Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite †
Abstract
:1. Introduction
2. Method
2.1. Bacterial Biological Activity Test
2.2. Preparation of Standard Solutions for Bacteria
2.3. Preparation of Standard Solutions for Fungus
3. Results and Discussion
3.1. Physicochemical Characterization of Adsorbents Surfaces
3.2. Effect of pH on the Release Ratio of Chlorazepam Drug In Vitro
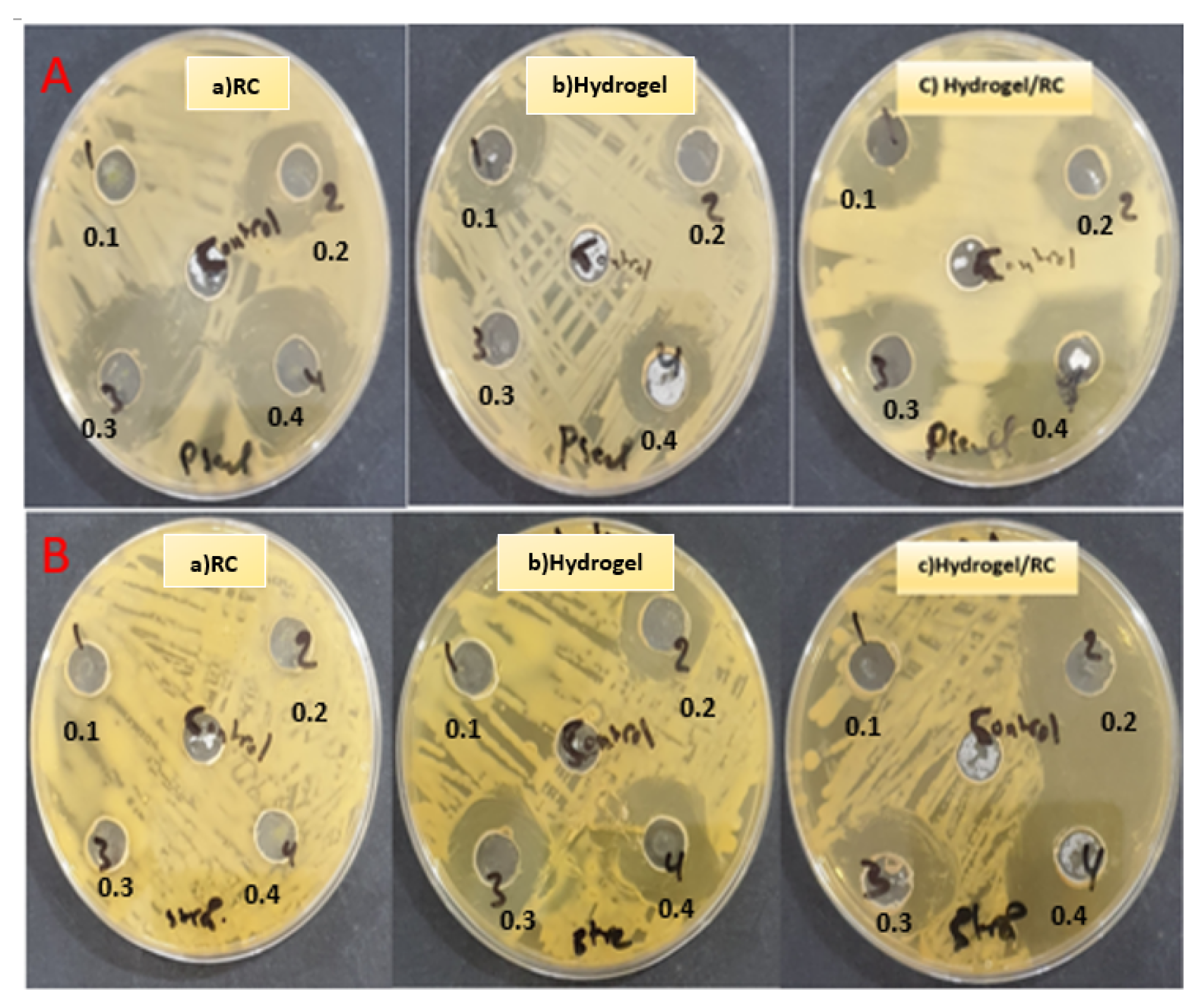
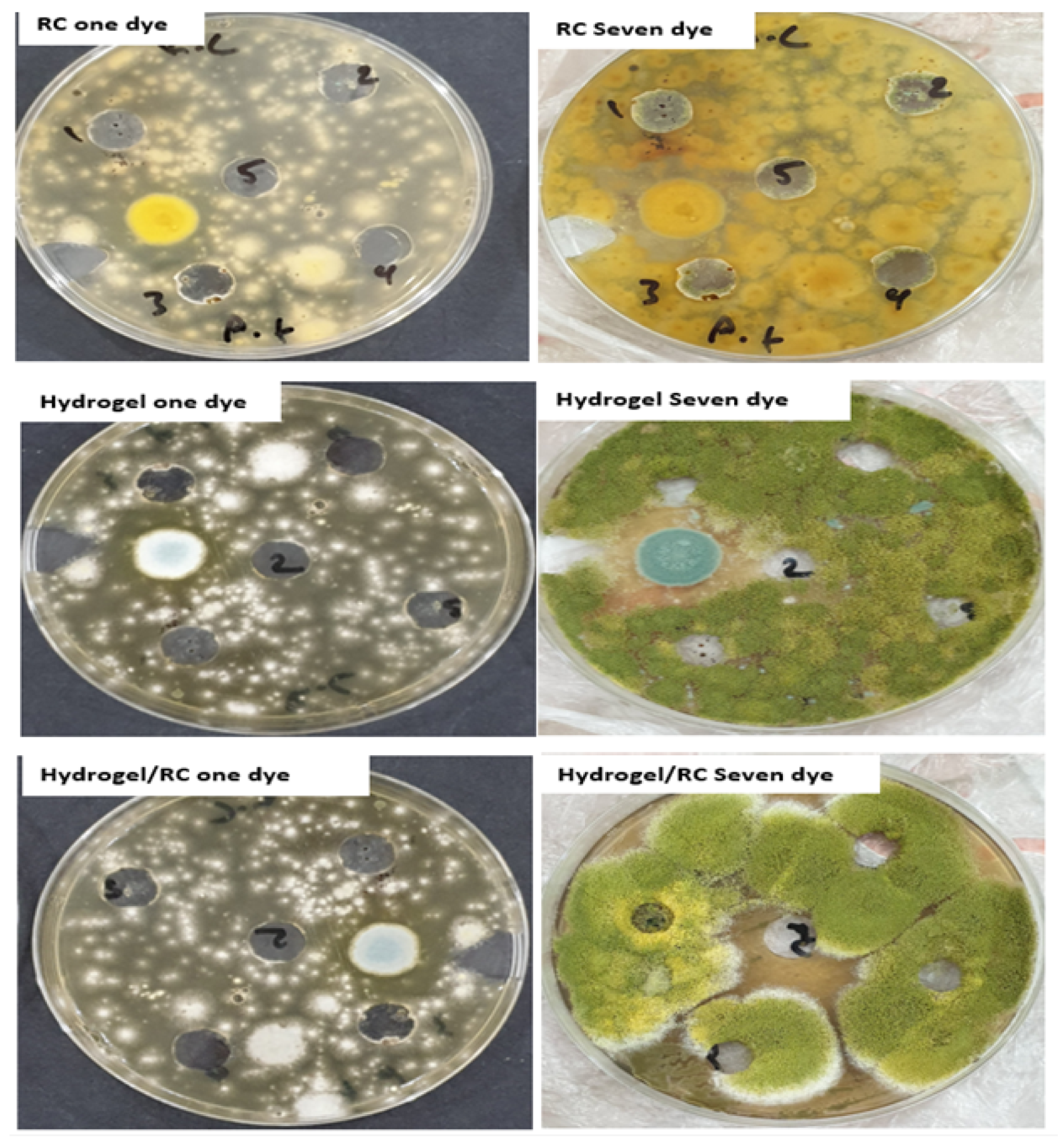
3.3. Biological Activity of Bacterial Test
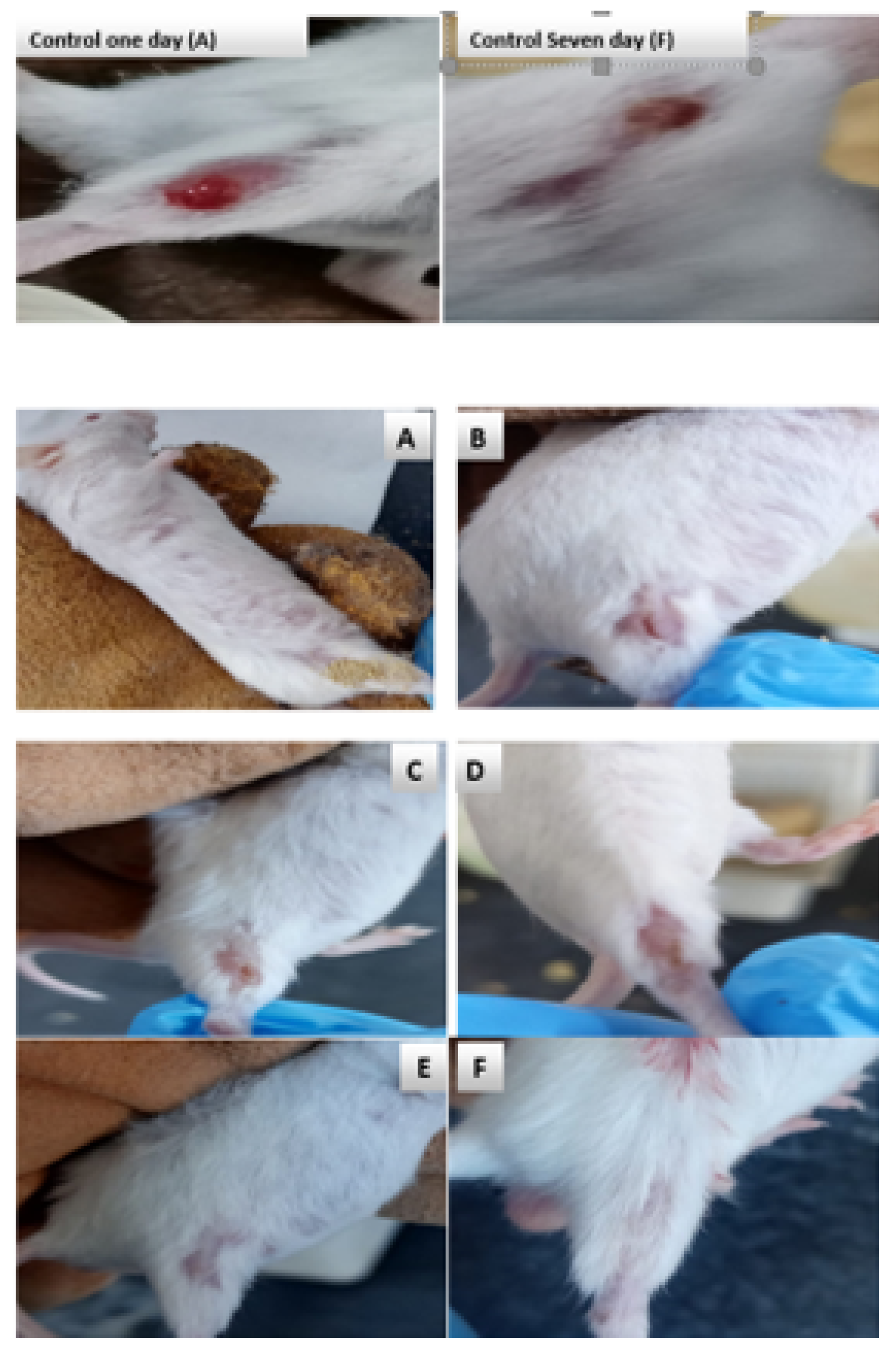
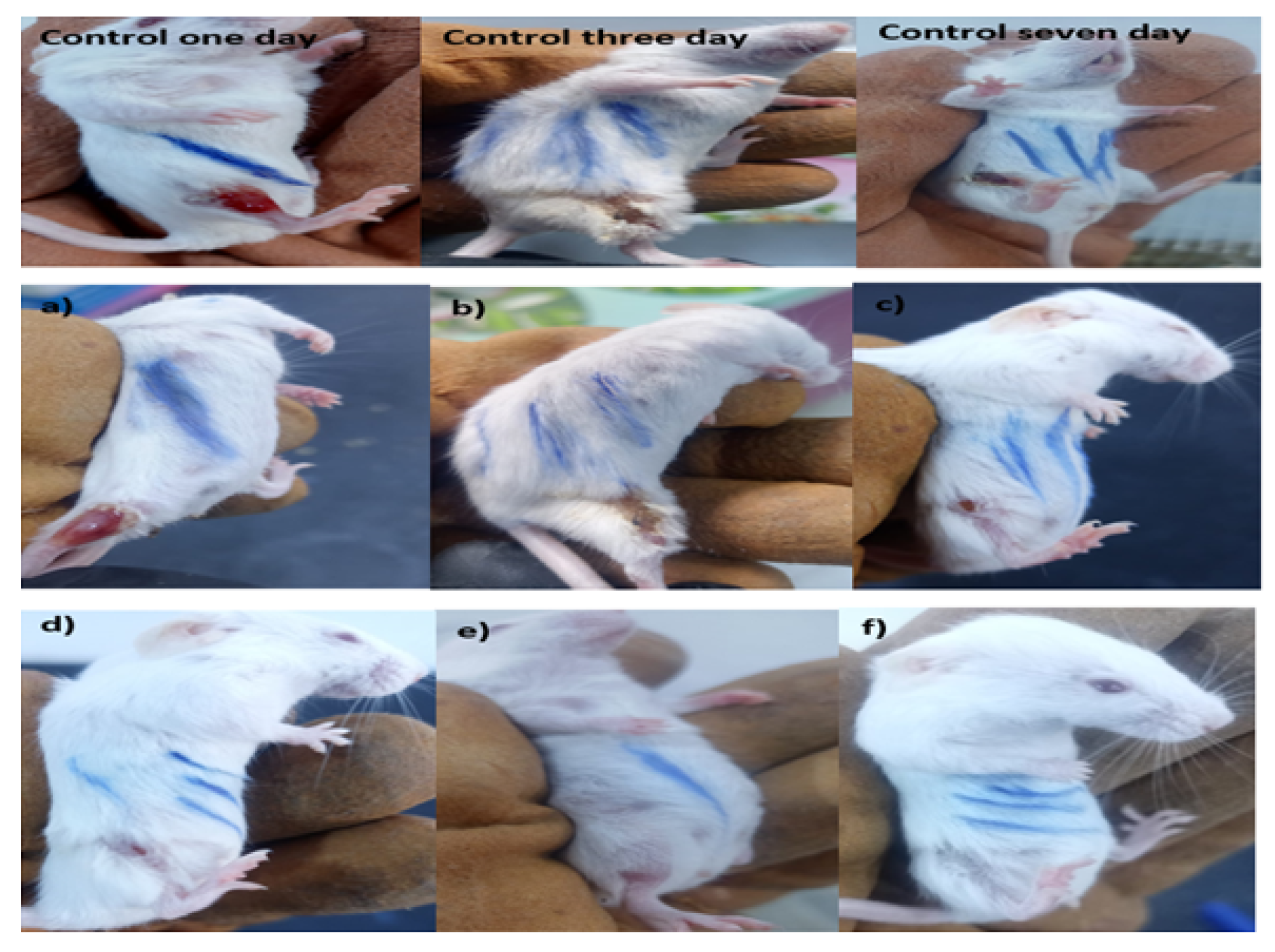
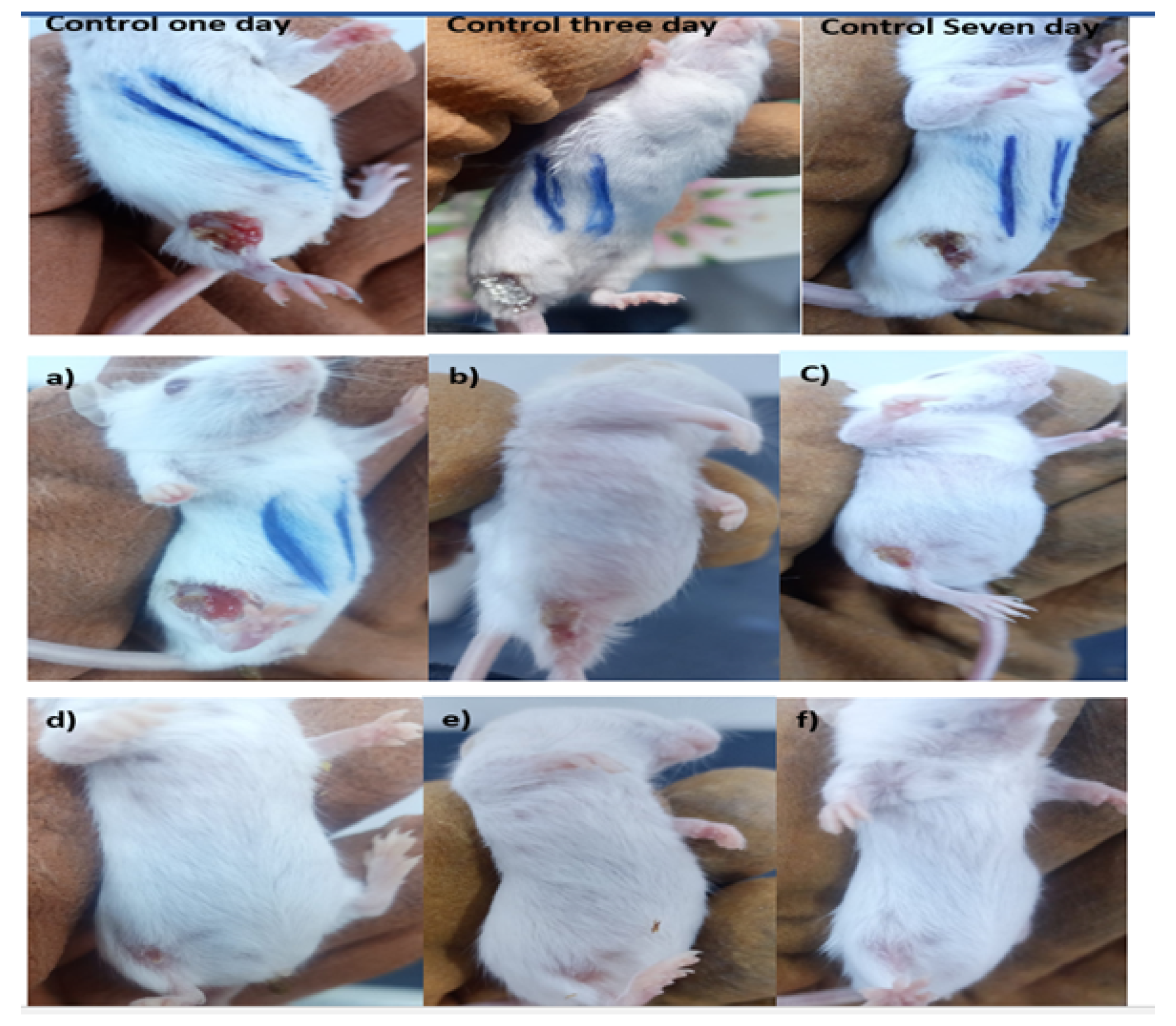
3.4. Efficacy of Surface Treatment in Mice Wound Healing
3.5. Future Scope and Generalizability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pashaei-Fakhri, S.; Peighambardoust, S.J.; Foroutan, R.; Arsalani, N.; Ramav, I.B. Crystal violet dye sorption over acrylamide/graphene oxide bonded sodium alginate nanocomposite hydrogel. Chemosphere 2021, 270, 129419. [Google Scholar] [CrossRef] [PubMed]
- Radia, N.D.; Mahdi, A.B.; Mohammed, G.A.; Sajid, A.; Altimari, U.S.; Shams, M.A.; Aljeboree, A.M.; Fh, A. Removal of Rose Bengal Dye from Aqueous Solution using Low Cost (SA-g-PAAc) Hydrogel: Equilibrium and Kinetic Study. Int. J. Drug Deliv. Technol. 2022, 12, 957–960. [Google Scholar] [CrossRef]
- Radia, N.D.; Kamona, S.M.H.; Jasem, H.; Abass, R.R.; Izzat, S.E.; Ali, M.S.; Ghafel, S.T.; Am, A. Role of Hydrogel and Study of its High-Efficiency to Removal Streptomycin drug from Aqueous Solutions. Int. J. Pharm. Qual. Assur. 2022, 13, 160–163. [Google Scholar]
- Naderi, P.; Kabiri, K.; Jahanmardi, R.; Zohuriaan-Mehr, M.J. Preparation of itaconic acid bio-based cross-linkers for hydrogels. J. Macromol. Sci. Part A 2022, 12, 12–18. [Google Scholar] [CrossRef]
- Thakur, S.; Chaudhary, J.; Thakur, A.; Gunduz, O.; Alsanie, W.F.; Makatsoris, C.; Thakur, V.K. Highly efficient poly(acrylic acid-co-aniline) grafted itaconic acid hydrogel: Application in water retention and adsorption of rhodamine B dye for a sustainable environment. Chemosphere 2022, 303, 134917. [Google Scholar] [CrossRef]
- Wared, S.H.H.; Radia, N.D. Synthesis and characterization of sodium alginate-g-polyacrylic acid hydrogel and its application for crystal violet dye adsorption. Int. J. Drug Deliv. Technol. 2021, 11, 556–565. [Google Scholar]
- Thakur, S. Synthesis, characterization and adsorption studies of an acrylic acid-grafted sodium alginate-based TiO2 hydrogel nanocomposite. Adsorpt. Sci. Technol. 2018, 36, 458–477. [Google Scholar] [CrossRef]
- Alkaim, A.F.; Ajobree, A.M. White marble as an alternative surface for removal of toxic dyes (Methylene blue) from Aqueous solutions. Int. J. Adv. Sci. Technol. 2020, 29, 5470–5479. [Google Scholar]
- Karam, F.F.; Kadhim, M.I.; Alkaim, A.F. Optimal conditions for synthesis of 1, 4-naphthaquinone by photocatalytic oxidation of naphthalene in closed system reactor. Int. J. Chem. Sci. 2015, 13, 650–660. [Google Scholar]
- Al-Gubury, H.Y.; Fairooz, N.Y.; Aljeboree, A.M.; Alqaraguly, M.B.; Alkaim, A.F. Photocatalytic degradation n-undecane using coupled ZnO-Co2O3. Int. J. Chem. Sci. 2015, 13, 863–874. [Google Scholar]
- Abd, F.G.; Mohsen, L.Y.; Al Shalah, L.A.M.; Alkaim, A.F. Silver nanoparticles synthesized by using Pseudomonas aeruginosa synergistically act with antibiotic. Asian J. Microbiol. Biotechnol. Environ. Sci. 2018, 20, S50–S52. [Google Scholar]
- Alabdali, Y.A.J.; Wali, H.F.; Alkaim, A.F. ZnO nanoparticles activity against the virulence gene of Pseudomonas aeruginosa isolated from patients with burn wounds infection in Al Muthanna population. Ann. Trop. Med. Public Health 2020, 23, 1032–1041. [Google Scholar] [CrossRef]
- Aljeboree, A.M.; Alkaim, A.F. Comparative removal of three textile dyes from aqueous solutions by adsorption: As a model (corn-cob source waste) of plants role in environmental enhancement. Plant Arch. 2019, 19, 1613–1620. [Google Scholar]
- Aljeboree, A.M.; Hussein, F.H.; Alkaim, A.F. Removal of textile dye (methylene blue mb) from aqueous solution by activated carbon as a model (corn-cob source waste of plant): As a model of environmental enhancement. Plant Arch. 2019, 19, 906–909. [Google Scholar]
- Bader, A.T.; Aljeboree, A.M.; Alkaim, A.F. Removal of Methyl Violet (MV) from aqueous solutions by adsorption using activated carbon from pine husks (plant waste sources). Plant Arch. 2019, 19, 898–901. [Google Scholar]
- Jawad, M.A.; Kadhim, A.J.; Radia, N.D. Role of sodium alginate-g-poly (Acrylic acid-fumaric acid) hydrogel for removal of pharmaceutical paracetamol from aqueous solutions by adsorption. Int. J. Pharm. Qual. Assur. 2021, 12, 202–205. [Google Scholar]
- Hammadi, A.H.; Habeeb, S.A.; Al-Jibouri, L.F.; Hussien, F.H. Synthesis, Characterization and Biological Activity of Zinc Oxide Nanoparticles (ZnO NPs). Syst. Rev. Pharm. 2020, 11, 431–439. [Google Scholar]
- Adam, A.M.A. Structural, thermal, morphological and biological studies of proton-transfer complexes formed from 4-aminoantipyrine with quinol and picric acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 104, 1–13. [Google Scholar] [CrossRef]
- Mundra, R.V.; Wu, X.; Sauer, J.; Dordick, J.S.; Kane, R.S. Nanotubes in biological applications. Curr. Opin. Biotechnol. 2014, 28, 25–32. [Google Scholar] [CrossRef]
- Mukha, I.P.; Eremenko, A.M.; Smirnova, N.P.; Mikhienkova, A.I.; Korchak, G.I.; Gorchev, V.F.; Chunikhin, A.Y. Antimicrobial activity of stable silver nanoparticles of a certain size. Appl. Biochem. Microbiol. 2019, 49, 199–206. [Google Scholar] [CrossRef]
- He, J.; Shi, M.; Liang, Y.; Guo, B. Conductive adhesive self-healing nanocomposite hydrogel wound dressing for photothermal therapy of infected full-thickness skin wounds. Chem. Eng. J. 2020, 394, 124888. [Google Scholar] [CrossRef]
- Liu, Y.; Song, S.; Liu, S.; Zhu, X.; Wang, P. Application of Nanomaterial in Hydrogels Related to Wound Healing. Adv. Nanocomposite Hydrogels Tissue Eng. 2022, 12, 4656037. [Google Scholar] [CrossRef]
- Xu, D.; Wang, S.; Li, B.; Qi, M.; Feng, R.; Li, Q.; Zhang, H.; Chen, H.; Kong, M.G. Effects of Plasma-Activated Water on Skin Wound Healing in Mice. Microorganisms 2020, 7, 1091. [Google Scholar] [CrossRef]
- Vågesjö, E.; Öhnstedt, E.; Mortier, A.; Lofton, H.; Huss, F.; Proost, P.; Roos, S.; Phillipson, M. Accelerated wound healing in mice by on-site production and delivery of CXCL12 by transformed lactic acid bacteria. Natl. Cent. Biotechnol. Inf. 2018, 7, 1–9. [Google Scholar] [CrossRef]
- Afrin, S.; Shahruzzaman, M.; Haque, P.; Islam, M.S.; Hossain, S.; Rashid, T.U.; Ahmed, T.; Takafuji, M.; Rahman, M.M. Advanced CNC/PEG/PDMAA Semi-IPN Hydrogel for Drug Delivery Management in Wound Healing. Gels 2022, 8, 340. [Google Scholar] [CrossRef] [PubMed]


| Surface Physical Parameters | Value |
|---|---|
| BET Surface Area (m g) | 19.225 |
| Langmuir Surface Area (m g) | 105.36 |
| Micro Pore Surface Area (m g) | 8.641 |
| Cumulative Pore Volume (cm g) | 1.936 |
| Micro Pore Volume (cm g) | 0.0037 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljeboree, A.M.; Hasan, I.T.; Jwaid, M.M.; Dawood, A.H.; Jawad, M.A. Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite. Eng. Proc. 2023, 59, 219. https://doi.org/10.3390/engproc2023059219
Aljeboree AM, Hasan IT, Jwaid MM, Dawood AH, Jawad MA. Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite. Engineering Proceedings. 2023; 59(1):219. https://doi.org/10.3390/engproc2023059219
Chicago/Turabian StyleAljeboree, Aseel M., Ishraq T. Hasan, Maher Mohammed Jwaid, Ashour H. Dawood, and Mohammed Abed Jawad. 2023. "Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite" Engineering Proceedings 59, no. 1: 219. https://doi.org/10.3390/engproc2023059219
APA StyleAljeboree, A. M., Hasan, I. T., Jwaid, M. M., Dawood, A. H., & Jawad, M. A. (2023). Enhanced Drug Delivery and Wound Healing with Novel Hydrogel Nanocomposite. Engineering Proceedings, 59(1), 219. https://doi.org/10.3390/engproc2023059219





