Suppression of Superficial Microglial Activation by Spinal Cord Stimulation Attenuates Neuropathic Pain Following Sciatic Nerve Injury in Rats
Abstract
:1. Introduction
2. Results
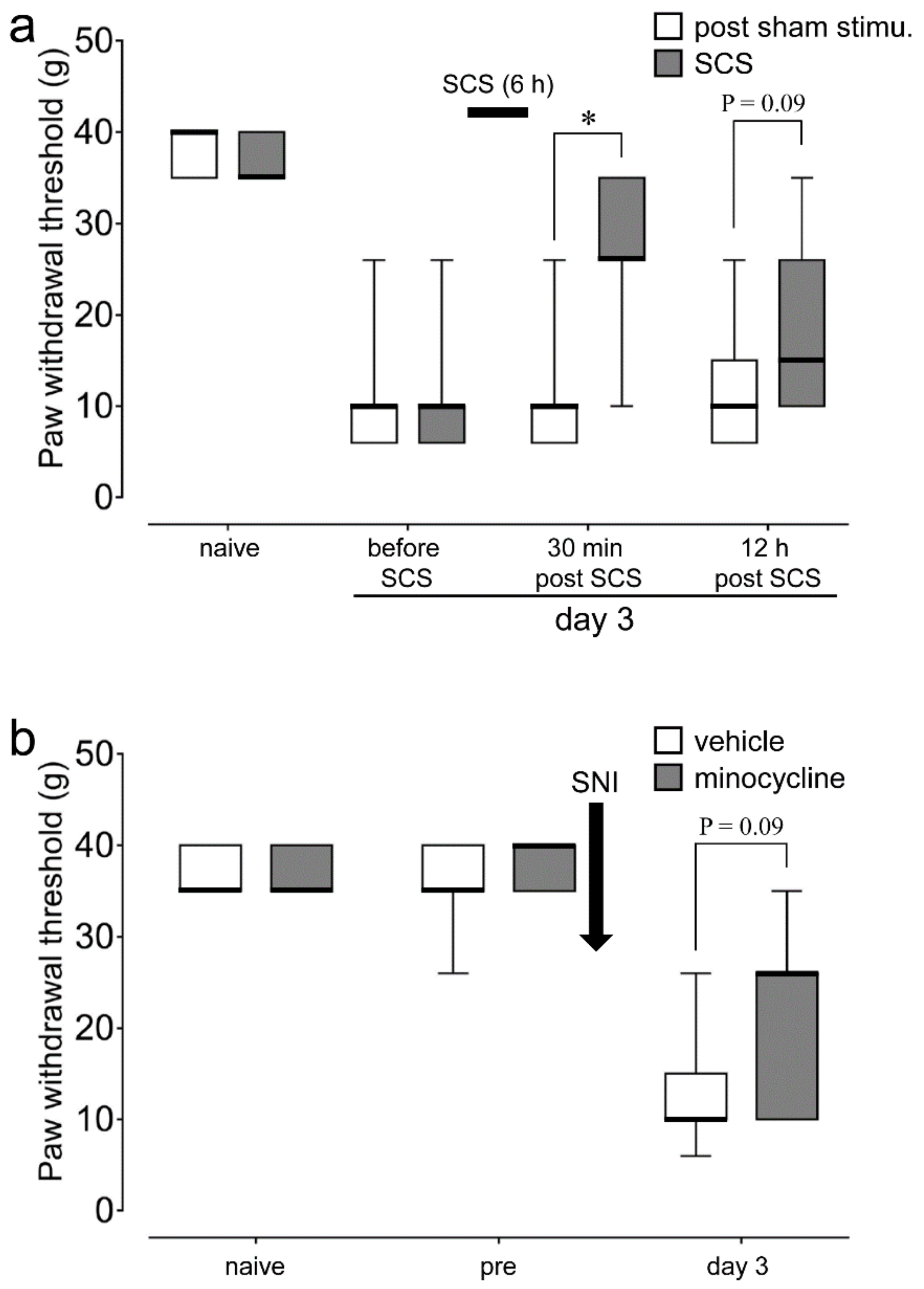
2.1. SCS and Minocycline Depressed Plantar Mechanical Hypersensitivity Following SNI
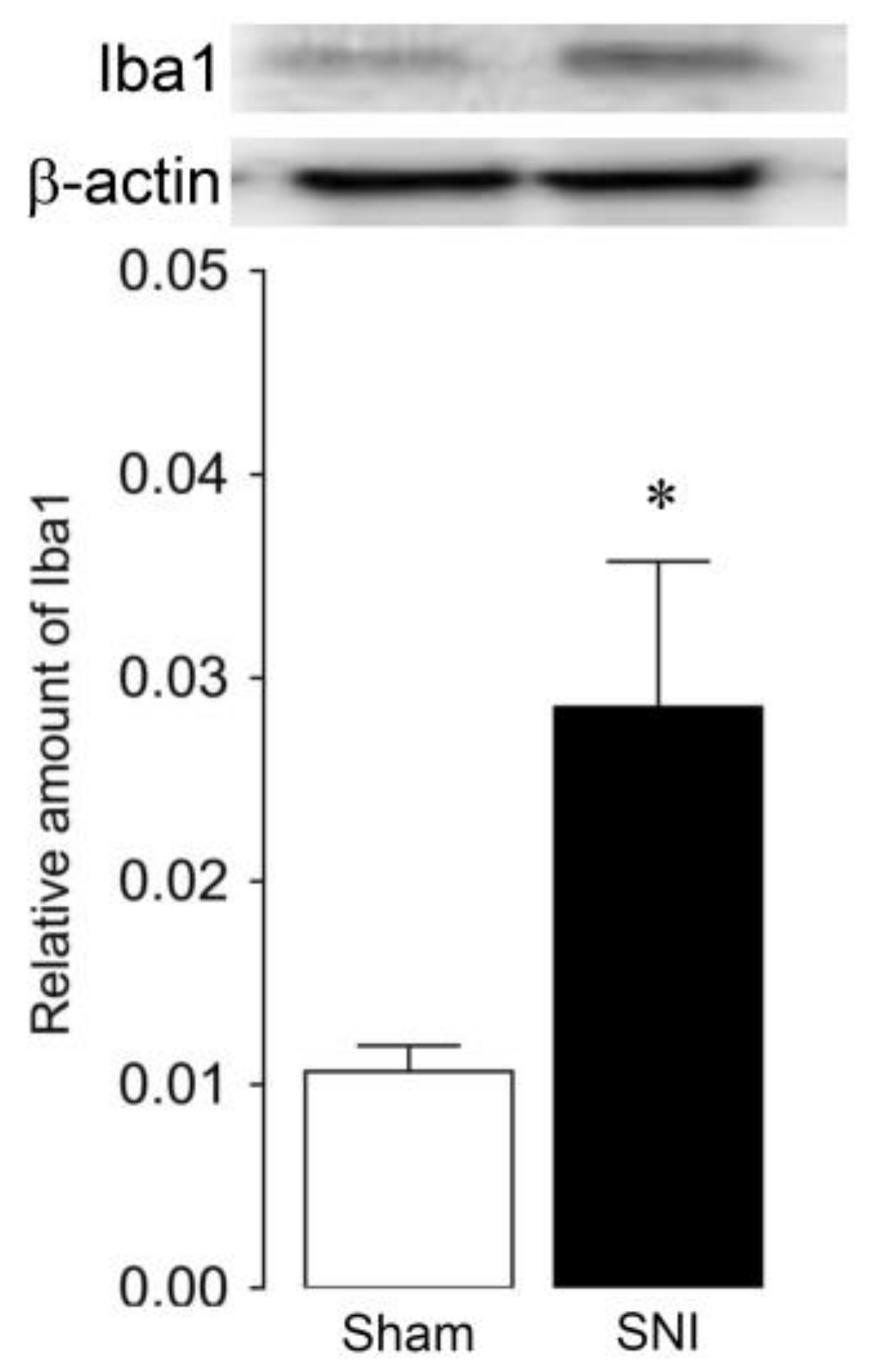
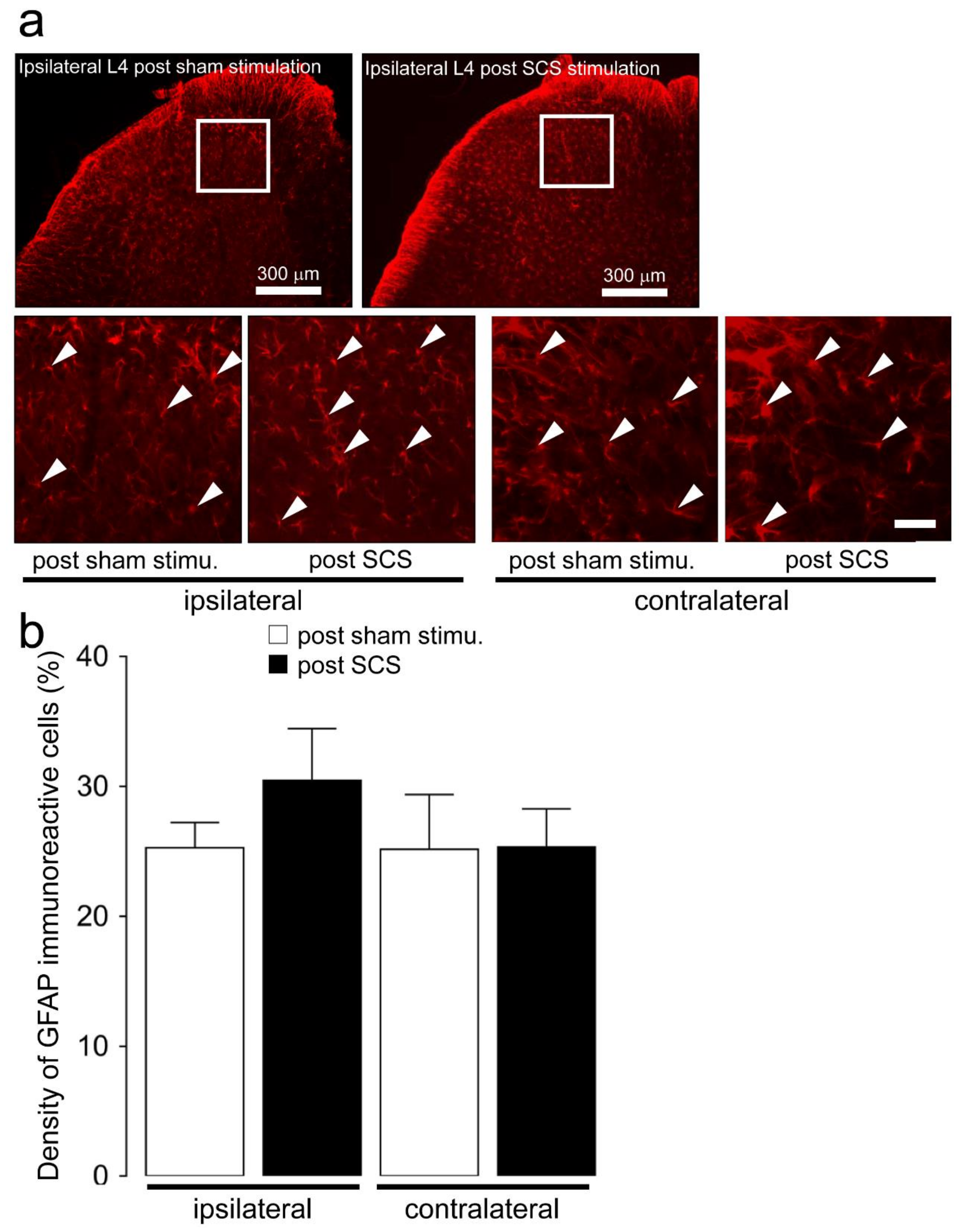
2.2. Microglial and Astroglial Activation in the L4 Dorsal Horn
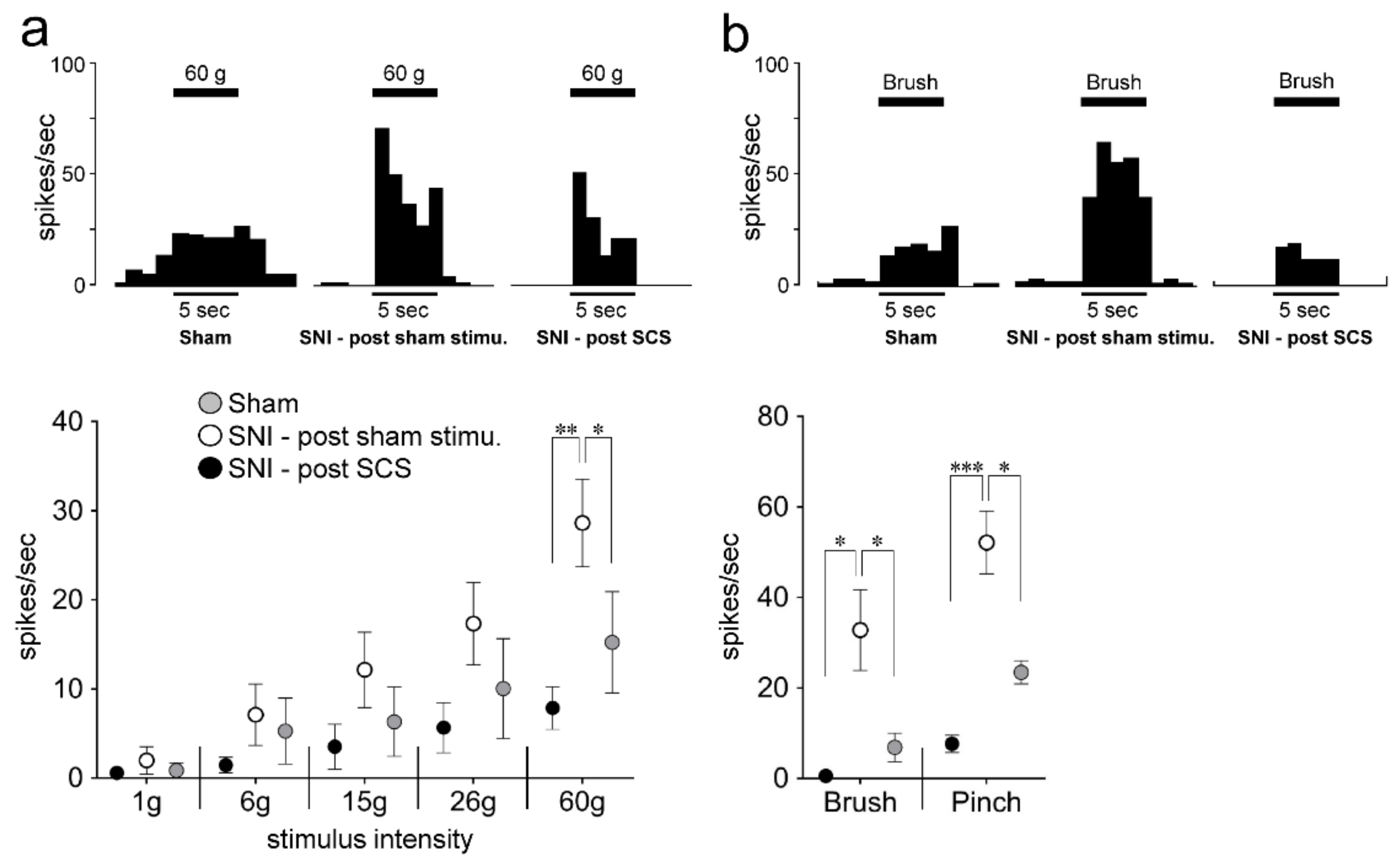
2.3. Effect of SCS on L4-S1 Nociceptive Neuronal Activity Following SNI
2.4. Cortical Responses to Electrical Stimulation of the Hind Paw
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Spared Nerve Injury Model Preparation and SCS Electrode Implantation
4.3. Plantar Mechanical Sensitivity
4.4. SCS
4.5. Effect of Minocycline Administration on Plantar Mechanical Sensitivity Following SNI
4.6. Immunohistochemistry
4.7. Western Blotting
4.8. Single Neuronal Recording
4.9. In Vivo Optical Imaging With a Voltage-Sensitive Dye
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SCS | spinal cord stimulation |
| SNI | spared nerve injury |
| SDH | spinal dorsal horn |
| CNS | central nervous system |
| i.p. | intraperitoneal |
| PWT | paw withdrawal threshold |
| MoT | motor threshold |
| PBS | phosphate-buffered saline |
| GFAP | glial fibrillary acidic protein |
| IR | immunoreactive |
| WDR | wide dynamic range |
| SD | standard deviations |
| ROI | region of interest |
| ANOVA | analysis of variance |
| SEM | standard error of the mean |
| TNFα | tumor necrosis factor alpha |
| NMDA | N-methyl-d-aspartic acid |
| IL-1β | interleukin-1 beta |
References
- Baron, R.; Binder, A.; Wasner, G. Neuropathic pain: Diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. 2010, 9, 807–819. [Google Scholar] [CrossRef]
- Sicras-Mainar, A.; Rejas-Gutierrez, J.; Perez-Paramo, M.; Navarro-Artieda, R. Cost of treatment of peripheral neuropathic pain with pregabalin or gabapentin in routine clinical practice: Impact of their loss of exclusivity. J. Eval. Clin. Pr. 2017, 23, 402–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Kaisy, A.; Van Buyten, J.P.; Amirdelfan, K.; Gliner, B.; Caraway, D.; Subbaroyan, J.; Rotte, A.; Kapural, L. Opioid-sparing effects of 10 kHz spinal cord stimulation: A review of clinical evidence. Ann. N. Y. Acad. Sci. 2020, 1462, 53–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rock, A.K.; Truong, H.; Park, Y.L.; Pilitsis, J.G. Spinal Cord Stimulation. Neurosurg. Clin. N. Am. 2019, 30, 169–194. [Google Scholar] [CrossRef] [PubMed]
- Decosterd, I.; Woolf, C.J. Spared nerve injury: An animal model of persistent peripheral neuropathic pain. Pain 2000, 87, 149–158. [Google Scholar] [CrossRef]
- Gong, W.Y.; Johanek, L.M.; Sluka, K.A. A Comparison of the Effects of Burst and Tonic Spinal Cord Stimulation on Hyperalgesia and Physical Activity in an Animal Model of Neuropathic Pain. Anesth. Analg. 2016, 122, 1178–1185. [Google Scholar] [CrossRef]
- Shechter, R.; Yang, F.; Xu, Q.; Cheong, Y.K.; He, S.Q.; Sdrulla, A.; Carteret, A.F.; Wacnik, P.W.; Dong, X.; Meyer, R.A.; et al. Conventional and kilohertz-frequency spinal cord stimulation produces intensity- and frequency-dependent inhibition of mechanical hypersensitivity in a rat model of neuropathic pain. Anesthesiology 2013, 119, 422–432. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.; Viisanen, H.; Meyerson, B.A.; Pertovaara, A.; Linderoth, B. Efficacy of kilohertz-frequency and conventional spinal cord stimulation in rat models of different pain conditions. Neuromodulation 2014, 17, 226–234. [Google Scholar] [CrossRef]
- Tilley, D.M.; Vallejo, R.; Kelley, C.A.; Benyamin, R.; Cedeno, D.L. A continuous spinal cord stimulation model attenuates pain-related behavior in vivo following induction of a peripheral nerve injury. Neuromodulation 2015, 18, 171–176. [Google Scholar] [CrossRef]
- Crosby, N.D.; Weisshaar, C.L.; Smith, J.R.; Zeeman, M.E.; Goodman-Keiser, M.D.; Winkelstein, B.A. Burst and Tonic Spinal Cord Stimulation Differentially Activate GABAergic Mechanisms to Attenuate Pain in a Rat Model of Cervical Radiculopathy. IEEE Trans. Biomed. Eng. 2015, 62, 1604–1613. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, Y.Q.; Qadri, Y.J.; Serhan, C.N.; Ji, R.R. Microglia in Pain: Detrimental and Protective Roles in Pathogenesis and Resolution of Pain. Neuron 2018, 100, 1292–1311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gui, Y.; Duan, S.; Xiao, L.; Tang, J.; Li, A. Bexarotent attenuated CCI-induced spinal neuroinflammation and neuropathic pain by targeting MKP-1. J. Pain 2019. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Wen, J.; Selvaraj, P.; Tanaka, M.; Moran, S.; Zhang, Y. Therapeutic Effect of the Substrate-Selective COX-2 Inhibitor IMMA in the Animal Model of Chronic Constriction Injury. Front. Pharm. 2018, 9, 1481. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.X.; Wang, S.K.; Yao, P.W.; Liao, G.J.; Na, X.D.; Li, Y.Y.; Zeng, W.A.; Liu, X.G.; Zang, Y. Early CALP2 expression and microglial activation are potential inducers of spinal IL-6 up-regulation and bilateral pain following motor nerve injury. J. Neurochem. 2018, 145, 154–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woller, S.A.; Eddinger, K.A.; Corr, M.; Yaksh, T.L. An overview of pathways encoding nociception. Clin. Exp. Rheumatol. 2018, 36, 172. [Google Scholar]
- Takeuchi, Y.; Osaki, H.; Yagasaki, Y.; Katayama, Y.; Miyata, M. Afferent Fiber Remodeling in the Somatosensory Thalamus of Mice as a Neural Basis of Somatotopic Reorganization in the Brain and Ectopic Mechanical Hypersensitivity after Peripheral Sensory Nerve Injury. eNeuro 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Morrison, H.W.; Filosa, J.A. A quantitative spatiotemporal analysis of microglia morphology during ischemic stroke and reperfusion. J. Neuroinflammation 2013, 10, 4. [Google Scholar] [CrossRef] [Green Version]
- Morales-Botello, M.L.; Aguilar, J.; Foffani, G. Imaging the spatio-temporal dynamics of supragranular activity in the rat somatosensory cortex in response to stimulation of the paws. PLoS ONE 2012, 7, e40174. [Google Scholar] [CrossRef]
- Nent, E.; Nozaki, C.; Schmole, A.C.; Otte, D.; Zimmer, A. CB2 receptor deletion on myeloid cells enhanced mechanical allodynia in a mouse model of neuropathic pain. Sci. Rep. 2019, 9, 7468. [Google Scholar] [CrossRef]
- Tsuda, M.; Mizokoshi, A.; Shigemoto-Mogami, Y.; Koizumi, S.; Inoue, K. Activation of p38 mitogen-activated protein kinase in spinal hyperactive microglia contributes to pain hypersensitivity following peripheral nerve injury. Glia 2004, 45, 89–95. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chen, Y.L.; Li, A.H.; Lu, J.C.; Wang, H.L. Minocycline, a microglial inhibitor, blocks spinal CCL2-induced heat hyperalgesia and augmentation of glutamatergic transmission in substantia gelatinosa neurons. J. Neuroinflammation 2014, 11, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonekatsu, M.; Taniguchi, W.; Yamanaka, M.; Nishio, N.; Tsutsui, S.; Yamada, H.; Yoshida, M.; Nakatsuka, T. Interferon-gamma potentiates NMDA receptor signaling in spinal dorsal horn neurons via microglia-neuron interaction. Mol. Pain 2016, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, S.; Kwon, Y.B. The CCL2 elevation in primary afferent fibers produces zymosan-induced hyperalgesia through microglia-mediated neuronal activation in the spinal dorsal horn. Brain Res. Bull. 2019, 149, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Ji, R.R.; Berta, T.; Nedergaard, M. Glia and pain: Is chronic pain a gliopathy? Pain 2013, 154, S10–S28. [Google Scholar] [CrossRef] [PubMed]
- Schafers, M.; Geis, C.; Svensson, C.I.; Luo, Z.D.; Sommer, C. Selective increase of tumour necrosis factor-alpha in injured and spared myelinated primary afferents after chronic constrictive injury of rat sciatic nerve. Eur. J. Neurosci. 2003, 17, 791–804. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.T.; Xin, W.J.; Zang, Y.; Wu, C.Y.; Liu, X.G. The role of tumor necrosis factor-alpha in the neuropathic pain induced by Lumbar 5 ventral root transection in rat. Pain 2006, 123, 306–321. [Google Scholar] [CrossRef]
- Park, C.K.; Lu, N.; Xu, Z.Z.; Liu, T.; Serhan, C.N.; Ji, R.R. Resolving TRPV1- and TNF-alpha-mediated spinal cord synaptic plasticity and inflammatory pain with neuroprotectin D1. J. Neurosci. 2011, 31, 15072–15085. [Google Scholar] [CrossRef]
- Xu, Z.Z.; Zhang, L.; Liu, T.; Park, J.Y.; Berta, T.; Yang, R.; Serhan, C.N.; Ji, R.R. Resolvins RvE1 and RvD1 attenuate inflammatory pain via central and peripheral actions. Nat. Med. 2010, 16, 592–597. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Nei, H.; Dougherty, P.M. A p38 mitogen-activated protein kinase-dependent mechanism of disinhibition in spinal synaptic transmission induced by tumor necrosis factor-alpha. J. Neurosci. 2010, 30, 12844–12855. [Google Scholar] [CrossRef]
- Yan, X.; Weng, H.R. Endogenous interleukin-1beta in neuropathic rats enhances glutamate release from the primary afferents in the spinal dorsal horn through coupling with presynaptic N-methyl-D-aspartic acid receptors. J. Biol. Chem. 2013, 288, 30544–30557. [Google Scholar] [CrossRef] [Green Version]
- Kawasaki, Y.; Zhang, L.; Cheng, J.K.; Ji, R.R. Cytokine mechanisms of central sensitization: Distinct and overlapping role of interleukin-1beta, interleukin-6, and tumor necrosis factor-alpha in regulating synaptic and neuronal activity in the superficial spinal cord. J. Neurosci. 2008, 28, 5189–5194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, R.X.; Li, A.; Liu, B.; Wang, L.; Ren, K.; Zhang, H.; Berman, B.M.; Lao, L. IL-1ra alleviates inflammatory hyperalgesia through preventing phosphorylation of NMDA receptor NR-1 subunit in rats. Pain 2008, 135, 232–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, K.; Dubner, R. Interactions between the immune and nervous systems in pain. Nat. Med. 2010, 16, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Kiyomoto, M.; Shinoda, M.; Honda, K.; Nakaya, Y.; Dezawa, K.; Katagiri, A.; Kamakura, S.; Inoue, T.; Iwata, K. p38 phosphorylation in medullary microglia mediates ectopic orofacial inflammatory pain in rats. Mol. Pain 2015, 11, 48. [Google Scholar] [CrossRef] [Green Version]
- Lu, Y.; Zhao, L.X.; Cao, D.L.; Gao, Y.J. Spinal injection of docosahexaenoic acid attenuates carrageenan-induced inflammatory pain through inhibition of microglia-mediated neuroinflammation in the spinal cord. Neuroscience 2013, 241, 22–31. [Google Scholar] [CrossRef]
- Berta, T.; Park, C.K.; Xu, Z.Z.; Xie, R.G.; Liu, T.; Lu, N.; Liu, Y.C.; Ji, R.R. Extracellular caspase-6 drives murine inflammatory pain via microglial TNF-alpha secretion. J. Clin. Invest. 2014, 124, 1173–1186. [Google Scholar] [CrossRef] [Green Version]
- Stephens, K.E.; Chen, Z.; Sivanesan, E.; Raja, S.N.; Linderoth, B.; Taverna, S.D.; Guan, Y. RNA-seq of spinal cord from nerve-injured rats after spinal cord stimulation. Mol. Pain 2018, 14. [Google Scholar] [CrossRef] [Green Version]
- Vallejo, R.; Tilley, D.M.; Cedeno, D.L.; Kelley, C.A.; DeMaegd, M.; Benyamin, R. Genomics of the Effect of Spinal Cord Stimulation on an Animal Model of Neuropathic Pain. Neuromodulation 2016, 19, 576–586. [Google Scholar] [CrossRef]
- Shinoda, M.; Ogino, A.; Ozaki, N.; Urano, H.; Hironaka, K.; Yasui, M.; Sugiura, Y. Involvement of TRPV1 in nociceptive behavior in a rat model of cancer pain. J. Pain 2008, 9, 687–699. [Google Scholar] [CrossRef]
- Asai, H.; Ozaki, N.; Shinoda, M.; Nagamine, K.; Tohnai, I.; Ueda, M.; Sugiura, Y. Heat and mechanical hyperalgesia in mice model of cancer pain. Pain 2005, 117, 19–29. [Google Scholar] [CrossRef]
- Ma, H.; Yao, C.; Ma, P.; Zhou, J.; Gong, S.; Tao, J.; Yu, X.M.; Jiang, X. Src activation in the hypothalamic arcuate nucleus may play an important role in pain hypersensitivity. Sci. Rep. 2019, 9, 3827. [Google Scholar] [CrossRef] [PubMed]
- Fujita, S.; Koshikawa, N.; Kobayashi, M. GABA(B) receptors accentuate neural excitation contrast in rat insular cortex. Neuroscience 2011, 199, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Chemla, S.; Chavane, F. Voltage-sensitive dye imaging: Technique review and models. J. Physiol. Paris 2010, 104, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, J.; Yu, T.; Du, D.; Jiang, W. Minocycline can delay the development of morphine tolerance, but cannot reverse existing tolerance in the maintenance period of neuropathic pain in rats. Clin. Exp. Pharm. Physiol. 2015, 42, 94–101. [Google Scholar] [CrossRef]
- Kumar, K.; North, R.; Taylor, R.; Sculpher, M.; Van den Abeele, C.; Gehring, M.; Jacques, L.; Eldabe, S.; Meglio, M.; Molet, J.; et al. Spinal Cord Stimulation vs. Conventional Medical Management: A Prospective, Randomized, Controlled, Multicenter Study of Patients with Failed Back Surgery Syndrome (PROCESS Study). Neuromodulation 2005, 8, 213–218. [Google Scholar] [CrossRef]
- Kumar, K.; Taylor, R.S.; Jacques, L.; Eldabe, S.; Meglio, M.; Molet, J.; Thomson, S.; O’Callaghan, J.; Eisenberg, E.; Milbouw, G.; et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: A multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain 2007, 132, 179–188. [Google Scholar] [CrossRef]
- Turner, J.A.; Loeser, J.D.; Bell, K.G. Spinal cord stimulation for chronic low back pain: A systematic literature synthesis. Neurosurgery 1995, 37, 1088–1095. [Google Scholar] [CrossRef]
- Kumar, K.; Wilson, J.R.; Taylor, R.S.; Gupta, S. Complications of spinal cord stimulation, suggestions to improve outcome, and financial impact. J. Neurosurg. Spine 2006, 5, 191–203. [Google Scholar] [CrossRef]
- Widerstrom-Noga, E. Neuropathic Pain and Spinal Cord Injury: Phenotypes and Pharmacological Management. Drugs 2017, 77, 967–984. [Google Scholar] [CrossRef]
- Dombovy-Johnson, M.L.; Hunt, C.L.; Morrow, M.M.; Lamer, T.J.; Pittelkow, T.P. Current Evidence Lacking to Guide Clinical Practice for Spinal Cord Stimulation in the Treatment of Neuropathic Pain in Spinal Cord Injury: A Review of the Literature and a Proposal for Future Study. Pain Pr. 2020, 20, 325–335. [Google Scholar] [CrossRef]
- Maheshwari, A.; Pope, J.E.; Deer, T.R.; Falowski, S. Advanced methods of spinal stimulation in the treatment of chronic pain: Pulse trains, waveforms, frequencies, targets, and feedback loops. Expert Rev. Med. Devices 2019, 16, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Sankarasubramanian, V.; Harte, S.E.; Chiravuri, S.; Harris, R.E.; Brummett, C.M.; Patil, P.G.; Clauw, D.J.; Lempka, S.F. Objective Measures to Characterize the Physiological Effects of Spinal Cord Stimulation in Neuropathic Pain: A Literature Review. Neuromodulation 2019, 22, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar] [CrossRef]
- Soma, K.; Shinoda, M.; Hayashi, Y.; Kanno, K.; Shirakwa, T.; Iwata, K. Involvement of TNFalpha in the enhancement of hypersensitivity in the adulthood-injured face associated with facial injury in infancy. Neurosci. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, S.; Shinoda, M.; Hayashi, Y.; Saito, H.; Asano, S.; Kubo, A.; Shibuta, I.; Furukawa, A.; Toyofuku, A.; Iwata, K. Increase in IGF-1 Expression in the Injured Infraorbital Nerve and Possible Implications for Orofacial Neuropathic Pain. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiyomoto, M.; Shinoda, M.; Okada-Ogawa, A.; Noma, N.; Shibuta, K.; Tsuboi, Y.; Sessle, B.J.; Imamura, Y.; Iwata, K. Fractalkine signaling in microglia contributes to ectopic orofacial pain following trapezius muscle inflammation. J. Neurosci. 2013, 33, 7667–7680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gong, W.; Johanek, L.M.; Sluka, K.A. Spinal cord stimulation reduces mechanical hyperalgesia and restores physical activity levels in animals with noninflammatory muscle pain in a frequency-dependent manner. Anesth. Analg. 2014, 119, 186–195. [Google Scholar] [CrossRef]
- Shibuta, K.; Suzuki, I.; Shinoda, M.; Tsuboi, Y.; Honda, K.; Shimizu, N.; Sessle, B.J.; Iwata, K. Organization of hyperactive microglial cells in trigeminal spinal subnucleus caudalis and upper cervical spinal cord associated with orofacial neuropathic pain. Brain Res. 2012, 1451, 74–86. [Google Scholar] [CrossRef]
- Rojewska, E.; Korostynski, M.; Przewlocki, R.; Przewlocka, B.; Mika, J. Expression profiling of genes modulated by minocycline in a rat model of neuropathic pain. Mol. Pain 2014, 10, 47. [Google Scholar] [CrossRef] [Green Version]
- Inoue, K.; Tsuda, M. Microglia in neuropathic pain: Cellular and molecular mechanisms and therapeutic potential. Nat. Rev. Neurosci. 2018, 19, 138–152. [Google Scholar] [CrossRef]
- Iwata, K.; Imai, T.; Tsuboi, Y.; Tashiro, A.; Ogawa, A.; Morimoto, T.; Masuda, Y.; Tachibana, Y.; Hu, J. Alteration of medullary dorsal horn neuronal activity following inferior alveolar nerve transection in rats. J. Neurophysiol. 2001, 86, 2868–2877. [Google Scholar] [CrossRef] [PubMed]
- Fujita, S.; Kaneko, M.; Nakamura, H.; Kobayashi, M. Spatiotemporal Profiles of Proprioception Processed by the Masseter Muscle Spindles in Rat Cerebral Cortex: An Optical Imaging Study. Front. Neural Circuits 2017, 11, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujita, S.; Zama, M.; Kaneko, K.; Murayama, S.; Ohtani, S.; Usui, M.; Noma, D.; Tsuda, H.; Yamamoto, K.; Kobayashi, M. Menthol-induced facilitation of cerebrocortical excitatory propagation induced by air puff stimulation of the nasal cavity in the rat: An optical imaging study. Neurosci. Lett. 2018, 696, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Fujita, S.; Yamamoto, K.; Kobayashi, M. Trigeminal Nerve Transection-Induced Neuroplastic Changes in the Somatosensory and Insular Cortices in a Rat Ectopic Pain Model. eNeuro 2019, 6. [Google Scholar] [CrossRef] [Green Version]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shinoda, M.; Fujita, S.; Sugawara, S.; Asano, S.; Koyama, R.; Fujiwara, S.; Soma, K.; Tamagawa, T.; Matsui, T.; Ikutame, D.; et al. Suppression of Superficial Microglial Activation by Spinal Cord Stimulation Attenuates Neuropathic Pain Following Sciatic Nerve Injury in Rats. Int. J. Mol. Sci. 2020, 21, 2390. https://doi.org/10.3390/ijms21072390
Shinoda M, Fujita S, Sugawara S, Asano S, Koyama R, Fujiwara S, Soma K, Tamagawa T, Matsui T, Ikutame D, et al. Suppression of Superficial Microglial Activation by Spinal Cord Stimulation Attenuates Neuropathic Pain Following Sciatic Nerve Injury in Rats. International Journal of Molecular Sciences. 2020; 21(7):2390. https://doi.org/10.3390/ijms21072390
Chicago/Turabian StyleShinoda, Masamichi, Satoshi Fujita, Shiori Sugawara, Sayaka Asano, Ryo Koyama, Shintaro Fujiwara, Kumi Soma, Takaaki Tamagawa, Tomoyuki Matsui, Daisuke Ikutame, and et al. 2020. "Suppression of Superficial Microglial Activation by Spinal Cord Stimulation Attenuates Neuropathic Pain Following Sciatic Nerve Injury in Rats" International Journal of Molecular Sciences 21, no. 7: 2390. https://doi.org/10.3390/ijms21072390





