Photoreactivity of Hair Melanin from Different Skin Phototypes—Contribution of Melanin Subunits to the Pigments Photoreactive Properties
Abstract
1. Introduction
2. Results
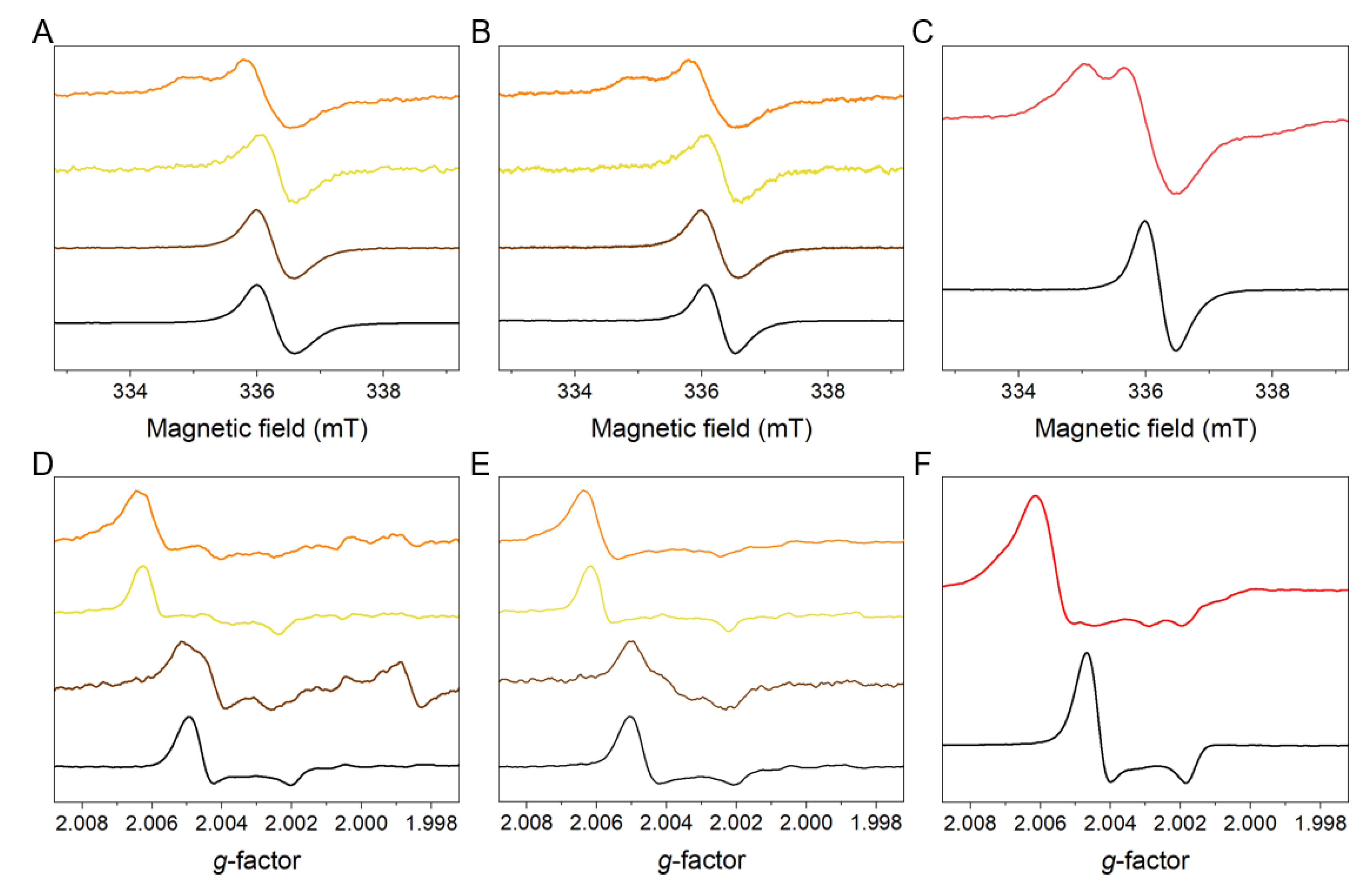
2.1. Paramagnetic Properties of the Isolated Melanins
2.2. Chemical Analysis of the Isolated Pigments
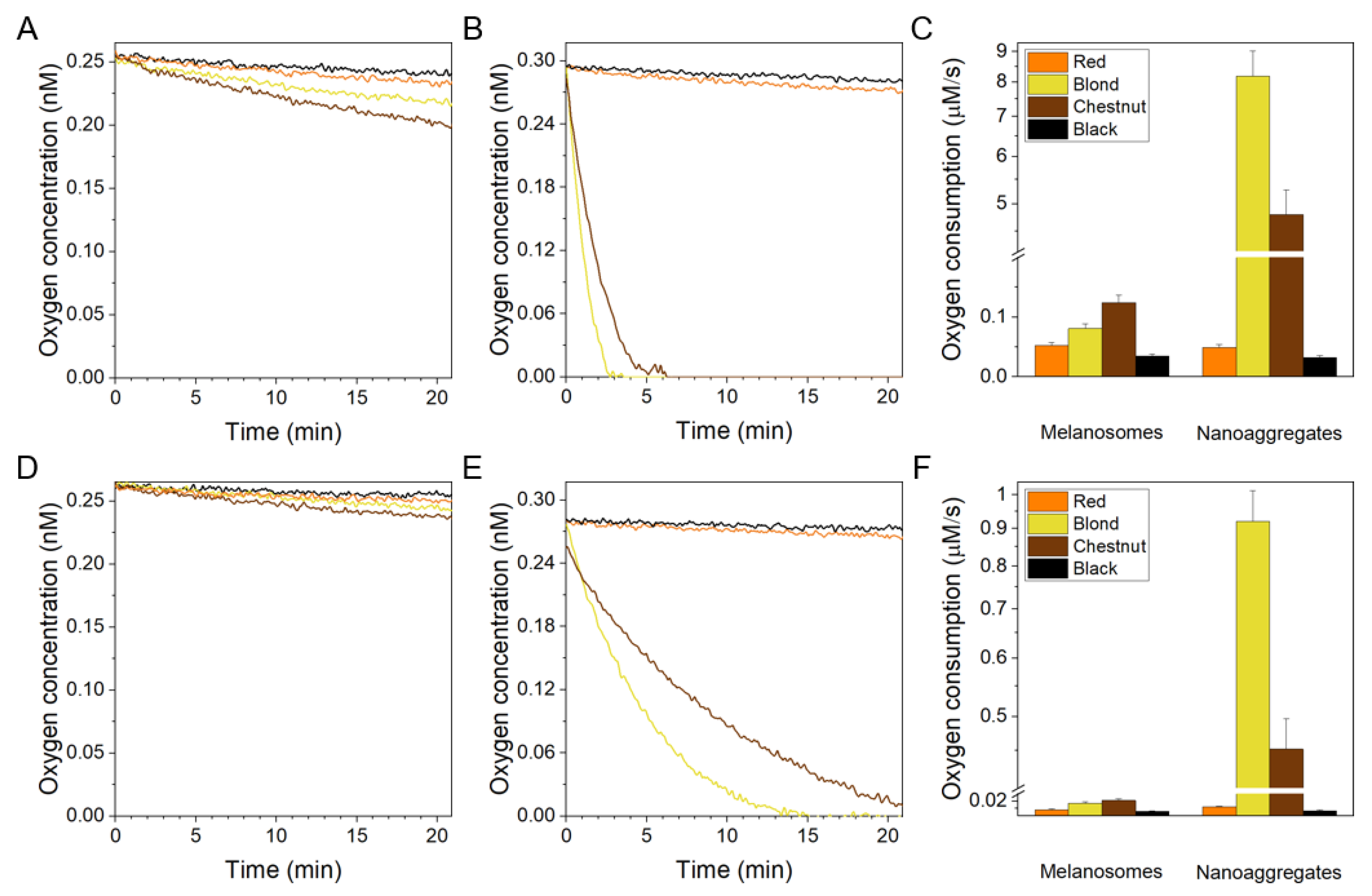
2.3. EPR Analysis of Oxygen Photoconsumption and Photogeneration of Superoxide Anion
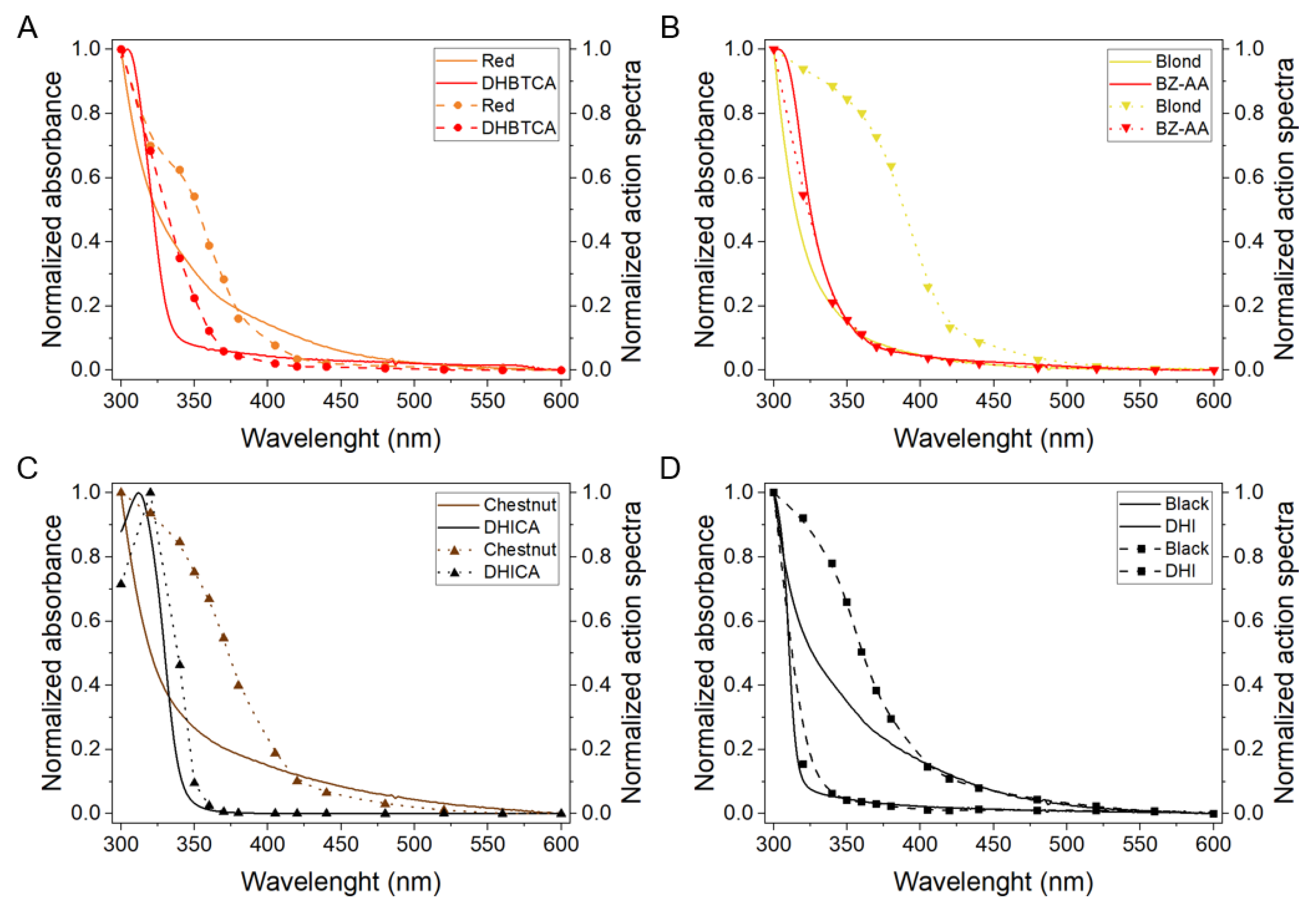
2.4. Time-Resolved Singlet Oxygen Phosphorescence Detection
3. Discussion
4. Materials and Methods
4.1. Isolation of Melanin Pigments
4.2. X-Band EPR Spectroscopy
4.3. W-Band EPR Spectroscopy
4.4. Chemical Analysis of Melanin Subunits
4.5. Oxygen Consumption Measurements
4.6. EPR Spin Trapping Studies
4.7. UV-Vis Spectroscopy
4.8. Time-Resolved Singlet Oxygen Phosphorescence
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| 3-AHP | 3-amino-4-hydroxyphenylalanine |
| 4-AHP | 4-amino-3-hydroxyphenylalanine |
| 5SCD-M | 5-S-cysteinyldopa melanin |
| A500 | absorbance at 500 nm |
| A650 | absorbance at 600 nm |
| AHPO | alkaline hydrogen peroxide oxidations |
| BT | benzothiazine |
| BZ | benzothiazole |
| BZ-AA | 6-(2-amino-2-carboxyethyl)-4-hydroxybenzothiazole |
| C/D-M | cysteine/DOPA melanin |
| D2O | deuterium oxide |
| DHBTCA | 7-(2-amino-2-carboxyethyl)-5-hydroxy-3,4-dihydro-2H-1,4-benzothiazine-3-carboxylic acid |
| DHI | 5,6-dihydroxyindole |
| DHICA | 5,6-dihydroxyindole-2-carboxylic acid |
| DMPO | 5,5-dimethyl-1-pyrroline N-oxide |
| DMSO | dimethyl sulfoxide |
| DOPA | L-3,4-dihydroxyphenylalanine |
| DTT | 1,4-dithiothreitol |
| EPR | electron paramagnetic resonance |
| H2O2 | hydrogen peroxide |
| HI | hydroiodic acid |
| HPLC | high-performance liquid chromatography |
| LED | light emitting diode |
| mHCTPO | 4-protio-3-carbamoyl-2,2,5,5-tetraperdeuteromethyl-3-pyrrolin-1-yloxy |
| PDCA | pyrrole-2,3-dicarboxylic acid |
| PTCA | pyrrole-2,3,5-tricarboxylic acid |
| ROS | reactive oxygen species |
| TTCA | thiazole-2,4,5-tricarboxylic acid |
| UV | ultraviolet |
| UVA | ultraviolet A |
| UV-Vis | ultraviolet-visible light |
References
- D’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; García-Borrón, J.-C.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26, 616–633. [Google Scholar] [CrossRef]
- Sarna, M.; Zadlo, A.; Hermanowicz, P.; Madeja, Z.; Burda, K.; Sarna, T. Cell elasticity is an important indicator of the metastatic phenotype of melanoma cells. Exp. Dermatol. 2014, 23, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Sarna, M.; Krzykawska-Serda, M.; Monika Jakubowska, M.; Zadlo, A.; Urbanska, K. Melanin presence inhibits melanoma cell spread in mice in a unique mechanical fashion. Sci. Rep. 2019, 9, 9280. [Google Scholar] [CrossRef] [PubMed]
- Nasti, T.H.; Timares, L. MC1R, Eumelanin and Pheomelanin: Their Role in Determining the Susceptibility to Skin Cancer. Photochem. Photobiol. 2014, 91, 188–200. [Google Scholar] [CrossRef]
- Leiter, U.; Eigentler, T.; Garbe, C. Epidemiology of skin cancer. Adv. Exp. Med. Biol. 2014, 810, 120–140. [Google Scholar] [CrossRef]
- Gandini, S.; Sera, F.; Cattaruzza, M.S.; Pasquini, P.; Picconi, O.; Boyle, P.; Melchi, C.F. Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure. Eur. J. Cancer. 2005, 41, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Pozzbon, F.C.; Acosta, A.E. Epidemiological profile of primary cutaneous melanoma over a 15-year old period at a private skin cancer center in Colombia. Rev. Salud Publica 2018, 20, 226–231. [Google Scholar] [CrossRef]
- Solano, F. Photoprotection and skin pigmentation: Melanin-related molecules and some other new agents obtained from natural sources. Molecules 2020, 25, 1537. [Google Scholar] [CrossRef]
- Noonan, F.P.; Zaidi, M.R.; Wolnicka-Glubisz, A.; Anver, M.R.; Bahn, J.; Wielgus, A.; Cadet, J.; Douki, T.; Mouret, S.; Tucker, M.A.; et al. Melanoma induction by ultraviolet A but not ultraviolet B radiation requires melanin pigment. Nat. Commun. 2012, 3, 884. [Google Scholar] [CrossRef]
- Meyskens, F.L., Jr.; Farmer, P.J.; Yang, S.; Anton-Culver, H. New Perspectives on Melanoma Pathogenesis and Chemoprevention. Cancer Prev. Recent Results in Cancer Res. 2007, 174, 191–195. [Google Scholar] [CrossRef]
- Szewczyk, G.; Zadlo, A.; Sarna, M.; Ito, S.; Wakamatsu, K.; Sarna, T. Aerobic photoreactivity of synthetic eumelanins and pheomelanins: Generation of singlet oxygen and superoxide anion. Pigment Cell Melanoma Res. 2016, 29, 669–678. [Google Scholar] [CrossRef]
- Chiarelli-Neto, O.; Ferreira, A.S.; Martins, W.K.; Pavani, C.; Severino, D.; Faiao-Flores, F.; Maria-Engler, S.S.; Aliprandini, E.; Martinez, G.R.; Di Masco, P.; et al. Melanin photosensitization and the effect of visible light on epithelial cells. PLoS ONE 2014, 9, e113266. [Google Scholar] [CrossRef]
- Suzukawa, A.A.; Vieira, A.; Winnischofer, S.M.B.; Scalfo, A.C.; Di Mascio, P.; da Costa Ferreira, A.M.; Ravanat, J.-L.; de Luna Martins, D.; Rocha, M.E.M.; Martinez, G.R. Novel properties of melanins include promotion of DNA strand breaks, impairment of repair, and reduced ability to damage DNA after quenching of singlet oxygen. Free Radic. Biol. Med. 2012, 52, 1945–1953. [Google Scholar] [CrossRef]
- Kozinska, A.; Oles, T.; Sarna, T. Photoactivation and detection of photoexcited molecules and photochemical products. Isr. J. Chem. 2012, 52, 745–756. [Google Scholar] [CrossRef]
- Wilkinson, F.; Abdel-Shafi, A.A. Mechanism of quenching of triplet states by molecular oxygen. Biphenyl derivatives in different solvents. J. Phys. Chem. A 1999, 103, 5425–5435. [Google Scholar] [CrossRef]
- Wang, A.; Marino, A.R.; Gasyna, E.M.; Sarna, T.; Norris, J.R. Investigation of photoexcited states in porcine eumelanin through their transient radical products. J. Phys. Chem. B 2009, 113, 10480–10482. [Google Scholar] [CrossRef]
- Premi, S.; Wallisch, S.; Mano, C.M.; Weiner, A.B.; Bacchiocchi, A.; Wakamatsu, K.; Bechara, E.J.; Halaban, R.; Douki, T.; Brash, D.E. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science 2015, 347, 842–847. [Google Scholar] [CrossRef]
- Brenner, M.; Hearing, V.J. The protective role of melanin against UV damage in human skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef]
- Takeuchi, S.; Zhang, W.; Wakamatsu, K.; Ito, S.; Hearing, V.J.; Kraemer, K.H.; Brash, D.E. Melanin acts as a potent UVB photosensitizer to cause an atypical mode of cell death in murine skin. Proc. Natl. Acad. Sci. USA 2004, 101, 15076–15081. [Google Scholar] [CrossRef]
- Del Bino, S.; Ito, S.; Sok, J.; Nakanishi, Y.; Bastien, P.; Wakamatsu, K.; Bernerd, F. Chemical analysis of constitutive pigmentation of human epidermis reveals constant eumelanin to pheomelanin ratio. Pigment Cell Melanoma Res. 2015, 28, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Nakanishi, Y.; Valenzuela, R.K.; Brilliant, M.H.; Kolbe, L.; Wakamatsu, K. Usefulness of alkaline hydrogen peroxide oxidation to analyze eumelanin and pheomelanin in various tissue samples: Application to chemical analysis of human hair melanin. Pigment Cell Melanoma Res. 2011, 24, 605–613. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Ohtara, K.; Ito, S. Chemical analysis of late stages of pheomelanogenesis: Conversion of dihydrobenzothiazine to a benzothiazole structure. Pigment Cell Melanoma Res. 2009, 22, 474–486. [Google Scholar] [CrossRef]
- Ito, S.; Suzuki, N.; Takebayashi, S.; Commo, S.; Wakamatsu, K. Neutral pH and copper ions promote eumelanogenesis after the dopachrome stage. Pigment Cell Melanoma Res. 2013, 26, 817–825. [Google Scholar] [CrossRef]
- Ito, S.; Wakamatsu, K.; Sarna, T. Photodegradation of eumelanin and pheomelanin and its pathophysiological implications. Photochem. Photobiol. 2018, 94, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Zadlo, A.; Szewczyk, G.; Sarna, M.; Camenisch, T.G.; Sidabras, J.W.; Ito, S.; Wakamatsu, K.; Sagan, F.; Mitoraj, M.; Sarna, T. Photobleaching of pheomelanin increases its phototoxic potential: Physicochemical studies of synthetic pheomelanin subjected to aerobic photolysis. Pigment Cell Melanoma Res. 2019, 32, 359–372. [Google Scholar] [CrossRef]
- Żądło, A.; Ito, S.; Sarna, M.; Wakamatsu, K.; Mokrzyński, K.; Sarna, T. The role of hydrogen peroxide and singlet oxygen in the photodegradation of melanin. Photochem. Photobiol. Sci. 2020, 19, 654–667. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Kikuta, M.; Koike, S.; Szewczyk, G.; Sarna, M.; Zadlo, A.; Sarna, T.; Wakamatsu, K. Roles of reactive oxygen species in UVA-induced oxidation of 5,6-dihydroxyindole-2-carboxylic acid- melanin as studied by differential spectrophotometric method. Pigment Cell Melanoma Res. 2016, 29, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Sarna, M.; Zadlo, A.; Czuba-Pelech, B.; Urbanska, K. Nanomechanical phenotype of melanoma cells depends solely on the amount of endogenous pigment in the cells. Int. J. Mol. Sci. 2018, 19, 607. [Google Scholar] [CrossRef] [PubMed]
- Ozeki, H.; Ito, S.; Wakamatsu, K.; Thody, A.J. Spectrophotometric characterization of eumelanin and pheomelanin in hair. Pigment Cell Melanoma Res. 1996, 9, 265–270. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Ito, S.; Rees, J.L. The usefulness of 4-amino-3-hydroxyphenylalanine as a specific marker of pheomelanin. Pigment Cell Melanoma Res. 2002, 15, 225–232. [Google Scholar] [CrossRef]
- Ozeki, H.; Ito, S.; Wakamatsu, K.; Hirobe, T. Chemical characterization of hair melanins in various coat-color mutants of mice. J. Investig. Dermatol. 1995, 105, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Wakamatsu, K.; Nakanishi, Y.; Miyazaki, N.; Kolbe, L.; Ito, S. UVA-induced oxidative degradation of melanins: Fission of indole moiety in eumelanin and conversion to benzothiazole moiety in pheomelanin. Pigment Cell Melanoma Res. 2012, 25, 434–445. [Google Scholar] [CrossRef]
- Itoh, T.; Ito, S.; Wakamatsu, K. Effects of aging on hair color, melanosome morphology, and melanin composition in Japanese females. Int. J. Mol. Sci. 2019, 20, 3739. [Google Scholar] [CrossRef] [PubMed]
- Redmond, R.W.; Gamlin, J.N. A compilation of singlet oxygen yields from biologically relevant molecules. Photochem. Photobiol. 1999, 70, 391–475. [Google Scholar] [CrossRef]
- Wilkinson, F.; Helman, W.P.; Ross, A.B. Quantum Yields for the Photosensitized Formation of the Lowest Electronically Excited Singlet State of Molecular Oxygen in Solution. J. Phys. Chem. Ref. Data 1993, 22, 113–262. [Google Scholar] [CrossRef]
- Di Mascio, P.; Martinez, G.R.; Miyamoto, S.; Ronsein, G.E.; Medeiros, M.H.G.; Cadet, J. Singlet molecular oxygen reactions with nucleic acids, lipids, and proteins. Chem. Rev. 2019, 119, 2043–2086. [Google Scholar] [CrossRef] [PubMed]
- Sarna, T.; Menon, I.A.; Sealy, R.C. Photoinduced oxygen-consumption in melanin systems. Action spectra and quantum yields for pheomelanins. Photochem. Photobiol. 1984, 39, 805–809. [Google Scholar] [PubMed]
- Zadlo, A.; Szewczyk, G.; Sarna, M.; Kozinska, A.; Pilat, A.; Kaczara, P.; Sarna, T. Photoaging of retinal pigment epithelial melanosomes: The effect of photobleaching on morphology and reactivity of the pigment granules. Free Radic. Biol. Med. 2016, 97, 320–329. [Google Scholar] [CrossRef]
- Zareba, M.; Szewczyk, G.; Sarna, T.; Hong, L.; Simon, J.D.; Henry, M.M.; Burke, J.M. Effects of photodegradation on the physical and antioxidant properties of melanosomes isolated from retinal pigment epithelium. Photochem. Photobiol. 2006, 82, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Novellino, L.; Napolitano, A.; Prota, G. Isolation and characterization of mammalian eumelanins from hair and irides. Biochim. Biophys. Acta Gen. Subj. 2000, 1475, 295–306. [Google Scholar] [CrossRef]
- Zadlo, A.; Pilat, A.; Sarna, M.; Pawlak, A.; Sarn, T. Redox active transition metal ions make melanin susceptible to chemical degradation induced by organic peroxide. Cell Biochem. Biophys. 2017, 75, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Sidabras, J.W.; Mett, R.R.; Froncisz, W.; Camenisch, T.G.; Anderson, J.R.; Hyde, J.S. Multipurpose EPR loop-gap resonator and cylindrical TE011 cavity for aqueous samples at 94 GHz. Rev. Sci. Instrum. 2007, 78, 034701. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Del Bino, S.; Hirobe, T.; Wakamatsu, K. Improved HPLC conditions to determine eumelanin and pheomelanin contents in biological samples using an ion pair reagent. Int. J. Mol. Sci. 2020, 21, 5134. [Google Scholar] [CrossRef] [PubMed]
- Olchawa, M.M.; Szewczyk, G.M.; Zadlo, A.C.; Krzysztynska-Kuleta, O.I.; Sarna, T.J. The effect of aging and antioxidants on photoreactivity and phototoxicity of human melanosomes: An in vitro study. Pigment Cell Melanoma Res. 2020, in press. [Google Scholar] [CrossRef]
- Olchawa, M.M.; Szewczyk, G.M.; Zadlo, A.C.; Sarna, M.W.; Wnuk, D.; Sarna, T.J. The Effect of antioxidants on photoreactivity and phototoxic potential of RPE melanolipofuscin granules from human donors of different age. Antioxidants 2020, 9, 1044. [Google Scholar] [CrossRef]
- Zadlo, A.; Mokrzyński, K.; Ito, S.; Wakamatsu, K.; Sarna, T. The influence of iron on selected properties of synthetic pheomelanin. Cell Biochem. Biophys. 2020, 78, 181–189. [Google Scholar] [CrossRef]







| Type of Hair Melanin | Quantum Yield at 300 nm | Quantum Yield at 332 nm | Quantum Yield at 365 nm | Quantum Yield at 445 nm | Quenching Rate Constant [(mg/mL)−1s−1] |
|---|---|---|---|---|---|
| Red | (6.67 ± 0.53) × 10−3 | (8.42 ± 0.69) × 10−3 | (1.46 ± 0.09) × 10−2 | (5.03 ± 0.27) × 10−3 | (1.33 ± 0.12) × 105 |
| Blond | (3.02 ± 0.21) × 10−2 | (8.38 ± 0.70) × 10−2 | (4.22 ± 0.25) × 10−2 | (1.18 ± 0.08) × 10−2 | (1.12 ± 0.09) × 105 |
| Chestnut | (3.22 ± 0.23) × 10−2 | (5.79 ± 0.39) × 10−2 | (2.87 ± 0.20) × 10−2 | (6.15 ± 0.41) × 10−3 | (2.24 ± 0.18) × 105 |
| Black | (1.87 ± 0.12) × 10−3 | (1.09 ± 0.07) × 10−3 | (4.08 ± 0.21) × 10−3 | (2.01 ± 0.14) × 10−3 | (1.83 ± 0.11) × 105 |
| Melanin Subunit | Quantum Yield at 300 nm | Quantum Yield at 332 nm | Quantum Yield at 365 nm | Quantum Yield at 445 nm |
|---|---|---|---|---|
| DHBTCA | (1.97 ± 0.14) × 10−3 | (2.48 ± 0.16) × 10−3 | (1.72 ± 0.09) × 10−3 | (3.69 ± 0.18) × 10−4 |
| BZ-AA | (4.65 ± 0.23) × 10−3 | (4.92 ± 0.25) × 10−3 | (4.07 ± 0.21) × 10−3 | (1.01 ± 0.04) × 10−4 |
| DHICA | (9.24 ± 0.56) × 10−3 | (1.11 ± 0.07) × 10−2 | (3.74 ± 0.19) × 10−3 | (1.92 ± 0.15) × 10−4 |
| DHI | (2.03 ± 0.12) × 10−3 | (2.91 ± 0.23) × 10−3 | (7.49 ± 0.40) × 10−4 | (1.08 ± 0.04) × 10−4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mokrzynski, K.; Ito, S.; Wakamatsu, K.; Camenish, T.G.; Sarna, T.; Sarna, M. Photoreactivity of Hair Melanin from Different Skin Phototypes—Contribution of Melanin Subunits to the Pigments Photoreactive Properties. Int. J. Mol. Sci. 2021, 22, 4465. https://doi.org/10.3390/ijms22094465
Mokrzynski K, Ito S, Wakamatsu K, Camenish TG, Sarna T, Sarna M. Photoreactivity of Hair Melanin from Different Skin Phototypes—Contribution of Melanin Subunits to the Pigments Photoreactive Properties. International Journal of Molecular Sciences. 2021; 22(9):4465. https://doi.org/10.3390/ijms22094465
Chicago/Turabian StyleMokrzynski, Krystian, Shosuke Ito, Kazumasa Wakamatsu, Theodore G. Camenish, Tadeusz Sarna, and Michal Sarna. 2021. "Photoreactivity of Hair Melanin from Different Skin Phototypes—Contribution of Melanin Subunits to the Pigments Photoreactive Properties" International Journal of Molecular Sciences 22, no. 9: 4465. https://doi.org/10.3390/ijms22094465
APA StyleMokrzynski, K., Ito, S., Wakamatsu, K., Camenish, T. G., Sarna, T., & Sarna, M. (2021). Photoreactivity of Hair Melanin from Different Skin Phototypes—Contribution of Melanin Subunits to the Pigments Photoreactive Properties. International Journal of Molecular Sciences, 22(9), 4465. https://doi.org/10.3390/ijms22094465










