The Role of Voltammetric Analysis in the Wine Industry †
Abstract
1. Introduction
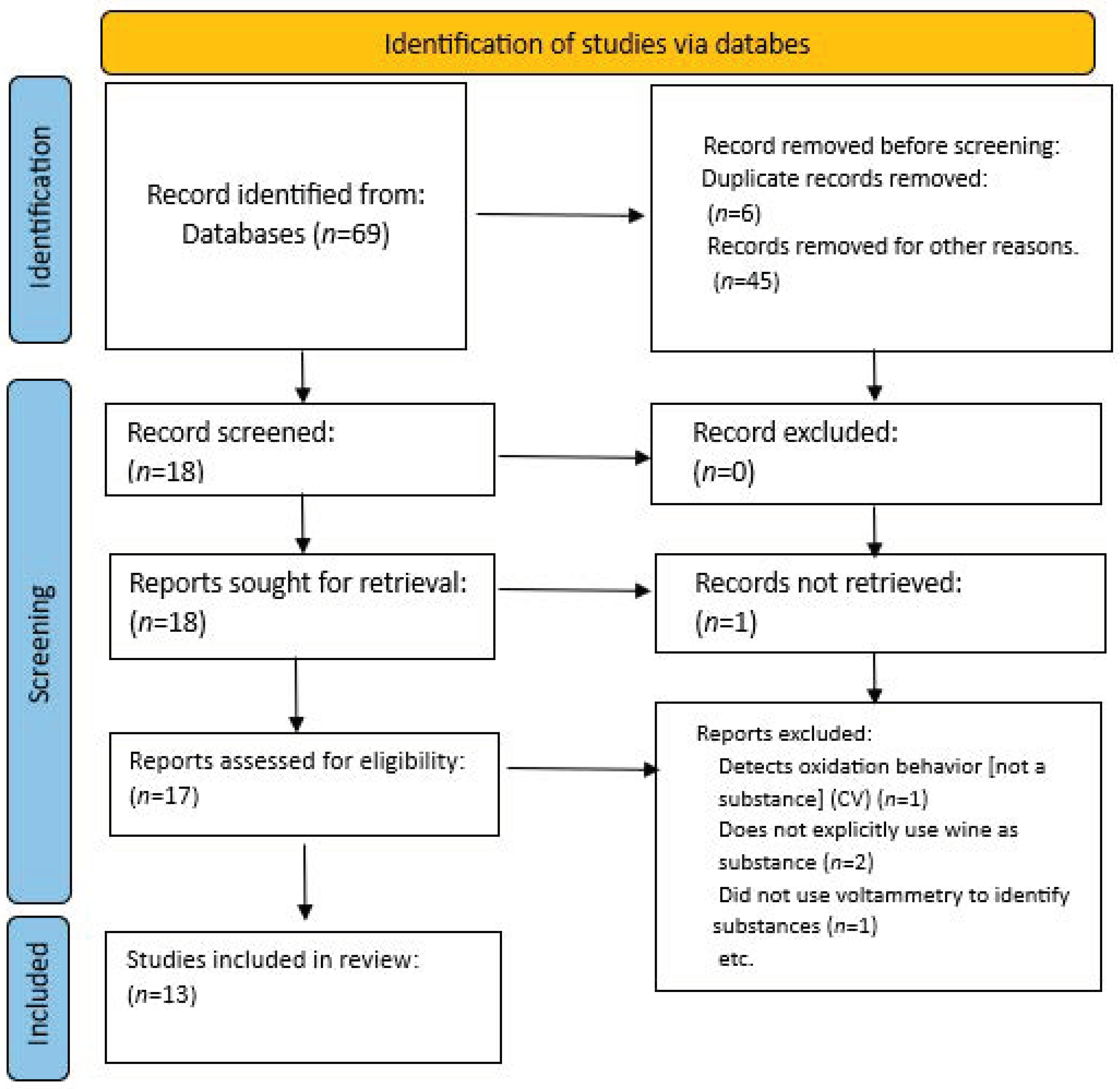
2. Methodology
3. Results and Discussion
3.1. Electrochemical Detection of Components
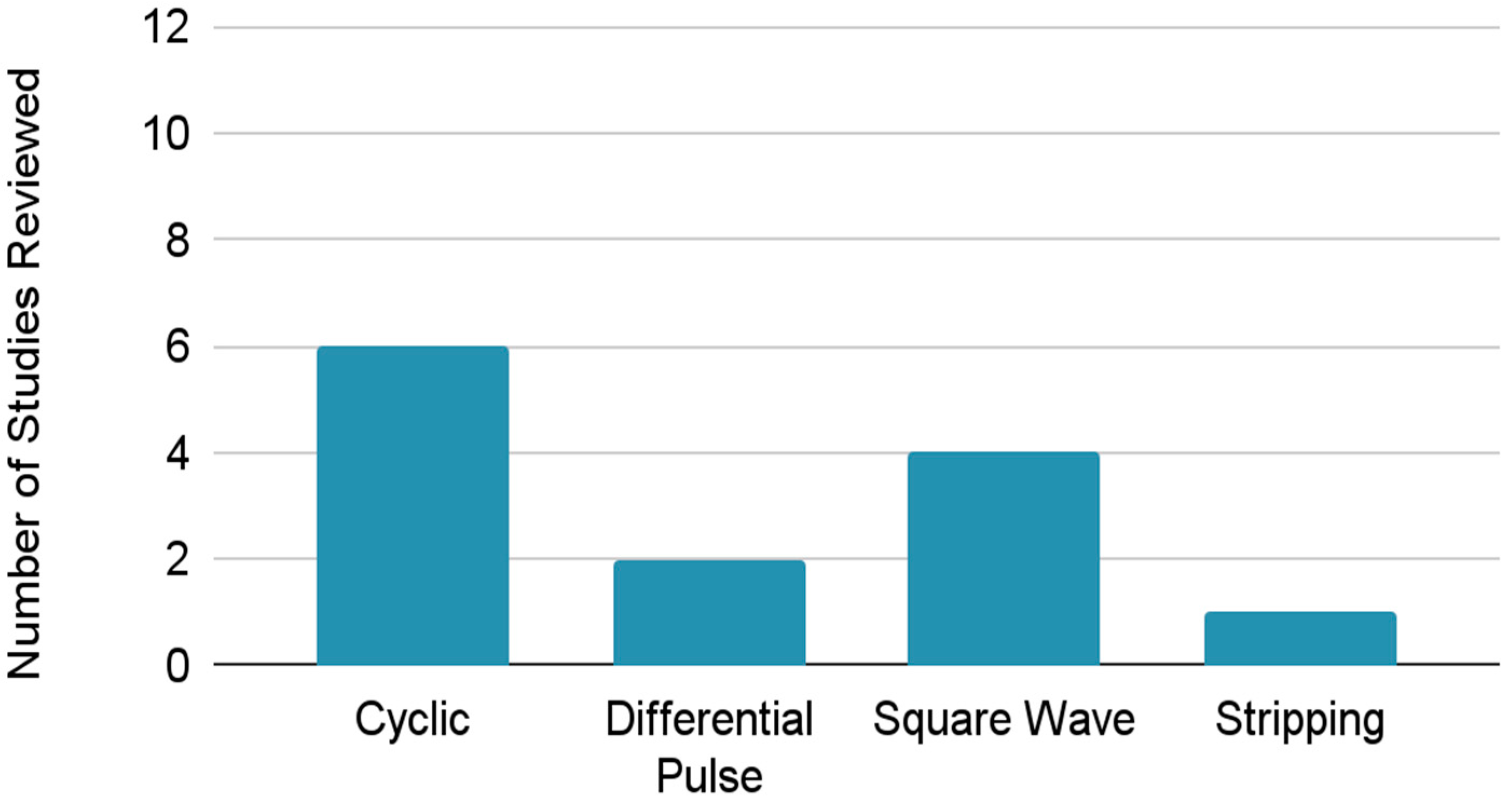
3.2. Method of Determination
3.3. Comparison of Electrode Performance
| Working Electrode | Component | Method | Peak | LOD | pH | Reference |
|---|---|---|---|---|---|---|
| AgCl/Ag (3M 78 KCl) | Free and total sulfides | SWV | *n.r. | 3 mg/L | n.d. | [20] |
| Glassy carbon (GC) electrode | Free sulfur dioxide | CV | 1120 mV | *n.r. | 3.3 | [14] |
| Graphite Electrode | Manganese (II) | SV | 550 mV | 0.6 mg/L | 5–5.2 | [17] |
| Screen-printed carbon | Sulfides | SWV | *n.r. | 0.4 mg L | >1.0 | [13] |
| Working Electrode | Component | Method | Peak | LOD | pH | Reference |
|---|---|---|---|---|---|---|
| Ag (Silver) | 2,4,6-trichloroanisole | CV | *n.r | 0.08–0.16 mg/L | n.d. | [9] |
| Glassy/screen-printed carbon electrode | Catechin | CV | 151 mV | *n.r. | 3.6 | [19] |
| Glassy carbon (GC) electrode | Catechins | DPV | 750 mV | 1.77 mg/L | n.r. | [16] |
| Glassy carbon (GC) electrode | Polyphenols | CV | 450 mV | *n.r. | 3.3 | [14] |
| Glassy carbon (GC) electrode | polyphenols | SWV | 395 and 730 mV | **n.r. | 3.46–3.74 | [15] |
| Glassy carbon (GC) electrode | Total polyphenol (TP) content | DPV | 440–475 mV | 1.77 mg/L | 3.6 | [21] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Avelino, K.Y.; Silva, R.R.; da Silva Junior, A.G.; Oliveira, M.D.; Andrade, C.A. Smart applications of bionanosensors for BCR/ABL fusion gene detection in leukemia. J. King Saud Univ. Sci. 2017, 29, 413–423. [Google Scholar] [CrossRef]
- Mabbott, G.A. An introduction to cyclic voltammetry. J. Chem. Educ. 1983, 60, 697. [Google Scholar] [CrossRef]
- Wawrzyniak, J.; Ryniecki, A.; Zembrzuski, W. Application of voltammetry to determine vitamin C in apple juices. Acta Sci. Pol. Technol. Aliment. 2005, 4, 5–16. [Google Scholar]
- McGorrin, R.J. One hundred years of progress in food analysis. J. Agric. Food Chem. 2009, 57, 8076–8088. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.Z.; Malakhova, N.A.; Stojko, N.Y. Stripping voltammetry in environmental and food analysis. Fresenius’ J. Anal. Chem. 2000, 368, 307–325. [Google Scholar] [CrossRef]
- Cordova, A.; Sumpio, B. Polyphenols are medicine: Is it time to prescribe red wine for our patients? Int. J. Angiol. 2009, 18, 111–117. [Google Scholar] [CrossRef]
- Ugalino, M.; Wirth, J.; Bégrand, S.; Dieval, J.B.; Vidal, S. Oxidation signature of grape must and wine by linear sweep voltammetry using disposable carbon electrodes. In Advances in Wine Researc, 1st ed.; Ebeler, S.B., Sacks, G., Vidal, S., Winterhalter, P., Eds.; ACS Publications: Washington, DC, USA, 2015; Volume 1203, Chapter 20; pp. 325–334. [Google Scholar]
- Okoli, C.; Schabram, K. A guide to conducting a systemic literature review of information system research. Commun. Assoc. Inf. Syst. 2015, 37, 879–910. [Google Scholar] [CrossRef]
- Freitas, P.; Dias, L.G.; Peres, A.M.; Castro, L.M.; Veloso, A.C. Determination of 2,4,6-trichloroanisole by cyclic voltammetry. Procedia Eng. 2012, 47, 1125–1128. [Google Scholar] [CrossRef]
- International Organization of Vine and Wine. Available online: https://www.oiv.int/public/medias/7840/oiv-collective-expertise-document-so2-and-wine-a-review.pdf (accessed on 15 February 2022).
- Danilewicz, J.C. Chemistry of manganese and interaction with iron and copper in wine. Am. J. Enol. Vitic. 2022, 67, 377–384. [Google Scholar] [CrossRef]
- Vilas-Boas, A.; Valderrama, P.; Fontes, N.; Geraldo, D.; Bento, F. Evaluation of total polyphenol content of wines by means of voltammetric techniques: Cyclic voltammetry vs Differential Pulse Voltammetry. Food Chem. 2019, 276, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.M.; Gonçalves, L.M.; Vyskočil, V.; Rodrigues, J.A. Free sulphite determination in wine using screen-printed carbon electrodes with prior gas-diffusion microextraction. Electrochem. Commun. 2016, 63, 52–55. [Google Scholar] [CrossRef]
- Makhotkina, O.; Kilmartin, P.A. The use of cyclic voltammetry for wine analysis: Determination of polyphenols and free sulfur dioxide. Anal. Chim. Acta 2010, 668, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Newair, E.F.; Kilmartin, P.A.; Garcia, F. Square wave voltammetric analysis of polyphenol content and antioxidant capacity of red wines using glassy carbon and disposable carbon nanotubes modified screen-printed electrodes. Eur. Food Res. Technol. 2018, 244, 1225–1237. [Google Scholar] [CrossRef]
- Ramalho, S.A.; Gualberto, N.C.; Neta, M.T.S.L.; Batista, R.A.; Araújo, S.M.; de Jesus da Silveira Moreira, J.; Narain, N. Catechin and Epicatechin contents in wines obtained from Brazilian exotic tropical fruits. Food Nutr. Sci. 2014, 5, 449–457. [Google Scholar] [CrossRef][Green Version]
- Burmakina, G.V.; Mokh, N.S.; Maksimov, N.G.; Zimonin, D.V.; Zhizhaev, A.M.; Rubaylo, A.I. Determination of manganese(II) in wines by stripping voltammetry on solid electrodes. J. Anal. Chem. 2016, 71, 71–76. [Google Scholar] [CrossRef]
- Taye, A.; Sergawie, A. Determination of ascorbic acid content of wine and soft drinks by voltammetric techniques at glassy carbon electrode. JSM Chem. 2019, 7, 1054. [Google Scholar]
- Deshaies, S.; Garcia, L.; Veran, F.; Mouls, L.; Saucier, C.; Garcia, F. Red Wine Oxidation Characterization by Accelerated Ageing Tests and Cyclic Voltammetry. Antioxidants 2021, 10, 1943. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, L.M.; Grosso Pacheco, J.; Jorge Magalhães, P.; António Rodrigues, J.; Araújo Barros, A. Determination of free and total sulfites in wine using an automatic flow injection analysis system with voltammetric detection. Food Addit. Contam. Part A 2010, 27, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Šeruga, M.; Novak, I.; Jakobek, L. Determination of polyphenols content and antioxidant activity of some red wines by differential pulse voltammetry, HPLC and spectrophotometric methods. Food Chem. 2011, 124, 1208–1216. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buenaventura, T.M.A.; Catangay, C.J.L.; Dolendo, C.D.C.; Soriano, A.N.; Lardizabal, D.D.; Rubi, R.V.C. The Role of Voltammetric Analysis in the Wine Industry. Eng. Proc. 2023, 56, 152. https://doi.org/10.3390/ASEC2023-16641
Buenaventura TMA, Catangay CJL, Dolendo CDC, Soriano AN, Lardizabal DD, Rubi RVC. The Role of Voltammetric Analysis in the Wine Industry. Engineering Proceedings. 2023; 56(1):152. https://doi.org/10.3390/ASEC2023-16641
Chicago/Turabian StyleBuenaventura, Therese Marie A., Cassandra Jayne L. Catangay, Christine Dominique C. Dolendo, Allan N. Soriano, Darvin D. Lardizabal, and Rugi Vicente C. Rubi. 2023. "The Role of Voltammetric Analysis in the Wine Industry" Engineering Proceedings 56, no. 1: 152. https://doi.org/10.3390/ASEC2023-16641
APA StyleBuenaventura, T. M. A., Catangay, C. J. L., Dolendo, C. D. C., Soriano, A. N., Lardizabal, D. D., & Rubi, R. V. C. (2023). The Role of Voltammetric Analysis in the Wine Industry. Engineering Proceedings, 56(1), 152. https://doi.org/10.3390/ASEC2023-16641






