Hydrogen production through water splitting is a popular technology. The reaction takes place in an electrolyzer unit. There are different types of electrolyzers available, including proton exchange membranes (PEMs), anion exchange membranes (AEMs), and alkaline and solid oxide electrolyzers. Although PEMs are a widely used commercial electrolyzer, they are very sensitive, not very durable, and have a high cost due to the membrane. To eliminate the cost of the membrane, the concept of membrane-less electrolysis technology was developed.
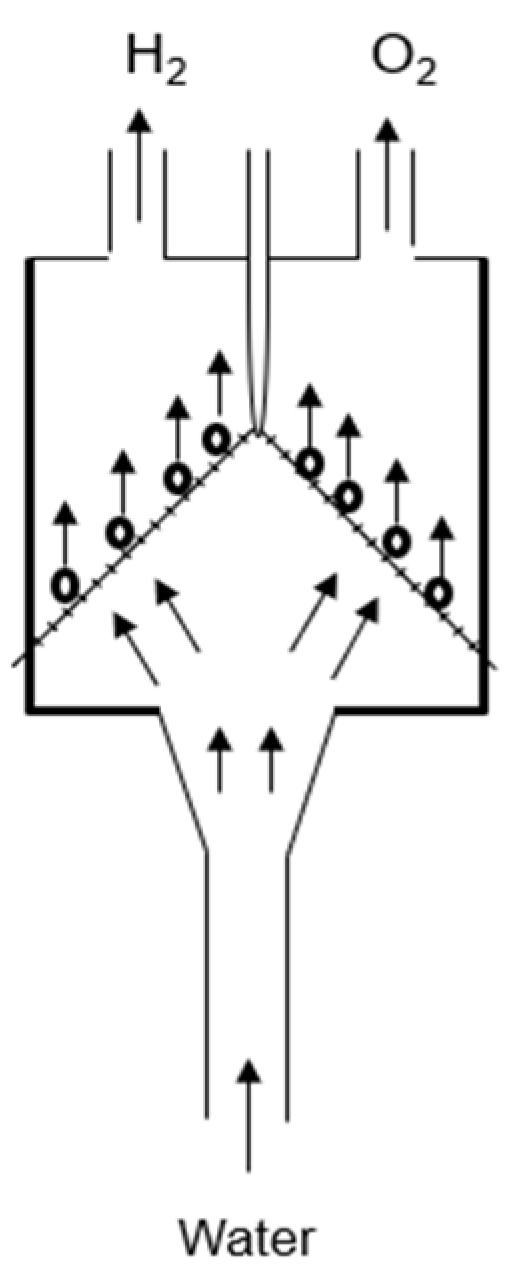
In this study, a new membrane-less electrolyzer design was proposed with a flow-through electrolyzer, as shown in Figure 1.
Figure 1.
Concept of water flow electrolyzer with gas separation mechanism.
The design was manufactured with the help of 3D printing technology with acrylonitrile butadiene styrene (ABS) polymer material that is more stable in basic media. To increase the conductivity of water, a 4 M KOH solution was provided for electrolysis. In this design, electrodes were placed at an angle of 30° to each other. The generated hydrogen and oxygen gases were separated from each other using the effect of the flow of water between the electrodes. The tapered inlet was provided to maintain a proper laminar flow through the inlet. In the middle of the electrolyzer, a divider was also provided for separating the generated hydrogen and oxygen gas. A Ni mesh with an 80-grit size was used in the place of electrodes. The purity of the generated gas was measured via a gas chromatography instrument. A H2 purity of about 99.85% was achieved by using a Ni mesh as an electrode at room temperature and normal pressure.
Author Contributions
N.U.K.: Experimental analysis, data acquisition, writing—original draft, approval of the version of the manuscript to be published. M.K.: Conception of investigation, funding acquisition, project administration, revising the manuscript critically for important intellectual content, Conception of investigation, formal analysis, revising original draft. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by NTPC India: RGIPT grant number 9100000216-151-1001.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflict of interest.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).