bacterial suspension in controlled conditions and exposure time might have given similar bacterial exposure to the fish.
The evaluation on the skin mucus protein of the susceptible and resistant groups showed viscosity variation. The susceptible group secreted much more and a viscous mucus than the resistant fish. The protein composition of the skin mucus between the infected group and control group was significantly different with the former showing more variable intensities of the protein bands (Appendix A Figure A2).
3.2. Skin Mucus Proteome Profiling and Differential Expression Analysis
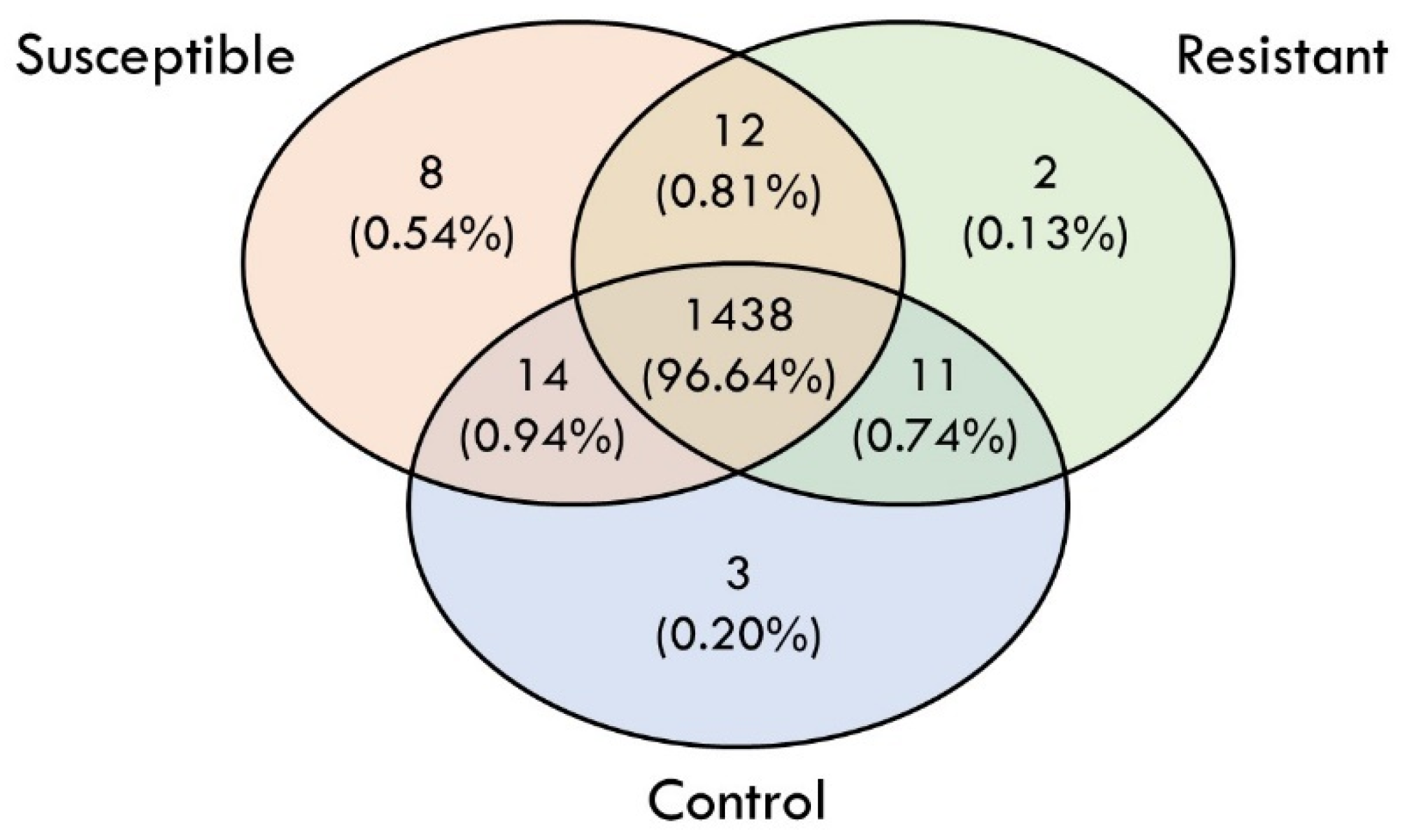
The label-free shotgun proteomic analysis using LCMS identified a total of 1488 proteins in the skin mucus samples. Out of the 1488 proteins, 3, 2 and 8 proteins were found to be exclusively expressed in the control, resistant and susceptible groups, respectively (Figure 2). The identified putative proteins are listed in Table 1.
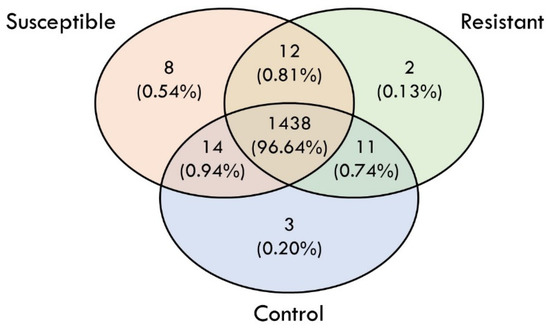
Figure 2.
Venn diagram that summarized the total number of putative proteins identified in control, resistant and susceptible groups, respectively, in response to Vibrio alginolyticus experimental infection.

Table 1.
Putative proteins were found to be exclusively expressed in the control, resistant and susceptible groups, respectively.
3.3. Differentially Expressed Proteins (DEPs) in Response to V. alginolyticus Experimental Infection
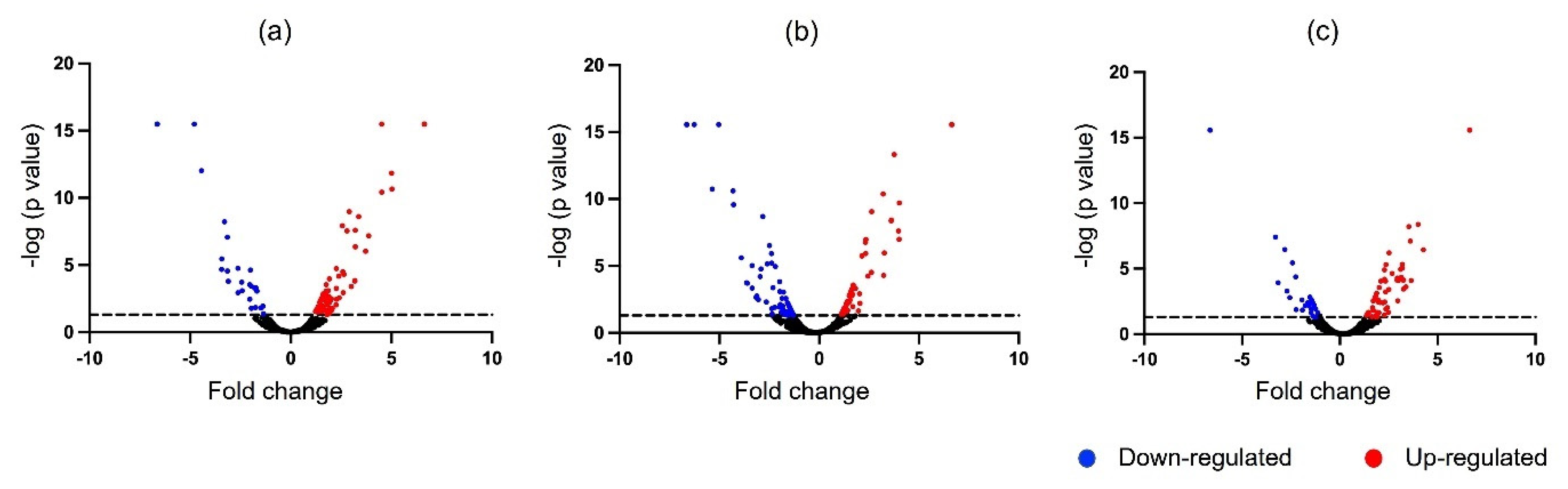
A pairwise comparison between the control, resistant and susceptible groups revealed different numbers of DEPs in response to the V. alginolyticus experimental infection (Figure 3). A comparison of the protein abundance between the control and resistant groups reveals 99 DEPs (Table 2), where 47 upregulated and 52 downregulated proteins were from the resistant group, while 60 upregulated and 42 downregulated proteins were identified in the susceptible group compared to the control group (Table 3). The comparison between the resistant and susceptible groups revealed 94 DEPs (Table 4), wherein 28 upregulated and 66 downregulated proteins were recognized in the resistant group. Based on the list of DEPs, the immune changes in V. alginolyticus infection could be deciphered. These can be seen through the alteration of the expression of an immune-related protein, namely Cystatin B, Complement Component C6, Complement factor 1, Allograft inflammatory factor 1, Deleted in malignant brain tumors protein, MHC class 1, Annexin A1, 3-hydroxybutyrate dehydrogenase type 2 and L rhamnose-binding lectin SML.
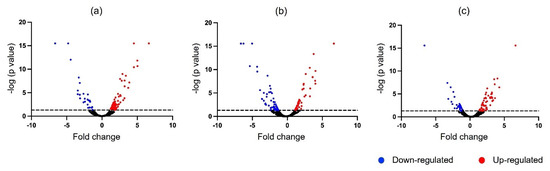
Figure 3.
The volcano plot showed a pairwise comparison of mucus proteome for the identification of significant differentially expressed proteins (p ≤ 0.05). (a) Control group versus resistant group; (b) control group versus susceptible group; (c) susceptible group versus resistant group.

Table 2.
Differentially expressed protein was found to be resistant when compared to the control.

Table 3.
Differentially expressed protein was found to be susceptible when compared to the control.

Table 4.
Differentially expressed proteins were found to be in resistant when compared to susceptible.
3.4. Gene Ontology Analysis of DEPs
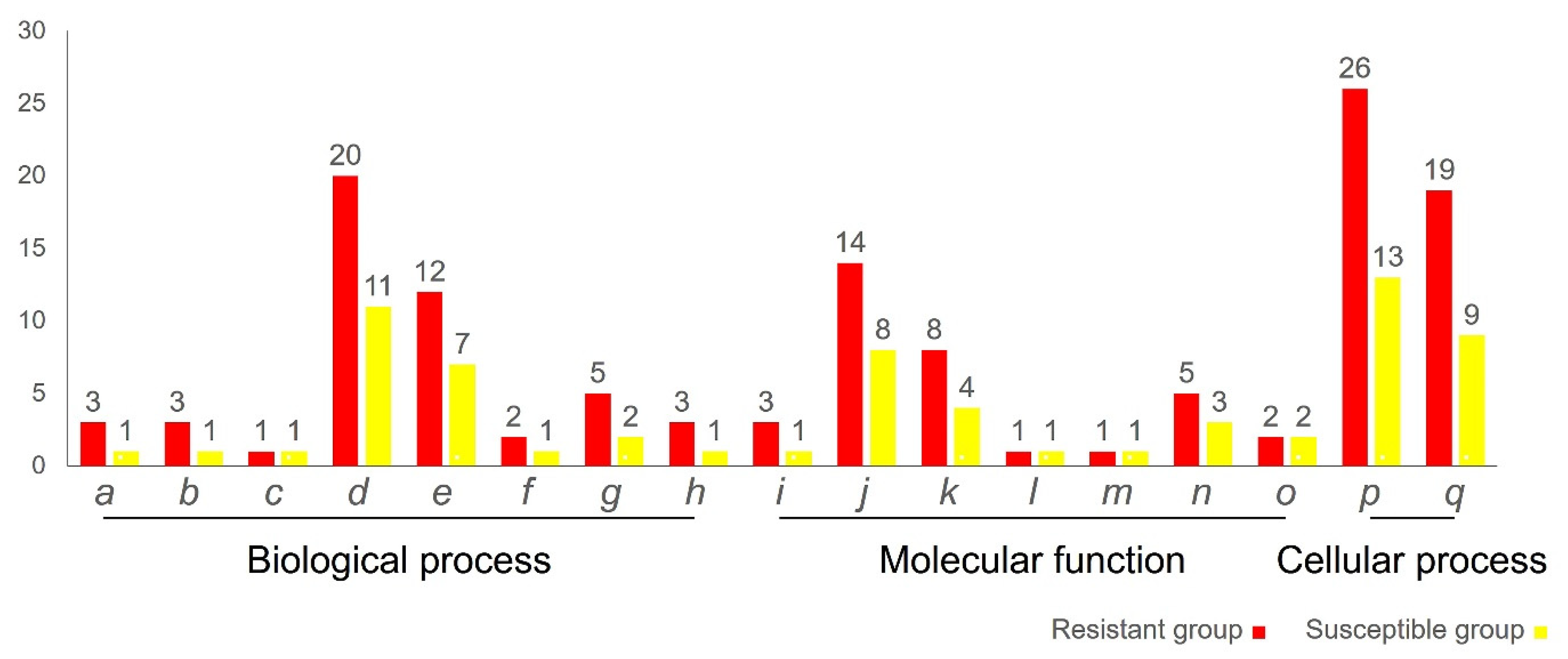
In order to gain further insight into the DEPs, they were categorized according to their biological process, molecular and cellular functions (Figure 4). The number of proteins annotated in most of the GO terms for the resistant group was relatively higher than the susceptible group, except for the developmental process (GO: 0032502), ATP-dependent activity (GO: 0140657), molecular function regulator (GO: 0098772) and transporter activity (GO: 0005215). The five GO terms annotated with the highest number of proteins were the cellular process, metabolic process, binding, cellular anatomical entity and protein-containing complex.
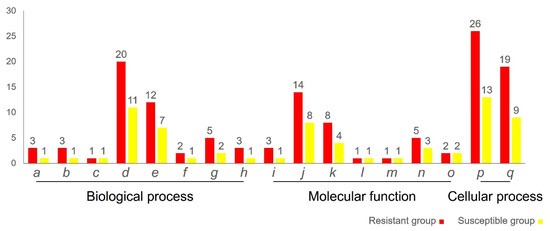
Figure 4.
Gene Ontology annotations of the differentially expressed protein in the mucus of Vibrio-resistant and -susceptible hybrid groupers based on the PANTHER classification system (p ≤ 0.05), which were categorized into biological process, molecular function and cellular function. y-axis: number of proteins; x-axis. GO terms. a: response to stimulus (GO: 0050896); b: signaling (GO: 0023052); c: developmental process (GO: 0032502); d: cellular process (GO: 0009987); e: metabolic process (GO: 0008152); f: locomotion (GO: 0040011); g: biological regulation (GO: 0065007); h: localization (GO: 0051179); i: molecular adaptor activity (GO: 0060090); j: binding (GO: 0005488); k: structural molecule activity (GO: 0005198); l: ATP-dependent activity (GO: 0140657); m: molecular function regulator (GO: 0098772); n: catalytic activity (GO: 0003824); o: transporter activity (GO: 0005215); p: cellular anatomical entity (GO: 0110165); q: protein-containing complex (GO: 0032991).
3.5. KEGG Enrichment
To ascertain the pathway-enriched proteins, the DEPs entailed in different biological pathways were annotated using the KEGG reference database. The DEPs were significantly (p-value ≤ 0.05) enriched in 17 pathways, with most of the DEPs highly associated with metabolic pathways followed by oxidative phosphorylation, glycolysis and carbon metabolism (Table 5).

Table 5.
KEGG pathways enrichment analysis of differentially expressed proteins that were associated with V. alginolyticus experimental infection.
More details of the differentially expressed proteins are presented in the appendix tables.
3.6. Identification of Potential Biomarker Candidates (PBCs)
The potential biomarkers that correspond to the Vibrio-resistant phenotype were identified and listed in Table 6. Thirteen proteins were found to be significantly altered in the resistant group when compared to the control, whereas no significant difference in these proteins was detected in the susceptible fish. This suggests disease-resistant properties against V. alginolyticus infection could be due to these identified non-invasive biomarkers.

Table 6.
Potential biomarker candidates.
4. Discussion
Skin mucus is known to be the first line of defense against diseases through the non-specific immune system [28]. Through proteome profiling, one can gain an understanding of molecular changes under certain events. Hence, it has been proven to be beneficial in gaining intuition about an organism’s biological response [29,30,31]. However, the immune response of hybrid groupers toward Vibrio sp. infection is poorly understood, owing to the little understanding of the underlying molecular mechanisms. This study assessed the acute inflammatory response of hybrid groupers by proteome analysis on skin mucus which allowed a better understanding of the fish immunity as well as to determine the possible disease biomarkers.
Currently, the execution of Vibrio sp. on hybrid groupers marked a positive effect on the innate immune response. Protein profiling on skin mucus showed several identified proteins in the susceptible group compared to the resistant group, which indicates a positive response to the infection [32]. The majority of DEPs were also found to be perturbed in the resistant and susceptible groups involved in the cellular process based on the GO classification and the metabolic and oxidoreductase phosphorylation activity as in the KEGG enrichment. In fish, skin mucus is crucial in providing respiratory and metabolic function [33]. In this regard, the present study exhibits most of the DEPs being enriched in metabolic pathways which could be classified as enzyme energy metabolisms, such as NADH dehydrogenase and ATP synthase. Moreover, two crucial proteins that may be involved in the pathological immune response, 3-hydroxybutyrate dehydrogenase type 2 (BDH2) and L rhamnose-binding lectin SML (L-RBL), were also observed.
It is notable that the expression of BDH2, a regulatory molecule that was found to be enriched in metabolic pathways and the synthesis and degradation of ketone bodies pathways, is increased only in the resistant group. BDH2 is a key component of ketone bodies, an endogenous molecule that is responsible for maintaining cellular energy and modulating the signaling cascade through the cellular process. The cell can derive energy from BDH2 during disease infection [34,35]. A slight increase in BDH2 in concentrations was modulated by signaling pathways as they became involved in the cell growth, proliferation and oxidative stress resistance [36]. BDH2 can be activated in immune cells that lead to an anti-inflammatory effect [37]. The present findings support that BDH2 is involved in providing the disease-resistant properties of hybrid groupers, which is advantageous as a basis for developing the non-invasive biomarkers.
In addition, the L-RBL protein was also discovered in this study. It was anticipated that this immune-related protein would have a distinct role in providing disease resistance. Focusing on its high expression in resistant fish, it can offer a non-invasive biomarker during the segregation of disease-resistant and -susceptible groupers. L-RBL is a member of the rhamnose-binding lectin proteins (RBLs) that belong to the animal lectin group, a sugar-specific binding protein that recognizes the carbohydrate structure domain. RBLs are widely distributed in viruses, prokaryotes and eucaryotes [38], exclusively found in teleost and aquatic invertebrates which have been previously identified in eggs, serums and skin mucus [39,40,41]. It has been reported that RBLs can provide innate immunity as they inhibit proliferation, cytotoxicity and opsonize nonself-cells and particles [42]. The present study is consistent with the reported data on the clearance of Gram-positive and -negative bacteria by phagocytosis, which is enhanced by the expression of RBLs in sea bass [40].
The proteomic analysis also revealed some immune-related DEPs which are Cystatin B, Complement Component C6, Complement factor 1, Allograft inflammatory factor 1, Deleted in malignant brain tumors protein, MHC class 1 and Annexin A1. These DEPs have provided us with an understanding of the molecular changes during Vibrio infection.
Cystatin B was found to be upregulated in the infected fish only. This suggests the idea of fish with naive immunity may produce this protein in response to pathogen invasion to maintain their physiological state. The role of Cystatin B is consistent with the previous study [43], as the overexpression of Cystatin B in mice interacted with various types of proteins to activate the JAK/STAT signaling pathway for the removal of pathogens and the killing of infected cells. Cystatin B may also be present and translocated into mitochondria, which helps to protect the mitochondria activity and increase the ROS generation, enhancing the activity of pathogen removal [44,45]. Another protein that shares the same pattern of expression with Cystatin B is Complement component C6 and Complement factor 1. As components of the complement system, they are believed to play an important role in forming the membrane attack complex (MAC) and could assist in the cytolytic killing of the pathogen [46,47].
Other immune-related proteins were also found to be upregulated after infection in the resistant compared to the susceptible group. Allograft inflammatory factor 1 (AIF-1), Deleted in malignant brain tumors protein (DMBT1) and MHC class 1 alpha were detected. In this study, the resistance to infection was associated with high levels of Allograft inflammatory factor 1 (AIF-1) expression. AIF-1 is a 17 kDa type II interferon (IFN-y), for which expression is most likely limited to macrophages and monocytes. The high expression of AIF-1 was previously found in the spleen of E. awaora treated with lipopolysaccharides [48]. The present findings agree with the previous studies wherein AIF-1 was noted to provide innate immunity in various ways. According to a previous report [49], the presence of AIF-1 on the body wall of the infected area showed the migration and recruitment of numerous counts of macrophages after the lipopolysaccharides’ stimulation. AIF-1 is also claimed to be involved in wound healing and shell repair after the tissue injury of abalone [50]. Theoretically, the protein recognition receptor on the surface of Gram-negative bacteria is able to recognize the molecular pattern associated to the pathogen and hence initiate the defense mechanism of the cellular process. AIF-1 also has been proven to be involved in the innate immunity in the head kidney, spleen and various body tissues of vertebrates, but it was first established in the skin mucus of an aquatic organism [50,51,52].
Another detected protein is DMBT1 which belongs to a scavenger receptor protein that can bind to both Gram-negative and -positive bacteria. DMBT1 expression is obvious upon inflammation and epithelial cell differentiation and is involved in innate immunity. This protein was previously found to be upregulated in intestinal epithelial cells after lipopolysaccharide or tumor necrosis factor induction. However, it can also inhibit LPS-induced NF-B activation and cytokine secretion, leading to anti-inflammatory functions that can trigger the acquired immunity later [53,54].
MHC class 1 is a major histocompatibility complex class 1. The overexpression of MHC class I in a resistant group compared to the control that involved cell adhesion molecule and phagosome pathways was observed in the present study. In teleost, MHC class 1 normally displays and presents itself as a surface antigen-presenting cell on an infected cell. Upon entering the environment, an unknown pathogen will infect the cell which will then be degraded by innate immunity. As a result, MHC class 1 is produced within the reticulum and the pathogen is marked to trigger the cytotoxic T-cell wherein this process will help in the killing of the pathogen [55,56]. This finding is in line with the previous report in which the V. alginolyticus was found to be cytotoxic and lethal to fish cell lines and able to degrade the mucus of sea bream [57].
The present finding also reveals AnxA1 as the significantly expressed protein detected in the resistant group after infection. The AnxA1 protein was found to have dual functions in inducing inflammation. Over 100 types of annexins have been discovered in various tissues of vertebrates and invertebrates. The binding properties of this protein are influenced by cytoskeleton interactions, membrane fusion, anticoagulation, intracellular signaling and phospholipase inhibition. Molecular studies of annexin in humans are sparse, but some research could be found on teleost [58,59]. In humans, the expression of AnxA1 is a pro-inflammatory and anti-inflammatory response increased by exogenous and endogenous glucocorticoids, respectively. It is capable of controlling cell apoptosis in various pathways [59,60]. In the infected human, the cell will undergo apoptosis that may cause inflammation in the host [61]. Therefore, AnxA1 serves in apoptotic cell removal by acting as a highly specific signaling ligand of an endogenous danger molecule to enhance phagocytic clearance without causing any inflammation [61,62,63,64,65]. AnxA1 has been detected upregulated in the gill mucus of Atlantic salmon infected with amoebic gill disease with no observed lesion [66]. However, this protein was also expressed in serum and provided immunity to Chinook salmon with skin lesions [60]. The anti-inflammatory role of AnxA1 is still largely unknown, even though its pro-inflammatory-induced protein role is well studied in fish
5. Conclusions
This study has successfully generated new knowledge which strongly suggests that 3-hydroxybutyrate dehydrogenase type 2 and L-rhamnose-binding lectin SML protein, as new non-invasive biomarkers, could be used in the discrimination between Vibrio-resistant and -susceptible groupers. In addition, the biological context exhibited by the mucus is also provided, likely to aid in enhancing the understanding of fish immunity under natural infection. Cystatin B, Complement Component C6, Complement factor 1, Allograft inflammatory factor 1, Deleted in malignant brain tumors protein, MHC class 1 and Annexin A1 protein are signified as having distinct functions and roles toward V. alginolyticus infection.
Author Contributions
Conceptualization, A.C.; Data curation, B.Y.C.L. and L.C.F.; Formal analysis, N., B.Y.C.L. and L.C.F.; Funding acquisition, A.C. and I.S.I.; Investigation, N., W.M.S.W.S. and L.C.F.; Methodology, N. and L.C.F.; Project administration, I.S.I.; Supervision, A.C., W.M.S.W.S., I.S.I. and L.C.F.; Visualization, A.C.; Writing—original draft, N.; Writing—review & editing, I.S.I. and L.C.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministry of Education (grant number TRGS/1/2020/UPM/02/1/3).
Institutional Review Board Statement
The study was conducted in accordance with the ethics guidelines approved by Universiti Putra Malaysia Animal Ethics Committee (Approval number: UPM/IACUC/AUP-R054/2022).
Data Availability Statement
Supporting data will be provided upon acceptable request.
Acknowledgments
This research was supported by the grant TRGS/1/2020/UPM/02/1/3. We thank the Institute of Biology System (INBIOSIS) for providing the experiment facilities and PROMET Laboratory, and MPOB for the research collaboration on the Orbitrap-MS System.
Conflicts of Interest
Authors declare that they have no conflict of interest.
Appendix A
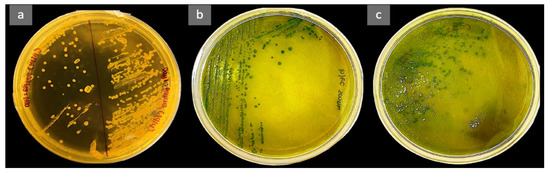
Figure A1.
Bacteria isolated on the TCBS agar from the mucus and spleen of dead fish (a), mucus of the resistant (b), and susceptible (c) hybrid groupers.
Figure A1.
Bacteria isolated on the TCBS agar from the mucus and spleen of dead fish (a), mucus of the resistant (b), and susceptible (c) hybrid groupers.
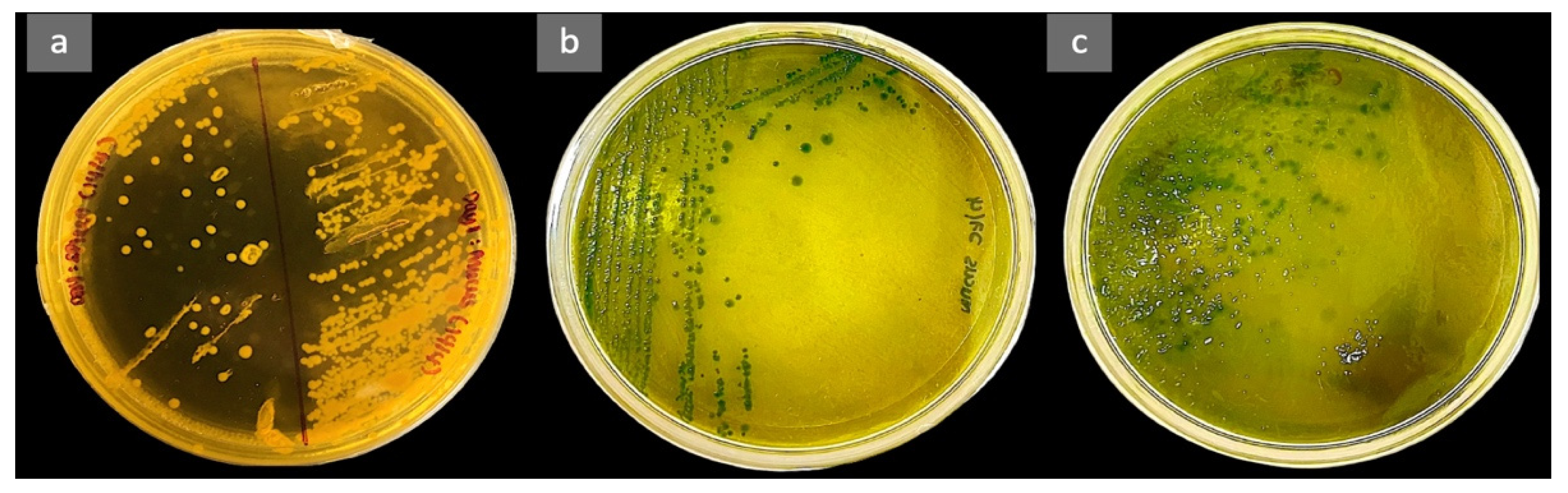
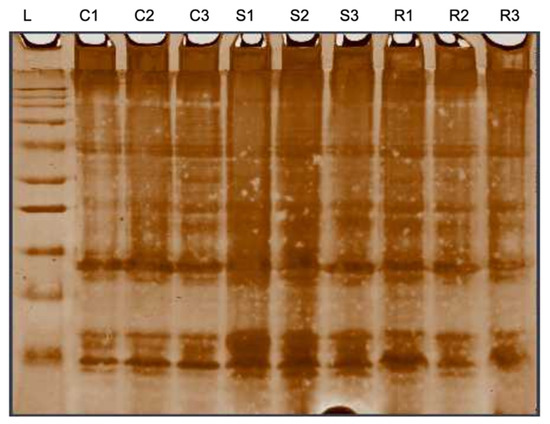
Figure A2.
Evaluation on mucus protein from different hybrid grouper groups by electrophoretically separated on 12% SDS polyacrylamide gel and double stained with Coomassie-silver. C1,C2,C3—control, S1,S2,S3—susceptible, and R1,R2,R3—resistant.
Figure A2.
Evaluation on mucus protein from different hybrid grouper groups by electrophoretically separated on 12% SDS polyacrylamide gel and double stained with Coomassie-silver. C1,C2,C3—control, S1,S2,S3—susceptible, and R1,R2,R3—resistant.
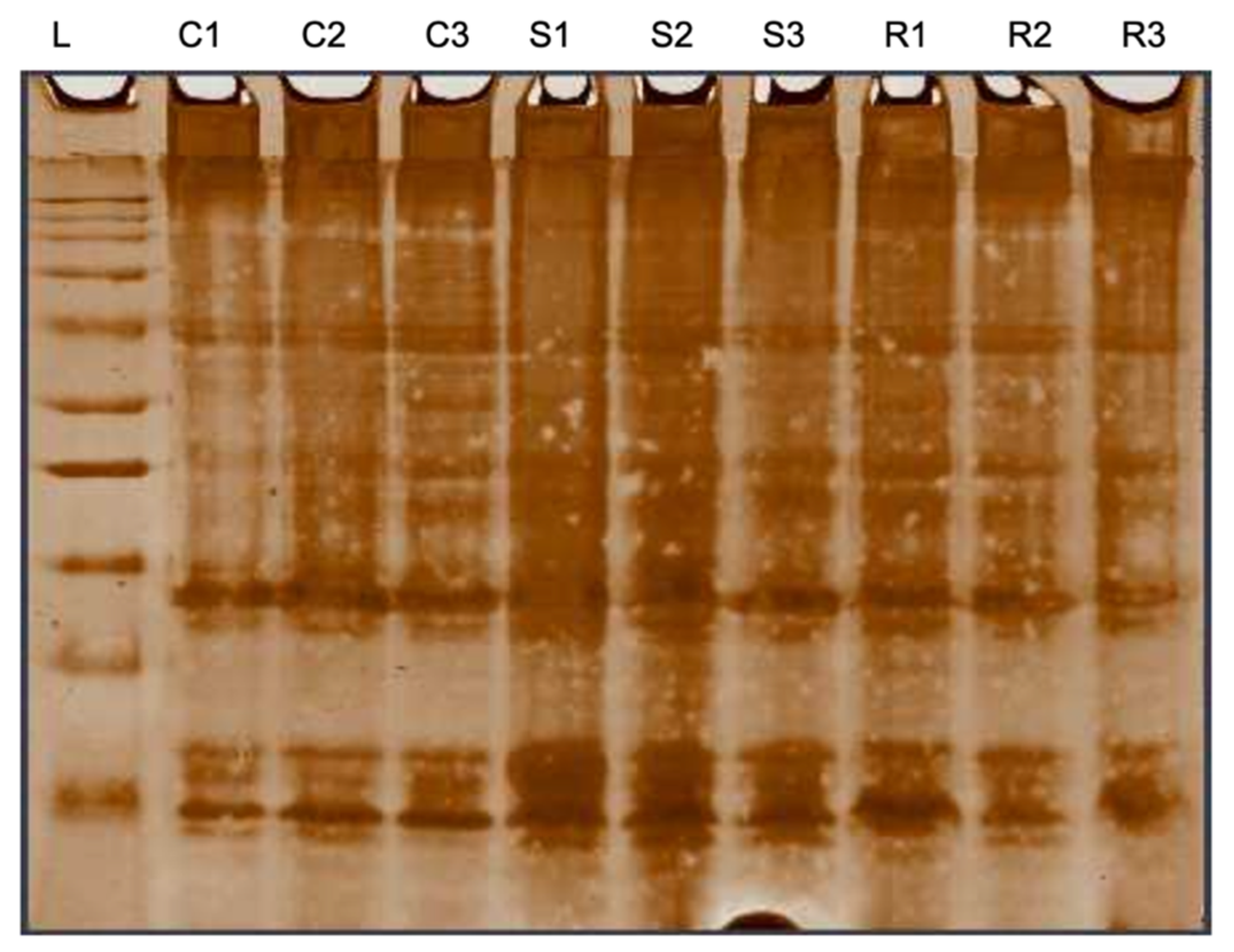

Table A1.
Differentially expressed protein-enriched in KEGG pathways.
Table A1.
Differentially expressed protein-enriched in KEGG pathways.
| Pathway ID | Description | Protein Count | p-Value | Protein | |
|---|---|---|---|---|---|
| Accession | Name | ||||
| 1100 | Metabolic pathways | 30 | 0.000 | 834906005 | glyceraldehyde-3-phosphate dehydrogenase |
| 295792242 | nucleoside diphosphate kinase B | ||||
| 328677247 | glyceraldehyde 3-phosphate dehydrogenase isoform, partial | ||||
| 221048061 | fructose-bisphosphate aldolase | ||||
| 834904058 | malate dehydrogenase | ||||
| 1832677686 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 4, mitochondrial | ||||
| 834904842 | phosphoglycerate kinase | ||||
| 1832683648 | nucleolin | ||||
| 1832687225 | 3-hydroxybutyrate dehydrogenase type 2 | ||||
| 1832613275 | cytochrome c oxidase subunit 5A, mitochondrial | ||||
| 197725770 | mitochondrial cytochrome C oxidase subunit Vb precursor | ||||
| 1832617105 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 6, mitochondrial | ||||
| 1832614309 | very-long-chain 3-oxoacyl-CoA reductase-A | ||||
| 1832627296 | isovaleryl-CoA dehydrogenase, mitochondrial | ||||
| 1832669039 | aldo-keto reductase family 1 member B1 isoform X1 | ||||
| 1832625753 | glyceraldehyde-3-phosphate dehydrogenase | ||||
| 343459175 | ATP synthase, H+ transporting, mitochondrial F0 complex, partial | ||||
| 295792326 | mitochondrial cytochrome c oxidase subunit 7C | ||||
| 1393214856 | type I glyceraldehyde-3-phosphate dehydrogenase | ||||
| 1832641526 | uroporphyrinogen decarboxylase | ||||
| 1832664977 | cytochrome c oxidase subunit 6B1 | ||||
| 1832644368 | peroxiredoxin-6 | ||||
| 1042045193 | glycine dehydrogenase (aminomethyl-transferring) | ||||
| 1832671044 | deoxyribose-phosphate aldolase | ||||
| 1832640714 | L-threonine dehydrogenase | ||||
| 1832608120 | cytochrome c oxidase subunit 4 isoform 1, mitochondrial | ||||
| 2067150115 | Thioredoxin domain-containing protein 12 | ||||
| 343459105 | NADH dehydrogenase 1 alpha, partial | ||||
| 1832680021 | betaine--homocysteine S-methyltransferase 1 | ||||
| 295792244 | fructose-bisphosphate aldolase A | ||||
| 190 | Oxidative phosphorylation | 9 | 0.000 | 1832613275 | cytochrome c oxidase subunit 5A, mitochondrial |
| 197725770 | mitochondrial cytochrome C oxidase subunit Vb precursor | ||||
| 1832608120 | cytochrome c oxidase subunit 4 isoform 1, mitochondrial | ||||
| 343459105 | NADH dehydrogenase 1 alpha, partial | ||||
| 343459175 | ATP synthase, H+ transporting, mitochondrial F0 complex, partial | ||||
| 295792326 | mitochondrial cytochrome c oxidase subunit 7C | ||||
| 1832617105 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 6, mitochondrial | ||||
| 1832664977 | cytochrome c oxidase subunit 6B1 | ||||
| 1832677686 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 4, mitochondrial | ||||
| 10 | Glycolysis/Gluconeogenesis | 7 | 0.000 | 834906005 | glyceraldehyde-3-phosphate dehydrogenase |
| 1393214856 | type I glyceraldehyde-3-phosphate dehydrogenase | ||||
| 1832625753 | glyceraldehyde-3-phosphate dehydrogenase | ||||
| 295792244 | fructose-bisphosphate aldolase A | ||||
| 328677247 | glyceraldehyde 3-phosphate dehydrogenase isoform, partial | ||||
| 221048061 | fructose-bisphosphate aldolase | ||||
| 834904842 | phosphoglycerate kinase | ||||
| 1200 | Carbon metabolism | 8 | 0.000 | 834906005 | glyceraldehyde-3-phosphate dehydrogenase |
| 1393214856 | type I glyceraldehyde-3-phosphate dehydrogenase | ||||
| 1832625753 | glyceraldehyde-3-phosphate dehydrogenase | ||||
| 295792244 | fructose-bisphosphate aldolase A | ||||
| 221048061 | fructose-bisphosphate aldolase | ||||
| 834904058 | malate dehydrogenase | ||||
| 834904842 | phosphoglycerate kinase | ||||
| 1042045193 | glycine dehydrogenase (aminomethyl-transferring) | ||||
| 1230 | Biosynthesis of amino acids | 6 | 0.000 | 834906005 | glyceraldehyde-3-phosphate dehydrogenase |
| 1393214856 | type I glyceraldehyde-3-phosphate dehydrogenase | ||||
| 1832625753 | glyceraldehyde-3-phosphate dehydrogenase | ||||
| 295792244 | fructose-bisphosphate aldolase A | ||||
| 221048061 | fructose-bisphosphate aldolase | ||||
| 834904842 | phosphoglycerate kinase | ||||
| 4260 | Cardiac muscle contraction | 6 | 0.000 | 1832606995 | tropomyosin alpha-4 chain-like isoform X4 |
| 1832613275 | cytochrome c oxidase subunit 5A, mitochondrial | ||||
| 197725770 | mitochondrial cytochrome C oxidase subunit Vb precursor | ||||
| 1832608120 | cytochrome c oxidase subunit 4 isoform 1, mitochondrial | ||||
| 295792326 | mitochondrial cytochrome c oxidase subunit 7C | ||||
| 1832664977 | cytochrome c oxidase subunit 6B1 | ||||
| 3010 | Ribosome | 5 | 0.001 | 1832612579 | 60S ribosomal protein L27a |
| 1832640175 | FAU ubiquitin-like and ribosomal protein S30 fusion a | ||||
| 1832673253 | 60S ribosomal protein L8 | ||||
| 1832610610 | 60S ribosomal protein L39 | ||||
| 157929900 | ribosomal protein L23 | ||||
| 30 | Pentose phosphate pathway | 3 | 0.001 | 295792244 | fructose-bisphosphate aldolase A |
| 1832671044 | deoxyribose-phosphate aldolase | ||||
| 221048061 | fructose-bisphosphate aldolase | ||||
| 4672 | Intestinal immune network for IgA production | 3 | 0.001 | 380006108 | MHC class II antigen |
| 326632479 | MHC class II antigen | ||||
| 62255674 | immunoglobulin mu heavy chain | ||||
| 51 | Fructose and mannose metabolism | 3 | 0.001 | 295792244 | fructose-bisphosphate aldolase A |
| 221048061 | fructose-bisphosphate aldolase | ||||
| 1832669039 | aldo-keto reductase family 1 member B1 isoform X1 | ||||
| 260 | Glycine, serine and threonine metabolism | 3 | 0.002 | 1832680021 | betaine--homocysteine S-methyltransferase 1 |
| 1042045193 | glycine dehydrogenase (aminomethyl-transferring) | ||||
| 1832640714 | L-threonine dehydrogenase | ||||
| 4514 | Cell adhesion molecules (CAMs) | 4 | 0.005 | 380006108 | MHC class II antigen |
| 161935902 | MHC class I alpha antigen | ||||
| 326632479 | MHC class II antigen | ||||
| 1832656308 | neural cell adhesion molecule 1a isoform X4 | ||||
| 4145 | Phagosome | 4 | 0.007 | 380006108 | MHC class II antigen |
| 161935902 | MHC class I alpha antigen | ||||
| 326632479 | MHC class II antigen | ||||
| 62255674 | immunoglobulin mu heavy chain | ||||
| 5168 | Herpes simplex virus 1 infection | 4 | 0.014 | 380006108 | MHC class II antigen |
| 161935902 | MHC class I alpha antigen | ||||
| 1832633330 | protein phosphatase 1, catalytic subunit, alpha isozyme a | ||||
| 326632479 | MHC class II antigen | ||||
| 630 | Glyoxylate and dicarboxylate metabolism | 2 | 0.015 | 834904058 | malate dehydrogenase |
| 1042045193 | glycine dehydrogenase (aminomethyl-transferring) | ||||
| 270 | Cysteine and methionine metabolism | 2 | 0.029 | 834904058 | malate dehydrogenase |
| 1832680021 | betaine--homocysteine S-methyltransferase 1 | ||||
| 72 | Synthesis and degradation of ketone bodies | 1 | 0.047 | 1832687225 | 3-hydroxybutyrate dehydrogenase type 2 |

Table A2.
Differentially expressed proteins pathways annotated in KEGG Automatic Annotation Server.
Table A2.
Differentially expressed proteins pathways annotated in KEGG Automatic Annotation Server.
| Accession | Name | KEGG Orthology |
|---|---|---|
| 1832702676 | von Willebrand factor A domain-containing protein 5A-like isoform X1 | K24510 |
| 1832606995 | tropomyosin alpha-4 chain-like isoform X4 | K10373 |
| 1832633330 | protein phosphatase 1, catalytic subunit, alpha isozyme a | K06269 |
| 1832613275 | cytochrome c oxidase subunit 5A, mitochondrial | K02264 |
| 1832681058 | antithrombin-III | K03911 |
| 1832628613 | EH domain-containing protein 4 | K12477 |
| 1832617553 | small nuclear ribonucleoprotein Sm D1 | K11087 |
| 1832634797 | transmembrane emp24 domain-containing protein 2 isoform X1 | K20347 |
| 1832661391 | complement factor I | K01333 |
| 1832671044 | deoxyribose-phosphate aldolase | K01619 |
| 1832645324 | arginine--tRNA ligase, cytoplasmic | K01887 |
| 1832640714 | L-threonine dehydrogenase | K15789 |
| 1832610610 | 60S ribosomal protein L39 | K02924 |
| 1832685492 | 39S ribosomal protein L45, mitochondrial | K17426 |
| 1832700793 | TATA-binding protein-associated factor 2N isoform X1 | K13098 |
| 1832687225 | 3-hydroxybutyrate dehydrogenase type 2 | K25939 |
| 1832690325 | uncharacterized protein LOC117256382 isoform X3 | K08870 |
| 1832685958 | arachidonate 15-lipoxygenase B-like isoform X1 | K18684 |
| 1832683648 | nucleolin | K11294 |
| 145105480 | interleukin enhancer-binding factor 2 | K13089 |
| 161935902 | MHC class I alpha antigen | K06751 |
| 1832684689 | trypsin-3 | K23011 |
| 1832608120 | cytochrome c oxidase subunit 4 isoform 1, mitochondrial | K02263 |
| 1832640175 | FAU ubiquitin-like and ribosomal protein S30 fusion a | K02983 |
| 343459105 | NADH dehydrogenase 1 alpha, partial | K03948 |
| 1832688344 | ADP/ATP translocase 1 | K05863 |
| 1832620903 | cystatin-B | K13907 |
| 1832667755 | elongation factor 1-alpha 1a | K03231 |
| 327239696 | complement component C6, partial | K03995 |
| 1832674019 | deleted in malignant brain tumors 1 protein-like isoform X1 | K13912 |
| 1832691617 | ubiquinol-cytochrome-c reductase complex assembly factor 2 | K17682 |
| 1832614309 | very-long-chain 3-oxoacyl-CoA reductase-A | K10251 |
| 1832627296 | isovaleryl-CoA dehydrogenase, mitochondrial | K00253 |
| 1832635569 | annexin A1 | K17091 |
| 1832617105 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 6, mitochondrial | K03939 |
| 1832631564 | E3 ubiquitin-protein ligase RBX1 | K03868 |
| 197725770 | mitochondrial cytochrome C oxidase subunit Vb precursor | K02265 |
| 1832626150 | C-reactive protein | K16143 |
| 2067150115 | Thioredoxin domain-containing protein 12 | K05360 |
| 1832680021 | betaine--homocysteine S-methyltransferase 1-like | K00544 |
| 1832680525 | ubiquitin-conjugating enzyme E2 L3 | K04552 |
| 1832606043 | ras-related protein Rab-27A | K07885 |
| 1710371347 | electron transfer flavoprotein subunit beta | K03522 |
| 1832702451 | LOW-QUALITY PROTEIN: apolipoprotein(a)- | K01315 |
| 1832645146 | pigment epithelium-derived factor | K19614 |
| 1832639949 | collagen alpha-1(X) chain | K19479 |
| 1832701092 | latexin | K23594 |
| 1832662114 | parvalbumin alpha | K23926 |
| 1832699882 | peptidyl-prolyl cis-trans isomerase FKBP10 | K09575 |
| 1832670505 | coatomer subunit gamma-2 | K17267 |
| 209981964 | alpha-1-antitrypsin | K04525 |
| 1832702196 | vasodilator-stimulated phosphoprotein isoform X1 | K06274 |
| 834904058 | malate dehydrogenase | K00026 |
| 1832656308 | neural cell adhesion molecule 1a isoform X4 | K06491 |
| 1393214856 | type I glyceraldehyde-3-phosphate dehydrogenase | K00134 |
| 306008591 | heat shock protein | K04077 |
| 295792244 | fructose-bisphosphate aldolase A | K01623 |
| 328677149 | hypothetical protein, partial | K01315 |
| 343459175 | ATP synthase, H+ transporting, mitochondrial F0 complex, partial | K02138 |
| 1832625753 | glyceraldehyde-3-phosphate dehydrogenase | K00134 |
| 1832630592 | glucosidase 2 subunit beta | K08288 |
| 1832669039 | aldo-keto reductase family 1 member B1 isoform X1 | K00011 |
| 1832632244 | parvalbumin beta | K23926 |
| 1832620905 | stefin-C | K13907 |
| 1832677686 | NADH dehydrogenase [ubiquinone] iron-sulfur protein 4, mitochondrial | K03937 |
| 1832638111 | hyaluronan and proteoglycan link protein 1-like | K06848 |
| 1832619571 | tetranectin-like | K17520 |
| 1832616138 | testin | K24270 |
| 1832604382 | histone H1 | K11275 |
| 834904842 | phosphoglycerate kinase | K00927 |
| 1832645835 | ruvB-like 2 | K11338 |
| 1832635997 | proteasome subunit beta type-7 isoform X1 | K02739 |
| 1832641526 | uroporphyrinogen decarboxylase | K01599 |
| 1832690337 | chitinase, acidic.1 | K01183 |
| 1832673253 | 60S ribosomal protein L8 | K02938 |
| 1832638138 | transcription factor BTF3 | K01527 |
| 1832643512 | rho GTPase-activating protein 12-like isoform X1 | K20636 |
| 1832644997 | coatomer subunit beta’ isoform X1 | K17302 |
| 1832642084 | neurogenic differentiation factor 6-B | K09080 |
| 1832685063 | hydroperoxide isomerase ALOXE3-like | K18684 |
| 1832644368 | peroxiredoxin-6 | K11188 |
| 1832670971 | LSM8 homolog, U6 small nuclear RNA associated | K12627 |
| 1832638785 | leucine-rich alpha-2-glycoprotein-like | K25431 |
| 1832664977 | cytochrome c oxidase subunit 6B1 | K02267 |
| 1832612090 | heat shock 70 kDa protein 4b | K09489 |
| 1042045193 | glycine dehydrogenase (aminomethyl-transferring) | K00281 |
| 1832622572 | acyl-CoA thioesterase 9, tandem duplicate 1 isoform X1 | K17361 |
| 1832656769 | vesicle-associated membrane protein 2 | K13504 |
| 328677247 | glyceraldehyde 3-phosphate dehydrogenase isoform, partial | K10705 |
| 1832679851 | drebrin-like b isoform X1 | K20520 |
| 1832650400 | prefoldin subunit 1 isoform X1 | K09548 |
| 221048061 | fructose-bisphosphate aldolase | K01623 |
| 1832658469 | fibrinogen beta chain | K03904 |
| 1832612579 | 60S ribosomal protein L27a | K02900 |
| 222087999 | fibrinogen beta chain precursor | K03904 |
| 1832689402 | fibrinogen gamma chain | K03905 |
| 1832662038 | leukocyte cell-derived chemotaxin-2 | K25755 |
| 1832685624 | microfibril-associated glycoprotein 4 | K25409 |
| 1832634516 | keratin, type I cytoskeletal 13 | K07604 |
| 1832685241 | beta-2-glycoprotein 1 | K17305 |
| 1832623025 | apolipoprotein Eb | K04524 |
| 405790938 | MHC class II antigen, partial | K06752 |
| 295792326 | mitochondrial cytochrome c oxidase subunit 7C | K02272 |
| 1832626146 | allograft inflammatory factor 1-like | K18617 |
| 1832610590 | DNA-directed RNA polymerase I subunit RPA1 | K02999 |
| 295792242 | nucleoside diphosphate kinase B | K00940 |
| 1832702726 | serpin H1b | K09501 |
| 1832665474 | sulfotransferase 2B1 | K01015 |
| 1832641369 | phosphotriesterase-related protein | K07048 |
| 1832686354 | puromycin-sensitive aminopeptidase | K08776 |
| 1832695641 | N-alpha-acetyltransferase 10 isoform X1 | K20791 |
| 1832682447 | dedicator of cytokinesis protein 7 isoform X1 | K21852 |
| 1832667938 | sideroflexin-3 | K23500 |
| 1832608362 | AP-1 complex subunit gamma-1 isoform X1 | K12391 |
| 1832621671 | rRNA 2’-O-methyltransferase fibrillarin | K14563 |
| 1832611868 | transmembrane 9 superfamily member 2 isoform X1 | K17086 |
| 1832671468 | coagulation factor VIIi | K01320 |
| 1832618423 | class I histocompatibility antigen, F10 alpha chain-like isoform X1 | K06751 |
| 62255674 | immunoglobulin mu heavy chain | K06856 |
| 157929900 | ribosomal protein L23 | K02894 |
| 2022331196 | immunoglobulin M heavy-chain constant mu variant 1 | K06856 |
| 834906005 | glyceraldehyde-3-phosphate dehydrogenase | K00134 |
| 1832604432 | erythroblast NAD(P)(+)--arginine ADP-ribosyltransferase | K19977 |
| 1832697311 | nucleobindin-2a isoform X1 | K20371 |
| 1832604652 | fibrinogen alpha chain-like | K03903 |
| 326632479 | MHC class II antigen | K06752 |
| 1832660271 | macrosialin | K06501 |
| 380006108 | MHC class II antigen | K06752 |
| 2022331198 | immunoglobulin M heavy-chain constant mu variant 2 | K06856 |
References
- Che Cob, Z.; Kumar Das, S. Optimum temperature for the growth form of Tiger grouper (Epinephelus fuscoguttatus ♀) × Giant grouper (Epinephelus lanceolatus ♂) hybrid. Sains Malays. 2016, 45, 541–549. [Google Scholar]
- Mo, Z.Q.; Wu, H.C.; Hu, Y.T.; Lu, Z.J.; Lai, X.L.; Chen, H.P.; He, Z.; Luo, X.; Li, Y.; Dan, X. Transcriptomic analysis reveals innate immune mechanisms of an underlying parasite-resistant grouper hybrid (Epinephelus fuscogutatus × Epinephelus lanceolatus). Fish Shellfish. Immunol. 2021, 119, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Glamuzina, B.; Rimmer, M.A. Grouper Aquaculture World Status and Perspectives. In Biology and Ecology of Groupers; CRC Press: Boca Raton, FL, USA, 2022; pp. 166–190. [Google Scholar]
- Rahman, M.A.; Lee, S.-G.; Yusoff, F.; Rafiquzzaman, S. Hybridization and Its Application in Aquaculture. In Sex Control in Aquaculture; John Wiley & Sons, Ltd.: Chichester, UK, 2018; pp. 163–178. [Google Scholar] [CrossRef]
- Ch ’ng, C.L.; Senoo, S. Egg and Larval Development of a New Hybrid Grouper, Tiger Grouper (Epinephelus fuscoguttatus) and Giant Grouper (Epinephelus lanceolatus). Aquac. Sci. 2008, 56, 505–512. [Google Scholar]
- Rimmer, M.A.; Glamuzina, B. A review of grouper (Family Serranidae: Subfamily Epinephelinae) aquaculture from a sustainability science perspective. Rev. Aquac. 2017, 11, 58–87. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Balasundaram, C.; Heo, M.-S. Molecular studies, disease status and prophylactic measures in grouper aquaculture: Economic importance, diseases and immunology. Aquaculture 2010, 309, 1–14. [Google Scholar] [CrossRef]
- Petersen, E.H.; My Chinh, D.T.; Diu, N.T.; Phuoc, V.V.; Phuong, T.H.; Dung, N.V.; Dat, N.K.; Giang, P.T.; Glencross, B.D. Bioeconomics of grouper, Serranidae: Epinephelinae, culture in Vietnam. Rev. Fish. Sci. 2013, 21, 49–57. [Google Scholar] [CrossRef]
- Bunlipatanon, P.; U-taynapun, K. Growth performance and disease resistance against Vibrio vulnificus infection of novel hybrid grouper (Epinephelus lanceolatus × Epinephelus fuscoguttatus). Aquac. Res. 2017, 48, 1711–1723. [Google Scholar] [CrossRef]
- Ye, G.; Dong, X.; Yang, Q.; Chi, S.; Liu, H.; Zhang, H.; Tan, B.; Zhang, S. Low-gossypol cottonseed protein concentrate used as a replacement of fish meal for juvenile hybrid grouper (Epinephelus fuscoguttatus♀ × Epinephelus lanceolatus♂): Effects on growth performance, immune responses and intestinal microbiota. Aquaculture 2020, 524, 735309. [Google Scholar] [CrossRef]
- Liao, J.; Cai, Y.; Wang, X.; Shang, C.; Zhang, Q.; Shi, H.; Wang, S.; Zhang, D.; Zhou, Y. Effects of a potential host gut-derived probiotic, Bacillus subtilis 6-3-1, on the growth, non-specific immune response and disease resistance of hybrid grouper (Epinephelus fuscoguttatus × Epinephelus lanceolatus♂). Probiotics Antimicrob. Proteins 2021, 13, 1119–1137. [Google Scholar] [CrossRef]
- Xiao, H.; Liu, M.; Li, S.; Shi, D.; Zhu, D.; Ke, K.; Xu, Y.; Dong, D.; Zhu, L.; Yu, Q. Isolation and characterization of a ranavirus associated with disease outbreaks in cultured hybrid grouper (♀ Tiger Grouper Epinephelus fuscoguttatus × giant grouper Epinephelus lanceolatus) in Guangxi, China. J. Aquat. Anim. Health 2019, 31, 364–370. [Google Scholar] [CrossRef]
- Zhu, Z.M.; Dong, C.F.; Weng, S.P.; He, J.G. The high prevalence of pathogenic Vibrio harveyi with multiple antibiotic resistance in scale drop and muscle necrosis disease of the hybrid grouper, Epinephelus fuscoguttatus (♀) × Epinephelus lanceolatus (♂), in China. J. Fish Dis. 2018, 41, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Suyanti, E.; Mahasri, G.; Lokapirnasari, W.P. Marine Leech Zeylanicobdella arugamensis Infestation As A Predisposing Factor For Vibrio alginolyticus Infection On The Hybrid Grouper “Cantang”(Epinephelus fuscoguttatus × Epinephelus lanceolatus) From Traditional Ponds In The Kampung Kerapu Lamongan Eas. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2021; p. 12035. [Google Scholar]
- Lee, K.-K. Pathogenesis studies on Vibrio alginolyticus in the grouper, Epinephelus malabaricus, Bloch et Schneider. Microb. Pathog. 1995, 19, 39–48. [Google Scholar] [CrossRef]
- Mohamed Alipiah, N.; Ramli, N.; Low, C.; Shamsudin, M.; Yusoff, F. Protective effects of sea cucumber surface-associated bacteria against Vibrio harveyi in brown-marbled grouper fingerlings. Aquac. Environ. Interact. 2016, 8, 147–155. [Google Scholar] [CrossRef]
- Mohamad, A.; Mursidi, F.-A.; Zamri-Saad, M.; Amal, M.N.A.; Annas, S.; Monir, S.; Loqman, M.; Hairudin, F.; Al-Saari, N.; Ina-Salwany, Y. Laboratory and Field Assessments of Oral Vibrio Vaccine Indicate the Potential for Protection against Vibriosis in Cultured Marine Fishes. Animals 2022, 12, 133. [Google Scholar] [CrossRef]
- Rodrigues, P.M.; Martin, S.A.; Silva, T.S.; Boonanuntanasarn, S.; Schrama, D.; Moreira, M.; Raposo, C. Proteomics in fish and aquaculture research. In Proteomics in Domestic Animals: From Farm to Systems Biology; Springer International Publishing: Cham, Switzerland, 2018; pp. 311–338. [Google Scholar]
- Mischak, H.; Ioannidis, J.P.A.; Argiles, A.; Attwood, T.K.; Bongcam-Rudloff, E.; Broenstrup, M. Implementation of proteomic biomarkers: Making it work. Eur. J. Clin. Investig. 2012, 42, 1027–1036. [Google Scholar] [CrossRef]
- Braceland, M.; McLoughlin, M.F.; Tinsley, J.; Wallace, C.; Cockerill, D.; McLaughlin, M.; Eckersall, P.D. Serum enolase: A non-destructive biomarker of white skeletal myopathy during pancreas disease (PD) in Atlantic salmon Salmo salar L. J. Fish Dis. 2015, 38, 821–831. [Google Scholar] [CrossRef]
- Coates, C.J.; Decker, H. Immunological properties of oxygen-transport proteins: Hemoglobin, hemocyanin and hemerythrin. Cell. Mol. Life Sci. 2016, 74, 293–317. [Google Scholar] [CrossRef]
- Hua, Y.; Qingsong, L.; Hui, L. Applications of transcriptomics and proteomics in understanding fish immunity. Fish Shellfish. Immunol. 2018, 77, 319–327. [Google Scholar]
- Leng, W.; Wu, X.; Shi, T.; Xiong, Z.; Yuan, L.; Jin, W.; Gao, R. Untargeted Metabolomics on Skin Mucus Extract of Channa argus against Staphylococcus aureus: Antimicrobial Activity and Mechanism. Foods 2021, 10, 2995. [Google Scholar] [CrossRef]
- Leng, W.; Wu, X.; Xiong, Z.; Shi, T.; Sun, Q.; Yuan, L.; Gao, R. Study on antibacterial properties of mucus extract of snakehead (Channa argus) against Escherichia coli and its application in chilled fish fillets preservation. LWT 2022, 167, 113840. [Google Scholar] [CrossRef]
- Lau, B.Y.C.; Amiruddin, M.D.; Othman, A. Proteomics analysis on lipid metabolism in Elaeis guineensis and Elaeis oleifera. Data Brief 2020, 31, 105714. [Google Scholar] [CrossRef] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Kammers, K.; Cole, R.N.; Tiengwe, C.; Ruczinski, I. Detecting significant changes in protein abundance. EuPA Open Proteom. 2015, 7, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Dash, S.; Das, S.; Samal, J.; Thatoi, H.N. Epidermal mucus, a major determinant in fish health: A review. Iran. J. Vet. Res. 2018, 19, 72–81. [Google Scholar] [CrossRef]
- Valdenegro-Vega, V.A.; Crosbie, P.; Bridle, A.; Leef, M.; Wilson, R.; Nowak, B.F. Differentially expressed proteins in gill and skin mucus of Atlantic salmon (Salmo salar) affected by amoebic gill disease. Fish Shellfish Immunol. 2014, 40, 69–77. [Google Scholar] [CrossRef]
- Raposo de Magalhães, C.S.F.; Cerqueira, M.A.C.; Schrama, D.; Moreira, M.J.V.; Boonanuntanasarn, S.; Rodrigues, P.M.L. A Proteomics and other Omics approach in the context of farmed fish welfare and biomarker discovery. Rev. Aquac. 2020, 12, 122–144. [Google Scholar] [CrossRef]
- Canellas, A.L.B.; Costa, W.F.; Freitas-Silva, J.; Lopes, I.R.; de Oliveira, B.F.R.; Laport, M.S. In sickness and in health: Insights into the application of omics in aquaculture settings under a microbiological perspective. Aquaculture 2022, 554, 738132. [Google Scholar] [CrossRef]
- Drago, F.; Sautière, P.E.; le Marrec-Croq, F.; Accorsi, A.; van Camp, C.; Salzet, M.; Lefebvre, C.; Vizioli, J. Microglia of medicinal leech (Hirudo medicinalis) express a specific activation marker homologous to vertebrate ionized calcium-binding adapter molecule 1 (Iba1/alias aif-1). Dev. Neurobiol. 2014, 74, 987–1001. [Google Scholar] [CrossRef]
- Lü, A.; Hu, X.; Wang, Y.; Shen, X.; Li, X.; Zhu, A.; Tian, J.; Ming, Q.; Feng, Z. iTRAQ analysis of gill proteins from the zebrafish (Danio rerio) infected with Aeromonas hydrophila. Fish Shellfish Immunol. 2014, 36, 229–239. [Google Scholar] [CrossRef]
- Mierziak, J.; Burgberger, M.; Wojtasik, W. 3-Hydroxybutyrate as a Metabolite and a Signal Molecule Regulating Processes of Living Organisms. Biomolecules 2021, 11, 402. [Google Scholar] [CrossRef]
- Stubbs, B.J.; Koutnik, A.P.; Goldberg, E.L.; Upadhyay, V.; Turnbaugh, P.J.; Verdin, E.; Newman, J.C. Investigating Ketone Bodies as Immunometabolic Countermeasures against Respiratory Viral Infections. Med 2020, 1, 43–65. [Google Scholar] [CrossRef] [PubMed]
- Dedkova, E.N.; Blatter, L.A. Role of Î2-hydroxybutyrate, its polymer poly-Î2-hydroxybutyrate and inorganic polyphosphate in mammalian health and disease. Front. Physiol. 2014, 17, 5. [Google Scholar] [CrossRef] [PubMed]
- Graff, E.C.; Fang, H.; Wanders, D.; Judd, R.L. Anti-inflammatory effects of the hydroxycarboxylic acid receptor 2. Metabolism 2016, 65, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Ballarin, L.; Cammarata, M.; Franchi, N.; Parrinello, N. Routes in Innate Immunity Evolution: Galectins and Rhamnose-binding Lectins in Ascidians. In Marine Protein and Peptides. Biological Activities and Applications; Wiley-Blackwell: Chichester, UK, 2013; pp. 185–205. [Google Scholar] [CrossRef]
- Shiina, N.; Tateno, H.; Ogawa, T.; Muramoto, K.; Saneyoshi, M.; Kamiya, H. Isolation and characterization of L-rhamnose-binding lectins from chum salmon (Oncorhynchus keta) eggs. Fish. Sci. 2002, 68, 1352–1366. [Google Scholar] [CrossRef]
- Cammarata, M.; Parisi, M.; Benenati, G.; Vasta, G.; Parrinello, N. A rhamnose-binding lectin from sea bass (Dicentrarchus labrax) plasma agglutinates and opsonizes pathogenic bacteria. Dev. Comp. Immunol. 2014, 44, 332–340. [Google Scholar] [CrossRef]
- Okamoto, M.; Tsutsui, S.; Tasumi, S.; Suetake, H.; Kikuchi, K.; Suzuki, Y. Tandem repeat l-rhamnose-binding lectin from the skin mucus of ponyfish, Leiognathus nuchalis. Biochem. Biophys. Res. Commun. 2005, 333, 463–469. [Google Scholar] [CrossRef]
- Vibhute, P.; Radhakrishnan, A.; Sivakamavalli, J.; Chellapandian, H.; Selvin, J. Antimicrobial and Immunomodulatory Role of Fish Lectins. In Aquat. Lectins; Springer: Singapore, 2022; pp. 257–286. [Google Scholar] [CrossRef]
- Maher, K.; Kokelj, B.J.; Butinar, M.; Mikhaylov, G.; Manček-Keber, M.; Stoka, V.; Vasiljeva, O.; Turk, B.; Grigoryev, S.A.; Kopitar-Jerala, N. A Role for Stefin B (Cystatin B) in Inflammation and Endotoxemia. J. Biol. Chem. 2014, 289, 31736–31750. [Google Scholar] [CrossRef]
- Rivera-Rivera, L.; Perez-Laspiur, J.; Colón, K.; Meléndez, L.M. Inhibition of interferon response by cystatin B: Implication in HIV replication of macrophage reservoirs. J. Neurovirol. 2012, 18, 20–29. [Google Scholar] [CrossRef][Green Version]
- Wickramasinghe, P.; Kwon, H.; Elvitigala, D.A.S.; Wan, Q.; Lee, J. Identification and characterization of cystatin B from black rockfish, Sebastes schlegelii, indicating its potent immunological importance. Fish Shellfish Immunol. 2020, 104, 497–505. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, S.; Wang, G. Response of complement expression to challenge with lipopolysaccharide in embryos/larvae of zebrafish Danio rerio: Acquisition of immunocompetent complement. Fish Shellfish Immunol. 2008, 25, 264–270. [Google Scholar] [CrossRef]
- Kimura, A.; Nonaka, M. Molecular cloning of the terminal complement components C6 and C8β of cartilaginous fish. Fish Shellfish Immunol. 2009, 27, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wu, X. Identification of differentially expressed genes in lipopolysaccharide-stimulated yellow grouper Epinephelus awoara spleen. Fish Shellfish Immunol. 2007, 23, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Schorn, T. Identification and Modulation of the Allograft Inflammatory Factor-1 (AIF-1) Homologous in the Medicinal Leech Hirudo Medicinalis. Doctoral Dissertation, Universita degli Studi dell’lnsubria, Varese, Italy, 2014. [Google Scholar]
- De Zoysa, M.; Nikapitiya, C.; Kim, Y.; Oh, C.; Kang, D.H.; Whang, I.; Kim, S.-J.; Lee, J.-S.; Choi, C.Y.; Lee, J. Allograft inflammatory factor-1 in disk abalone (Haliotis discus): Molecular cloning, transcriptional regulation against immune challenge and tissue injury. Fish Shellfish Immunol. 2010, 29, 319–326. [Google Scholar] [CrossRef]
- Miyata, M.; Iinuma, K.; Miyazaki, T. DNA cloning and characterization of an allograft inflammatory factor-1 homologue in red sea bream (Chrysophrys major). Aquaculture 2001, 194, 63–74. [Google Scholar] [CrossRef]
- Ying, Z.Y.; Qiu, L.Y.; You, X. Functional Identification of Allograft Inflammatory Factor 1-Like Gene in Luning Chicken. Anim. Biotechnol. 2018, 29, 234–240. [Google Scholar]
- Ligtenberg, A.J.M.; Karlsson, N.G.; Veerman, E.C.I. Deleted in Malignant Brain Tumors-1 Protein (DMBT1): A Pattern Recognition Receptor with Multiple Binding Sites. Int. J. Mol. Sci. 2010, 11, 5212–5233. [Google Scholar] [CrossRef]
- Müller, H.; Nagel, C.; Weiss, C.; Mollenhauer, J.; Poeschl, J. Deleted in malignant brain tumors 1 (DMBT1) elicits increased VEGF and decreased IL-6 production in type II lung epithelial cells. BMC Pulm. Med. 2015, 15, 32. [Google Scholar] [CrossRef]
- Stenger, S.; Modlin, R.L. Cytotoxic T cell responses to intracellular pathogens. Curr. Opin. Immunol. 1998, 10, 471–477. [Google Scholar] [CrossRef]
- Grimholt, U. MHC and Evolution in Teleosts. Biology 2016, 5, 6. [Google Scholar] [CrossRef]
- Balebona, M.C.; Andreu, M.J.; Bordas, M.A.; Zorrilla, I.; Moriñigo, M.A.; Borrego, J.J. Pathogenicity of Vibrio alginolyticus for Cultured Gilt-Head Sea Bream (Sparus aurata L.). Appl. Environ. Microbiol. 1998, 64, 4269–4275. [Google Scholar] [CrossRef]
- Solito, E.; De Coupade, C.; Canaider, S.; Goulding, N.J.; Perretti, M. Transfection of annexin 1 in monocytic cells produces a high degree of spontaneous and stimulated apoptosis associated with caspase-3 activation. J. Cereb. Blood Flow Metab. 2001, 133, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Barton, G.J.; Newman, R.H.; Freemont, P.S.; Crumpton, M.J. Amino acid sequence analysis of the annexin super-gene family of proteins. JBIC J. Biol. Inorg. Chem. 1991, 198, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Linke, B.; Abeler-Dörner, L.; Jahndel, V.; Kurz, A.; Mahr, A.; Pfrang, S.; Linke, L.; Krammer, P.H.; Weyd, H. The Tolerogenic Function of Annexins on Apoptotic Cells Is Mediated by the Annexin Core Domain. J. Immunol. 2015, 194, 5233–5242. [Google Scholar] [CrossRef]
- Carson, D.A.; Ribeiro, J.M. Apoptosis and disease. Lancet 1993, 341, 1251–1254. [Google Scholar] [CrossRef]
- Wang, H.-G.; Pathan, N.; Ethell, I.M.; Krajewski, S.; Yamaguchi, Y.; Shibasaki, F.; McKeon, F.; Bobo, T.; Franke, T.F.; Reed, J.C. Ca 2+ -Induced Apoptosis Through Calcineurin Dephosphorylation of BAD. Science 1999, 284, 339–343. [Google Scholar] [CrossRef]
- Solito, E.; Kamal, A.; Russo-Marie, F.; Buckingham, J.C.; Marullo, S.; Perretti, M. A novel calcium-dependent proapoptotic effect of annexin 1 on human neutrophils. FASEB J. 2003, 17, 1–27. [Google Scholar] [CrossRef]
- Roh, J.S.; Sohn, D.H. Damage-Associated Molecular Patterns in Inflammatory Diseases. Immune Netw. 2018, 18, e27. [Google Scholar] [CrossRef] [PubMed]
- Maderna, P.; Yona, S.; Perretti, M.; Godson, C. Modulation of Phagocytosis of Apoptotic Neutrophils by Supernatant from Dexamethasone-Treated Macrophages and Annexin-Derived Peptide Ac2–26. J. Immunol. 2005, 174, 3727–3733. [Google Scholar] [CrossRef]
- Young, N.; Cooper, G.; Nowak, B.; Koop, B.; Morrison, R. Coordinated down-regulation of the antigen processing machinery in the gills of amoebic gill disease-affected Atlantic salmon (Salmo salar L.). Mol. Immunol. 2008, 45, 2581–2597. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).