Abstract
We developed a new Bach-type reaction in the presence of oxy-hemoglobin as an oxygen supplier to synthesize polyazobenzene by traditional Bach reaction. The resultant product is a form of polymeric dye/hemoglobin copolymer. The advantage of this research is that it involves a new reaction using the function of biomolecules, as well as the formation of plastics and biomaterials. The bio-based material may have good affinity with life forms, which may lead to applications in medical science.
1. Introduction
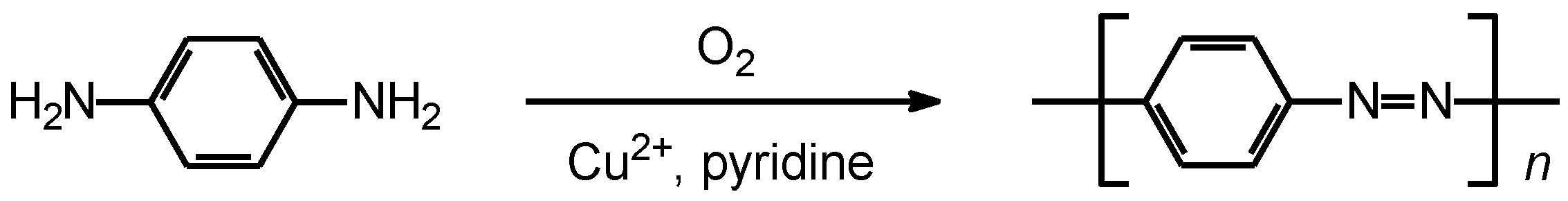
In 1966, Bach synthesized main chain-type polyazobenzene by Cu2+−catalyzed oxidative coupling (Scheme 1) [1]. This reaction requires oxygen for the polycondensation.
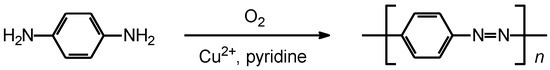
Scheme 1.
Synthesis of polyazobenzene.
Polyazobenzene is the simplest polymeric dye [2,3,4,5,6,7,8,9,10,11,12,13]. The range of applications can be expanded by processing azo-polymer dyes into films and tapes.
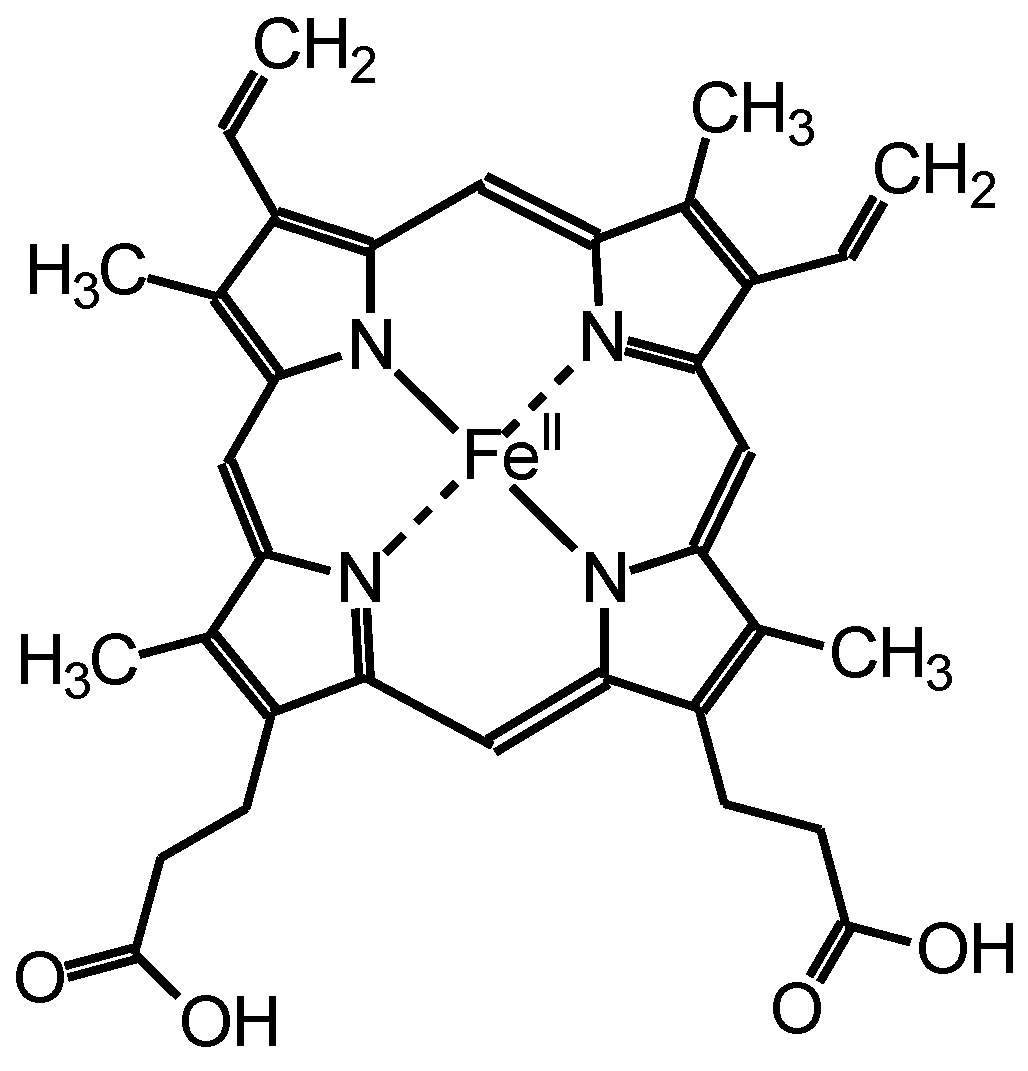
Hemoglobin (Hb) is a protein found in the red blood corpuscle that bonds to oxygen molecules and transports them throughout the body. The chemical structure of hem as a center of Hb is displayed in Figure 1. Hb has high oxidation activity, stability, redox properties, and inexpensiveness [14,15,16]. We performed the Bach oxidative coupling reaction using Hb as oxygen supplier in place of O2 bubbling during the reaction. The resultant material is a composite form of polyazobenzene and Hb, abbreviated as PAZ/Hb.
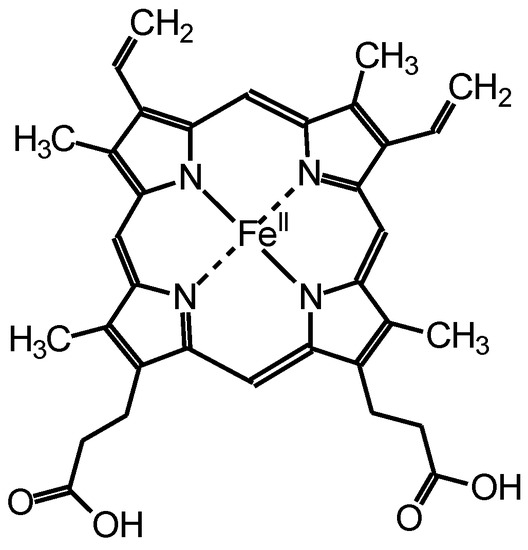
Figure 1.
Chemical structure of hem.
2. Materials and Methods
2.1. Materials
Hemoglobin was purchased from Nacalai Tesque (Kyoto, Japan). p-Phenylendiamine was obtained from Tokyo Chemical Industry (TCI, Tokyo, Japan). CuI was purchased from Sigma Aldrich (Saint Louis, MO, USA).
2.1.1. Synthesis of Polyazobenzene
Polyazobenzene as a standard sample was prepared by the Bach method under oxygen gas atmosphere. A solution of p-phenylenediamine (1.05 g, 9.73 mmol), and CuI (0.21 g, 1.1 mmol) in pyridine (20 mL) was stirred for 24 h at 25 °C. A large volume of methanol was poured into the solution to wash the polymer. After centrifugation, the resultant was dried to yield 0.451 g of the product as a black powder.
2.1.2. Synthesis of Polyazobenzene/Hemoglobin
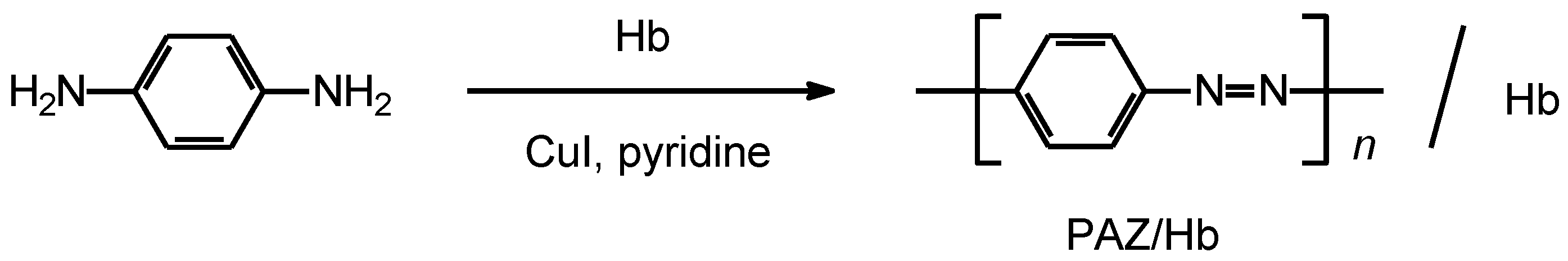
The synthetic route for the preparation of polymer is shown in Scheme 2. p-Phenylenediamine (1.06 g, 9.83 mmol), CuI (0.218 g, 1.15 mmol), hemoglobin (1.06 g), and pyridine (20 mL) were added to a 200-mL Erlenmeyer flask and stirred for 24 h at 25 °C. A large volume of methanol was poured into the solution to wash the polymer. After filtration, the product was dried under vacuum to yield 1.74 g as a black powder, abbreviated as PAZ/Hb.
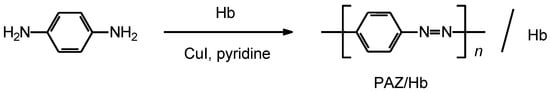
Scheme 2.
Synthesis of polyazobenzene (PAZ)/hemoglobin (Hb) composite (PAZ/Hb).
3. Results and Discussion
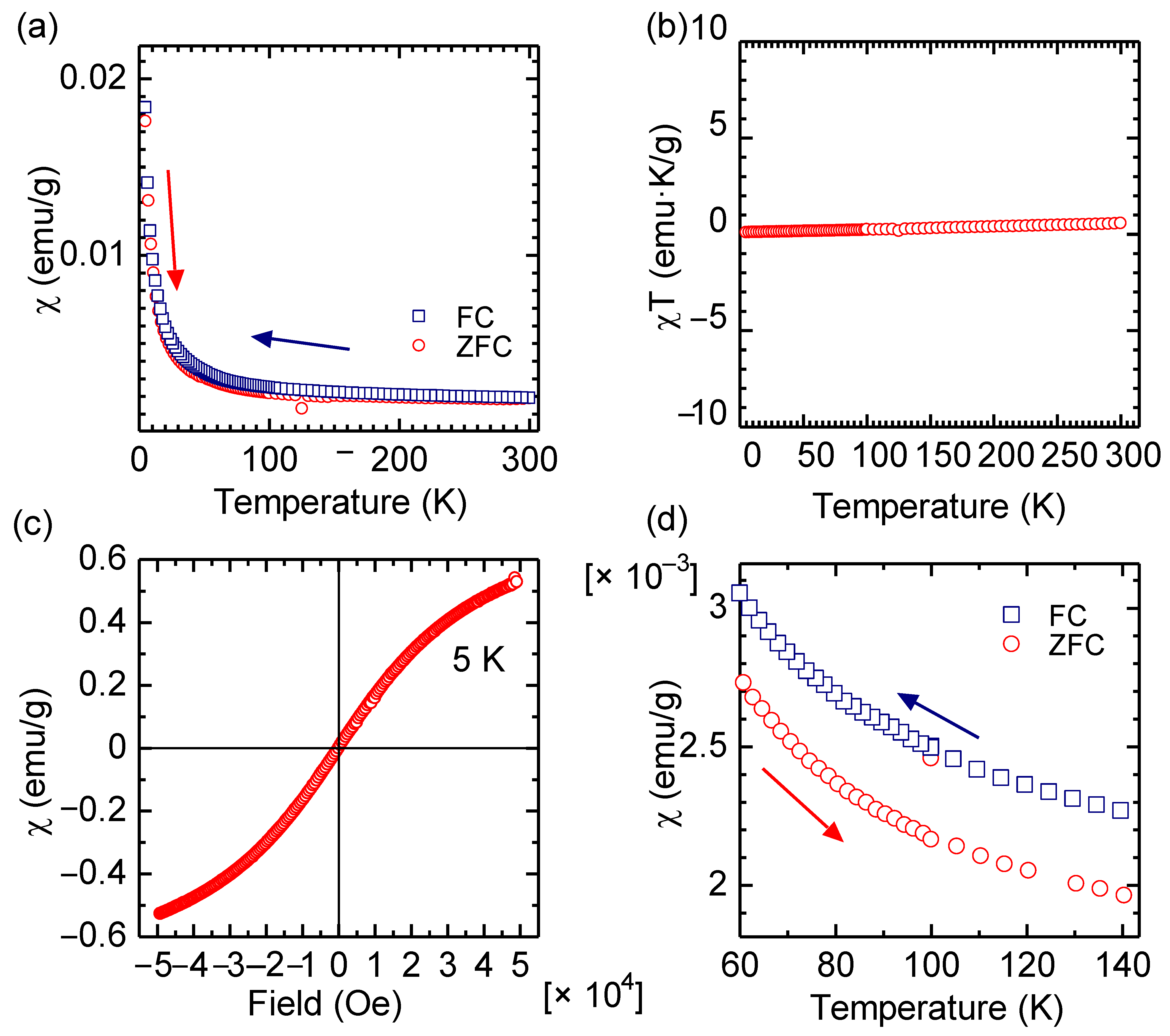
First, from a materials science research-based viewpoint, the magnetic measurement of Hb was performed. Figure 2a shows the results of superconductor quantum interference device (SQUID) measurements for a pure Hb. Figure 2b as a χT vs. T plot (Currie plot) indicated that the Hb is a paramagnetic material. However, the Hb exhibited a tendency toward soft ferromagnetic-like saturation magnetization, with no hysteresis, and no remanent magnetization at 5 K, as shown in Figure 2c (χ vs. magnetic field, M-H curve). This result indicates that the Hb may have a tendency toward super-paramagnetism at low temperature range, although a blocking temperature of the sample was not observed. Iron at the center of the hem unit is magnetically isolated from other Hb units at the molecular level, showing paramagnetic behavior as a form of isolated ferromagnet. The Hb shows a higher χ value in field cooling (FC) than zero field cooling (ZFC), as shown in Figure 2d.
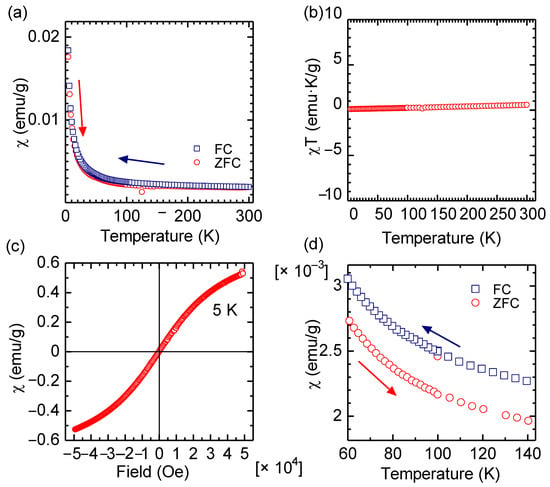
Figure 2.
Magnetic measurements of Hb with a superconductor quantum interference device (SQUID). (a) χ vs. T curve. (b) χT vs. T. (c) χ vs. magnetic field at 5 K. (d) Magnification of χ vs. T curve (a). χ: Magnetic susceptibility.
3.1. Infrared Absorption Spectroscopy (IR)
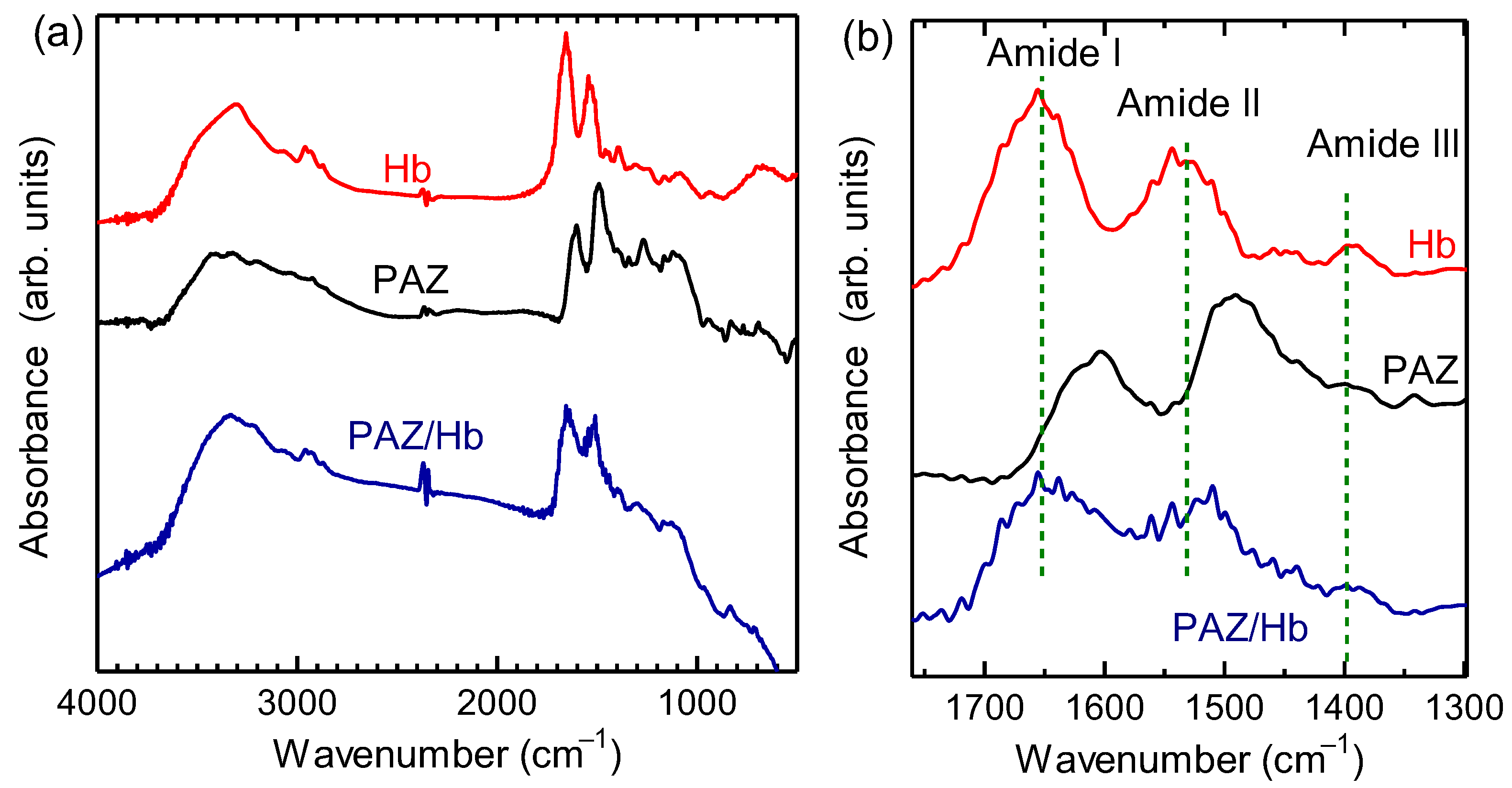
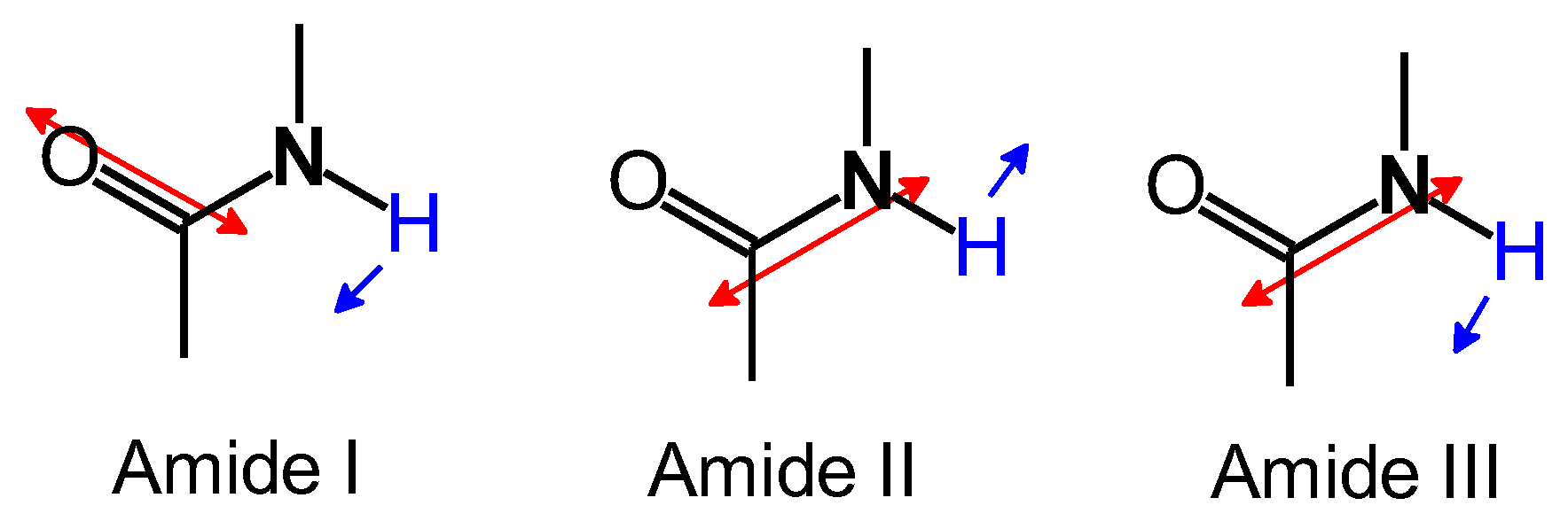
The results of Fourier transform infrared (FT-IR) absorption spectroscopy measurements for the obtained material and hemoglobin are shown in Figure 3. From the polymer, the stretching vibration of C-H in the aromatic ring (3335 cm–1), the stretching vibration of C=C in the aromatic ring (1638 cm–1), the stretching vibration of N=N (1560 cm–1), the in-plane bending vibration of C-H in the aromatic ring (1170 cm–1), and the out-of-plane bending vibration of C-H in the aromatic ring (832 cm–1) were observed. Three absorption bands at 1300–1700 cm–1 (Amide I, Amide II, and Amide III, Figure 4) were indicative of the amide bonds derived from hemoglobin. The polymer signal became broad because vibrations of N=N and amide bonds were overlapped. FT-IR spectroscopy measurements for Hb, PAZ, and PAZ/Hb confirmed the chemical structures.
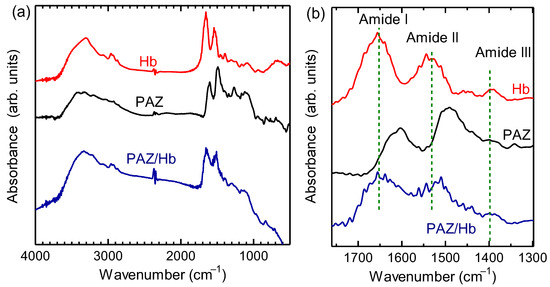
Figure 3.
(a) Fourier-transform infrared (FT–IR) absorption spectra of hemoglobin (Hb), polyazobenzene (PAZ), and polyazobenzene/Hb (PAZ/Hb). (b) Magnifications of the IR absorption spectra.
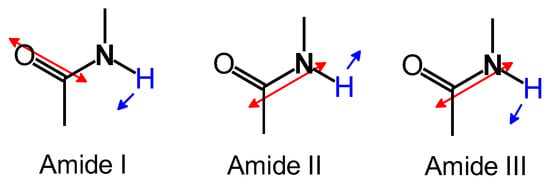
Figure 4.
Vibrational modes of Amide I, Amide II, and Amide III.
3.2. Electrical Conductivity
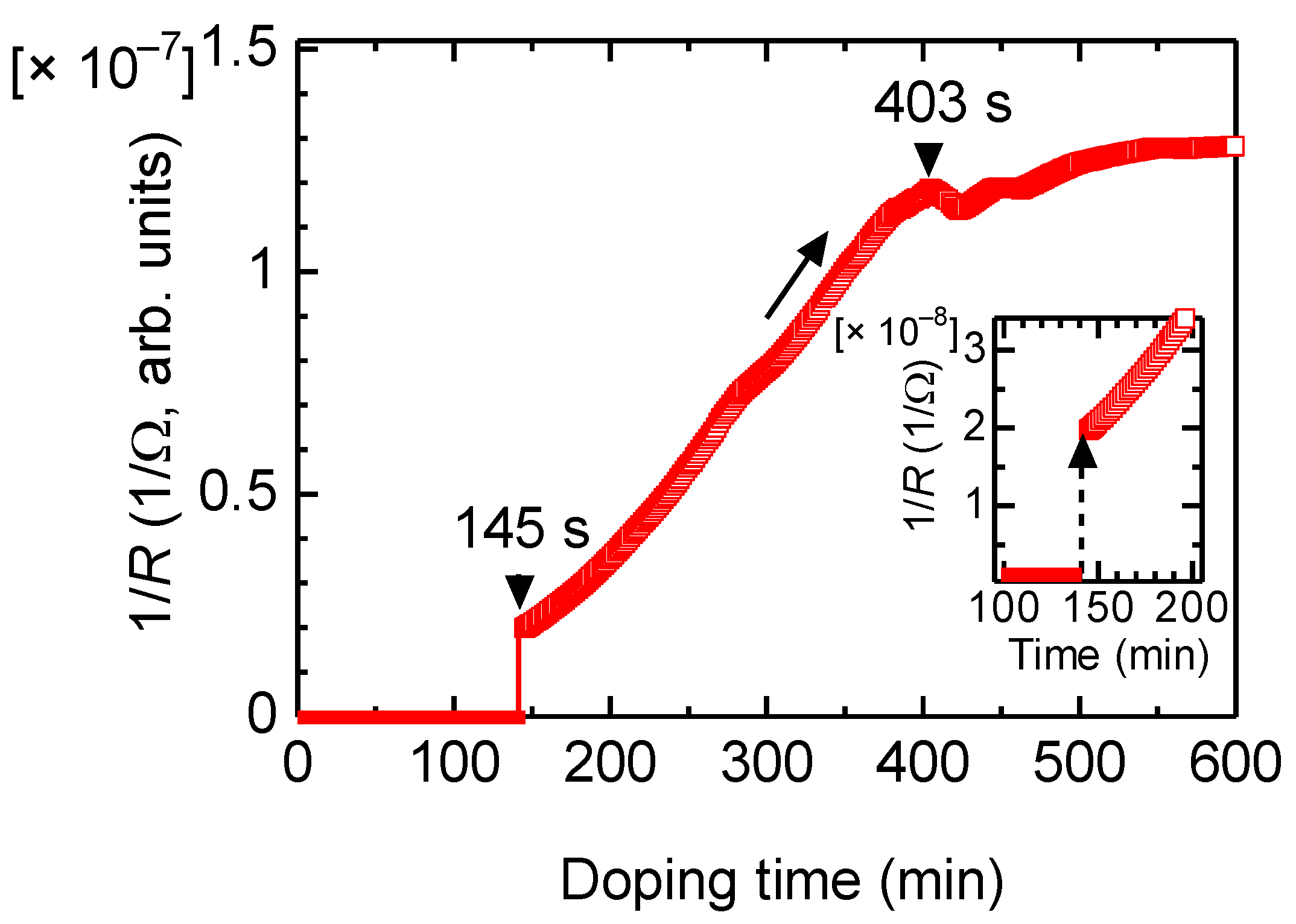
Change in electrical conductivity was examined upon vapor-phase doping of iodine. Electrical conduction as 1/R was observed with the two-probe method. The reciprocal value of resistance is proportional to the electrical conductivity. Figure 5 shows the change in electric conductivity as a function of the doping time.
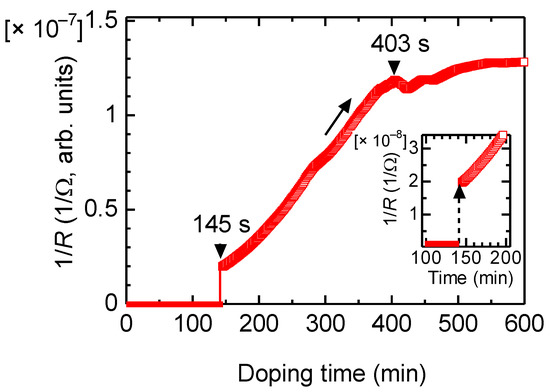
Figure 5.
Change in electrical conductivity of PAZ/Hb vs. vapor phase iodine doping.
The electrical conduction of PAZ/Hb was increased after 145 s upon vapor-phase iodine doping via a small maximum at 403 s. Although the increase in the conductivity was slow, the iodine doping allows the polymer to increase conductivity as a typical characteristic of conductive polymers.
3.3. Surface Temperature
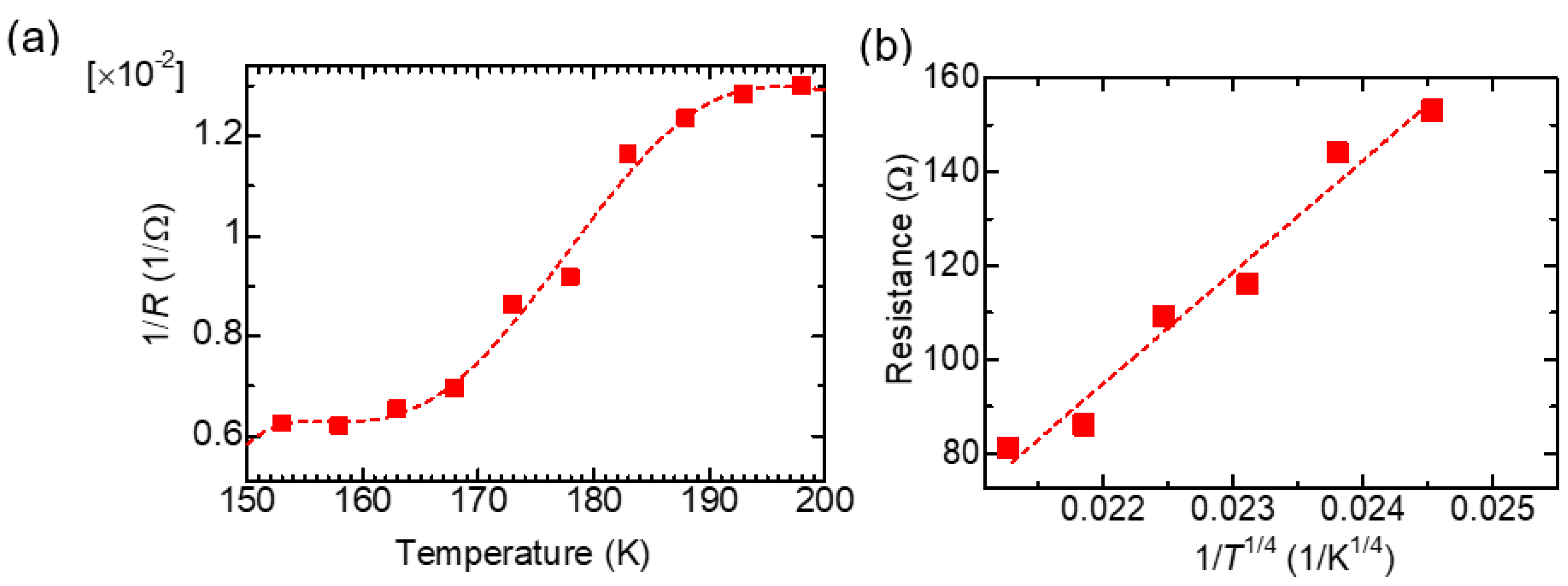
Figure 6a shows 1/R (∝ σ) as a function of the temperature of PAZ/Hb, demonstrating that an increase in the surface temperature increases conductivity due to the behavior of a semiconductor. Figure 6b depicts 1/T1/4 as a Mott plot [17]. The linear increment of resistance with 1/T1/4 value indicates 3-D variable range hopping containing inter-main chain electron hopping and inter-domain electron hopping [18].
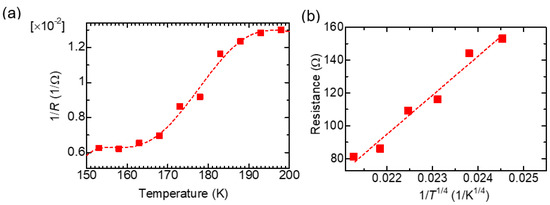
Figure 6.
1/R (∝ σ) as a function of the temperature of PAZ/Hb (a). Resistance vs. 1/T1/4 as a Mott plot (b) for PAZ/Hb.
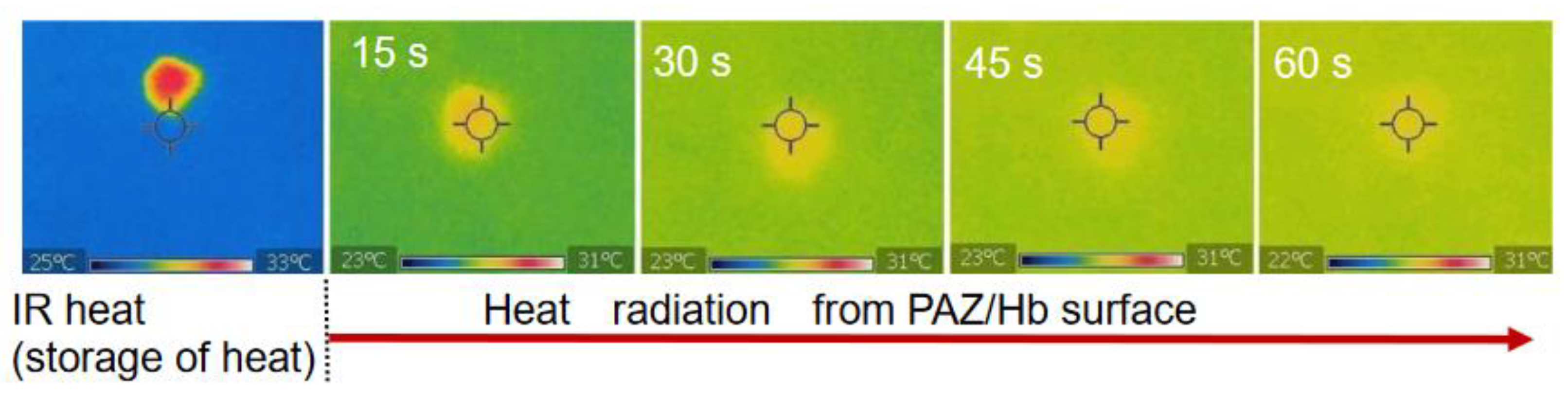
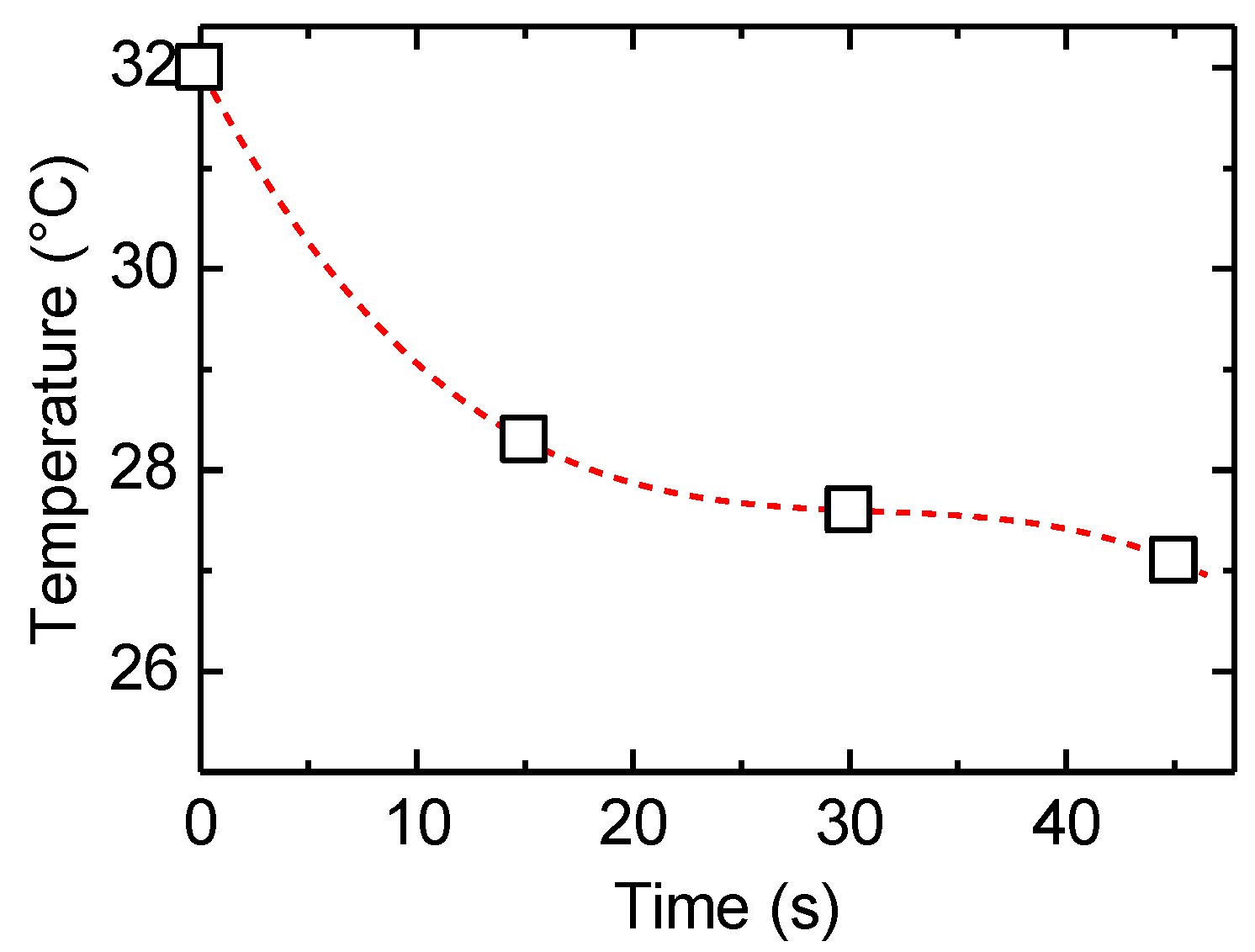
After heating of the PAZ/Hb with infrared light, the temperature change due to natural heat discharge was observed by thermographic images at room temperature (Figure 7). Although the temperature decreased immediately after the irradiation, the surface temperature was moderately maintained (Figure 8), suggesting a heat storage function of PAZ/Hb.
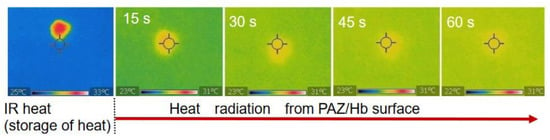
Figure 7.
Surface thermographic images of PAZ/Hb accompanied by heat discharge after IR heating.
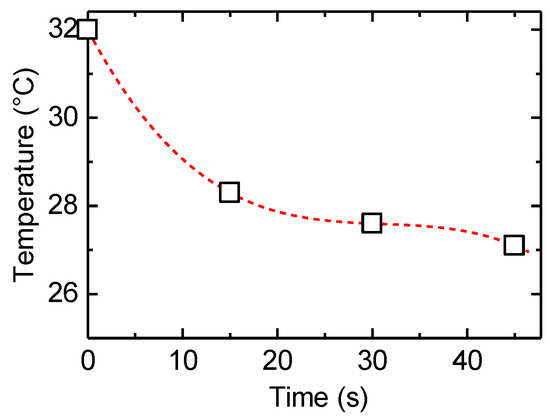
Figure 8.
Change in surface temperature as a function of time for PAZ/Hb accompanied by heat discharge after heat treatment.
3.4. Thermal Analysis
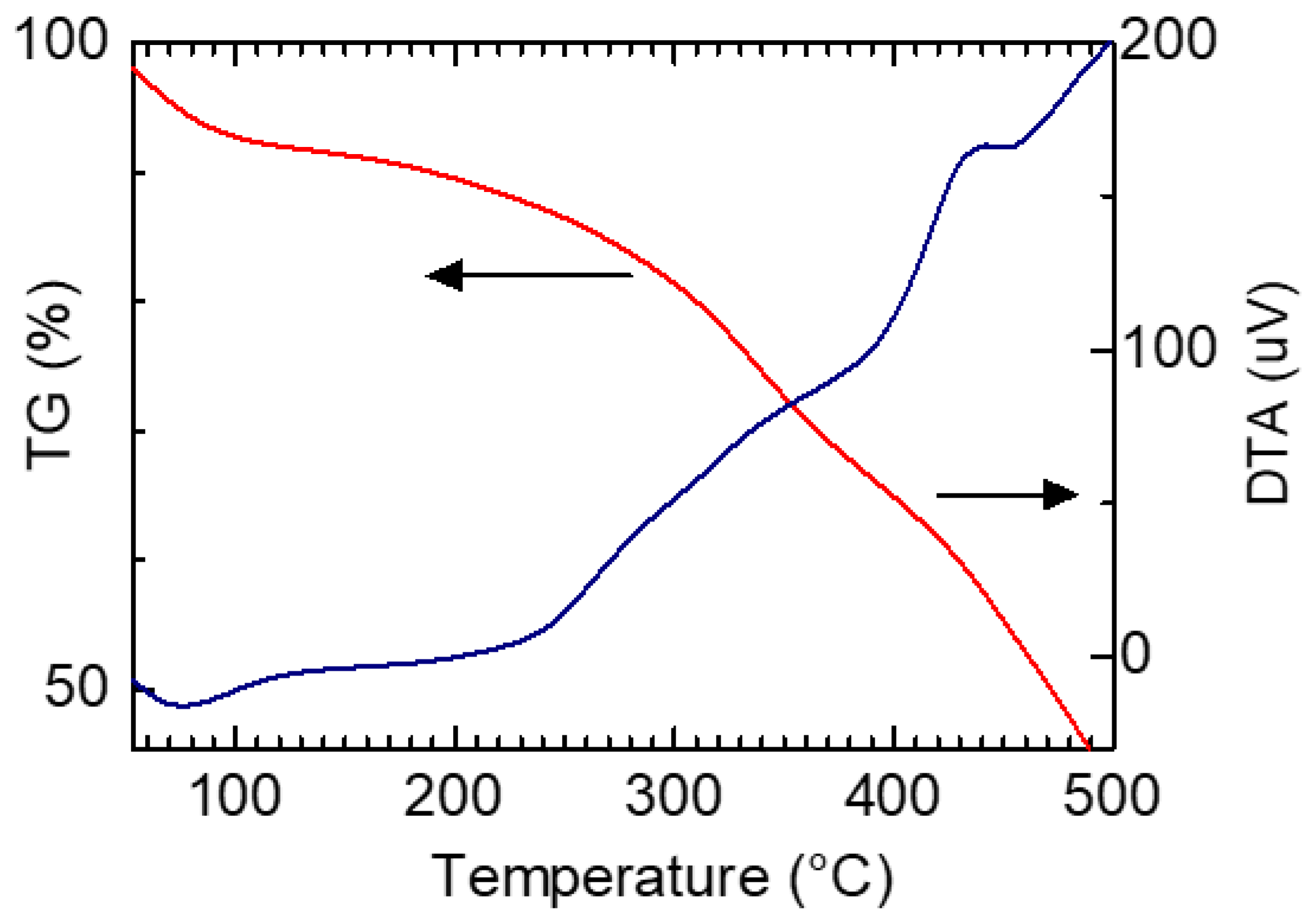
Figure 9 shows thermogravimetric (TG) analysis and differential thermal analysis (DTA) measurement results of PAZ/Hb. TG was used to measure the weight losses upon heating. An inflection point was observed at approximately 240 °C. The PAZ/Hb was carbonized with heating at 500 °C.
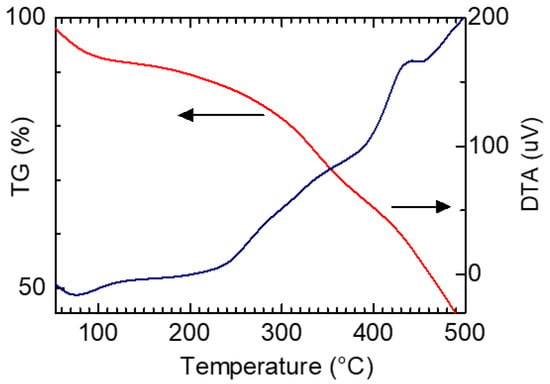
Figure 9.
Thermogravimetric (TG) analysis and differential thermal analysis (DTA) measurement results of PAZ/Hb.
3.5. Biocompatibility
To examine the biocompatibility of PAZ/Hb, PAZ/Hb fine powder was set on a sprout in a sterilized flask. After 3 days, the tissue of the sprout was maintained (Figure 10). Moreover, a form of helical vessel was maintained in the presence of PAZ/Hb. This implies that PAZ/Hb may have good affinity with the sprout and an antibacterial function derived from the π-conjugated structure.

Figure 10.
Optical microscopy image of a sprout tissue with PAZ/Hb.
4. Conclusions
We developed a new Bach-type reaction with the aid of oxy-hemoglobin to synthesize polyazobenzene as a polymeric-dye/hemoglobin composite. The advantage of this research is that it involved a new reaction using the function of biomolecules (Hb: oxygen supplier), as well as composite formation for plastics and biomaterials. The bio-based material displayed good affinity with life forms, which may lead to applications in medical science. In addition, the combination of Hb and polyazobenzene as a magnetically active composite can be applied to the field of biomedical science.
5. Techniques
The FT–IR 4600 (JASCO, Tokyo, Japan) instrument used the KBr method. Magnetic susceptibility measurements of the polymer were carried out using a superconductor interference device (SQUID, Quantum Design CA, Magnetic property measurement system, MPMS). FLIR i5 was used for obtaining thermographic images.
Author Contributions
M.I. synthesized the PAZ and PAZ/Hb. M.I. and H.G. performed the IR analysis. H.G. carried out conductivity measurements. R.M. performed magnetic measurements of the Hb with the SQUID. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Japan Society for the Promotion of Science (JSPS), Grants-in-Aid for Scientific Research (No. 20K05626).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We would like to thank the OPEN FACILITY, Research Facility Center for Science and Technology, University of Tsukuba.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bach, H.C. Oxidative coupling of primary aromoatic diamines-aromatic azopolymers. ACS Polym. Prep. 1966, 7, 576–581. [Google Scholar]
- Selvaraj, V.; Karthika, T.S.; Mansiya, C.; Alagar, M. An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. J. Mol. Struct. 2021, 1224, 129195. [Google Scholar] [CrossRef]
- Mehvish Ajaz, M.; Shakeel, S.; Rehman, A. Microbial use for azo dye degradation a strategy for dye bioremediation. Int. Microbiol. 2020, 23, 149–159. [Google Scholar] [CrossRef]
- Rawat, D.; Sharma, R.S.; Karmakar, S.; Arora, L.S.; Mishra, V. Ecotoxic potential of a presumably non-toxic azo dye. Ecotoxicol. Environ. Saf. 2018, 148, 528–537. [Google Scholar] [CrossRef]
- Harichandran, G.; Prasad, S. SonoFenton degradation of an azo dye, Direct Red. Ultrason. Sonochem. 2016, 29, 178–185. [Google Scholar] [CrossRef]
- Masashi Otaki, M.; Kumai, R.; Sagayama, H.; Goto, H. Synthesis of Polyazobenzenes Exhibiting Photoisomerization and Liquid Crystallinity. Polymers 2019, 11, 348. [Google Scholar] [CrossRef] [Green Version]
- Kuenstler, A.S.; Clark, K.D.; Alaniz, J.R.; Hayward, R.C. Reversible actuation via photoisomerization-induced melting of a semicrystalline poly(azobenzene). ACS Macro Lett. 2020, 9, 902–909. [Google Scholar] [CrossRef]
- Dowds, M.; Bank, D.; Strueben, J.; Soto, D.P.; Sönnichsen, F.D.; Renth, F.; Temps, F.; Staubitz, A. Efficient reversible photoisomerisation with large solvodynamic size-switching of a main chain poly(azobenzene-alt-trisiloxane). J. Mater. Chem. 2020, 8, 1835–1845. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, X.; Yuan, J.; An, X.; Sun, X.; Yi, J.; Song, X.M. Fast and self-recoverable photoinduced deformation behavior of azobenzene-containing poly(arylene ether)s with binaphthalene groups. J. Mater. Chem. C 2021, 9, 14139–14145. [Google Scholar] [CrossRef]
- Mosim Ansari, M.; Bera, R.; Mondal, S.; Das, N. Triptycene-Derived Photoresponsive Fluorescent Azo-Polymer as Chemosensor for Picric Acid Detection. ACS Omega 2019, 4, 9383–9392. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.O.; Seraphim, P.M.; Teixeira, M.F.S. Methylated DNA impedimetric immunosensor based on azo-polymer-AuNPs dots and 5-methylcytosine antibody using dissolved oxygen as a redox probe. Electrochem. Commun. 2022, 136, 107242. [Google Scholar] [CrossRef]
- Alauddin, S.M.; Aripin, N.F.K.; Velayutham, T.S.; IrakliChaganava, I.; Alfonso, M.F. The role of conductivity and molecular mobility on the photoanisotropic response of a new azo-polymer containing sulfonic groups. J. Photochem. Photobiol. A 2020, 389, 112268. [Google Scholar] [CrossRef]
- Masuda, K.; Shinozaki, R.; Shiraishi, A.; Ichijo, M.; Yamane, K.; Miyamoto, K.; Omatsu, T. Picosecond optical vortex-induced chiral surface relief in an azo-polymer film. J. Nanophotonics 2020, 14, 016012. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Tang, K.; Liu, S.G.; Zhang, Y.Y.; Zou, G.L. Hemoglobin biocatalysts synthesis of a conducting polyaniline. Reac. Funct. Polym. 2005, 65, 239–248. [Google Scholar] [CrossRef]
- Dong, J.; Li, X.; Zhou, Y.; Lu, Y.; Lv, Y.; Chi, Y.; He, O. Interactions of gallic acid with porcine hemoglobin: Effect on the redox state and structure of hemoglobin. J. Agric. Food Chem. 2021, 69, 397–403. [Google Scholar] [CrossRef]
- Xu, X.; Cui, Y.; Bu, H.; Chen, J.; Li, Y.; Tang, G.; Wang, L.Q. A photosensitizer loaded hemoglobin–polymer conjugate as a nanocarrier for enhanced photodynamic therapy. J. Mater. Chem. B 2018, 6, 1825–1833. [Google Scholar] [CrossRef]
- Mott, N.F.; Davis, E.A. Electronic Process in Non-Crystalline Materials, 2nd ed.; Oxford University Press: Oxford, UK, 1971. [Google Scholar]
- Komaba, K.; Goto, H. Soliton excitations in liquid crystal polyacetylene. Mol. Cryst. Liq. Cryst. 2020, 703, 69–78. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).