1. Introduction
The exploration of plant-based ingredients as a protein source continues to garner significant attention within the aquafeed industry. Soybean residue is the byproduct obtained during the production of soy-based products like soy milk and soybean curd. Soybean residue is nutritionally valuable, containing abundant bioactive compounds such as saponins, phytosterols, and isoflavones [
1,
2]. However, unprocessed wet soybean residue is susceptible to microbial contamination [
3]. The lipoxidase in soybean residue can oxidize unsaturated fatty acids, resulting in the development of a beany odor [
4]. Furthermore, soybean residue contains antinutritional factors, which can impact digestion and absorption in animals [
1]. Consequently, a considerable portion of generated soybean residue is discarded as agricultural waste [
5].
Soybean residue fermentation involves processes such as grinding, steam sterilization, and microbial inoculation, followed by a designated fermentation period [
6]. The fermentation of soybean residue can effectively enhance its nutritional composition and palatability while reducing levels of anti-nutritional factors [
7,
8,
9]. The utilization of fermented soybean residue as a feed resource for aquatic animals is an economical and environmentally friendly choice. In largemouth bass (
Micropterus salmoides), substituting 40 g/kg of soybean meal with fermented soybean residue significantly increased the specific growth rate (SGR) of fish. As the level of fermented soybean residue increased, largemouth bass exhibited a significant decrease in their hepatosomatic index (HSI), viscera ratio, and liver lipid content [
10]. In African catfish (
Clarias gariepinus), similar results were observed, with the replacement of 50% of fish meal using fermented soybean residue showing the highest specific growth rate [
11]. Zulhisyam et al. [
12] reported that fermented soybean residue altered the probiotic profile in the intestine of African catfish.
Furong crucian carp is obtained through the hybridization of Furong carp (
Cyprinus carpio Furong) as the maternal parent and red crucian carp (
Carassius auratus red var.) as the paternal parent, and it is extensively cultured in China. Furong crucian carp offers the advantages of rapid growth, excellent flesh quality, and strong stress resistance [
13]. So far, there is no existing research on the utilization of fermented soybean residue in Furong crucian carp. In the present study, various proportions of fermented soybean residue were employed as substitutes for rapeseed meal and rice bran in their feed. Despite the nutritional similarity between rapeseed meal and soybean meal, the practical application of rapeseed meal is constrained due to its elevated levels of harmful substances and numerous anti-nutritional factors such as glucosinolates, sinapine and its derivatives, tannin, etc. [
14]. Moreover, the price of rice bran has also been consistently rising. Therefore, the substitution of rapeseed meal and rice bran with fermented soybean residue proves advantageous in reducing aquaculture costs and enhancing economic benefits.
2. Materials and Methods
2.1. Ethical Statement
This study obtained approval from the Ethics Committee of Hunan Agricultural University, Changsha, China. The study’s procedures were meticulously conducted in strict adherence to applicable laws and guidelines.
2.2. Experimental Design
The fermented soybean residue was supplied by Hunan Junhui International Agriculture and Animal Husbandry Co., Ltd. (Changsha, China). The fermented soybean residue was generated through a 72-h fermentation process with lactic acid bacteria. The nutrient contents measured after drying were 20.31% crude protein, 11.12% crude fat, 21.14% crude fiber, and 4.5% ash. The feeds were formulated to contain 0%, 6%, 12%, 18%, and 24% fermented soybean residue (CON, FSR6, FSR12, FSR18, and FSR24, respectively), respectively, to form five isonitrogenous (crude protein: 33%) and isolipidic (crude lipid: 5%) diets. Following complete grinding, the raw materials were sieved through a screen with a 0.425 mm aperture, weighed, blended, pelletized, dried, and then stored at 4 °C until required. The formula and composition of the diets are displayed in
Table 1.
2.3. Fish and Experimental Conditions
The experiment was carried out at the aquaculture facility located at Hunan Agricultural University. The Furong crucian carp used for the experiment were obtained from the Xiangyin Fisheries Science Institute, Yueyang, Hunan, China. A total of 750 healthy fish (25.00 ± 2.08 g) were randomly allocated into twenty-five cages (1.5 m3) suspended in a cement pool. The experiment comprised five treatments, each with five replicates (cages) per treatment. All fish were fed CON for 14 days, following which each group was fed diets of CON, FSR6, FSR12, FSR18, and FSR24, respectively. The feeding trial lasted for 56 days. The fish were fed to apparent satiation twice (8:30 and 16:30) daily. Throughout this duration, the water temperature was 27 ± 5 °C. The pH, dissolved oxygen, nitrite, and NH4-N were 7.7 ± 0.2, 7.5 ± 1.5 mg/L, <0.005 mg/L, and <0.2 mg/L, respectively.
2.4. Collecting Samples
At the trial’s conclusion, a 24-h fasting period was observed for the fish in each cage before individual counting and weighing. Nine fish per cage were randomly picked and anesthetized using eugenol (Macklin Biochemical Technology Co., Ltd., Shanghai, China). Following the measurement of their body length and weight, four fish per cage (20 fish per treatment) were chosen for a whole-fish proximate analysis. For the remaining five fish per cage (25 fish per treatment), after collecting blood samples, the serum was separated. Following dissection, the viscera and liver were isolated and weighed. The hindgut contents of each fish were collected to identify their intestinal microbiota composition. The foregut was collected to identify intestinal digestive enzymes, and the midgut was prepared for intestinal slices.
2.5. Analyzing the Composition of Diets and Fish
The crude protein, crude lipid, and moisture of the experimental diets and whole fish were analyzed using standard methods [
15].
2.6. Serum Biochemical and Antioxidant Parameters
The activities of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), and the levels of albumin (ALB), total protein (TP), globulin (GLB), total cholesterol (TC), glucose (GLU), and triglyceride (TG) in serum were measured using a BS450 automatic biochemical analyzer (Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China).
The total antioxidant capacity (T-AOC) (A015-1-2), the total superoxide dismutase activity (T-SOD) (A001-1-1), and the content of malondialdehyde (MDA) (A003-1-1) in serum were analyzed using detection kits purchased from Nanjing Jiancheng (Nanjing, China).
2.7. Intestinal Digestive Enzymes
The activities of amylase (C016-1-1), trypsin (A080-2-2), and lipase (A054-1-1) were measured using detection kits obtained from Nanjing Jiancheng. For the determination of amylase activity, iodine–starch colorimetry was utilized [
16]. Tissue samples were accurately weighed and mixed with nine times their volume of saline solution. The mixture was homogenized in an ice bath, followed by centrifugation at 2500 rpm for 10 min to collect the supernatant. In a test tube, 100 μL of the supernatant was combined with 500 μL of 0.4 mg/mL of soluble starch in a 20 mM phosphate buffer. After incubation at 37 °C for 7.5 min, the reaction was halted by adding 500 μL of a 0.01 M iodine solution, followed by the addition of 3000 μL of distilled water. In blank samples, 100 μL of distilled water was added after the iodine solution. The absorbance of the reaction mixture was measured at 660 nm. One unit of amylase activity was defined as the amount of amylase in 100 mL of supernatant that hydrolyses 10 mg of the substrate (starch) in 30 min at 37 °C.
For the determination of trypsin activity, the method described by Gong et al. [
17] was used. The preparation method of the sample supernatant was the same as that of amylase. The reaction was initiated by mixing a 3 mL substrate solution (67 mM sodium phosphate buffer with 0.25 mM N-α-Benzoyl-L-Arginine ethyl ester) and 0.2 mL of supernatant in quartz cuvettes (1 cm light path). The increase in the A253 nm value was recorded for 5 min. One unit (U) of enzyme activity was defined as a ΔA253 nm of 0.001 per min at a pH of 7.6 and 25 °C.
Lipase activity was evaluated using a modified method described by Lessinger et al. [
18]. Tissue samples were accurately weighed and mixed with four times their volume of saline solution. The mixture was homogenized in an ice bath and then centrifuged at 2500 rpm for 10 min to collect the supernatant. An oil emulsion was prepared containing 35 mM of Na-deoxycholate, 8 mL of olive oil, 100 mM of CaCl
2, and ultrapure water to a final volume of 250 mL. This emulsion served as the substrate for lipase hydrolysis, leading to a reduction in turbidity. The supernatant was thoroughly mixed with the oil emulsion. Absorbance measurements were taken before and after incubating the mixture in a 37 °C water bath for 10 min. Lipase activity was expressed as units per gram of protein (U/g protein), with one unit defined as the consumption of 1 μmol of substrate per minute at 37 °C.
2.8. Intestinal Histomorphology
The intestinal tissues underwent dehydration using a gradient of ethanol concentrations that increased progressively, followed by equilibration in xylene and embedding in paraffin (PPDT-12C1, Ceike, Shanghai, China). Sections were cut to 4 μm using a paraffin microtome (RM2235, Leica Camera AG, Stuttgart, Germany), stained with hematoxylin and eosin, mounted with neutral resin, and examined under a light microscope (E600, Nikon Corporation, Tokyo, Japan). Villous height (Vh), muscularis thickness (Mt), and goblet cell (GC) numbers were recorded using image analysis software (version 1.7) (Olympus CellSens Standard, Tokyo, Japan).
2.9. The Intestinal Microbial Composition
DNA was extracted from the hindgut contents, and its purity and quality were subsequently assessed. PCR reactions were conducted to amplify the V3–V4 hypervariable region of the bacterial 16S rRNA gene. The PCR products were submitted to Beijing Ovisen Genetech Co., Ltd. (Beijing, China), for paired-end sequencing using the Illumina-MiSeq PE300 high-throughput sequencing platform. Sequences were clustered into Operational Taxonomic Units (OTUs) with a 97% sequence similarity threshold. Venn diagrams were generated based on the differential OTUs in each group. The Mothur software (Version 1.31.2) was employed for an alpha diversity analysis. Statistical comparisons of the taxa abundance at the phylum and genus levels among groups were conducted using the Metastats Program.
2.10. Statistical Analysis
Data were expressed as means ± standard errors. Statistical analysis was conducted using the SPSS 22.0 software. Statistical comparisons between diets were carried out using one-way ANOVA and Tukey’s multiple range tests. Significance was considered at p < 0.05.
4. Discussion
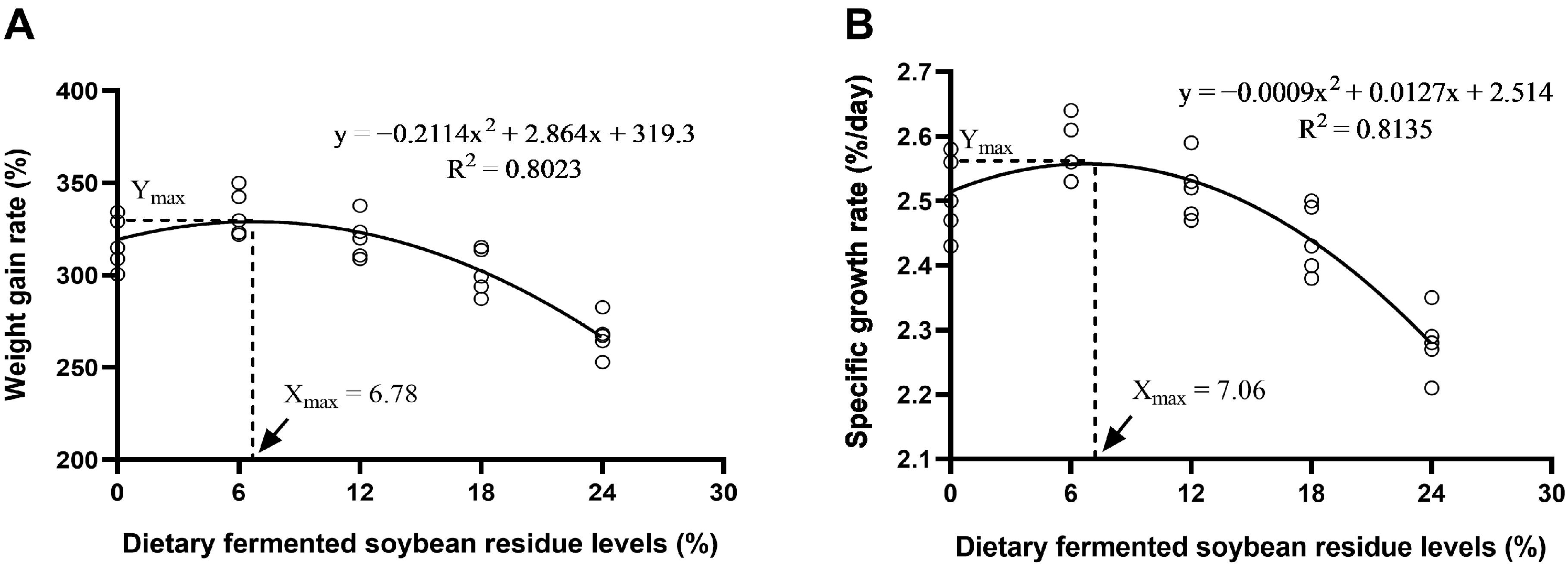
After fermentation, soybean residue exhibited an increase in crude protein content accompanied by an effective reduction in anti-nutritional factors [
7]. Additionally, soluble dietary fiber, polyphenol isoflavones, and other active substances increased [
6], potentially promoting the growth of the animals. Accordingly, the findings from this study indicated that incorporating 6.78% to 7.06% fermented soybean residue into the diet significantly enhanced the WGR and SGR of Furong crucian carp. A study reported that, through a regression analysis of SGR, it was determined that the optimal inclusion level of fermented soybean residue in the diet of
Jian carp (
Cyprinus carpio var. Jian) is 10.2% [
19]. In an investigation of largemouth bass, the optimal growth performance was observed when fermented soybean residue, at a level of 9%, replaced 4% of soybean meal in the diet [
10]. In this study, the inclusion of fermented soybean residue at 12% resulted in an increase in the FCR for Furong crucian carp, accompanied by marked decreases in WGR and SGR. Similar to the findings of this study, the earlier study found that a high level of fermented soybean residue in the diet is detrimental to the growth of African catfish [
20]. In this study, in conjunction with the analysis of microbial communities, it was observed that, when the fermentation of soybean residue reached 12%, there was a significant increase in the abundance of
Vibrio, while beneficial bacteria such as
Romboutsia and
Clostridium_sensu_stricto_1 significantly decreased. Previous studies have reported that certain species of
Vibrio act as opportunistic pathogens in aquaculture [
21]. Therefore, the observed decline in the growth of Furong crucian carp when soybean residue substitution levels exceeded 12% may be associated with a decrease in the fish’s resistance to pathogens.
Certain components in soybeans, including protein, isoflavones, and dietary fiber, have been documented to impact cholesterol metabolism [
22,
23]. The results of the present study indicated that incorporating fermented soybean residue into the diet notably reduced the lipid content in Furong crucian carp. Additionally, with the increasing level of fermented soybean residue, the serum GLU, TC, and TG levels of Furong crucian carp significantly decreased. This suggested that the fermented soybean residue in the diet promoted the carbohydrate and lipid metabolism in Furong crucian carp. In largemouth bass, as the level of fermented soybean residue replacing soybean meal increased, there was a decrease in liver fat content, as well as a reduction in plasma GLU, TG, and TC levels [
10]. In Syrian hamsters, Villanueva et al. [
24] noted a substantial decrease in the TC and TG concentrations in plasma and the liver by adding fermented soybean residue to the high-fat diet. It was suggested that the dietary fiber and protein, being the primary constituents of fermented soybean residue, might have influenced cytochrome P450 14α-sterol demethylases and peroxisome proliferator-activated receptor alpha in the liver, thereby regulating lipid metabolism. In this study, the precise mechanism through which fermented soybean residue promoted carbohydrate and lipid metabolism remained to be further investigated.
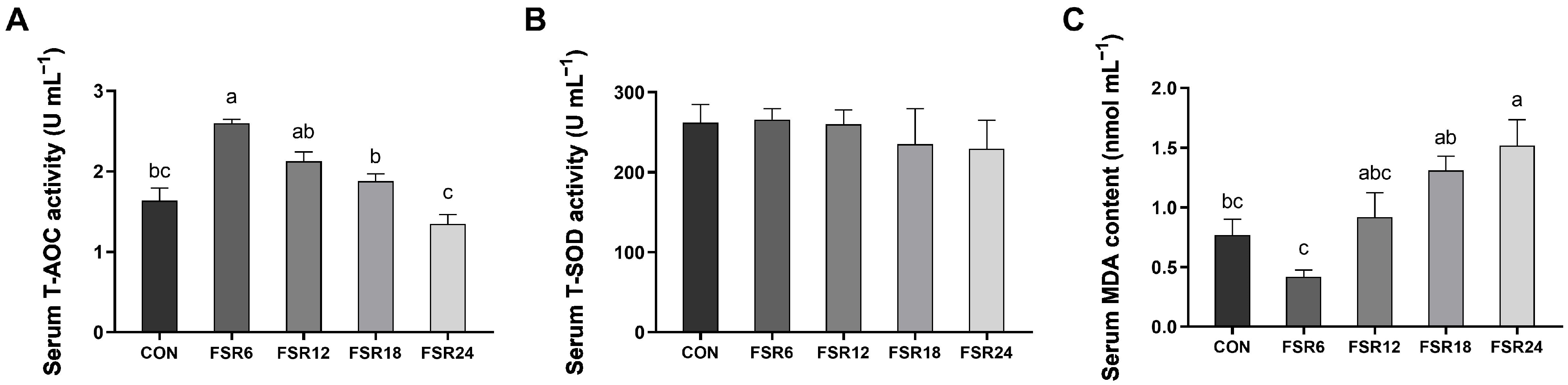
A study employing the yeast
Yarrowia lipolytica to ferment soybean residue demonstrated substantial increases in lipid, succinate, and free amino acid levels, consequently enhancing the antioxidant capacity [
9]. Furthermore, the increase in phenolic compounds and isoflavone content in fermented soybean residue also contributed to its antioxidant properties [
25]. MDA serves as an indicator reflecting the extent of damage induced via oxygen free radicals, SOD neutralizes superoxide radicals, and T-AOC reflects the overall antioxidant capacity of a biological system. In the present study, incorporating 6% fermented soybean residue into the diet resulted in the highest serum SOD and T-AOC and the lowest MDA content. This indicated that 6% fermented soybean residue enhanced the antioxidant capacity of Furong crucian carp. Similarly, enhanced antioxidant capacities were also observed in black sea bream (
Acanthopagrus schlegeli) and white shrimp (
Litopenaeus vannamei) fed diets containing fermented soybean meal [
26,
27].
The activities of digestive enzymes in the intestine reflect the ability of fish to digest and absorb nutrients. In the present study, 6% fermented soybean residue significantly improved the amylase activity of the intestine of Furong crucian carp, which might partially explain the improved growth performance using 6% fermented soybean residue. In African catfish, the substitution of 50% fermented soybean residue for fish meal significantly upregulated the activity of intestinal amylase [
20]. Soybean-derived peptides are small protein fragments that could be generated via lactic acid–bacteria-mediated fermentation [
28,
29]. The variations in soybean-derived peptides might be the reason why incorporating fermented soybean residue into the diet could influence the intestinal digestive activities in Furong crucian carp. Jiang et al. [
30] found that supplementing the diet with bioactive peptides derived from soybeans enhanced the alpha-amylase and trypsin activities in the intestine of Chinese mitten crab (
Eriocheir sinensis).
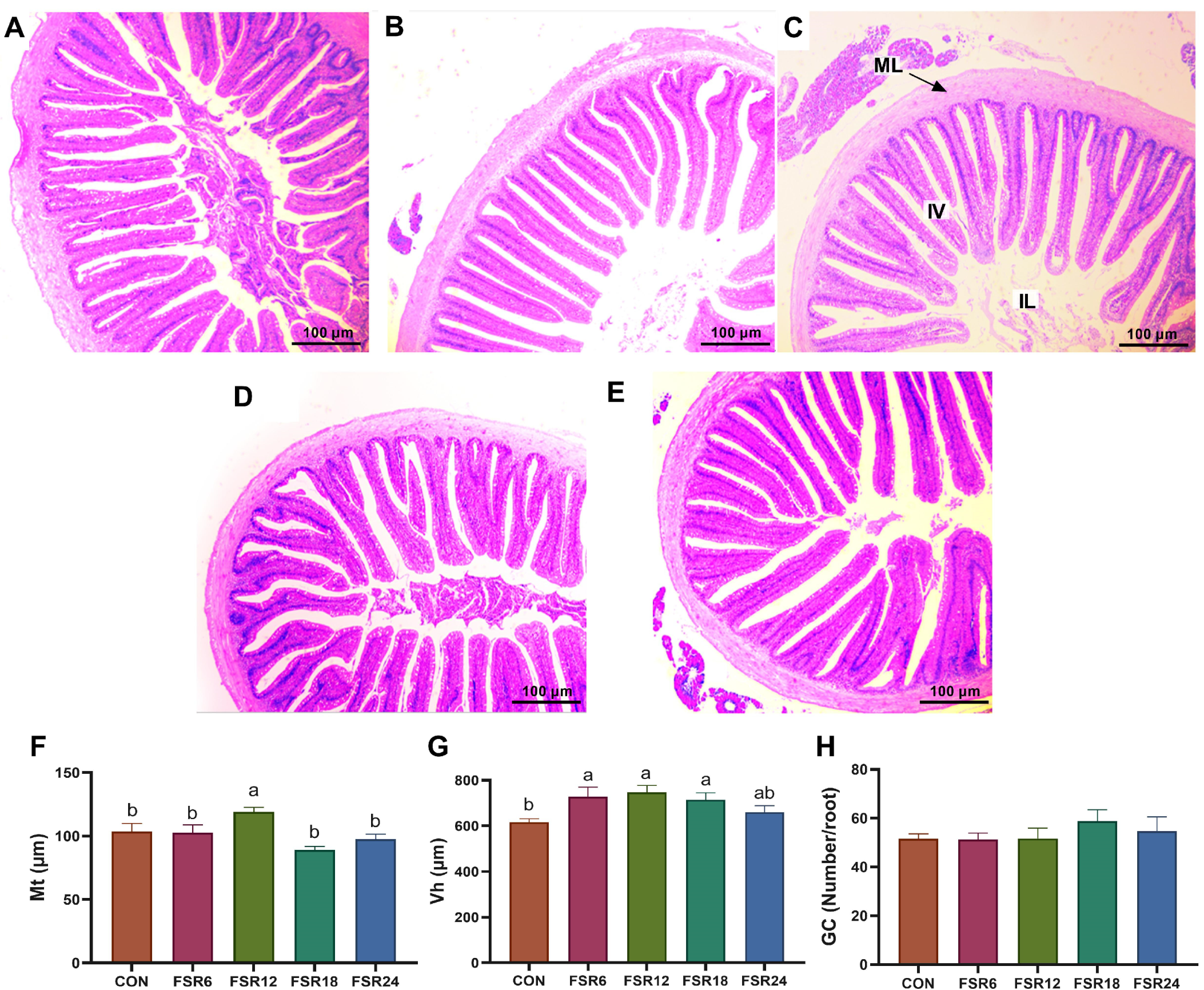
Mt, Vh, and GC are important indicators for evaluating intestinal structural characteristics. In the present study, incorporating 6% fermented soybean residue into the diet significantly increased the Vh in the intestine of Furong crucian carp. When the level of fermented soybean residue in the diet reached 12%, the Mt in the intestine of Furong crucian carp showed a significant increase. In the study on African catfish, it was demonstrated that the substitution of 25% fishmeal with fermented soybean residue (at a level of 9% in the diet) markedly increased the Vh in the intestine [
11]. In the investigation carried out by Refstie et al. [
31], it was observed that the pathological changes in the intestine induced via soybean meal were less prominent in fish that were fed fermented soybean white flakes, as opposed to those fed non-fermented soybean white flakes. Research studies have indicated that diverse antinutritional components present in soybeans, such as saponins, play a role in the development of morphological alterations in the intestines of salmonids [
32,
33]. On one hand, fermentation possesses the capacity to decrease or eliminate anti-nutritional components in soybean residue. On the other hand, some literature, including studies by Feng et al. [
34] and Tellez et al. [
35], has reported a direct correlation between Vh and intestinal microbial populations.
It was noted that the predominant phyla in the hindgut of different treatments for Furong crucian carp were consistent, comprising Fusobacteriota, Proteobacteria, Cyanobacteria, and Firmicutes, respectively. This pattern resembled the dominant microbial composition observed in the intestinal tract of tilapia (
Oreochromis niloticus) [
36]. In this study, the dominant bacterial genera in the hindgut of Furong crucian carp included the
Cetobacterium,
Aeromonas, and ZOR0006. Among them,
Cetobacterium is overwhelmingly dominant, with abundances exceeding 50% in all treatment groups.
Cetobacterium is a core microbial group in the intestinal tract of herbivorous fish. This genus not only facilitates cellulose hydrolysis but also engages in the fermentation of proteins and carbohydrates, producing vitamin B
12 [
37]. Abundant
Cetobacterium demonstrated the ability to use dietary fiber in the diet of Furong crucian carp. In the present study, 6% fermented soybean residue significantly increased the relative abundances of
Romboutsia and
Clostridium_sensu_stricto_1 in the intestine of Furong crucian carp.
Romboutsia represents a novel class of probiotics that could potentially enhance the host’s intestinal health. A significant reduction in its presence may serve as an indicator of dysbiosis within the host’s intestinal microbiota [
38]. In addition,
Romboutsia possesses the capability to metabolize diverse carbohydrates into beneficial compounds such as short-chain fatty acids, oligosaccharides, and other prebiotics [
39]. The changes in the abundance of
Romboutsia noted in this study could be linked to the content of soybean saponin in fermented soybean residue [
40]. Microbial species belonging to Clostridiales were tightly correlated with the generation of short-chain fatty acids and demonstrated distinct anti-inflammatory properties [
41]. A study also reported that the inclusion of microbial fermented and enzymatically hydrolyzed soybean meal in the diet increased the abundance of
Clostridium_sensu_stricto_1 in the intestine of weaned piglets [
42]. The predominant representation of
Vibrio within the Proteobacteria phylum suggested its potential role as an intestinal pathogen, contributing to the occurrence of vibriosis outbreaks [
43]. In the present study, incorporating 6% fermented soybean residue into the diet could reduce the abundance of
Vibrio in the intestines of Furong crucian carp, increase the abundance of beneficial bacteria such as
Romboutsia and
Clostridium_sensu_stricto_1, promote the metabolism of carbohydrates and fatty acids, and contribute to intestinal health.