1. Introduction
Cerium (Ce) is a member of the lanthanide series of metals on the periodic table. The discovery of Ce in an oxide form was first reported in 1803 by scientists in both Sweden and Germany [
1]. This oxide was given the name “ceria” by discoverer Jons Jacob Berzelius in Sweden. Berzelius named the compound after the dwarf planet Ceres, which itself was named for the Roman goddess of agriculture. Cerium is the most abundant of the rare-earth metals found in the Earth’s crust [
2], where it comprises about 0.0046% by weight. Cerium and other lanthanide metals naturally occur in an array of minerals in the environment including carbonates (e.g., bastnäsite, (Ce,La)(CO
3)F), silicates (e.g., allanite, (Ca,Ce)(Al
2Fe
2+)(Si
2O
7)(SiO
4)O(OH)), cerite (Ce,Ca)
9(Fe,Mg)(SiO
4)
3(HSiO
4)
4(OH)
3), phosphates (e.g., monazite,(Ce,La,Nd,Th)(PO
4)), and oxides (e.g., euxenite, (Y,Ca,Ce,U,Th) (Nb,Ti,Ta)
2O
6)). Of these minerals, however, only bastnäsite and monazite have been serving a major role for commercial uses. In the last several years, Ce-based compounds have received much attention because of their popular applications of CeO
2 nanoparticles (NPs) in industrial applications. Unfortunately, due to the relative newness of CeO
2 NP environmental chemistry as a field, the impact of CeO
2 NPs on ecosystem health (e.g., toxicological effects on aquatic-terrestrial organisms) has not been well understood. This review (discusses the geochemistry of Ce, geological occurrence, its applications and uses, environmental fate and the contradicting findings in recent toxicological studies.
3. Geological Occurrence
Cerium is found in a variety of mineral classes, primarily including carbonates, phosphates, silicates, oxides and hydroxides. Main sources of industrial cerium include the carbonate mineral bastnäsite and the phosphate mineral monazite. These key cerium minerals and other lanthanide associated minerals are discussed in detail below.
3.1. Carbonates
Carbonates, the largest group of Ce-containing minerals, can be grouped in anhydrous, hydroxyl or halogen, and hydrated groups (
Table 4). Some of anhydrous carbonates are burbankite, carbocernaite, remondite, sahamalite, and tundrite. Carbonates with hydroxyl or halogen are bastnäsit, daqingshanite, huanghoite, hydroxylbastnäsite, parasite, rontgenite, and synchysite. Hydrate carbonates are ancylit, calcioancylite, lanthanit, rhabdophane, and thorbastnäsite.
Table 4.
Several members of the rare earth anhydrous and hydrous carbonates. Modified after [
16].
Table 4.
Several members of the rare earth anhydrous and hydrous carbonates. Modified after [16].
| Mineral | Chemical Formula | Type Locality |
|---|
| Anhydrous | | |
| Bastnäsite-(Ce) | | Sweden |
| Burbankite | | USA |
| Carbocernaite | | Russia |
| Cordylite-(Ce) | | Greenland |
| Huanghoite | | China |
| Hydroxylbastnäsite-(Ce) | | Russia |
| Parisite-(Ce) | | Colombia |
| Remondite-(Ce) | | Cameroon |
| Rontgenite | | Greenland |
| Sahamalite | | USA |
| Synchysite-(Ce) | F | Greenland |
| Hydrous | | |
| Ancylite-(Ce) | | Greenland |
| Calcioancylite | | Russia |
| Calkinsite | | USA |
| Daqingshanite-(Ce) | | China |
| Lanthanite-(Ce) | | Wales |
| Rhabdophane | | England |
| Thorbastnäsite | | Russia |
| Tundrite-(Ce) | | Russia |
As stated earlier, bastnäsite (Ce,La)(CO
3)F) is one of two most important mineral sources of cerium and other rare-earth metals (such as Th). Bastnäsite is of particular importance to the discovery of Ce. An early mining of bastnäsite began at the Mountain Pass rare earth mine in San Bernardino, California with the discovery of a new class of rare earth deposit: a large carbonaceous mineral deposit containing significant amounts of bastnäsite. The major composition of bastnäsite consisted of approximately 49% cerium, 33% lanthanum, 12% neodymium with some praseodymium, samarium and gadolinium. Bastnäsite minerals also exhibited at least twice the Eu concentrations of a typical monazite. As more carbonatite deposits were discovered in Africa and China, mining processes were developed and these deposits became vital to the global lanthanide supply. From 1965 to the mid- 1980s, the Mountain Pass site emerged as the world’s major source of lanthanides. Currently China plays a leading role in the global rare earth supply (as high as 97%); the Bayan Obo Mining District is the source of the majority of the world’s lanthanides [
16].
3.2. Phosphates
Though relatively few cerium phosphate minerals exist, they are very important to consider when identifying sources of cerium (
Table 5). Until bastnäsite began to be processed around 1965, the phosphate mineral monazite, which usually forms small reddish-brown crystals, was the only significant commercial source of lanthanides such as cerium and thorium. Monazite is most often found in placer deposits, which are mineral accumulations often in the form of black sedimentary rocks. Monazite is fairly dense (about 4.6 to 5.7 g/cm
3), a property consistent with that of most black sand minerals such as zircon and cassiterite. Placer deposits in India are particularly rich in monazite as well as deposits in Australia, Madagascar and South Africa [
17].
Table 5.
Several members of the rare earth phosphates. Modified after [
17].
Table 5.
Several members of the rare earth phosphates. Modified after [17].
| Mineral | Chemical Formula | Type Locality |
|---|
| Agardite-(Ce) | | Germany |
| Monazite-(Ce) | ) | Russia |
One of the major reasons that bastnäsite came to replace monazite in lanthanide production in the 1960’s is the radioactive nature of monazite, which contains Th and produces radioactive daughter products. This radioactive character is useful because it allows monazite to be used as a geological dating tool; thorium was also considered as a potential nuclear fuel in the 1960s. When this interest began to decline, bastnäsite was an attractive replacement option because it contained much lower concentrations of Th than were found in monazite. Monazite is also more difficult to store than bastnäsite since it must be kept away from any mineral samples that are sensitive to radiation.
Monazite and bastnäsite minerals are both broken down into further classifications based on relative elemental composition. Four types of monazite and three types of bastnäsite are commonly referred to in literature. Monazite occurs in monazite-(Ce) [meaning Ce is the most predominant rare earth metal in monazite-(Ce)( (Ce,La,Nd,Th)(PO
4)], monazite-(La), monazite-(Nd), and monazite-(Pr); bastnäsite occurs in bastnäsite-(Ce), bastnäsite-(La), and bastnäsite-(Y). The –(Ce) compositions constitute most of the bastnäsite and monazite minerals in existence [
17].
3.3. Silicates
Silicates comprise the most abundant group of minerals in the earth’s crust. Silicates play an extremely important role in soil minerals (
Table 6). A significant amount of Ce is found in the silicate mineral allanite,(Ca,Ce,Y)
2(AlFe
3+)
3(SiO
4)
3(OH), Cerite-(Ce), (Ce,Ca)
9(Fe,Mg)(SiO
4)
3(HSiO
4)
4(OH)
3, gadolinite-(Ce) (Ce,La,Nd,Y)
2Fe
2+Be
2Si
2O
10 and Joaquinite-(Ce), NaBa
2Ce
2FeTi
2[Si
4O
12]
2O
2(OH,F)·H
2O.
Table 6.
Several members of the rare earth silicates. Modified after [
17].
Table 6.
Several members of the rare earth silicates. Modified after [17].
| Mineral | Chemical Formula | Type Locality |
|---|
| Allanite-(Ce) | | Greenland |
| Cerite-(Ce) | | Sweden |
| Gadolinite-(Ce) | | Japan, Norway |
| Joaquinite-(Ce) | | USA |
Like bastnäsite and monazite, allanite can be broken down into three further classifications: allanite-(Ce) ((CaCe)(Al2Fe2+)(Si2O7)(SiO4)O(OH)), allanite-(La), and allanite-(Y). Allanite is a useful source of rare earth elements because it can contain up to 20% rare earths by composition. The high concentrations of thorium and other radioactive elements in the mineral lead to radiative degradation and structure disruption of allanite and surrounding minerals. The distribution of allanite minerals is widespread with large, high-quality deposits occurring in Greenland (where allanite was discovered), Sweden, Norway, Finland, and Russia. Allanite has hardness values ranging from 5.5–6 Mohs and is found mainly in metamorphic sedimentary and felsic igneous rocks. Allanite is also commonly referred to as orthite.
3.4. Oxides and Hydroxides
Cerium (hyr)oxide minerals are least common of all Ce minerals such as aeschynite-(Ce) (Ce,Ca,Fe,Th)(Ti,Nb)
2(O,OH)
6, euxenite-(Y) (Y,Ca,Ce,U,Th)(Nb,Ti,Ta)
2O
6, and perovskite CaTiO
3 with Ce varieties (
Table 7). Of the few oxides and hydroxides that contribute to commercial cerium sources, euxenite is the most significant oxide phase despite its low composition percent of cerium (~4%). Unlike allanite, monazite and bastnäsite, euxenite exists almost entirely in the euxenite-(Y) form, featuring yttrium as its major component instead of cerium. Like allanite, euxite also exhibits radiation damage to itself and nearby minerals due to high radioactive content. With hardness values varying between 5.5 and 6.5 Mohs, euxenite features comparable to slightly higher hardness than its other cerium-containing counterparts, allanite, monazite, and bastnäsite. Euxenite occurs along with monazite in black sands as well as in granite pegmatites. Euxenite is found primarily in Norway, where it was discovered, as well as in Russia, Brazil, Madagascar, Canada, and the US. Euxenite functions as an ore for many of the other rare earth elements contained in the mineral as well [
16].
As discussed in the previous section, Ce is commonly present with other lanthanides and metals forming phosphate and carbonate minerals. The heterogeneity of natural minerals makes the isolation process of pure Ce extremely difficult. Some ores are directly used for metallurgical purposes. Other Ce enriched ores undergo the purification process prior to industrial and commercial uses. The separation of Ce from natural minerals is conducted via oxidation and variable solubility based filtration steps.
In general, the mineral ores are leached with sulfuric acid and then titrated with sodium hydroxide to remove thorium precipitates. This step is followed by precipitation with oxalate to remove to most of rare earth elements (e.g., Ce, La, Th, Nd) as insoluble oxalate forms. These oxalates undergo annealing/oxidation process to produce lanthanide (tri-and tetra-valent state) oxides. Acidification of these mixed oxides by nitric acid makes all of the lanthanide (III) oxides soluble except for Ce(IV)(OH)
4. Insoluble Ce(IV) hydroxides are separated through the filtration process [
3,
18]. Once the purity of Ce is improved from the mixed lanthanide/metal oars, a variety of Ce compounds are synthesized for commercial and industrial applications. The historic and current uses of Ce compounds are discussed below.
Table 7.
Several members of the rare earth oxides and hydroxides. Modified after [
16].
Table 7.
Several members of the rare earth oxides and hydroxides. Modified after [16].
| Mineral | Chemical Formula | Type Locality |
|---|
| Aeschynite-(Ce) | | Russia |
| Euxenite-(Y) | | Norway |
| Perovskite | CaTiO3 with Ce varieties | Russia |
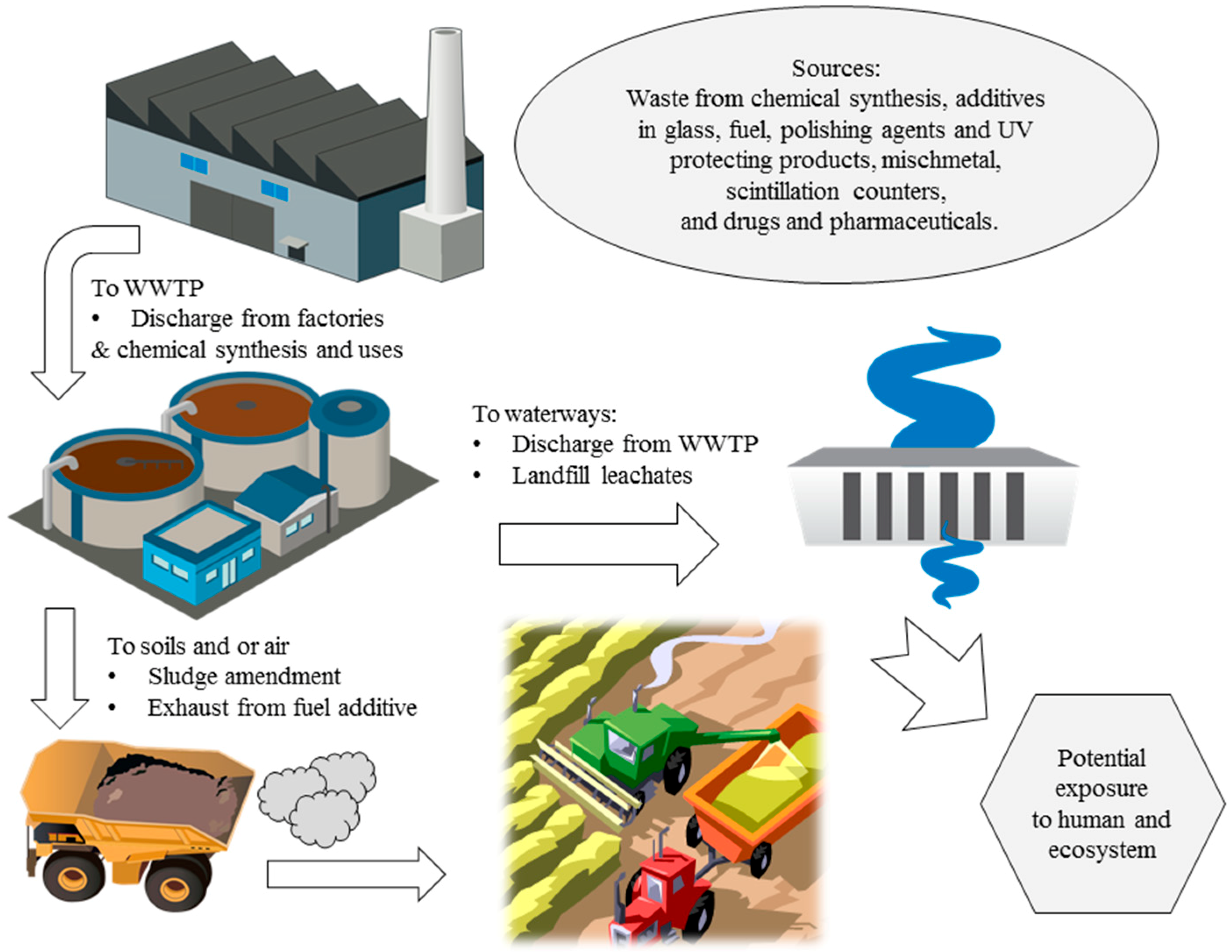
6. Environment Fate of CeO2 Nanoparticles
The environmental fate of CeO
2 NPs is dependent on its applications and uses. A schematic illustration of environmental fate of cerium based industrial and consumer products are shown in
Figure 1. As previously stated, the use of cerium as a diesel fuel additive coupled with a particulate filter drastically decreases particulate matter emissions in automobile exhaust, up to 90% by weight [
47]. Some Ce escaping in the emissions could accumulate in soils. Exhaust particulate matter exists as very fine particles, normally < 1 μm, leading to extremely diffuse pollution through air exposure [
48]. Cerium is also introduced to landfills as solid wastes of electronics, soils via recycling of sewage sludge, and the aquatic environment via wastewater discharge from ceramic manufacturing plants [
49].
When particles are not efficiently removed from wastewater streams, they can become a long-term problem for the environment. A study by Limbach,
et al. (2008) has shown that, for a model wastewater treatment plant, a significant fraction of CeO
2 NPs were able to escape the clearing system and avoid adsorption to clearing sludge [
50]. The surface charge of the NPs was found to play a key role in the stabilization against clearance, leading to detectable concentrations of CeO
2 NPs up to 6 wt % in the model plant’s exit stream [
50].
At the wastewater treatment plant, about 95% of CeO
2 NPs undergo biosolid/sludge accumulation in wastewater treatment plants. Thus CeO
2 discharge into the aquatic environment is fairly limited as compared to accumulation levels in the terrestrial environment including landfills [
50]. In general, engineered NP partitioning is the most significant in wastewater treatment plant biosolids/sludge, it follows that Ce pollution could occur after the amendment of agricultural soil via application of wastewater biosolids. As in the case of many other metal NPs such as TiO
2, ZnO, and Ag NPs [
51], it is believed that biosolids application to agricultural soils serves as one of the largest environmental exposure pathways for CeO
2 NPs [
52].
Unfortunately, due to the relativeness newness of discovery of CeO
2 NP chemistry as a field, the fate and transport of NPs in general are not still well-understood. Much work has been done with TiO
2 and silver NPs but most NPs have not been studied as extensively. Nanoparticulate CeO
2, while greatly useful for a wide variety of applications, is not without the risks and hazards associated with NPs in general. The toxicity of cerium NPs and cerium in general is discussed in depth in
Section 5.
Figure 1.
Environmental fate of cerium based consumer and industrial products including CeO
2 nanoparticles. Illustrations of water treatment plant, factory and water discharge are from integration and application network, [
53].
Figure 1.
Environmental fate of cerium based consumer and industrial products including CeO
2 nanoparticles. Illustrations of water treatment plant, factory and water discharge are from integration and application network, [
53].
Cerium oxide NPs are of great interest biologically for many reasons, including their many starkly contrasting toxicity effects in different conditions. As previously mentioned, increased Ce(III):Ce(IV) ratios result in increased SOD mimetic activity and thus further inactivation of superoxide free radicals [
6]. Unlike SOD, CeO
2 NPs can directly react with hydroxyl radicals [
54] as well as superoxide radicals in order to inactivate both short-lived and stable radicals [
55]. A study by Ivanov,
et al. showed that the rate of inactivation of stable radicals by CeO
2 NPs is dependent directly upon particle size and that smaller particle sizes result in increased rates of radical inactivation [
56]. Discussed below are effects of CeO
2 NP exposure to biological organisms including plants, bacteria/aquatic organisms, and mammals, as well as the recorded and possible effects of these exposures.
6.1. Plants
Once in the soil, cerium NPs may be absorbed into vegetation or contaminate water. Cerium has been shown to be likely to enter the produce food chain through take-up by alfalfa, corn, tomato, cucumber, and soy [
57,
58]. Nanoparticles tend to accumulate on root surface of plants, decreasing hydraulic conductivity and often inhibiting root growth [
59]. Nanoceria has been reported to significantly reduce germination rates in corn, tomato, and cucumber; the same study reported enhanced root growth in cucumber and corn species after introduction of CeO
2 NPs, but reduced root growth in tomato and alfalfa plants. Shoot elongation was also found to be enhanced in all four species [
57]. Another study displayed differing effects, finding that CeO
2 NPs did not affect root growth in radish, tomato, lettuce, wheat, rape, cabbage, and cucumber plants [
60]. Concentration-dependent absorption by plant roots has been observed, but most of the NPs are instead adsorbed to the surface of the root; entry into the roots is limited and very difficult [
61]. In a recent spectroscopic investigation, the fate of CeO
2 NPs in soybean plants was investigated in fully grown plant tissues [
62]. The X-ray absorption spectroscopy studies showed that Ce remained mostly as Ce(IV)O
2 NPs within the plant (pods and tissues), and a small fraction of Ce(IV) was reduced to Ce(III).
Priester and co-workers (2012) recently examined the effect of CeO
2 NPs on soybean [
62]. The researchers found an interesting dose-response relationship. Plants with low CeO
2 NPs (100 mg/kg) grew slowly and had reduced leaf cover, whereas plants grown with high CeO
2 NPs (1000 mg/kg) yielded less soybean biomass. They also observed the shutdown of nitrogen fixation in root nodules when plants were exposed to high CeO
2 concentrations.
Cerium dioxide NPs have also been shown to protect anthocyanins in grapes from ROS by inhibiting oxidative degradation in neutral and alkaline solutions. However, in acidic solutions, no inhibitory effects were observed after exposure of the anthocyanins to NP CeO
2 [
63]. Similar pH-dependent effects have been demonstrated across a variety of plants and other organisms.
Cerium dioxide NPs have also shown translocation ability; that is, the capability to cross biological barriers such as the blood-brain barrier. When applied to maize leaves, CeO
2 NPs was demonstrated to either adsorb to the leaf surface in agglomerated form (larger particles) or penetrate the leaf and become incorporated into the leaf structure (smaller particles). However, in plants cultivated after airborne exposure to CeO
2 NPs, newly grown leaves exhibited no translocation. This lack of evidence for translocation is important since maize is a major agricultural crop and knowledge of NP ability to translocate into newly grown, non-exposed plants is needed. Nanoparticles most commonly enter plants via root uptake or air exchange into the leaves. Nanoparticles larger than 1.1 µm were found by the study to be unable to penetrate the leaf surface and enter the leaves [
64].
6.2. Bacteria and Algae
Nanoparticle effects on both Gram-positive and Gram-negative bacteria have been studied extensively. Cerium dioxide NPs were shown to exhibit size-dependent and concentration-dependent inhibition of both
Escherichia coli and
Bacillus subtilis; it is suggested that the NPs do not penetrate the bacterial cells, but rather adsorb to them, inducing deleterious effects. The same study found that CeO
2 NPs induced no inhibitory effects on the growth of
Shewanella oneidensis, demonstrating that bacterial responses to NPs and interactions between the two vary greatly among bacterial species [
65].
Another study on cytotoxicity of CeO
2 NPs to Gram-negative
E. coli also reported that the NPs were adsorbed in large amounts on the outer membrane [
66]. The NPs are positively charged at neutral pH values, meaning they are subject to a strong electrostatic attractive force toward bacterial outer membranes such as that of
E. coli. Close contact was reported between the NPs and
E. coli, indicating that oxidative response may have been responsible for the observed toxicity, which was significant after the adsorption of the NPs and their reduction from Ce(IV) to Ce(III). The survival rate of
E. coli was reduced to half for a 5 mg/L exposure to CeO
2 [
66]. This study and other investigations have found that cytotoxicity of CeO
2 NPs for
E. coli can be reversed by changing the exposure media, indicating that surface charge density is largely responsible for NP CeO
2 cytotoxic effects [
67].
Zeyons and co-workers (2009) also observed that toxicity requires direct contact between CeO
2 NPs and cells of
Synechocystis PCC6803 cyanobacteria and the RR1 strain of E. coli [
68].
More recently, Dahle and Arai investigated a subchronic toxicity study of CeO
2 NPs on soil-denitrification process as a function of particle size (33 and 78 nm), total Ce concentration (50–500 mg/L), and speciation [Ce(IV)
vs. Ce(III)] [
69]. Soluble Ce(III) was far more toxic than Ce(IV)O
2 NPs when an equal total concentration of Ce was evaluated. Particle size-dependent toxicity, species-dependent toxicity, and concentration-dependent toxicity were all observed in this study for both the steady-state and the kinetic evaluations. They suggested that changes in physicochemical properties of Ce(IV)O
2 NPs in soil solution might be important in assessing the environmental fate and toxicity of NPs in aquatic and terrestrial environments.
In a study on the effect of NPs on microbial communities in wastewater treatment plants, it was found that CeO
2 NPs exhibited stronger inhibitory effects on biogas production by the microbes than TiO
2, gold, or silver NPs [
70]. After release into the waterway, CeO
2 NPs are far more likely to partition into aquatic plants and sediment than to stay in the water, but some aquatic species such as algae and fish are still at risk and the possible toxicity and effects of CeO
2 NP uptake in these organisms must be investigated [
71]. When CeO
2 NPs do remain in the sediment, concentrations tend to be locally high due to the adsorption and aggregation tendencies of NPs, as stated earlier.
Rodea-Palomares and co-workers observed the species specific aquatic toxicity to CeO
2 NPs [
72]. The aquatic cyanobacteria
Anabaena CPB4337 exhibited toxicity to CeO
2 NPs values ranging from 0.27 to 6.3 mg/L while the aquatic green alga
Pseudokirchneriella subcapitata exhibited values from 2.4 to 29.6 mg/L. A few studies reported that the growth inhibition of
P. subcapitata by CeO
2 NPs was at 10.3 mg/L [
73] and 7.6 mg/L [
74]. Cerium dioxide NPs were shown to cause membrane disruption and severe damage to the cells of both organisms though uptake of the NPs by the cells was not indicated. As in the case of
E. coli and
B. subtilis mentioned above, the
Anabaena cells became coated with adsorbed CeO
2 NPs. It is suspected that cell wall and membrane disruption was likely the cause of cell damage rather than oxidative activity [
72]. From these and other similar studies, it remains unclear whether nanoceria toxicity can be largely attributed to cellular adsorption or if uptake of the particles lends to intracellular effects, especially because negatively charged particles should be repelled by the large negatively charged domains of cell membranes. Importantly, it has been demonstrated that CeO
2 NP toxicity in
P. subcapitata is also influenced by pH, ionic strength and amounts of natural organic matter. Increasing ionic strength levels result in increased CeO
2 NP aggregation, while higher levels of natural organic matter sharply lowered average NP aggregate sizes, severely decreasing aggregation from particle sizes of ~4500 nm to sizes of 100–200 nm. Adsorption of the NPs to natural organic matter reduced their bioavailability and thus lowering the toxicity of CeO
2 NPs to
P. subcapitata [
75]. However, Mariner and co-workers (2013) reported that changes in agglomeration state of CeO
2 NPs did not significantly inhibit the growth of
P. subcapitata [
76]. The EC
50 values were 5.6, 4.1 and 6.2 mg/L, after exposure to non-aged (non-aggregated), 3 and 30 days aged aggregated NP suspensions, respectively.
6.3. Invertebrates
Caenorhabditis elegans is a commonly-studied soil nematode that serves as an important aquatic indicator. Ceria NPs have been shown to induce size-dependent toxicity effect on the survival and fertility of
C. elegans; while all CeO
2 NP exposure sizes in one study led to decreased reproductive ability and increased fatality, the smaller NPs had a markedly more toxic effect than the larger NPs. This effect was also observed for TiO
2 NPs [
77]. Another study on nanoceria exposure in
C. elegans indicated that CeO
2 NPs could lead to reduced lifespans via catalysis of ROS generation and subsequent oxidative damage. The study observed a 12% reduction in mean lifespan even at the lowest CeO
2 NPs exposure level of 1 nM [
43].
While an early study did not report the acute toxicity of CeO
2 NPs to two crustaceans
Daphnia magna and
Thamnocephalus platyurus, even ranging up to 5000 mg/L [
76], few recent studies showed some toxicological effects of CeO
2 NP to
Daphnia. Artells and co-workers (2013) evaluated the effects of CeO
2 NPs ([CeO
2]
total = 1–100 mg/L, exposure time: 48 h) on the survival and the swimming performance of
Daphnia similis and
Daphnia pulex [
78]. EC
50 for
D. similis (0.26 mg/L) was 350 times more sensitive than
D. pulex. They suggested that the interspecific toxic effects are probably due to morphological variations such as the presence of reliefs on the cuticle and a longer distal spine in
D. similis that effectively trap NP aggregates, yielding in a greater sorption of NP in
D. similis. Swimming velocities were significantly affected both species. Biodistribution of CeO
2 NP in
Daphnia pulex was also investigated during a molting stage [
79]. A CeO
2 NP uptake was increased by a factor of three when algae were present during the NP exposure. Although NPs were localized in the gut content, the depuration was not efficient to remove the NPs. Interestingly, shedding of the chitinous exoskeleton (every 59 ± 21 h with growth rates about 1.1 or 1.8 μm per stage) was the crucial mechanism controlling the release of NPs regardless of the feeding regime during exposure.
Using an integrated multibiomarker approach, short-term (<100 h) toxicological effects of CeO
2 NPs on two freshwater invertebrates; the amphipod
Gammarus roeseli and the bivalve
Dreissena polymorpha, were investigated [
80]. Under the environmentally realistic concentrations (1–100 µg/L),
G. roeseli accumulated more Ce than
D. polymorpha, though no strong adverse effects were observed on G. roeseli. However, the exposure led to decreases in the size of the lysosomal system, catalase activity and lipoperoxidation in mussel digestive glands.
Conway
et al. studied the effects of short-term (5 wk) exposure of CeO
2 NPs in a marine mussel,
Mytilus galloprovincialis [
81]. The direct and the indirect exposure (NP sorbed phytoplankton) exposure of CeO
2 NPs (CeO
2: 0–3 mg/L) did not make any significantly different in the [Ce]
total accumulation in dry mussel tissue or pseudofeces. Although ~99% of CeO
2 was excreted, the depuration rates decreased with increasing time in groups exposed to CeO
2 directly, indicating stress.
6.4. Vertebrates
Larger-scale organisms should also be considered when evaluating the ecotoxicological effects of CeO
2 NPs. In a study exposing multiple nanoscale metal oxides (zinc oxide, CeO
2, and titanium dioxide) to zebrafish (
Danio rerio) and rainbow trout (
Oncorhynchus mykiss) both via water and via diet, the oxide NPs were largely not found to be able to penetrate the fish cell barriers. The zinc, cerium, and titanium content was measured in the gill, liver, skin, brain, but, blood, and kidneys of the two fish species and only two areas of significant NP uptake were observed. Ceria NP uptake was discovered in the liver of the zebrafish exposed via water and ionic titanium was detected in the gut of the trout exposed via diet. It is likely that this inability of the metal oxide NPs, including CeO
2, was due to limited bioavailability caused by accumulation of large aggregates (up to 3 μm) in the aqueous exposure route [
82].
Mammals are among the lesser-studied organisms for CeO
2 NP toxicity effects; unlike the widespread variety of plants, bacteria, and aquatic organisms, the only mammal cell lines that have been evaluated for CeO
2 NP toxicity currently include those of rodents and humans. Cerium dioxide NPs have been demonstrated to be neuroprotective in mouse hippocampal brain slices, indicating that the NPs could be useful as therapeutic agents for strokes due to reduced ischemic cell death [
83]. Another study of rodent cell line HT22 verified that CeO
2 NPs (as well as yttrium NPs) prevent oxidative stress to cells and are neuroprotective [
84]. However, it has also been shown that a single CeO
2 NP exposure leads to reduced liver weight, hepatocyte enlargement, reduced albumin levels, and diminished sodium-potassium ratios in male Sprague-Dawley rats [
44].
Cerium dioxide NPs have demonstrated a range of activity in human cells as well. Unlike bacterial and algal cells, effective uptake of CeO
2 NPs by human cells has been demonstrated repeatedly. It has been shown that CeO
2 NPs modulate the brain-derived neurotrophic factor (BDNF) pathway in human Alzheimer’s disease models in order to trigger neuronal survival [
85]. The NPs have been shown to exhibit toxicity to bronchial epithelial cells due to ROS production [
86], although another study showing the same results for bronchial epithelial cells also found no toxic effects for human brain cells and rat cardiomyocytes [
45]. Another study exhibited low to moderate toxicity of CeO
2 NPs to human alveolar epithelial and macrophage cell lines [
87]. In a study comparing CeO
2 to Al
2O
3 and ZnO NPs, CeO
2 NPs were found to induce membrane damage and inhibit colony formation to differing degrees across human lung epithelial cells, carcinoma cells, and normal cell lines. The CeO
2 NPs did not demonstrate notable adverse effects on cell proliferation and viability; they were shown to be much less toxic than ZnO in terms of cell viability, proliferation, colony formation, and membrane integrity, but more toxic than Al
2O
3 even after long-term exposure [
88].
As previously stated, the results of studies across similar cell lines are not always in agreement. Cerium dioxide NPs have been reported to both induce and prevent oxidative stress in a variety of organisms. Studies have shown that existing levels of CeO
2 in the environment are far “lower than the estimated no effect dose for chronic human exposure” [
45], leading to the assertion by some that CeO
2 NP, along with a variety of other NPs, may not exhibit any environmental effects at all [
75]. However, until the ecotoxicological effects of CeO
2 NPs are well-understood, studies must continue to investigate the generation, transport, uptake, and toxicity mechanisms of these potentially harmful materials.
7. Conclusions
A cerium compound of particular interest is nanoparticulate ceria, also known as cerium oxide (CeO
2). By far, CeO
2 NPs are the most widely commercially used cerium compound for electronics, automotive, and solar panels for energy. As the production of CeO
2 NP increases, so does the usage in the industry and consumer markets. The recent study by Keller and Lazareva (2013) predicted more than 60%–86% of engineered NPs including CeO
2 NPs will end up in landfills and soils, and the pathway of WWTP effluent and biosolids to the soil-water environment is fairly limited [
49]. The estimated concentrations of CeO
2 NPs in WWTP in the San Francisco Bay (SFB) area are 10
−2 to 1 µg/L in effluent and 1 to 10 µg/kg in dry biosolids. In other study by Gottschalk
et al. (2013) [
89], even much smaller concentration of CeO
2 NPs in WWTP effluent and biosolid are reported (0.5 × 10
−4 µg/L and 10
−6 mg/kg, respectively). While there are needs in improving the detection and quantification of CeO
2 NPs in different environmental media, it is imminent that organisms in ecosystem will be exposed to these low concentrations (micro gram per liter or kilogram).
In the last decade, scientific community has begun to uncover the effect of engineered NPs on human and ecosystem health through environmental chemistry and toxicology research in plants, bacteria, aquatic organisms, and mammals. However, the research findings are still difficult to extrapolate to natural aquatic and terrestrial systems since (1) some toxicological data are contradicting (e.g., CeO
2 as antioxidant or reactive oxygen species production-inducing agent) [
40,
41,
44,
45]; (2) inconsistent characterization of NPs (e.g., ratio of Ce
3+ and Ce
4+); and (3) the results are not often generated under the environmentally relevant geochemical conditions (e.g., concentration, chemical species) that directly affect the NP reactivity (e.g., solubility and complexation with inorganic and organic ligands) and toxicity. In understanding the geochemical fate and toxicity of CeO
2 NPs, it might be important in considering the relevant geochemical conditions (pH and common ligands, phosphate) [
90] in the future toxicological and environmental chemistry investigations.