Primary Characterization of a Life-Cycle Mutant akasusabi of the Red Alga Neopyropia yezoensis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation of a Mutant
2.2. Algal Strain, Culture Conditions and Calculation of the Growth Rate
2.3. Quantitative Measurement of Photosynthetic Pigments
2.4. Stress Treatments
2.5. Statistics Analysis
3. Results
3.1. Increased Growth of aks Gametophytes
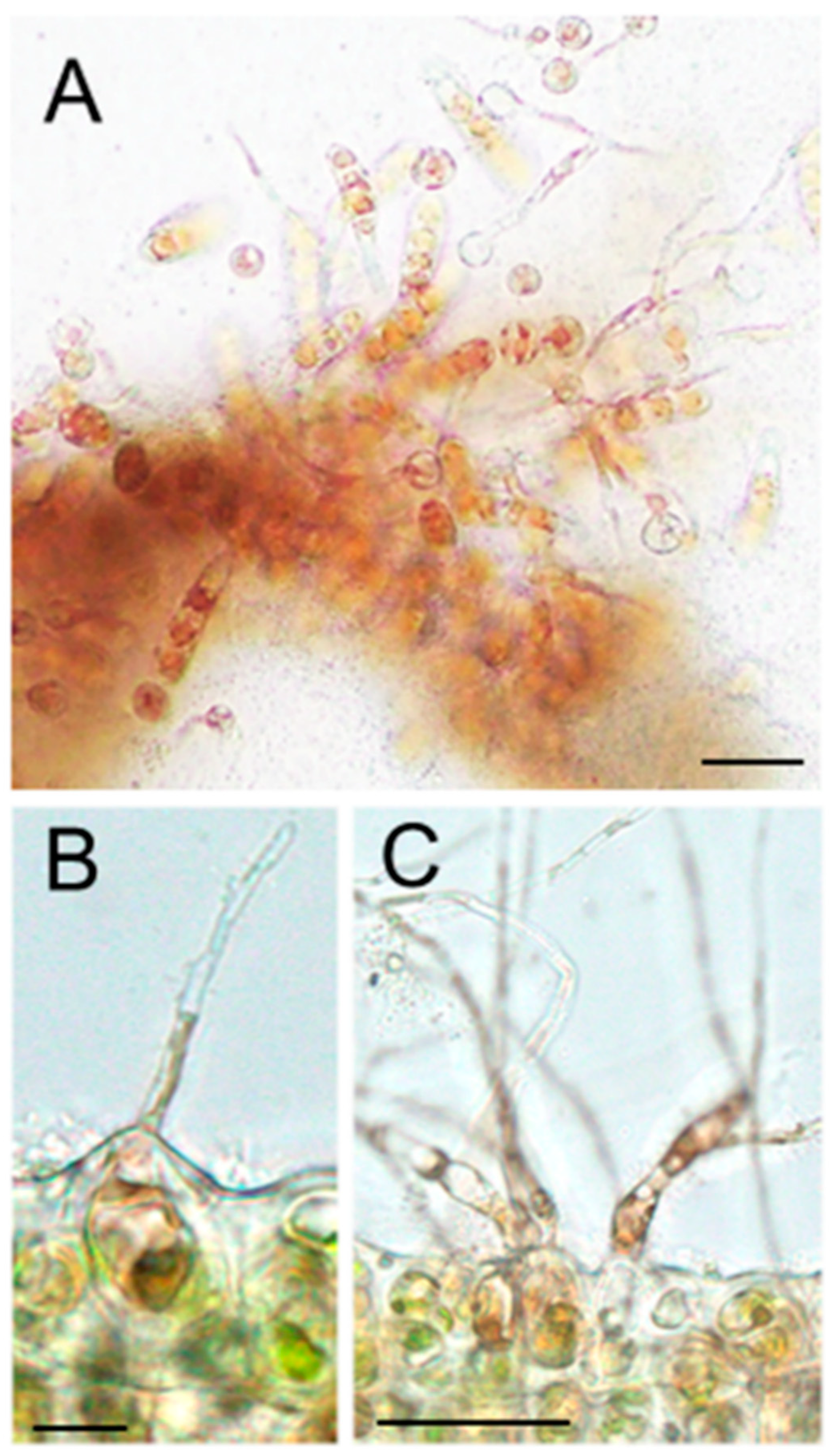
3.2. Sporophyte Production with Mitotic Defect in aks Carposporangia
3.3. High Spore Productivity after Wounding Stress in aks
3.4. Heat Stress Inducibility of Callus Formation in aks
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Friedman, W.E. One genome, two ontogenies. Science 2013, 339, 1045–1046. [Google Scholar] [CrossRef]
- Bowman, J.L.; Sakakibara, K.; Furumizu, C.; Dierschke, T. Evolution in the cycles of life. Annu. Rev. Genet. 2016, 50, 133–154. [Google Scholar] [CrossRef]
- Blouin, N.A.; Brodie, J.A.; Grossman, A.C.; Xu, P.; Brawley, S.H. Porphyra: A marine crop shaped by stress. Trends Plant Sci. 2011, 16, 29–37. [Google Scholar] [CrossRef]
- Takahashi, M.; Mikami, K. Oxidative stress promotes asexual reproduction and apogamy in the red seaweed Pyropia yezoensis. Front. Plant. Sci. 2017, 8, 62. [Google Scholar] [CrossRef] [Green Version]
- Drew, K.M. Conchocelis-phase in the life-history of Porphyra umbilicalis (L.) Kütz. Nature 1949, 164, 748–749. [Google Scholar] [CrossRef]
- Cock, J.M.; Godfroy, O.; Macaisne, N.; Peters, A.F.; Coelho, S.M. Evolution and regulation of complex life cycles: A brown algal perspective. Curr. Opin. Plant Biol. 2014, 17, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Bogaert, K.; Engelen, A.H.; Leliaert, F.; Roleda, M.Y.; de Clerck, O. Seaweed reproductive biology: Environmental and genetic controls. Bot. Mar. 2017, 60, 89–108. [Google Scholar] [CrossRef]
- Ma, J.H.; Miura, A. Observations of the nuclear division in the conchospores and their germlings in Porphyra yezoensis Ueda. Jpn. J. Phycol. 1984, 32, 373–378. [Google Scholar]
- Ohme, M.; Kunifuji, Y.; Miura, A. Cross experiments of the color mutants in Porphyra yezoensis Ueda. Jpn. J. Phycol. 1986, 34, 101–106. [Google Scholar]
- Burzycki, G.M.; Waaland, J.R. On the position of meiosis in the life history of Porphyra torta. Bot. Mar. 1987, 30, 5–10. [Google Scholar] [CrossRef]
- Ohme, M.; Miura, A. Tetrad analysis in conchospore germlings of Porphyra yezoensis (Rhodophyta, Bangiales). Plant Sci. 1988, 57, 135–140. [Google Scholar] [CrossRef]
- Tseng, C.K.; Sun, A. Studies on the alternation of the nuclear phases and chromosome numbers in the life history of some species of Porphyra from China. Bot. Mar. 1989, 32, 1–8. [Google Scholar] [CrossRef]
- Mitman, G.G.; van der Meer, J.P. Meiosis, blade development, and sex determination in Porphyra purpurea (Rhodophyta). J. Phycol. 1994, 30, 147–159. [Google Scholar] [CrossRef]
- Yan, X.; Li, L.; Aruga, Y. Genetic analysis of the position of meiosis in Porphyra haitanensis Chang et Zheng (Bangiales, Rhodophyta). J. Appl. Phycol. 2005, 17, 467–473. [Google Scholar] [CrossRef]
- Wang, J.; Dai, J.; Zhang, Y. Nuclear division of the vegetative cells, conchosporangial cells and conchospores of Porphyra yezoensis (Bangiales, Rhodophyta). Phycol. Res. 2006, 54, 201–207. [Google Scholar] [CrossRef]
- Shimizu, A.; Morishima, K.; Kobayashi, M.; Kunimoto, M.; Nakayama, I. Identification of Porphyra yezoensis (Rhodophyta) meiosis by DNA quantification using confocal laser scanning microscopy. J. Appl. Phycol. 2007, 20, 83–88. [Google Scholar] [CrossRef]
- Yan, X.; Huang, M. Identification of Porphyra haitanensis (Banglales, Rhodophyta) meiosis by simple sequence repeat markers. J. Phycol. 2010, 46, 982–986. [Google Scholar] [CrossRef]
- Strasburger, E. The periodic reduction of the number of the chromosomes in the life history of living organisms. Ann. Bot. 1984, 8, 281–316. [Google Scholar]
- Mehra, P.N.; Sulklyan, D.S. In vitro studies on apogamy, apospory and controlled differentiation of rhizome segments of the fern, Ampelopteris prolifera (Retz.) Copel. Bot. J. Linn. Soc. 1969, 62, 431–443. [Google Scholar] [CrossRef]
- Bell, P.R. Apospory and apogamy: Implications for understanding the plant life cycle. Int. J. Plant Sci. 1992, 153 Pt 2, S123–S136. [Google Scholar] [CrossRef]
- Mikami, K.; Li, C.; Irie, R.; Hama, Y. A unique life cycle transition in the red seaweed Pyropia yezoensis depends on apospory. Commun. Biol. 2019, 2, 229. [Google Scholar] [CrossRef]
- Coelho, S.M.; Godfroy, O.; Arun, A.; Le Corguillé, G.; Peters, A.F.; Cock, J.M. OUROBOROS is a master regulator of the gametophyte to sporophyte life cycle transition in the brown alga Ectocarpus. Proc. Natl. Acad. Sci. USA 2011, 108, 11518–11523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arun, A.; Coelho, A.M.; Peters, A.F.; Bourdareau, S.; Pérès, L.; Scornet, D.; Strittmatter, M.; Lipinska, A.P.; Yao, H.; Godfroy, O.; et al. Convergent recruitment of TALE homeodomain life cycle regulators to direct sporophyte development in land plants and brown algae. Elife 2019, 8, e43101. [Google Scholar] [CrossRef]
- van der Meer, J.P.; Patwary, M.U.; Bird, C.J. Genetics of Gracilaria tikvahiae (Rhodophyceae). X. Studies on a bisexual clone. J. Phycol. 1984, 20, 42–46. [Google Scholar] [CrossRef]
- van der Meer, J.P. Genetics of Gracilaria tikvahiae (Rhodophyceae). XI. Further characterization of a bisexual mutant. J. Phycol. 1986, 22, 151–158. [Google Scholar]
- Li, C.; Ariga, I.; Mikami, K. Difference in nitrogen starvation-inducible expression patterns among phylogenetically diverse ammonium transporter genes in the red seaweed Pyropia yezoensis. Am. J. Plant Sci. 2019, 10, 1325–1349. [Google Scholar] [CrossRef] [Green Version]
- Seely, G.R.; Duncan, M.J.; Vidaver, W.E. Preparative and analytical extraction of pigments from brown algae with dimethyl sulfoxide. Mar. Biol. 1972, 12, 184–188. [Google Scholar] [CrossRef]
- Beer, S.; Eshel, A. Determining phycoerythrin and phycocyanin concentrations in aqueous crude extracts on red algae. Mar. Freshw. Res. 1985, 36, 785–792. [Google Scholar] [CrossRef]
- Suda, M.; Mikami, K. Reproductive responses to wounding and heat stress in gametophytic thalli of the red alga Pyropia yezoensis. Front. Mar. Sci. 2020, 7, 394. [Google Scholar] [CrossRef]
- Zhong, C.; Aruga, Y.; Yan, X. Morphogenesis and spontaneous chromosome doubling during the parthenogenetic development of haploid female gametophytes in Pyropia haitanensis (Bangiales, Rhodophyta). J. Appl. Phycol. 2019, 31, 2729–2741. [Google Scholar] [CrossRef]
- Zhong, C.; Yan, X. Haploid spontaneous diploidization during apogamy of male gametophytes in Pyropia haitanensis (Bangiales, Rhodophyta). J. Appl. Phycol. 2020, 32, 1395–1403. [Google Scholar] [CrossRef]
- Mikami, K. Diploid apogamy in red algal species of the genus Pyropia. J. Aquat. Res. Mar. Sci. 2019, 2019, 206–208. [Google Scholar]
- Gao, G.; Clare, A.S.; Rose, C.; Caldwell, G.S. Reproductive sterility increases the capacity to exploit the green seaweed Ulva rigida for commercial applications. Algal Res. 2017, 24, 64–71. [Google Scholar] [CrossRef] [Green Version]
- Gao, G.; Clare, A.S.; Rose, C.; Caldwell, G.C. Intrinsic and extrinsic control of reproduction in the green tide-forming alga, Ulva rigida. Environ. Exp. Bot. 2017, 139, 14–22. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, M.; Kumari, P.; Li, C.; Mikami, K. Low temperature causes discoloration by repressing growth and nitrogen transporter gene expression in the edible red alga Pyropia yezoensis. Mar. Environ. Res. 2020, 159, 105004. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Chen, X.; Yi, Q.; Wang, G.; Pan, G.; Lin, A.; Peng, G. A strategy for the proliferation of Ulva prolifera, main causative species of green tides, with formation of sporangia by fragmentation. PLoS ONE 2010, 5, e8571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wichard, T.; Oertel, W. Gametogenesis and gamete release of Ulva mutabilis and Ulva lactuca (chlorophyta): Regulatory effects and chemical characterization of the “swarming inhibitor”. J. Phycol. 2010, 46, 2248–2259. [Google Scholar] [CrossRef]
- Vesty, E.F.; Kessler, R.W.; Wichard, T.; Coates, J.C. Regulation of gametogenesis and zoosporogenesis in Ulva linza (Chlorophyta): Comparison with Ulva mutabilis and potential for laboratory culture. Front. Plant Sci. 2015, 6, 15. [Google Scholar] [CrossRef] [Green Version]
- Katsaros, C.; Weiss, A.; Llangos, I.; Theodorou, I.; Wichard, T. Cell structure and microtubule organisation during gametogenesis of Ulva mutabilis Føyn (Chlorophyta). Bot. Mar. 2017, 60, 123–135. [Google Scholar] [CrossRef] [Green Version]
- Mosquna, A.; Katz, A.; Decker, E.L.; Rensing, S.A.; Reski, R.; Ohad, N. Regulation of stem cell maintenance by the Polycomb protein FIE has been conserved during land plant evolution. Development 2009, 136, 2433–2444. [Google Scholar] [CrossRef] [Green Version]
- Okano, Y.; Aono, N.; Hiwatashi, Y.; Murata, T.; Nishiyama, T.; Ishikawa, T.; Kubo, M.; Hasebe, M. A polycomb repressive complex 2 gene regulates apogamy and gives evolutionary insights into early land plant evolution. Proc. Natl. Acad. Sci. USA 2009, 106, 16321–16326. [Google Scholar] [CrossRef] [Green Version]
- Horst, N.A.; Katz, A.; Pereman, I.; Decker, E.L.; Ohad, N.; Reski, R. A single homeobox gene triggers phase transition, embryogenesis and asexual reproduction. Nat. Plants 2016, 2, 15209. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, M.; Brocchierin, L. A comprehensive classification and evolutionary analysis of plant homeobox genes. Mol. Biol. Evol. 2009, 26, 2775–2794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joo, S.; Wang, M.H.; Lui, G.; Lee, J.; Barnas, A.; Kim, E.; Sudek, S.; Worden, A.Z.; Lee, J.-H. Common ancestry of heterodimerizing TALE homeobox transcription factors across Metazoa and Archaeplastida. BMC Biol. 2018, 16, 136. [Google Scholar] [CrossRef] [PubMed]
- Shaver, S.; Casas-Mollano, J.A.; Cerny, R.H.; Cerutti, H. Origin of the polycomb repressive complex 2 and gene silencing by an E(z) homolog in the unicellular alga Chlamydomonas. Epigenetics 2010, 5, 301–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Chen, D.-H.; Liu, B.-Y.; Shen, W.-H.; Ruan, Y. Conservation and diversification of polycomb repressive complex 2 (PRC2) proteins in the green lineage. Brief. Funct. Genom. 2017, 16, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Mikulski, P.; Komarynets, O.; Fachinelli, F.; Weber, A.P.M.; Schubert, D. Characterization of the Polycomb-group mark H3K27me3 in unicellular algae. Front. Plant Sci. 2017, 8, 607. [Google Scholar] [CrossRef]
- Schubert, D. Evolution of Polycomb-group function in the green lineage. F1000Res 2019, 8. [Google Scholar] [CrossRef] [Green Version]





| Strain | Carposporangia 1 | Carpospores 2 | ||||||
|---|---|---|---|---|---|---|---|---|
| WT | Two-cell layer | Four-cell layer | ||||||
| Upper | Lower | Top to bottom | ||||||
| 11.4 ± 2.6 (n = 14) | 9.61 ± 2.1 (n = 14) | 4.69 ± 0.6 (n = 7) | 4.65 ± 0.5 (n = 7) | 4.68 ± 0.4 (n = 7) | 5.23 ± 0.6 (n = 7) | 10.26 ± 1.7 (n = 62) | ||
| aks | One-cell layer | Two-cell layer | ||||||
| Upper | Lower | |||||||
| 17.5 ± 3.3 | 11.68 ± 1.6 | 11.18 ± 1.4 | 11.84 ± 1.4 | |||||
| (n = 41) | (n = 30) | (n = 30) | (n = 76) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikami, K.; Matsumura, T.; Yamamoto, Y. Primary Characterization of a Life-Cycle Mutant akasusabi of the Red Alga Neopyropia yezoensis. Phycology 2021, 1, 14-26. https://doi.org/10.3390/phycology1010002
Mikami K, Matsumura T, Yamamoto Y. Primary Characterization of a Life-Cycle Mutant akasusabi of the Red Alga Neopyropia yezoensis. Phycology. 2021; 1(1):14-26. https://doi.org/10.3390/phycology1010002
Chicago/Turabian StyleMikami, Koji, Takaharu Matsumura, and Yuji Yamamoto. 2021. "Primary Characterization of a Life-Cycle Mutant akasusabi of the Red Alga Neopyropia yezoensis" Phycology 1, no. 1: 14-26. https://doi.org/10.3390/phycology1010002
APA StyleMikami, K., Matsumura, T., & Yamamoto, Y. (2021). Primary Characterization of a Life-Cycle Mutant akasusabi of the Red Alga Neopyropia yezoensis. Phycology, 1(1), 14-26. https://doi.org/10.3390/phycology1010002







