1. Introduction
The grass carp (
Ctenopharyngodon idella) is the most widespread freshwater fish species in China, with an annual yield exceeding 5.5 million tons, accounting for approximately 20% of the country’s total freshwater fish production [
1]. It is widely consumed as a good source of high-quality proteins. Grass carp is an herbivorous fish that has been widely introduced throughout the world, and it consumes up to 27.6 kg of vegetation per kg of fish every year [
2]. The grass carp is not only widely farmed in ponds, but it also widely distributed in streams, rivers, reservoirs, lakes, and wetlands to regulate water quality [
1]. However, the grass carp, especially in ponds, is susceptible to various diseases, including bacterial septicemia caused by
Aeromonas spp., viral hemorrhagic disease caused by grass carp reovirus (GCRV), and columnaris disease caused by
Flavobacterium columnare [
3,
4,
5]. Although GCRV severely affects grass carp aquaculture, vaccination is widely used in China to prevent and control GCRV. Bacterial diseases are common in grass carp aquaculture, and the main pathogens are
F. columnare [
3],
Aeromonas hydrophila [
4],
Acinetobacter lwoffii [
5],
Vibrio mimicus [
6], and
Vibrio vulnificus [
7]. Compared with the other freshwater fish species (e.g. crucian carp and bighead carp), the grass carp is more susceptible to
F. columnare and
Aeromonas sp. in China [
3,
8].
F. columnare causes serious clinical symptoms, including gill lesions, skin lesions, and fin erosions, resulting in severe economic losses relative to grass carp [
3].
Recently, bacterial diseases in grass carp in China have been prevented and controlled using antibiotics and disinfectants. The large-scale and repeated application of these chemical drugs can cause a series of problems, including drug residues, bacterial resistance, and environmental pollution [
9]. Thus, biological control using probiotics, which causes less contamination and has a low risk of resistance, is an alternative method for preventing diseases.
Bacillus is ubiquitous in water, soil, air, animals, and plants and is an important biocontrol bacterium in crops, livestock, animals, and fish.
Bacillus probiotics have been extensively used in aquaculture as alternatives to antibiotics for fish farming [
10].
Bacillus velezensis exhibits broad-spectrum antibacterial activity, making it a vital biocontrol agent against various plant and animal diseases [
11]. For instance,
B.
velezensis demonstrates potent biocontrol effects on plant diseases including potato scab [
12], lotus root rot [
13], and wheat
Fusarium head blight [
14], as well as on infections in crucian carp (
Carassius auratus) caused by
A. hydrophila [
15], those in Nile tilapia (
Oreochromis niloticus) caused by
Streptococcus agalactiae [
16], in hybrid grouper resulting from
Vibrio harveyi [
17], and in grass carp from
A. hydrophila [
18].
B. velezensis FZB42 strain is successfully used in agriculture to biocontrol rhizobacteria and to promote plant growth [
11]. Additionally,
B. velezensis CPA1-1 is a potential probiotic for inhibiting non-O1
Vibrio cholerae and improving host immunity in oriental river prawn (
Macrobrachium nipponense) [
19].
B. velezensis Bs916 is used as a potential probiotic for the biological control of white spot disease in crayfish [
20].
B.
velezensis could produce anti-microbial active compounds, such as bacilysin, bacillibactin, bacillaene, fengycin, marcolactin H, and surfactin, exhibiting a strong antagonistic action against animal and plant pathogens [
21]. These secondary metabolites also induce systemic resistance in plants. Genome sequence analysis indicated that
B.
velezensis harbors many gene clusters encoding secondary metabolites, i.e.,
B.
velezensis VJH504 contains several gene clusters encoding NRPS, transAT-PKS, T3PKS, and PKS-like types of secondary metabolites [
22]; and
B. velezensis HNA3 possesses 12 gene clusters related to 14 secondary metabolites with bioactive compounds [
23]. Furthermore,
B.
velezensis shows high hydrolase activity due to the presence of protease, chitinase, cellulase, and glucanase, which is associated with carbon source and cellulose utilization [
24].
The aim of this study is to isolate probiotics that can control aquaculturally important pathogens found in the intestines of the healthy grass carp and to evaluate the biocontrol potential and safety of the strain. In this study, a representative strain CYS06 of Bacillus, with antagonistic activity, was isolated from the intestines of healthy grass carp. The broad-spectrum bacteriostatic activity, extracellular enzyme activity, environmental adaptability, and biocontrol efficacy of this strain were analyzed. The whole genome of this strain was sequenced, and key functional and biosafety-related genes were analyzed. The strain CYS06 is an excellent candidate for commercialization as a biocontrol agent, especially in grass carp farming.
4. Discussion
Probiotics, including bacteria, yeast, and actinomycetes, are considered to be environment-friendly biological control agents for the prevention and control of fish diseases. Antagonistic probiotics are commonly used to biocontrol aquatic animals pathogens [
17,
19,
37], including
A. hydrophila,
Aeromonas salmonicida,
S. agalactiae,
V. harveyi, and
V.
cholerae.
B.
velezensis exhibits highly antagonistic effects against various aquatic animals pathogens, representing a new research hotspot for the biological control of bacterial diseases in aquatic animals [
21]. However, limited probiotics are available for the prevention and control of grass carp bacterial diseases. Given that
Bacillus could form endospores to protect it from extreme stresses, the
Bacillus strain with antagonistic activity against
Aeromonas sp. and
F. columnare was screened to control fish bacterial diseases. Among 112 strains of
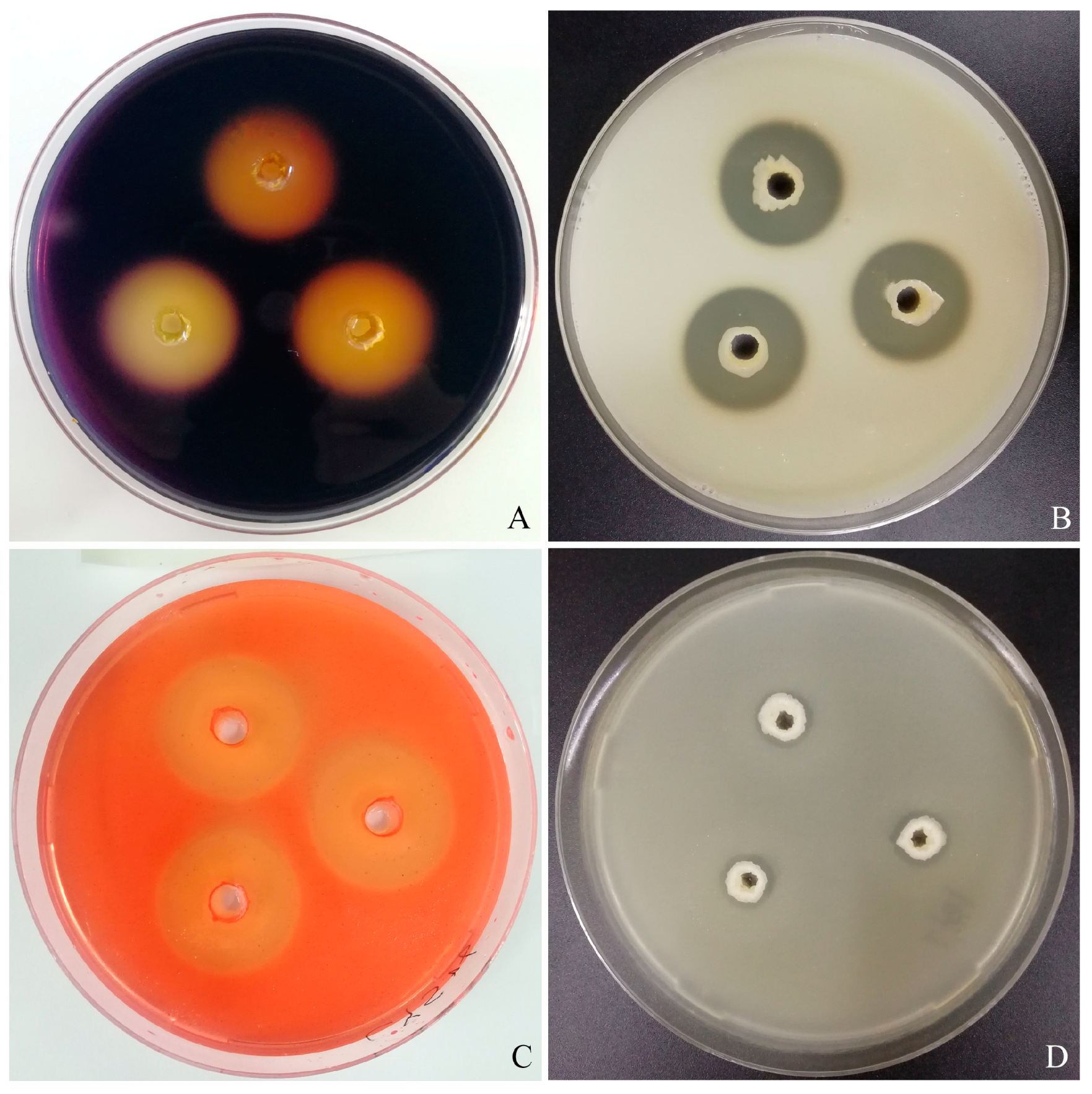
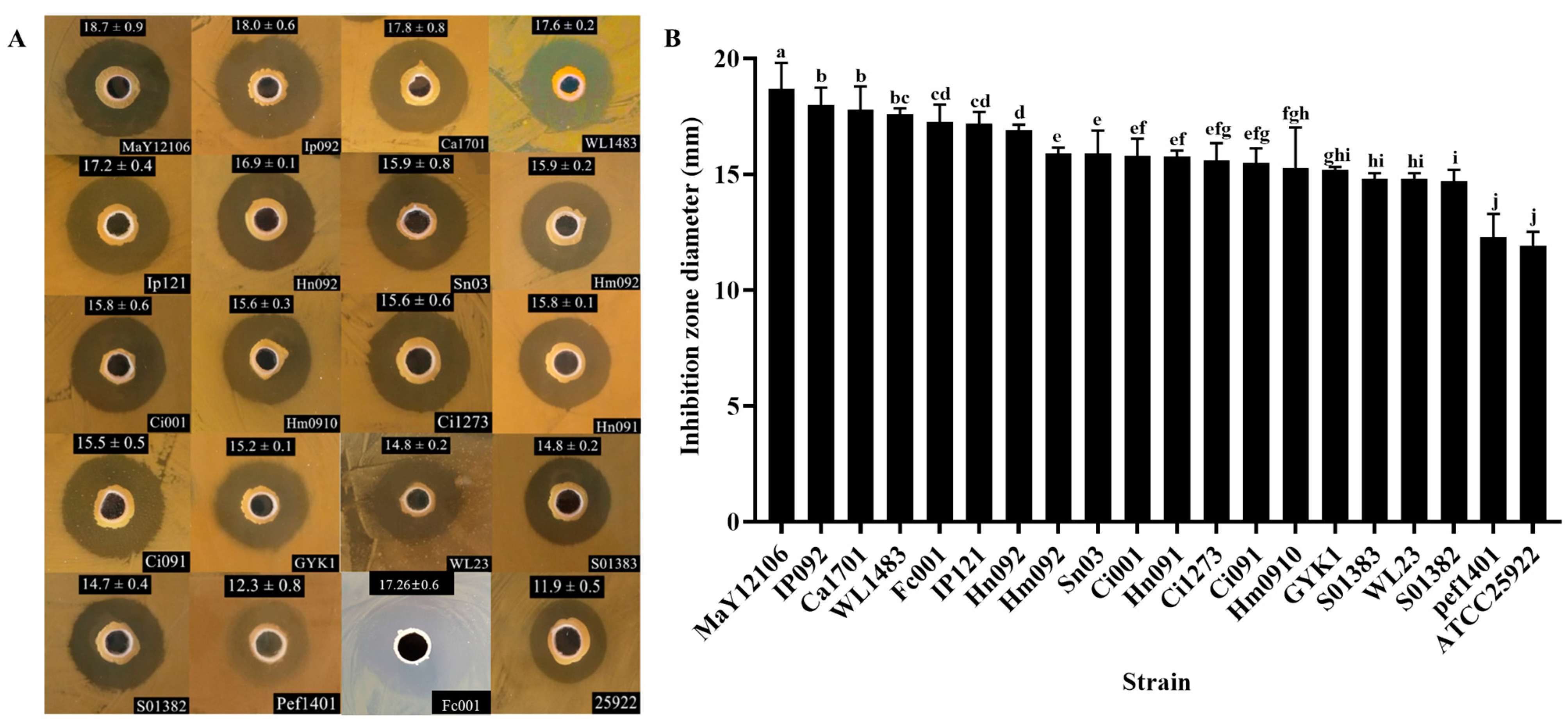
Bacillus, 9 strains showed antagonistic activity, and strain CYS06 exhibits the strongest antagonistic activity. The strain CYS06, exhibiting several highly antagonistic fish pathogens, was isolated from the intestine of healthy grass carp, and this strain was identified as
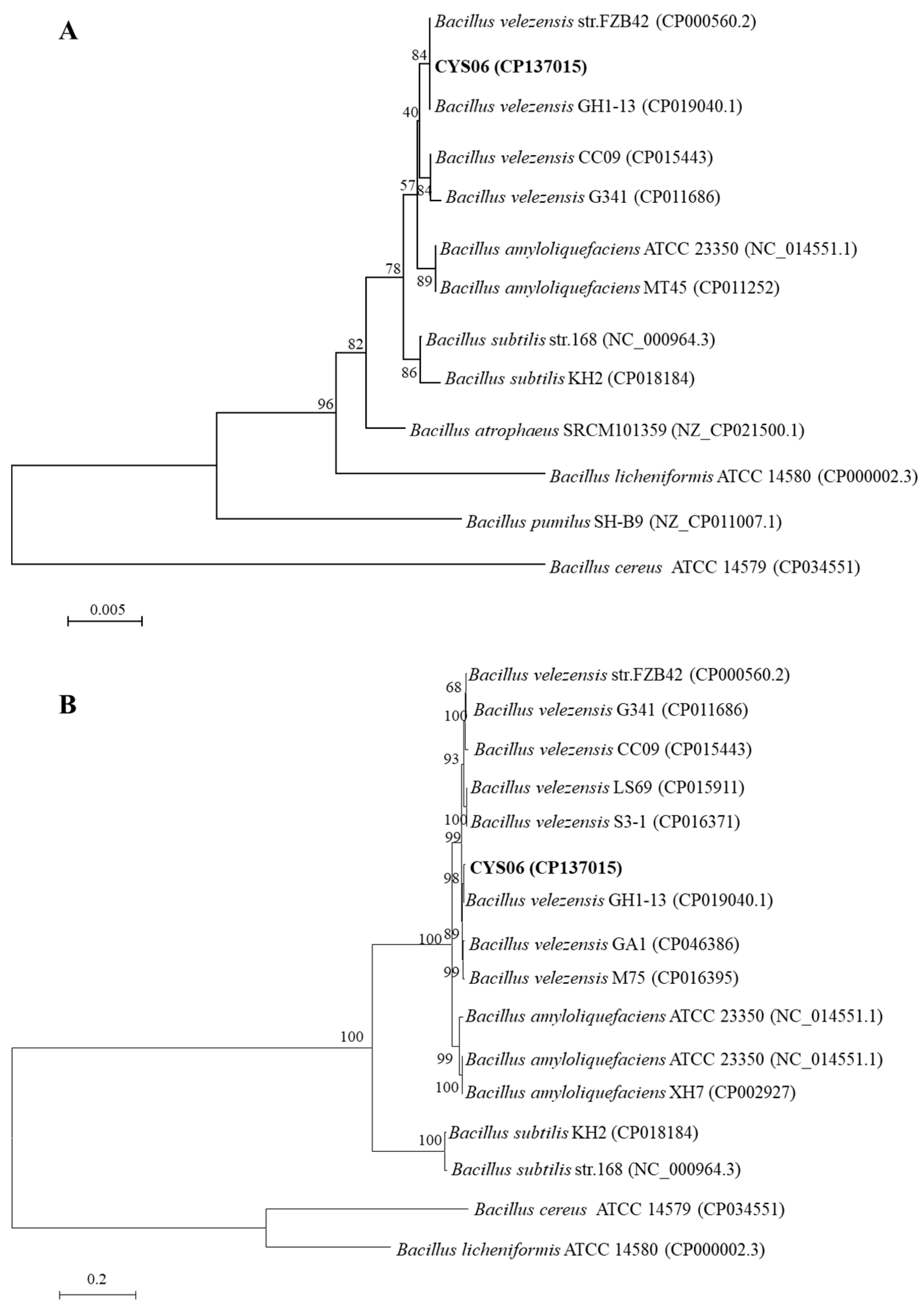
B. velezensis, based on molecular identification, including sequence analysis of the 16S rRNA,
gyrA, ANI, and DDH.
Previous reports showed that
B. velezensis possessed anti-microbial active compounds against a broad range of fish pathogens, including the bacteria belonging to the genus of
Aeromonas,
Edwardsiella,
Streptococcus,
Vibrio,
Nocardia, and
Salmonella [
10,
16,
27]. In this study,
B. velezensis CYS06 exhibited broad-spectrum antagonistic activity against
Aermonas spp.,
E. ictaluri, and
F. columnare. Specifically, the
A.
schubertii WL-23 strain showed multiple resistance characteristics, such as resistance to macrolides, tetracyclines, aminoglycosides, β-lactams, chloramphenicols, lincomycin, sulfonamides, and rifampicin (data published in Chinese). The strain CYS06 can significantly inhibit the WL-23 strain, indicating a difference in the antibacterial mechanism between CYS06 and common antibiotics. These results suggest that the antibacterial substances secreted by the strain CYS06 can be used for screening new drugs.
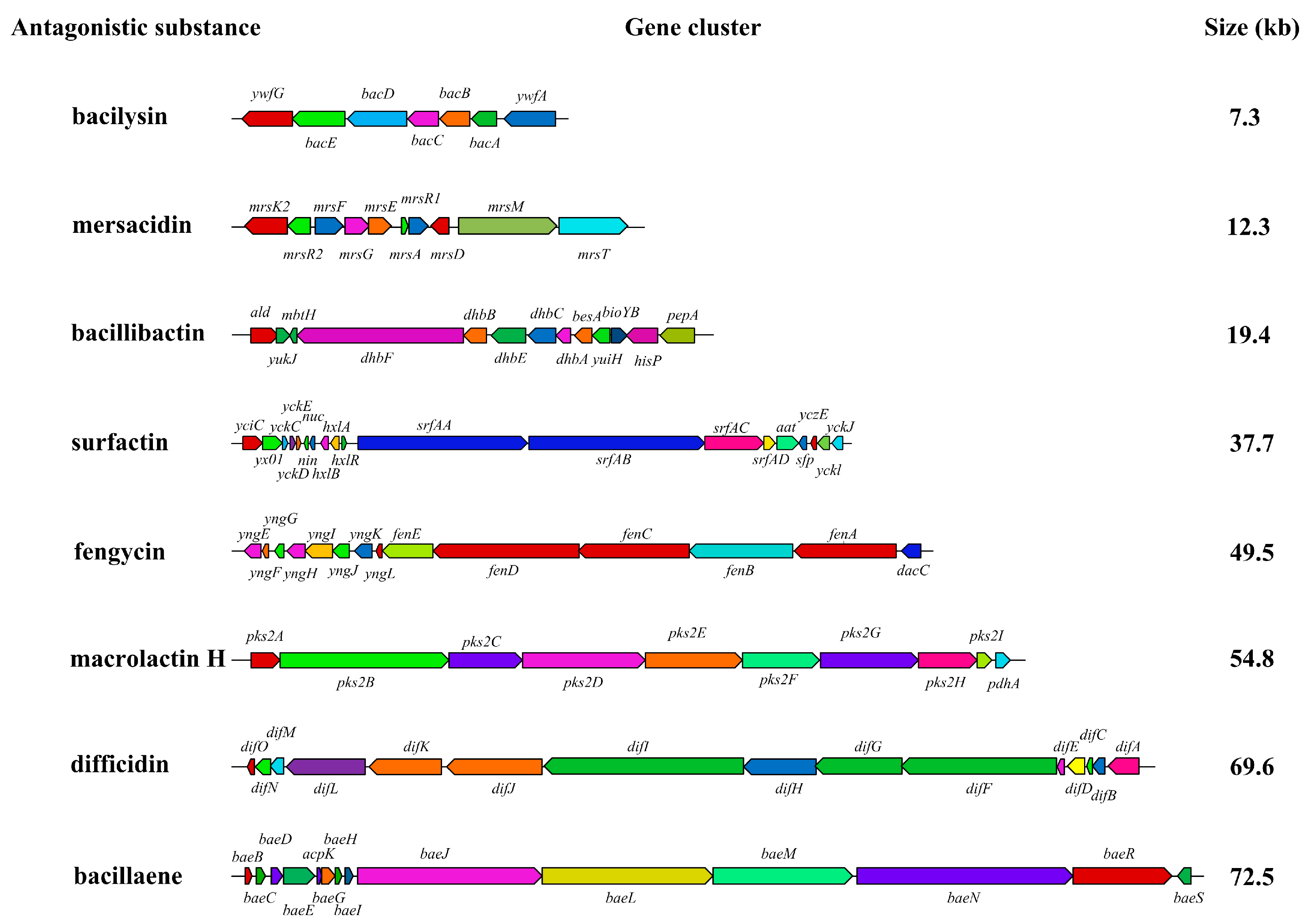
B. velezensis could produce antimicrobial substances such as polyketides, lipopeptides, and peptides, which inhibit bacteria and fungi [
21]. The secondary metabolites produced by
B. velezensis involve bacillibactin, fengycin, bacilysin, surfactin, difficidin, and mersacidin, which exhibit broad antagonistic activities against pathogenic bacteria or fungi [
22]. For example, anti-microbial substances such as bacillibactin chelated iron, fengycin altered cell wall permeability, bacilysin hindered glucosamine synthesis, surfactin destroyed cell membrane, and difficidin inhibited the protein biosynthesis of bacteria [
23]. Mersacidin belongs to the type B lantibiotics, which can inhibit bacteria by two methods: the inhibition of cell wall biosynthesis by scavenging the peptidoglycan precursor lipid Ⅱ, and lysis of the cell membrane by forming pores [
38]. In this study, eight biosynthesis gene clusters related to bacilysin, mersacidin, bacillibactin, surfactin, fengycin, macrolactin H, difficidin, and bacillaene in the genome of strain CYS06 were predicted using antiSMASH software, indicating that strain CYS06 has the ability to produce a wealth of secondary metabolites with antagonistic activity. Moreover, the antimicrobial substances of
B. velezensis showed high thermal stability, broad pH tolerance, and resistant to enzyme digestion in the previously described study [
27,
39]. Thus, we deduce that the potential antimicrobial substances produced by CYS06 could exert antibacterial effects in various environments, indicating that this strain has promising applications. The application of strain CYS06 or its secondary metabolites could reduce the use of antibiotics in aquaculture.
B. velezensis could secrete hydrolases, such as protease, chitinase, cellulase, and glucanase, which could significantly control plant diseases and promote plant growth [
24,
40]. The strain CYS06 possesses high protease activity, which can promote its antibacterial properties; moreover, the protease, amylase, cellulase, and lipase secreted by CYS06 could promote the digestion and utilization of nutrient substances in the fish gastrointestinal tract, if this strain is used as a feed additive. In this study, the genes related to protease, α-amylase, cellulase, and lipase were confirmed by genome CAZymes analysis. The gene families related to cellulase and hemi-cellulase (GH5, GH11, GH26, GH43, GH51, and GH53), starch hydrolases (GH13 and GH126), and chitin degradation (GH18, GH23, CE9, and CBM50) were found in the genome of strain CYS06, indicating that it has a broad range of applications in agriculture and industry.
The pathogenicity of the strain CYS06 was evaluated, suggesting that this strain is safe for grass carp. Additionally, the presence of risk associated genes, including antibiotic resistance genes, and biogenic amine and enterotoxin genes in the CYS06 genome, were evaluated. The results showed that the major biogenic amine encoding genes and enterotoxin genes were absent in the genome of strain CYS06. Furthermore, there was a low associated risk of resistance gene lateral transfer due to the absence of mobile elements within the gene vicinity. In the previous report, most
B. velezensis strains (96.7%) possessed potential tetracycline resistance gene (
tetL), but no strains harbored any acquired antimicrobial resistance genes, indicating that
B. velezensis possesses a low risk in terms of antibiotic resistance [
41]. These results showed that the strain CYS06 has good biosafety.
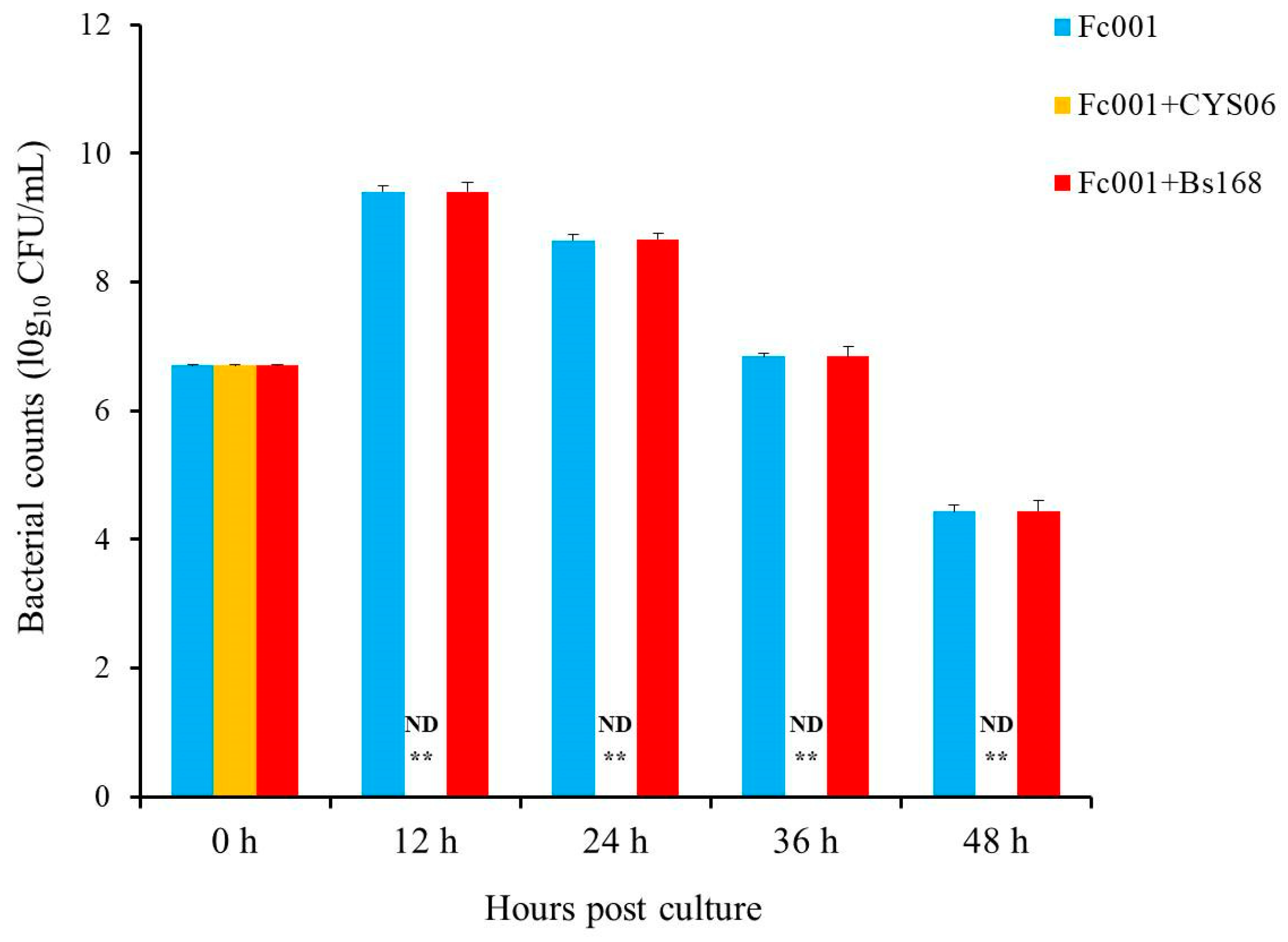
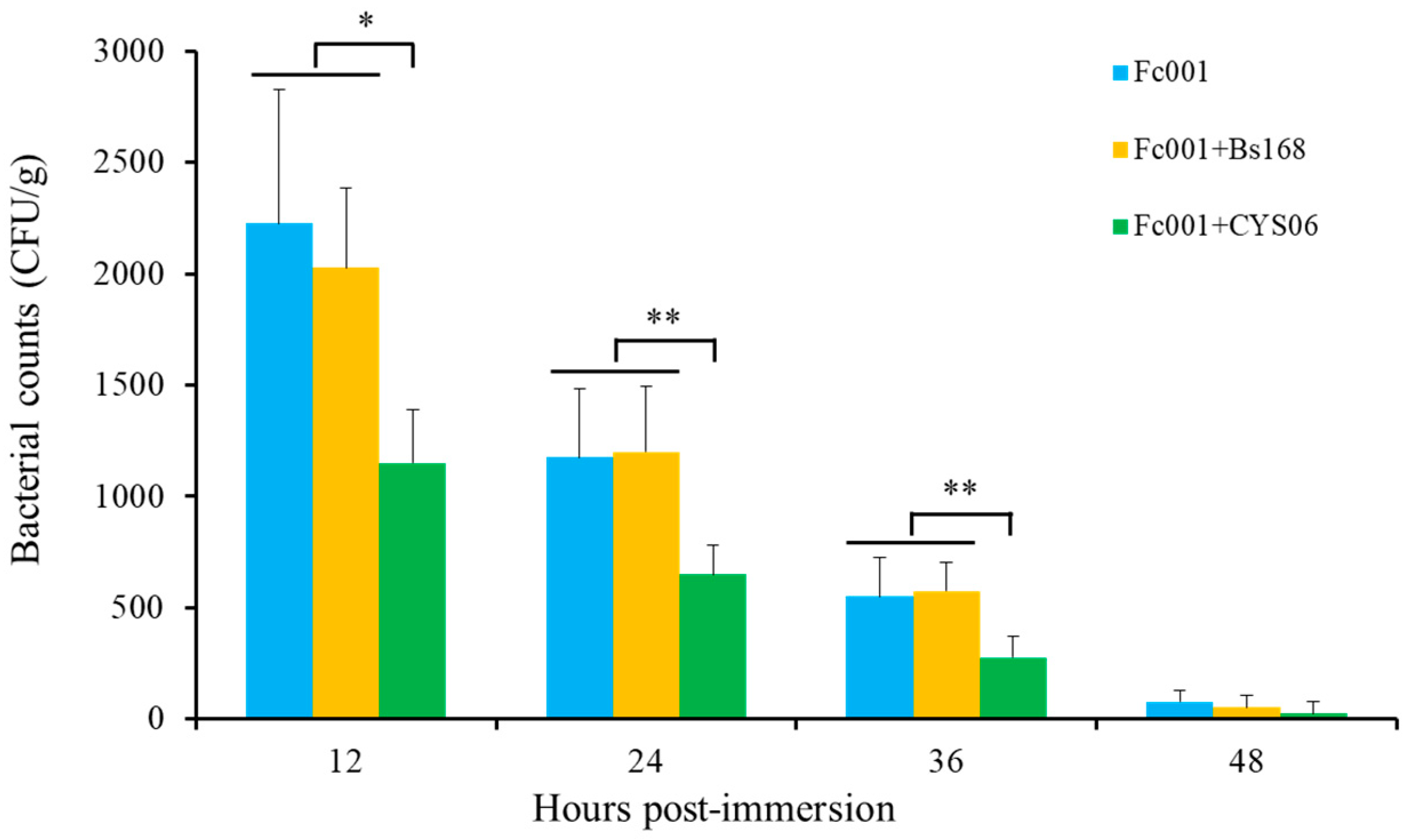
The commercial puffed diet supplemented with strain CYS06 significantly enhanced the ability of grass carp to fight A. hydrophila infection compared with that noted for the control diet, and the RPS was 63.63% at the concentration of 107 CFU/g, indicating that dietary supplementation with strain CYS06 enhances disease resistance. In addition, co-cultivation of the CYS06 and Fc001 strains in vitro resulted in the death of F. columnare within 12 h; moreover, the immersion experiment showed that strain CYS06 could inhibit the growth of F. columnare Fc001 on the gills of grass carp, indicating that strain CYS06 has the ability to prevent and control columnaris disease caused by F. columnare. Therefore, strain CYS06 can be used as a promising biocontrol agent for bacterial disease control in grass carp aquaculture.