Abstract
A first assessment of genetic diversity and expansion of the invasive species Amorpha fruticosa L. was estimated in the 91E0* (Annex I Dir. 92/43/EEC) priority habitat of Nestos. A. fruticosa is a perennial deciduous shrub belonging to the family Fabaceae and is native to the south-western part of North America, which recently was introduced to Europe as an ornamental plant. Its expansion through the cross-border river of Nestos has significant negative effects on the 91E0* habitat (a habitat with Alnus glutinosa, Populus alba, and Ulmus minor). Herein, we studied the genetic diversity with amplified fragment length polymorphisms fragments of eastern and western populations in order to see if there is one genetic pool for the species. Furthermore, we estimated the regeneration index (IR) in order to see the potential expansion of this invasive species. Results showed that eastern and western populations have almost equal heterozygosity (He) and with a slightly higher value for the western population, which was in agreement with the results of the IR. This is an innovative study that discusses the implications of A. fruticosa of 91E0* habitat in Nestos and proposes management measures to eliminate its expansion and improve the resilience of the habitat.
1. Introduction
Alien and invasive plants threaten the integrity of natural ecosystems because most of them are invasive, compete with native plants, and in many cases take their place in the ecosystem. Invasive species are characterized by rapid spread due to the production of large numbers of progeny, which can be dispersed over long distances [1]. Moreover, they have a high survival rate due to their plasticity to respond to different environments. In addition, some natural phenomena can further help them spread, such as floods, wind, surface erosion, etc.
Τhe spread of invasive species has also been significantly affected by human activities [2]. Since ancient times, alien plants have been inadvertently or intentionally transported to new environments and cultivated as horticultural or ornamental plants [3]. Their spread is also facilitated by human activities, the role of which is twofold: transport propagating material and habitat alteration. The main advantage is that they do not have physical enemies that would control their populations so that they fully exploit their competitive potential. Some invasive plants also develop new mechanisms of interaction with natural ecosystems [4]. One of these mechanisms is the production of allelopathic substances and their release in the ecosystem [5].
Amorpha L. (Fabaceae Juss.: Amorpheae Boriss.) is a genus of 16 species native to North America [6]. These plants can be shrubs, subshrubs, or herbaceous perennials and occur in a variety of habitats, including riparian corridors, savannas, and grasslands. Many species of Amorpha have very restricted distributions, and several are considered threatened or endangered [7].
Amorpha fruticosa L. (indigo bush) is a perennial deciduous shrub belonging to the family Fabaceae and is native to the south-western part of North America [8]. The indigo bush was introduced in Europe as an ornamental plant [5]. It is used as a medicine for the treatment of burns and eczema [9]. In recent years, special attention has been paid to it due to its bioactivities. Studies have shown that it is a plant rich in polyphenols and contains bioactive ingredients. It also has anticancer, anti-inflammatory, and antimicrobial activity [10]. Indigo bush is a plant with great decorative value and is used in the construction of urban landscapes and in the protection of slopes. It can survive in dry soils, but prefers riverbanks and the edges of flooded forests, where it is easily spread by water as its seed pods are buoyant [11]. Its great tolerance to various climatic conditions as well as its great ability to multiply, gives it the advantage to grow outside its natural distribution with great ease [12]. Today, it is recognized as one of the most invasive species in Europe, with severe impacts on local ecosystems [11]. It is a photophilic plant that competes for light, forming dense thickets that overshadow native flora, and this leads to a decrease in biodiversity [11,12,13]. Due to the damage it causes to natural ecosystems and water management, measures should be taken immediately to reduce/control its populations.
In Greece, the cross-border rivers such as the Nestos, the Axios, etc., are transport corridors for alien plant species, which expand their range using water [14]. However, apart from the specific mode of transport, in recent years, many foreign plant species have been introduced into Greece, both intentionally (e.g., ornamental, and medicinal plants) and unintentionally, using various ways of introduction [2]. Although the number of alien species is large, not all are invasive. Out of the 343 alien species, only 50 show invasive behavior, and most of them are of American origin [15].
The Nestos is a cross-border river of Bulgaria and Northern Greece. It originates from Mount Rila in southern Bulgaria and passes between the mountain ranges of Aimos and Rhodope. Its length is 234 km, of which 130 km is located in Greece [16]. Before it empties into the sea, it stretches across the Chrysoupoli plain and expands as a deltaic system with freshwater lakes forming the Delta of Nestos. In that area, priority habitat 91E0 (Annex I Dir. 92/43/EEC) exists which shows two subtypes: the subtype of alder stands (Alnus glutinosa) and the subtype of white poplar (Populus alba) and elm (Ulmus minor) stands. The first subtype is composed of pure alder stands, which grow in light to moderately light soils with very good physical properties, which can be characterized as Calcaric fluvisols. Moreover, the subsoil water level is relatively high (90–95 cm). This subtype is found between the dykes in places next to the riverbanks or in places a few dozen meters away from them. The second subtype constitutes a significant part of the riparian forest and is located outside of the dykes. It is composed of white poplar and elm stands, which grow in deep light to moderately light soils with good physical properties, which belong to the Eutric Fluvisol soil type, without alkalinity and salinity problems. The subsoil water level is low but shows a large variation [17].
The area along the river has changed significantly since the Second World War [18]. In 1945, forests and wetlands dominated the area. In the 1960s, agricultural land increased, resulting in a decrease in forested areas. There was also a large reduction in wetlands due to their conversion to grassland. However, despite the great decline of the natural ecosystem, the area still maintains the most extensive riverine forest in Greece. Furthermore, the Nestos River Plain is of significant ecological importance due to its many important habitats. It has been classified as a wetland of international importance and has been included in the Ramsar Convention. It is also protected by the Berne Convention, European Commission Directives, and Greek legislation. In addition, it belongs to the Natura 2000 Network [19].
In Greece, Amorpha fruticosa L. is considered as an invasive species because it is a plant that has the ability to spread over a wide area. In the present study, the genetic diversity of Amorpha fruticosa L. populations growing along the Nestos River was investigated. Our objectives were (1) to determine the level of genetic differentiation, if it exists, between the two sides of the river: west and east; (2) to estimate the degree of inbreeding in each population as well as the effective population size; and (3) to discuss the implications of the expansion of the specific species in the natural ecosystem and more specifically in the populations of native species in the area.
2. Materials and Methods
2.1. Plant Material
Leaf sample collection was carried out on 20 Amorpha fruticosa individuals from 2 populations of the Nestos River area (indicative coordinates for west population A: X: 565671, Y: 4527927 and east population: X: 566918, Y: 4525481). DNA extraction from the fresh needles was performed with the Macherey Nagel Plant kit (Düren in North Rhine-Westphalia, Germany) according to the manufacturer’s protocol.
2.2. AFLP Procedure
A total of 200 ng of genomic DNA was fragmented by digestion with 4 U of EcoRI and MseI enzymes at a temperature of 37 °C for 3 h. The digested DNA fragments were then combined with EcoRI and MseI adapters, and the resulting mixture was ligated using T4 DNA ligase from New England Biolabs (Ipswich, MA, USA). This ligation step was carried out at a temperature of 26 °C for 3 h. The resulting DNA from this process was used as the primary template for AFLP analysis.
For the pre-amplification step, a pair of primers based on the sequences of the EcoRI and MseI adapters, with an additional selective nucleotide at the 3′ end (EcoRI+A and MseI+C), were used. The pre-amplification PCR was conducted in a total volume of 20 μL, which contained 1X Kapa Taq Buffer, 0.2 mM of each dNTP, 2.5 mM MgCl2, 30 ng of each primer (EcoRI+A and MseI+C), 1 unit of Taq DNA polymerase from Kapa Biosystems (located in Wilmington, MA, USA), and 5 μL of the diluted DNA fragments obtained from the digestion and ligation reaction. The PCR cycling conditions were as follows: initial denaturation at 95 °C for 30 s, followed by 32 cycles of denaturation at 95 °C for 30 s, annealing at 56 °C for 30 s, extension at 72 °C for 1 min, and a final extension step at 72 °C for 10 min. To confirm the amplification, a 5 μL sample of the reaction was subjected to agarose gel electrophoresis, while the remaining 15 μL was diluted 5-fold with a TE buffer.
Selective amplifications were performed in 10 μL reaction volumes, consisting of 3 μL of the diluted preselective template, and using the same reaction conditions as the preselective amplification step, with the exception of using 30 ng of a MseI primer and 5 ng of an EcoRI primer per reaction. The selective amplification cycling program was carried out using a BioRad thermocycler (Hercules, CA, USA), and followed this pattern: an initial cycle of denaturation at 95 °C for 30 s, annealing at 65 °C for 30 s, and extension at 72 °C for 1 min. This was followed by twelve cycles of denaturation at 95 °C for 30 s, annealing starting at 65 °C for 30 s and decreasing by 0.75 °C each cycle, and extension at 72 °C for 1 min. Finally, twenty-three cycles of denaturation at 95 °C for 30 s, annealing at 56 °C for 30 s, and extension at 72 °C for 1 min were performed, with a final hold at 72 °C for 30 min.
The f-AFLP product mixtures were subjected to denaturation by treating them with formamide at a temperature of 94 °C for a duration of 2 min. Subsequently, these denatured mixtures were separated through electrophoresis using an ABI Prism 3730xl Genetic Analyzer, which is manufactured by Applied Biosystems (Waltham, MA, USA). Ten AFLP selective primer combinations were employed. Each individual hybrid was then examined to determine the presezze or absence of specific fragments. The size of the detected fragments was determined by utilizing the GeneMapper 4.0 program, along with an internal standard known as GS 500 LIZ, which was also provided by Applied Biosystems (Waltham, MA, USA). In order to minimize the impact of potential size homoplasy, only fragments within the size range of 150 to 500 bases were considered and subjected to further analysis and counting [20].
2.3. Index of Regeneration
A. fruticosa presence and density were assessed in priority habitat 91E0* within the Nestos area, north-eastern Greece (Figure 1). Fifteen (15) plots, each covering an area of 706.86 m2, were selected at random and placed evenly throughout habitat 91E0*. Subsequently, an analysis was conducted to examine the composition and structure of tree species in these plots. A. fruticosa was present in four out of 15 plots, of which three were located in the west riverbank and one in the east. Tree height (m), density (individuals ha−1), and the index of regeneration (IR) were measured for A. fruticosa individuals. IR was calculated by multiplying the A. fruticosa density (per m2) by the average A. fruticosa height (cm) and provides a measure of the regeneration extent [20].

Figure 1.
Amorpha fruticosa extended by the 91Ε0* priority habitat in Nestos Delta, north-eastern Greece.
2.4. Data Collection and Statistical Analysis for AFLP Data
To transform allele size information obtained from GeneMapper 4.0 (Applied Biosystems, USA) into a binary format, an AFLP Excel Macro was employed. This macro assigned a value of “1” to fragments that were present and “0” to those that were absent. To minimize the impact of potential size homoplasy, only consistent fragments falling within the 150 to 500 base pair range were tallied and subjected to additional scrutiny [21]. In addition, GenAlex v6.0 [22] was used in order to calculate parameters such as percentage of polymorphic bands (P), Shannon’s information index (I), and genetic diversity (He). GenAlex v6.0 was also used to conduct an analysis of molecular variance (AMOVA) and to calculate genetic distances and principal coordinate analyses (PCoA).
In detail, principal coordinate analysis (PCoA) is a multivariate technique that allows one to find and plot the major patterns within a multivariate data set (e.g., multiple loci and multiple samples). The mathematics are complex, but in essence, PCoA is a process by which the major axes of variation are located within a multidimensional data set. For multidimensional data sets, each successive axis explains proportionately less of the total variation, such that when there are distinct groups, the first two or three axes will typically reveal most of the separation among them. Four different options are provided, two based on the conversion of the distance matrix to a covariance matrix, and two working directly from the input distance matrix. The distance matrix is used as the pool for calculating the distribution on axes [22].
2.5. Habitat Information of 91E0*
Habitat 91E0* in the Nestos Delta expands in low altitudes (<100 m above sea level), with surfaces of a mild to flat relief (gradient 0–10%) (Korakaki E. and Karetsos G. personal data sampling). It constitutes from dominant tree species such as Fraxinus excelsior L. and Alnus glutinosa (L.) Gaertn., but also Fraxinus angustifolia Vahl, Salix alba L., Populus alba L., Pinus nigra J.F. Arnold, Juglans regia L., Cornus sanguinea L., Ulmus minor Mill., and Quercus robur subsp. pedunculiflora (K. Koch) Menitsky. There is a profusion of climbing plants (e.g., Periploca graeca L., etc.), and the herbaceous layer invariably includes many species (e.g., Aegopodium podagraria L., Hedera helix L., Lysimachia punctata L., Circaea lutetiana L., Arum maculatum L., etc.) (Solomou A. and Karetsos G. personal data sampling). The Nestos Delta has been created by alluvial deposits of Nestos River. Fluvial sediments consist of alluvial deposits of clays, sands, and gravels, whereas floodplain deposits consist mainly of clays and sands [23].
3. Results
3.1. Genetic Results
Seven AFLP primer combinations yielded 313 loci for the two populations. According to AFLP results, the eastern population had 83.39% polymorphism, while the western population had 83.07%. The mean expected heterozygosity (He) was 0.127, and the western population presented a mean heterozygosity slightly higher (0.136) than the eastern (0.119). Parameters for genetic diversity are being presented in Table 1 along with the epigenetic results.

Table 1.
Mean and standard error (SE) for Na (number of alleles), Ne (number of effective alleles), I (Shannon information index), and He (expected heterozygosity).
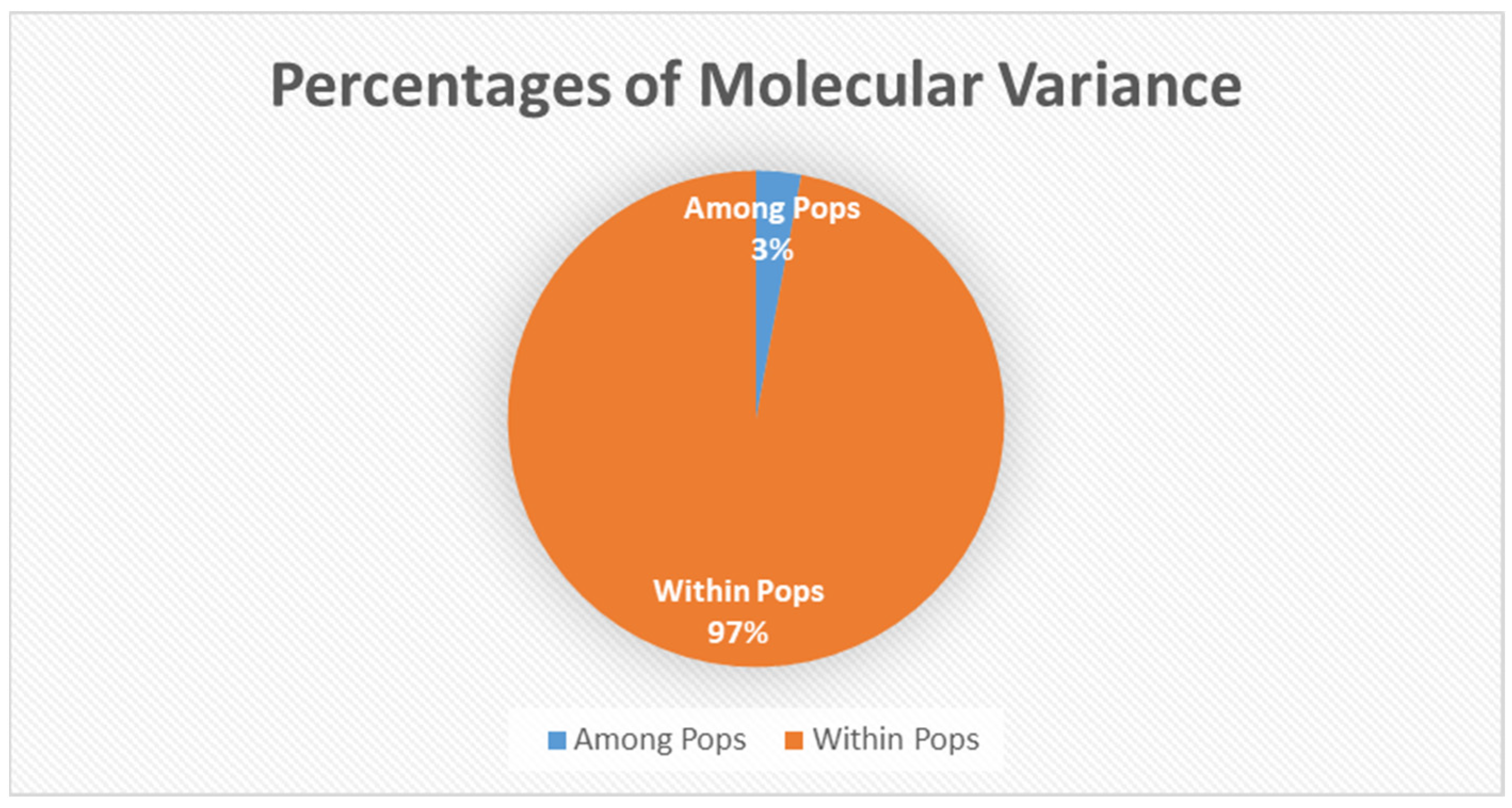
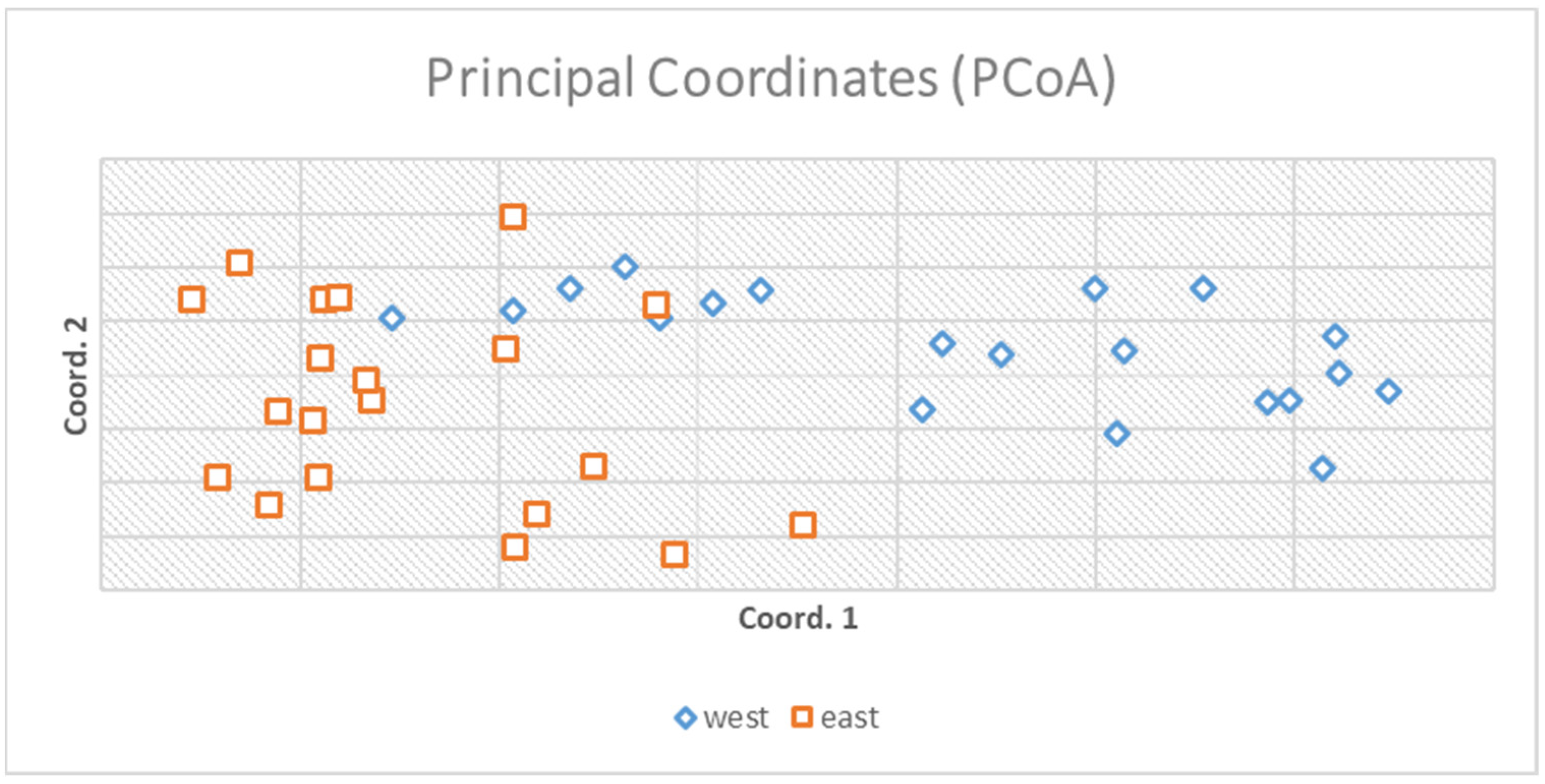
The AMOVA (analysis of molecular variance) portioned 97% of the genetic variation within populations and 3% only among populations (Figure 2). Principal coordinate analysis explained 16.89% of the variance (Figure 3).
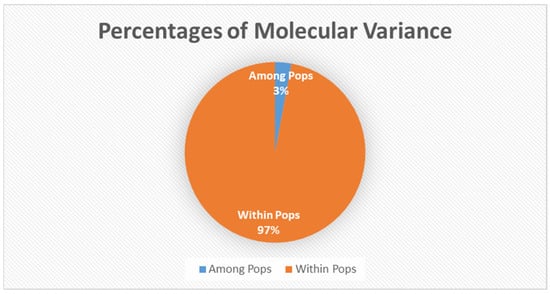
Figure 2.
Analysis of molecular variance among and within populations, among population variance was 3% while within was 97%. Genetic variation from AFLP fragments used for the analysis of AMOVA.
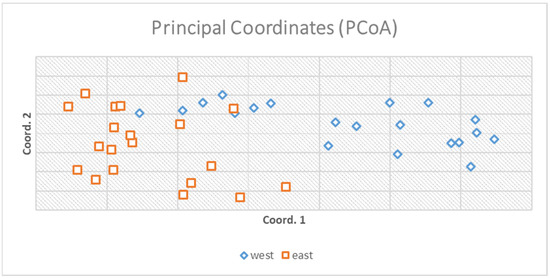
Figure 3.
Principal coordinate analysis explained 16.89% of the total variation and two small groups according to the origin of populations were formulated. The figure indicates a small grouping for the two populations derived from the matrix of genetic distances of the genotypes.
3.2. Density and Index of Regeneration
Across the four plots, a total of 233 A. fruticosa individuals were documented, resulting in an average density of 824.07 plants per hectare. Notably, the density of A. fruticosa and the index of regeneration (IR) were found to be higher in the population residing on the west river side compared to the east river side. However, it is worth mentioning that the mean tree height was greater on the east river side, as indicated in Table 2. The regeneration of alien species A. fruticosa and A. negundo is most widespread in the alluvial forest (Korakaki personal communication).

Table 2.
Mean height, mean density, and mean index of regeneration (IR).
4. Discussion
False Indigo (Amorpha fruticosa L.) has been reported as non-native [15], and recent observations have documented its presence in significant areas along the Ardas, Evros, and Nestos rivers in north-east Greece, indicating its introduction from neighboring Bulgaria, possibly through river transport of genetic material. In the riparian forests of the Nestos River area in NE Greece, A. fruticosa exhibits significant regeneration dynamics and appears to suppress vegetation in important priority habitats.
In this study, we assessed the genetic diversity of two populations from the west and east sides of the river Nestos in order to estimate if they exhibit differences in genetic parameters or share similar indexes of genetic diversity. AFLP loci estimated that heterozygosity for the western population was slightly higher but not significantly different from the eastern population. Furthermore, as the AMOVA analysis pointed out, the majority of genetic variation was within populations (97%). Principal coordinate analysis explained only 16.89% of the total variation, and two small groups according to the origin of populations were formulated. The main conclusion from the genetic analysis was that the western and eastern populations share a common genetic pool, which comes through the river Nestos with the water.
Regarding similar research, the authors did not find any reference to genetic diversity using molecular markers for plants of A. fruticosa. There is however one study by [24] in which they measured 47 agronomical and morphological traits for biomass energy. Results showed that all traits were influenced by accessions, a key that may also demonstrate genetic content that probably influences variation. Furthermore, studies have been conducted on the expression of genes associated with drought [25], phenolic constituents [26], proteomic analysis of symbiotic proteins [27], and cancer research [28,29]. The aforementioned research signifies from one point of view the significance of A. fruticosa, although we cannot eliminate the negative effects that it has on ecosystems due to its rapid expansion. This invasive species has the potential to invade meadow communities and rapidly transform the habitat into a dense and uniform thicket. It outcompetes native plants and disrupts natural plant succession and biodiversity. This poses a serious threat by suppressing native tree species in the Nestos Delta’s priority alluvial forest (91E0*). Additionally, it puts the survival of species Emys orbicularis, Testudo hermanni, and Callimorpha quadripunctaria* at risk, as they rely on these habitats.
According to our findings, A. fruticosa is more vigorous at the west riverside of Nestos, which poses a significant risk to biodiversity and the suppression of native tree species within the alluvial forest of the Nestos region. Thus, any conservation measures and forest management actions to limit the expansion of A. fruticosa should be prioritized on this river bank. Conservation measures are essential to safeguard and maintain this natural habitat, which is of significant interest to the community. Actions should also aim to raise awareness about the threats faced by the alluvial forest and ensure its long-term protection and preservation. This IR result gives us the first indication that A. fruticosa will be more expansive on the west side, and the result of the small but higher heterozygosity is in agreement with the current result. Since 2021, the Institute of Mediterranean Forest Ecosystems (ELGO-DIMITRA) has been establishing different experimental treatments in order to explore possible solutions to limit A. fruticosa expansion.
Finally, in this study, we found a similar genetic pool from the eastern and western sides of the river Nestos that probably originates from Bulgaria. Future research should also cover a genetic analysis of those Bulgarian populations. It is considered necessary to continue the research and study of A. fruticosa in the coming years so that there is more comprehensive knowledge of the species and the effect it has on the ecosystem so that a system of management of the species can then be created and its advantages can be exploited without having negative effects on the ecosystem.
5. Conclusions
A first report about genetic diversity and expansion of the alien species Amorpha fruticosa L. was employed across the river of Nestos for the first time in Greece. We studied two populations, one from the western side and one from the eastern side, with AFLP markers, and we did not find differentiation among them. This indicates that probably a single genetic pool was transmitted through the river from Bulgaria, and future research should also implement Bulgarian populations. Furthermore, the first assessment of the regeneration status was accomplished also indicates the expansion of the species. It is a mandatory step to evaluate and propose management actions in order to preserve and limit the expansion of the species, which significantly has negative effects in 91E0* habitat.
Author Contributions
Conceptualization, E.V.A. and E.K.; methodology, E.V.A., E.K., E.M., A.D.S., G.M. and G.K.; software, E.V.A., E.M. and E.K.; validation, all; resources, E.V.A. and E.K.; data curation, E.V.A. and E.K.; writing—original draft preparation, E.V.A., E.K., E.M. and A.D.S.; writing—review and editing, all; supervision, E.V.A. and E.K.; project administration, E.K.; funding acquisition, E.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the LIFE PRIMED project, LIFE17 NAT/GR/000511, which is co-financed by the Greek Green Fund (GRFU).
Data Availability Statement
Data not available.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Richardson, D.M.; Pyšek, P.; Rejmanek, M.; Barbour, M.G.; Panetta, F.D.; West, C.J. Naturalization and invasion of alien plants: Concepts and definitions. Divers. Distrib. 2000, 6, 93–107. [Google Scholar] [CrossRef]
- Korakaki, E.; Legakis, A.; Katsanevakis, S.; Koulelis, P.; Avramidou, E.; Soulioti, N.; Petrakis, P. Invasive alien species of greece. Invasive Alien Species Obs. Issues Around World 2021, 3, 124–189. [Google Scholar]
- Miyawaki, S.; Washitani, I. Invasive alien plant species in riparian areas of japan: The contribution of agricultural weeds, revegetation species and aquacultural species. Glob. Environ. Res.-Engl. Ed. 2004, 8, 89–101. [Google Scholar]
- Callaway, R.M.; Aschehoug, E.T. Invasive plants versus their new and old neighbors: A mechanism for exotic invasion. Science 2000, 290, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Ivanescu, B.; Lungu, C.; Vlase, L.; Gradinaru, A.C.; Tuchilus, C. Hplc analysis of phenolic compounds, antioxidant and antimicrobial activity of Amorpha fruticosa L. Extracts. J. Plant Dev. 2019, 26, 77. [Google Scholar] [CrossRef]
- Straub, S.C.; Sorrie, B.A.; Weakley, A.S. Amorpha confusa, a new name for an old Amorpha (Fabaceae: Amorpheae). J. Bot. Res. Inst. Tex. 2009, 151–155. [Google Scholar]
- Straub, S.C.; Doyle, J.J. Molecular phylogenetics of Amorpha (Fabaceae): An evaluation of monophyly, species relationships, and polyploid origins. Mol. Phylogenetics Evol. 2014, 76, 49–66. [Google Scholar] [CrossRef]
- Liu, J.; Wang, E.T.; Chen, W.X. Diverse rhizobia associated with woody legumes Wisteria sinensis, Cercis racemosa and Amorpha fruticosa grown in the temperate zone of China. Syst. Appl. Microbiol. 2005, 28, 465–477. [Google Scholar] [CrossRef]
- Chinese, P. A Dictionary of Chinese Materia Medica; Chinese Science and Technology Press of Medicine: Beijing, China, 1997. [Google Scholar]
- Mitscher, L.A.; Park, Y.H.; Alshamma, A.; Hudson, P.B.; Haas, T. Amorfrutin a and b, bibenzyl antimicrobial agents from Amorpha fruticosa. Phytochemistry 1981, 20, 781–785. [Google Scholar] [CrossRef]
- Krpan, A.P.; Tomašić, Ž.; Bašić Palković, P. Biopotential of indigobusch (Amorpha fruticosa L.)–second year of investigation. Šumarski List. 2011, 135, 103–112. [Google Scholar]
- Kozuharova, E.; Matkowski, A.; Woźniak, D.; Simeonova, R.; Naychov, Z.; Malainer, C.; Mocan, A.; Nabavi, S.M.; Atanasov, A.G. Amorpha fruticosa–a noxious invasive alien plant in europe or a medicinal plant against metabolic disease? Front. Pharmacol. 2017, 8, 333. [Google Scholar] [CrossRef] [PubMed]
- Szigetvári, C. Initial steps in the regeneration of a floodplain meadow after a decade of dominance of an invasive transformer shrub, Amorpha fruticosa L. Tiscia 2002, 33, 67–77. [Google Scholar]
- Pedashenko, H.P.; Apostolova, I.I.; Vassilev, K.V. Amorpha fruticosa invasibility of different habitats in lower danube. Phytol. Balc. 2012, 18, 285–291. [Google Scholar]
- Arianoutsou, M.; Bazos, I.; Delipetrou, P.; Kokkoris, Y. The alien flora of Greece: Taxonomy, life traits and habitat preferences. Biol. Invasions 2010, 12, 3525–3549. [Google Scholar] [CrossRef]
- Samaras, A.G.; Koutitas, C.G. Modelling the impact on coastal morphology of the water management in transboundary river basins: The case of river Nestos. Manag. Environ. Qual. Int. J. 2008, 19, 455–466. [Google Scholar] [CrossRef]
- Décamps, H.; Décamps, O. Mediterranean Riparian Woodlands; Conservation of Mediterranean Wetlands; Tour du Valat: Arles, France, 2001. [Google Scholar]
- Mallinis, G.; Emmanoloudis, D.; Giannakopoulos, V.; Maris, F.; Koutsias, N. Mapping and interpreting historical land cover/land use changes in a natura 2000 site using earth observational data: The case of Nestos delta, Greece. Appl. Geogr. 2011, 31, 312–320. [Google Scholar] [CrossRef]
- Dafis, S.; Papastergiadou, E.; Georghiou, K.; Babalonas, D.; Georgiadis, T.; Papageorgiou, M.; Lazaridou, T.; Tsiaoussi, V. The Greek ‘habitat’project Natura 2000: An overview. In Life Contract B4-32000/94/756; Commission of the European Communities, The Goulandris Natural History Museum-Greek Biotope/Wetland Centre; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Bianchi, L.; Paci, M. Dinamica evolutiva e gestione delle abetine toscane: Sintesi di quarant’anni di ricerche. For.-J. Silvic. For. Ecol. 2008, 5, 122. [Google Scholar] [CrossRef]
- Vekemans, X.; Beauwens, T.; Lemaire, M.; Roldán-Ruiz, I. Data from amplified fragment length polymorphism (AFLP) markers show indication of size homoplasy and of a relationship between degree of homoplasy and fragment size. Mol. Ecol. 2002, 11, 139–151. [Google Scholar] [CrossRef]
- Peakall, R.O.D.; Smouse, P.E. Genalex 6: Genetic analysis in excel. Population genetic software for teaching and research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Taftsoglou, M.; Valkaniotis, S.; Papathanassiou, G.; Klimis, N.; Dokas, I. A detailed liquefaction susceptibility map of nestos river delta, thrace, Greece based on surficial geology and geomorphology. Geosciences 2022, 12, 361. [Google Scholar] [CrossRef]
- DeHaan, L.; Ehlke, N.; Sheaffer, C.; Wyse, D.; DeHaan, R. Evaluation of diversity among north american accessions of false indigo (Amorpha fruticosa L.) for forage and biomass. Genet. Resour. Crop Evol. 2006, 53, 1463–1476. [Google Scholar] [CrossRef]
- Li, M.; Liu, Z.; Liu, C.; Zhu, F.; Wang, K.; Wang, Z.; Li, X.; Lan, X.; Guan, Q. Drought resistance of tobacco overexpressing the afnac1 gene of Amorpha fruticosa Linn. Front. Plant Sci. 2022, 13, 980171. [Google Scholar] [CrossRef] [PubMed]
- Dat, N.T.; Lee, J.-H.; Lee, K.; Hong, Y.-S.; Kim, Y.H.; Lee, J.J. Phenolic constituents of Amorpha fruticosa that inhibit nf-κb activation and related gene expression. J. Nat. Prod. 2008, 71, 1696–1700. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Qi, D.; Liu, X.; Kong, X.; Gao, Y.; Zhou, Z.; Wu, Q. Proteomic analysis of symbiotic proteins of Glomus mosseae and Amorpha fruticosa. Sci. Rep. 2015, 5, 18031. [Google Scholar] [CrossRef] [PubMed]
- Todorova, T.; Boyadzhiev, K.; Shkondrov, A.; Parvanova, P.; Dimitrova, M.; Ionkova, I.; Krasteva, I.; Kozuharova, E.; Chankova, S. Screening of Amorpha fruticosa and Ailanthus altissima extracts for genotoxicity/antigenotoxicity, mutagenicity/antimutagenicity and carcinogenicity/anticarcinogenicity. BioRisk 2022, 17, 201–212. [Google Scholar] [CrossRef]
- Cvetković, D.M.; Jovankić, J.V.; Milutinović, M.G.; Nikodijević, D.D.; Grbović, F.J.; Ćirić, A.R.; Topuzović, M.D.; Marković, S.D. The anti-invasive activity of Robinia pseudoacacia L. and Amorpha fruticosa L. On breast cancer mda-mb-231 cell line. Biologia 2019, 74, 915–928. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).