1. Introduction
The overuse of antibiotics has afflicted society with an epidemic of antibiotic-recalcitrant infections and subsequent harmful inflammation, resulting in lowered immunity, cellular damage, secondary infections, and intestinal microbial imbalances [
1]. Thus, the search for alternative antibacterial and noninflammatory agents is urgently needed. As an essential protectiveness organ, the intestinal tract is subjected to potential pathogens and consequent inflammation, which can cause diarrhea, slow growth, and even death in commercially raised animals like chickens if the intestinal flora becomes imbalanced [
2]. Because of this risk, probiotic preparations have attracted considerable research attention because they could be used to reestablish the intestinal flora balance and restore immunity quickly.
Probiotic preparations are formulations containing live microorganisms that can modify the composition of the intestinal macrobiotic within the body, thus improving the organism’s health, fostering better digestion and absorption and restoring a healthy immune response [
3]. Abaya-Loyola et al. [
4] found that Bacillus coagulant (
B. coagulant) can improve physical fitness by enhancing the body’s digestion and absorption of nutrients. This is because
B. coagulant can scavenge free radicals and boost the activity of antioxidant enzymes in the host, resulting in an antioxidant effect.
B. coagulant can synthesize anti-inflammatory cytokines as immune regulatory agents by activating the CONFAB signaling pathway through direct interaction between the bacterial cell wall’s outer layer and immune cell receptors [
5]. Our previous studies showed that BC-HYI improved heat resistance and chicken feed utilization and reduced LPS-induced inflammatory damage by activating the TLR4/NF-AB-signaling pathway to promote cytokine production [
6]. However, the mechanism of BC-HYI’s reduction in oxidative stress (OS) in laying chickens has yet to be elucidated.
Reactive oxygen species (ROS) is a toxic substance produced during the body’s metabolic process. According to [
7], accumulating excessive amounts of these substances leads to oxidative stress (OS), which disrupts the balance within cells and results in mitochondria dysfunction. Fao et al. showed that the presence of heavy metals like Cr(VI) could increase the level of cellular ROS, resulting in abnormal mitochondrial membrane potential (MMP) and decreased catalepsy activity [
8]. We evaluated cellular oxidative stress levels by detecting reactive oxygen species (ROS) content combined with antioxidant enzyme results in the article.
Wei et al. found that ROS content increased in diabetic rats, and the abundance of Firmicutes and Bacteroides in the gut microbiota was higher [
9]. High levels of ROS and increased abundance of Enterococcus faecalis were found during the development of inflammatory bowel disease (IBD) [
10]. Bacterial lipopolysaccharide (LPS) increased ROS and damaged chicken embryonic lung cells by activating NF-κB and Nrf2 signaling pathways [
11]. The probiotic,
B. coagulans, has been reported to have multiple effects in animals, including shifting the intestinal population of microbes towards the beneficial types, accelerating growth, promoting healthy immune responses, and inhibiting pathogen invasion [
12]. In a dextran sulfate sodium (DSS)-induced mouse model of IBD, treatment with
B. coagulans significantly modulated the cytokines, IL-4, IL-6, IL-8 and IL-10, upregulated the expression of claudin and mucin, and promoted repair of the intestinal barrier [
13]. Furthermore, 200 mg/kg of
B. coagulans improved growth performance and increased the activity of antioxidant enzymes and the abundance of intestinal bifidobacteria and bacilli in pigs [
14]. Laying hens administered with
B. coagulans R11 showed increased total antioxidant capacity (T-AOC) and superoxide dismutase (SOD) activity and reduced the MDA concentration [
15].
B. coagulans also reduced the prevalence of harmful bacteria in cows with diarrhea and prevented oxidative damage [
16]; however, the mechanism by which gut microbiota lower OS has not been worked out in detail. In our experiments, the beneficial effect of BC-HYI on the antioxidant stress of laying chicks was evaluated in vivo by detecting changes in intestinal tissue structure and intestinal microbiota quantity combined with the contents of antioxidant enzymes in blood and tissue. In in vitro experiments, the antioxidant mechanism of BC-HYI was explored by detecting the amount of reactive oxygen species (ROS) combined with the content of antioxidant enzymes in cells and its impact on gene and protein expression.
The normal growth of laying chickens during the rearing period is essential to ensure their economic value for later egg laying. Laying chickens are susceptible to OS during husbandry resulting in decreased immunity, diarrhea, vomiting, weight loss, and even death [
17]. If BC-HYI can change the metabolites in the intestine by increasing the content of beneficial bacteria, it can even change the expression of genes and proteins through this pathway to increase antioxidant capacity. First of all, BC-HYI is a newly discovered strain of Bacillus coagulans with powerful functions. We comprehensively illustrated the role of BC-HYI through in vitro and in vivo experiments. We used functional prediction to combine intestinal microbiota functions and signaling pathways, which serves as a basis for subsequent exploration. Therefore, for laying chickens, feeding probiotic BC-HYI in advance can not only improve immunity, but also reduce mortality and increase survival rate when oxidative stress occurs. This provides some data support for the research and development of antibiotic alternatives and ensuring the economic benefits of animal husbandry. Therefore, this study aimed to explore the research goal of this investigation, which was to ascertain the ways in which BC-HYI balanced the gut macrobiotic and reduced LPS-induced oxidative stress. In this paper, we presented data on the functional mechanism underlying the use of BC-HYI in promoting the steady state of gut macrobiotic and reducing OS in laying chickens.
2. Materials and Methods
2.1. Bacteria Strains and Cultivation
B. coagulant HYI (BC-HY1) was obtained from the Chinese General Microorganism Culture Collection Center which produced exopolysaccharides (CGMCC No. 24423). B. coagulant HYI was inoculated into an LBS broth medium (Boombox Biotechnology Co., Ltd., Beijing, China) and grown at 37 °C in a shaker incubator (210 rpm) for 22 h. The pH was kept at 6.2. Lactobacillus rhamnosus GG (LGG) was obtained from Hebei Agricultural University and cultured statically at 37 °C in a D Man-Rosa-Sharpe (MRS) medium (Boombox Biotechnology Co., Ltd., Beijing, China) to stationary phase. The purity and identity of the strains were checked, and bacterial liters were determined by serial dilution, spreading on agar plates, and colony counting. Serial dilutions were performed to obtain the desired inoculate (1.0 × 105, 1.0 × 106, 1.0 × 107 and 1.0 × 108 CFU/L).
2.2. Animals and Sample Collection
A total of 288 one-day-old laying chickens were donated by the Beijing Sadhu Yuk ou Poultry Industry Co., Ltd. (Beijing, China). After a 10-day adaptation, the animals were split into five groups and assigned randomly to different feeding conditions. (1) The basal diet was administered to the control (CON) group; (2) the LPS group, which received basal diet feeding; (3) B6 group, which received basal diet feeding supplemented with 1.0 × 106 CFU/mL of BC-HY1; (4) the B7 group, which received basal diet feeding supplemented with 1.0 × 107 CFU/mL of BC-HY1; (5) the B8 group, which received basal diet feeding supplemented with 1.0 × 108 CFU/mL of BC HY1. On Day 28, Groups 2–5 received 2 mg/kg of LPS administered by gavage. Blood, intestinal and fecal samples were collected within 6 h of LPS gavage, and the samples were collected and stored at −80 °C for future analysis.
2.3. Histological Analysis
Upon collection, intestinal tissue samples from chickens were immediately fixed in 10% neutral buffered formalin to preserve their structural integrity. The paraffin-embedded samples were thinly sliced into sections measuring approximately 5 μm thick to obtain thin sections suitable for microscopic examination. To analyze the cellular morphology and tissue composition, thin sections of the intestinal tissue were prepared and subjected to local staining using the widely employed hematoxylin and eosin (H&E) technique. Furthermore, the stained sections were examined under an IX71 visible light microscope (Olympus, Japan). This microscope provides high-resolution imaging capabilities, allowing for detailed observation and analysis of the stained tissue sections.
2.4. High-Throughput Sequencing Was Performed on the 16S rRNA Genes
To analyze the microbial composition of fecal samples, DNA extraction was performed using the Fast DNA
® SPIN for salt kit (MP Biomedicals, Solon, OH, USA) following the recommended protocol. To target bacterial 16S rRNA genes, the V3–V4 region was selected for amplification. To achieve this, specific primers were used, namely 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′). These primers were selected based on their ability to amplify the target region, allowing a comprehensive assessment of bacterial communities in fecal samples. After PCR amplification, the resulting product was purified using a preparative DNA gel extraction kit (Oxygen BioSciences, Union City, CA, USA). The libraries were sequenced on the Illumina MiSeq PE300 platform, a widely used NGS system known for its high-throughput sequencing capabilities. In order to analyze the obtained sequencing data, the online service provided by the Majorbio cloud platform (
WWW.Majorbio.com) (accessed on 15 March 2023) was used. Lease analysis, a statistical method commonly used in ecological research, was used to further study the relationship between microbial taxa and environmental factors.
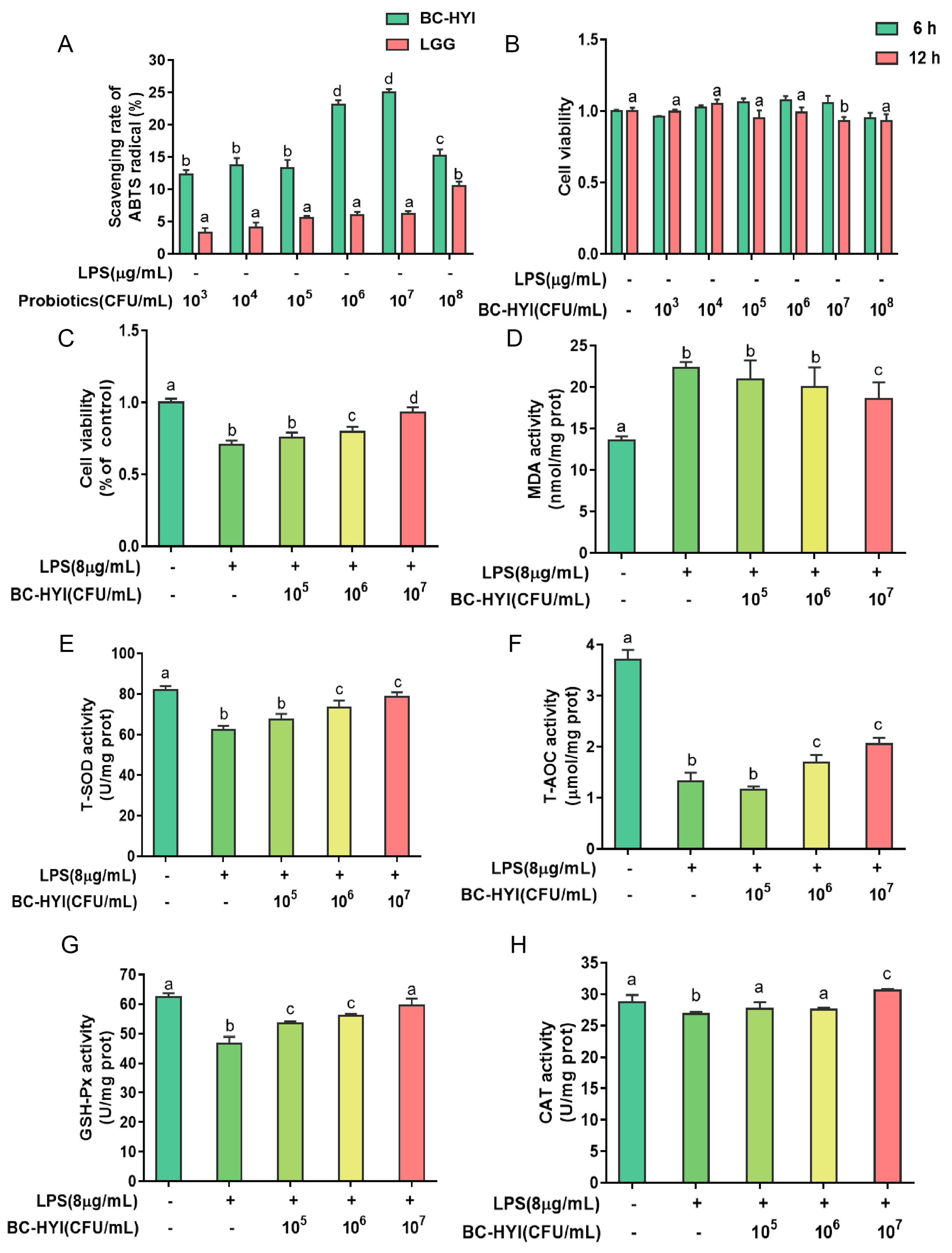
2.5. ABTS Free Radical Scavenging Ability
According to the study protocol of Jo et al. [
18], we used a similar method to that of Kim et al. (2019) to assess the ABTS radical scavenging activity of the samples. In summary, bacterial cell suspensions, treated with varying BC-HYI or LGG concentrations, were combined with a diluted ABTS solution (1 L). After thoroughly mixing and preparing the mixture, it was incubated in a dark environment at room temperature for 6 min.
2.6. Cell Lines and Culture Conditions
The DF-1 cell line, derived from glass fibers and obtained from ATCC (SCRC-1040), has been widely used in various studies. In this particular experiment, cells were cultured in a DF-1 medium supplemented with 13% fetal bovine serum (FBS), which provides essential nutrients and growth factors for cell proliferation and maintenance. The temperature of the incubator was maintained at 37 °C, which is the ideal temperature for the growth of avian cells. Additionally, the incubator provides a humidity level of 90% to prevent cell dehydration and promote a suitable cellular environment. To maintain proper atmospheric composition, the incubator maintains a 5% CO2 concentration, which is essential to maintaining the pH balance of the culture medium. To prevent microbial contamination and ensure the sterility of cell cultures, 1% (v/v) penicillin DF-1 medium was supplemented with streptomycin (Solarbio Science and Technology Co., Ltd., Beijing, China). The culture medium was refreshed every 48 h to provide fresh nutrients and remove metabolic waste products produced by the cells.
2.7. Cell Treatments
To evaluate the antioxidant properties of BC-HYI, DF-1 cells were categorized into different groups: Control (CON), LPS treatment (LPS), CB5 (1.0 × 105 BC-HYI), CB6 (1.0 × 106 BC-HYI), and CB7 (1.0 × 107 BC-HYI). DF-1 cells were seeded at a density of 1.2 × 106 cells/L (100 µL per well) in a 96-well plate and incubated for 24 h. The control and LPS groups were cultured in DMEM devoid of FBS and antibiotics for 6 h. In the BC-HYI + LPS group, cells were co-cultured with BC-HYI for 6 h, followed by washing with PBS to remove the macrobiotics. Then, DMEM without FBS containing LPS (at the concentration determined by the CCK-8 assay) was added for 6 h to induce oxidative stress, excluding the control group.
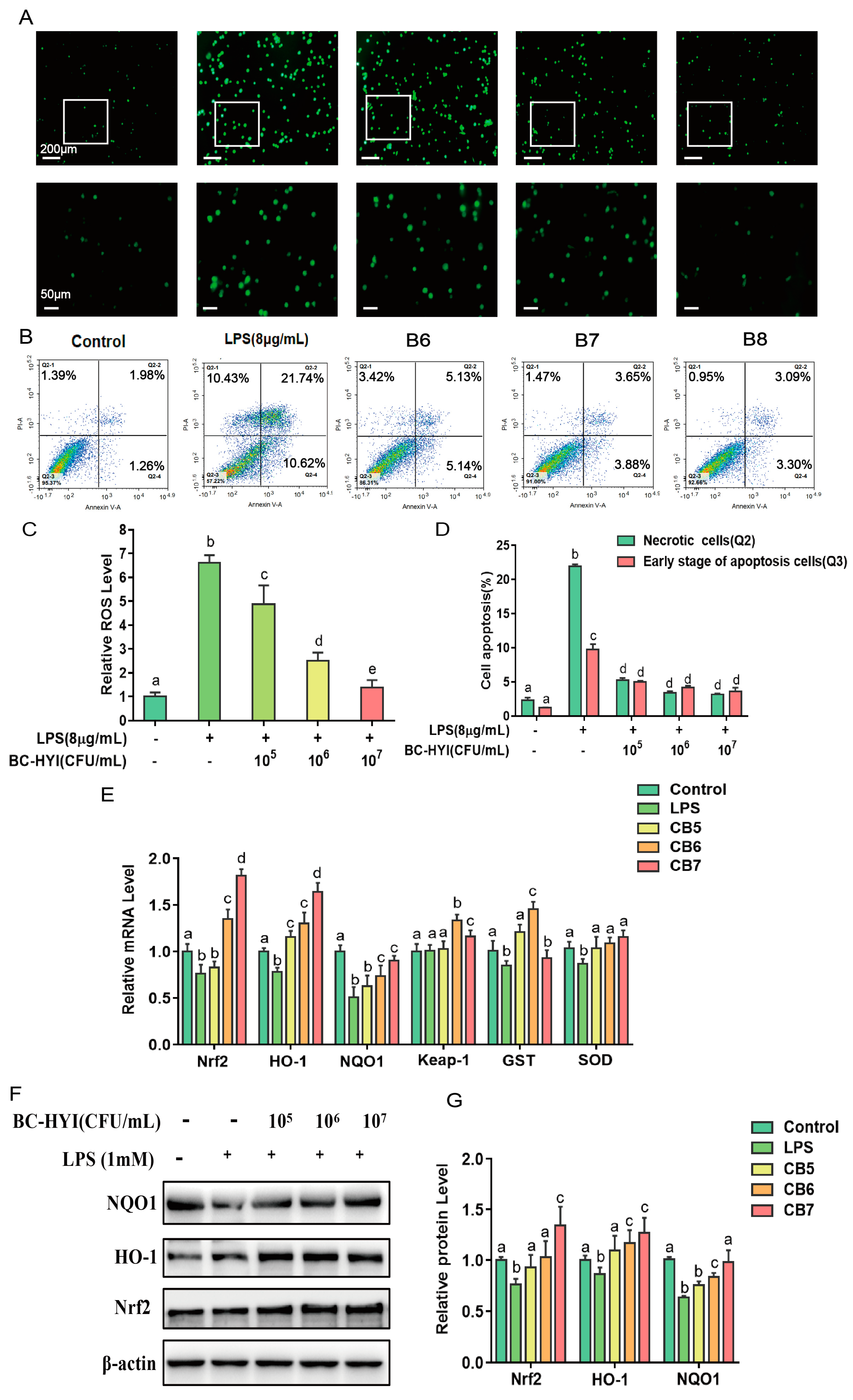
2.8. Reactive Oxygen Species (ROS) Assay
The levels of reactive oxygen species (ROS) in DF-1 cells were determined utilizing a fluorescent dye-based assay employing 2′,7′-dichlorofluorescein acetate (DCFH-DA, D2215-25 mg, Landholder, Beijing, China). DF-1 cells were cultivated in a 6-well plate, with each well containing approximately 1.2 × 106 cells, and the culture medium was replenished upon reaching 80% confluence. After the respective treatments, the cell culture medium was aspirated, and afterward, the cells underwent a single phosphate-buffered saline (PBS) wash.
Following the initial steps of the experiment, the DF-1 cells were subjected to further treatment to assess the level of reactive oxygen species (ROS) present within the cellular environment. To accomplish this, the cells were treated with a solution of 2′,7′-dichlorofluorescein diacetate (DCFH-DA), which is a non-fluorescent compound that can be converted to a fluorescent form in the presence of ROS.
The DCFH-DA solution, prepared at 10 µM and diluted in serum-free medium, was added to the cells and incubated at 37 °C for 20 min. During this incubation period, the DCFH-DA molecules entered the cells and became trapped within the cytoplasm.
After the incubation period, the DCFH-DA solution was carefully discarded, and the cells underwent three consecutive washes with a fresh supply of serum-free medium. This washing step was essential to remove any residual DCFH-DA solution and ensure accurate measurement of the ROS-induced fluorescence.
It is important to note that the initially non-fluorescent DCFH-DA molecule can undergo oxidation in the presence of ROS, generating a fluorescent compound known as 2′,7′-dichlorofluorescein (DCF). The formation of DCF fluorescence is directly proportional to the levels of ROS present in the cellular environment.
To visualize and capture images of the fluorescence, a fluorescence microscope manufactured by Olympus Corporation (Olympus, Japan) was utilized. The microscope was equipped with appropriate filters, including an excitation filter with a wavelength of 488 nm and an emission filter with a wavelength of 525 nm. These filters were specifically chosen to maximize the detection and visualization of the DCF fluorescence.
Under the fluorescence microscope, the DF-1 cells were observed, and images were captured using suitable magnification settings. The intensity of the fluorescence observed in the captured images directly corresponded to the levels of ROS within the cells. Higher fluorescence intensity indicated higher levels of ROS production, while lower intensity suggested lower ROS levels.
This fluorescence microscopy-based method allowed for qualitatively assessing ROS levels in the DF-1 cells. By analyzing the captured images, researchers could gain insights into the cellular oxidative stress status and evaluate the effects of different treatments or interventions on ROS generation.
In conclusion, treating DF-1 cells with the DCFH-DA solution followed by fluorescence microscopy enabled the measurement and visualization of ROS levels within the cells. This technique provided valuable information regarding cellular redox status and contributed to a better understanding of the role of ROS in cellular processes and diseases.
2.9. Antioxidant Enzyme Activity and MDA Content in Jejunum Samples, Serum, and Cultured Cells
DF-1 cells were seeded at a density of 1.2 × 10
6 cells/mL in a 6-well plate and incubated for 24 h. The jejunum samples were added to a tissue lysis solution (Nanjing Jiancheng) and macerated. After the abovementioned treatment, the superoxide dismutase (SOD), catalase (CAT), Glutathione peroxidase (GSH-Px), total antioxidant capacity (T-AOC) and malondialdehyde (MDA) assay kits (Nanjing Jiancheng Biological Engineering Research Institute Co., Ltd., Nanjing, China) were used to measure the content in cells, serum and jejunum samples [
19]. The BCA assay kit (Nanjing Jiancheng) was used to determine the protein content of cells and tissues.
2.10. Measurement of Cell Apoptosis
To quantify the population of apoptotic cells, we employed the Annex V apoptosis detection kit I (BD Biosciences, San Diego, CA, USA) labeled with FITC, following the instructions provided by the manufacturer. After rinsing the cells twice with chilled PBS, we resuspended them in a 1X binding buffer at a 1 × 106 cell/L density. Next, 1 × 105 cells in a 100 µL aliquot were transferred to a 5 mL culture tube, and 5 µL of Annexin V and 5 µL of propidium iodide were added. Gentle vortexing ensured thorough mixing, and the mixture was then incubated at room temperature (25 °C), shielded from light, for 15 min. Following the incubation, the cells were analyzed using a flow cytometer (BD LSRII, San Diego, CA, USA) within 1 h. The acquired flow cytometry data were processed using BD Facsimile software version 8.0. The numbers of late apoptotic and early apoptotic cells were recorded in the Q2 and Q3 quadrants, respectively.
2.11. Real-Time Quantitative Polymerase Chain Reaction
Total RNA extraction and cDNA synthesis using the SteadyPure Universal RNA Extraction Kit (Accurate Biology, Changsha, China) and the Pro Taq HS SYBR Green premixed qPCR kit (Accurate Biology, Changsha, China) were performed following the manufacturer’s instructions. PCR primer sequences for the gallus genes were designed and selected by Primer3Plus and NCBI Website (
https://www.ncbi.nlm.nih.gov (accessed on 18 September 2023)) as presented in
Table S1. GAPDH as housekeeping gene was used to normalize target gene transcript levels. Real-time PCR was performed using Premix Ex TaqTM with SYBR Green (Accurate Biology, Changsha, China) and ABI Stepone Real-Time PCR System 7500 Fast Real-Time PCR System (Applied Biosystems, Carlsbad, CA, USA). The thermocycle protocol included 30 s at 95 °C followed by 40 cycles of 5 s denaturation at 95 °C, 30 s annealing/extension at 60 °C, and then a final melting curve analysis to monitor the purity of the PCR product. The 2−ΔΔCt method was used to estimate messenger RNA (mRNA) abundance. Relative gene expression levels were normalized by eukaryotic reference gene GAPDH.
2.12. Western Immunoblot Analysis
To extract total cellular protein from DF-1 cells and jejunum tissue, we utilized RIPA dialysis buffer obtained from Nanjing Kanchenjunga Bioengineering Institute in Nanjing, China. The protein concentration in the supernatant was determined using a BCA protein assay kit provided by the same institute. Equal amounts of protein (30 µg) were taken from each sample and separated by 10% SDS-PAGE. Subsequently, the proteins were transferred to a polyvinylidene fluoride (PVDF) membrane (Immobile, Shanghai, China) after blocking the membrane. After that, the membranes were incubated overnight at 4 °C with specific primary antibodies, followed by a 1 h incubation with secondary antibodies at room temperature. Based on the manufacturer’s instructions, bioluminescence was detected using an ECL luminescence reagent. The internal control alpha-tubulin (α-tubulin) was used to normalize the relative protein expression. Quantitative analysis of fluorescence intensity was conducted using Image software.
2.13. Statistical Analyses
Data shown in the study were obtained from at least three independent experiments and all data in different experimental groups were expressed as the mean ± SD. Statistical analyses were performed using GraphPad Prism version 7.0 (San Diego, CA, USA). The flow cytometry data were analyzed by BD FACSDiva Software version 8.0. One-way analysis of variance (ANOVA) followed by a least significant difference multiple comparison test was used. Values of p < 0.05 were considered statistically significant while p < 0.01 was considered a trend.
4. Discussion
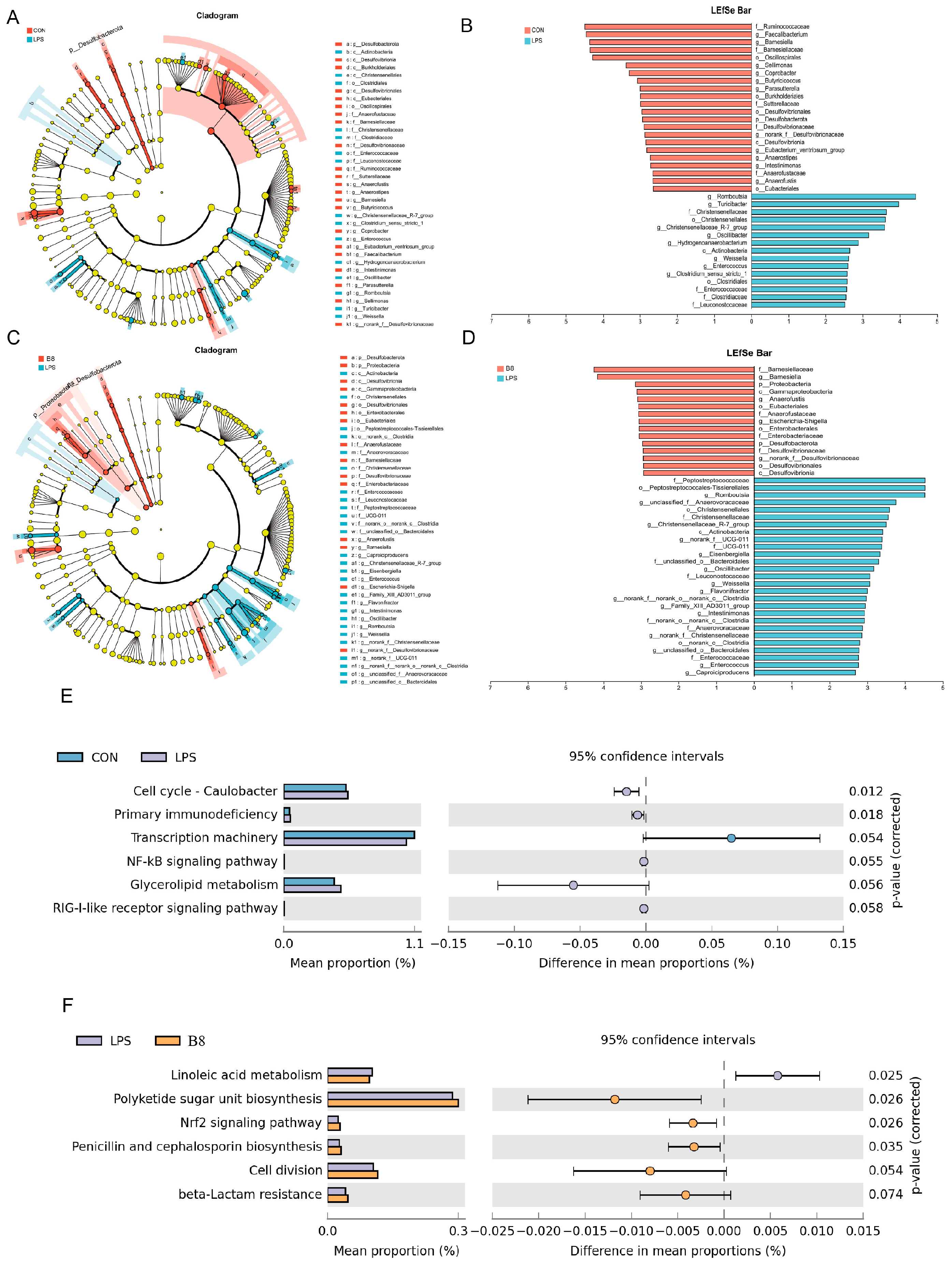
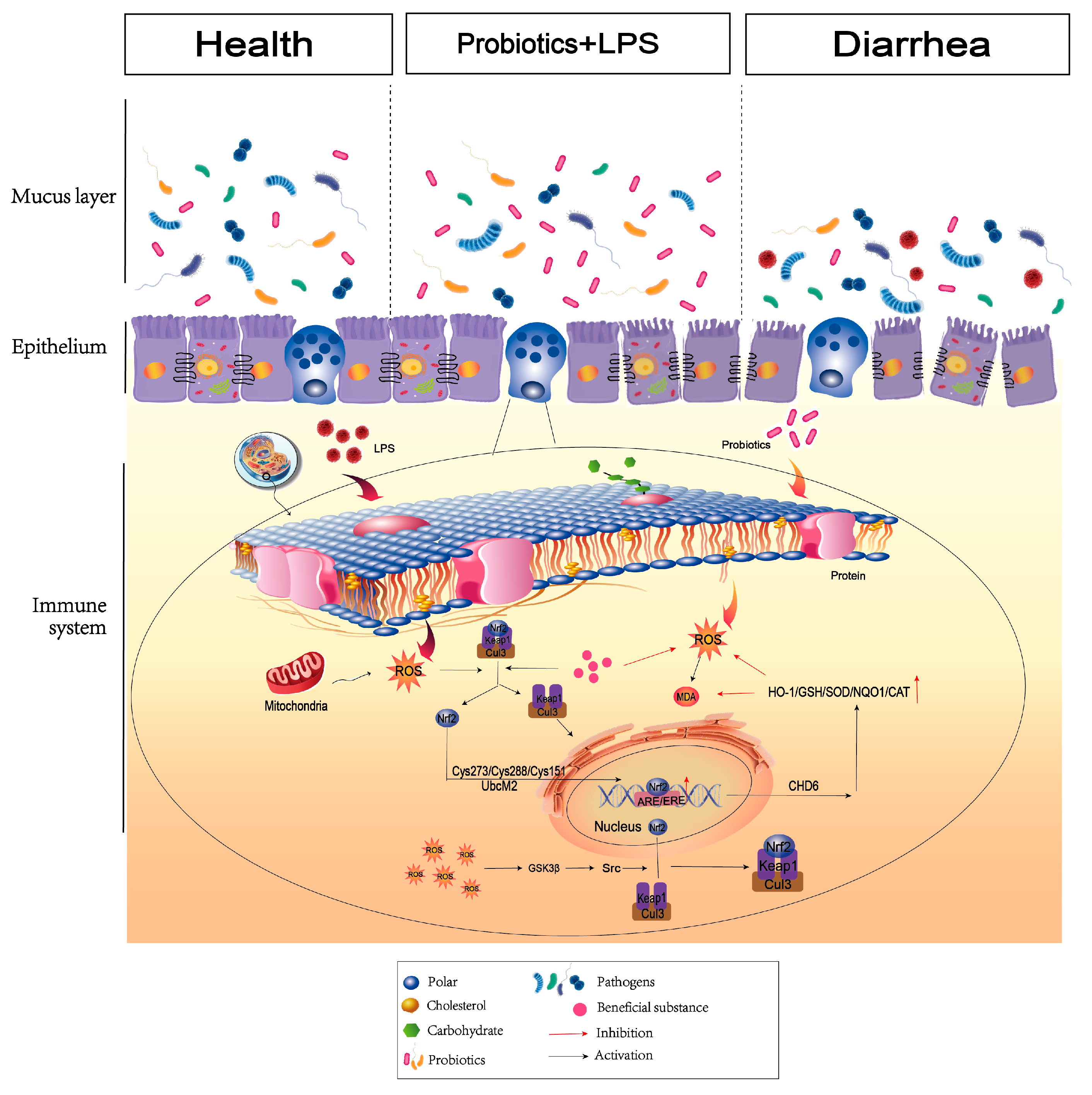
Dietary supplementation of probiotic strategies helps control the development of enteritis by altering intestinal epithelial integrity and microbiota structure. Our results clearly indicated that supplementation of BC-HYI reduces LPS damage to maintain the intestinal structural integrity in laying chickens by increasing the activity of antioxidant enzymes Pasteurella and Lactobacillus and abundance of beneficial bacteria in Faecalibacterium, as well as reduced abundance of harmful bacteria in Bacteroides and Clostridium. The results of the article indicate that BC-HYI can change the intestinal environment by increasing the content of beneficial bacteria, and may also affect the expression of genes and proteins in the Nrf2 signaling pathway to increase antioxidant capacity.
With the development of colitis, the accompanying inflammation generates a large amount of ROS, and the oxidative stress causes damage to intestinal epithelial cells. In the present study, LPS-induced oxidative stress is similar to the pathological characteristics of the stress responses in the body to curative colitis, as well as LPS-induced oxidative stress commonly adopted in the process of drug discovery [
20]. It is well known that effective scavenging of hydroxide radicals is beneficial for living organisms [
21]. BC-HYI showed higher free radical scavenging ability than LGG, especially in the CB6 and CB7 groups. These results are consistent with previous antioxidant studies of B. coagulant on HT-29 cells [
22]. In addition, York-Duran et al. [
23] reported that the scavenging of ROS includes both enzymatic and non-enzymatic systems.
The activity of antioxidant enzymes was increased by BC-HYI with a concomitant reversal of the apoptotic response induced by LPS.ROS. It is an active signaling molecule which plays a regulatory role in oxidative stress by regulating oxidative indicators, increasing formaldehyde (MDA) content and decreasing the expression of antioxidant enzyme genes [
24]. Researchers also found that Se-enriched Bacillus subtitles significantly reduced MDA content and increased SOD, CAT, and GPX activities in oxidative stressed fish [
25]. Correspondingly, Bacillus coagulant T242 increased T-SOD and CAT activities by inhibiting oxygen radical production and activating the Nrf2 signaling pathway in HT-29 cells [
26], consistent with our findings. In addition, Chaos et al. showed that Bacillus coagulant 12 increased the expression of Nrf2, HO-1, AKT, P-ACCT, and Bcl-2 proteins, thereby reversing antitoxin-induced apoptosis in mouse kidney cells [
27]. This result may be due to the activation of antioxidant stress response by
B. coagulant, mainly through Nrf2 and MAPK signaling pathways.
Consistent with the findings of Kai et al., the present study found that BC-HYI reduced the number of harmful bacteria while increasing the relative abundance of beneficial bacteria [
17] It has been reported that OS was induced in mice by whole-body radiation, the ROS content was significantly increased, the antioxidant enzyme activity was decreased, the numbers of Bactericide and
Streptococcus fatalistic were increased, and the abundance of
Arachnophobia was decreased in the gut [
28]. Similarly, studies by Wu et al. demonstrated that treatment with 2 × 10
7 CFU/L
B. coagulant increased the abundance of lactobacillus and decreased cloistral in porcine colon [
29]. In our previous investigations, it was demonstrated that BC-HYI has the capability to generate organic acids, including lactic acid, satyric acid, and propionic acid [
30]. These organic acids play a crucial role in connecting the gut macrobiotic with various diseases. For instance, Saks et al. [
31] revealed that Bacillus coagulant SANK 70258 could effectively treat colitis by increasing the abundance of Arachnophobia and the levels of satyric acid in the gut. Based on our current findings, we propose that BC-HYI modifies the composition of the gut macrobiotic by producing metabolites such as short-chain fatty acids and interactions. Moreover, BC-HYI regulates the Erna and protein expression levels of TLR4, reducing intestinal permeability and improving LPS-induced colitis and overall gut health. These results are consistent with the findings reported by [
32].
In conclusion, this study highlights the efficacy of BC-HYI in reducing LPS-induced oxidative damage and reducing its severity in laying chickens. BC-HYI reduced mortality by enhancing the abundance of beneficial intestinal microbes, maintaining intestinal homeostasis, and relieving oxidative stress caused by environmental challenges. Thus, BC-HYI has the potential to be a safe natural antioxidant in feed and also can be used to develop new storage-stable probiotics aimed at improving intestinal health, providing more possibilities for replacing antibiotics. However, the mechanism of Bacillus coagulant antioxidant activity needs to be further studied to provide a theoretical basis for research leading to improved production, which is of great value to the livestock and poultry breeding industry.