Shift of Choline/Betaine Pathway in Recombinant Pseudomonas for Cobalamin Biosynthesis and Abiotic Stress Protection
Abstract
1. Introduction
2. Results and Discussion
2.1. The Effect of Recombinant N-Methyltransferases Expression on Bacterial Growth Rate under Stress Conditions
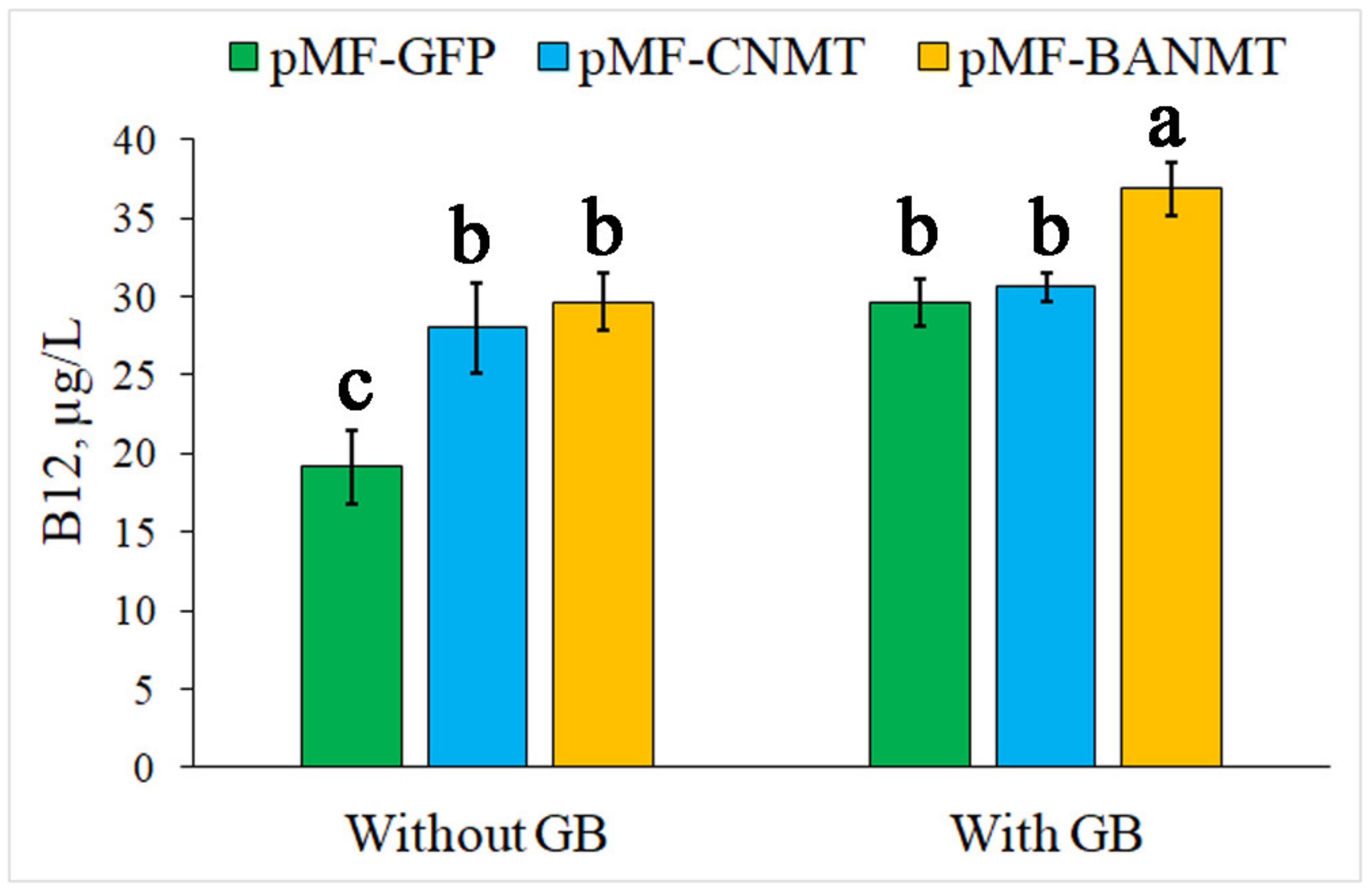
2.2. The Effect of Recombinant N-Methyltransferases Expression on Vitamin B12 Production
3. Materials and Methods
3.1. Bacterial Strains and Culture Conditions
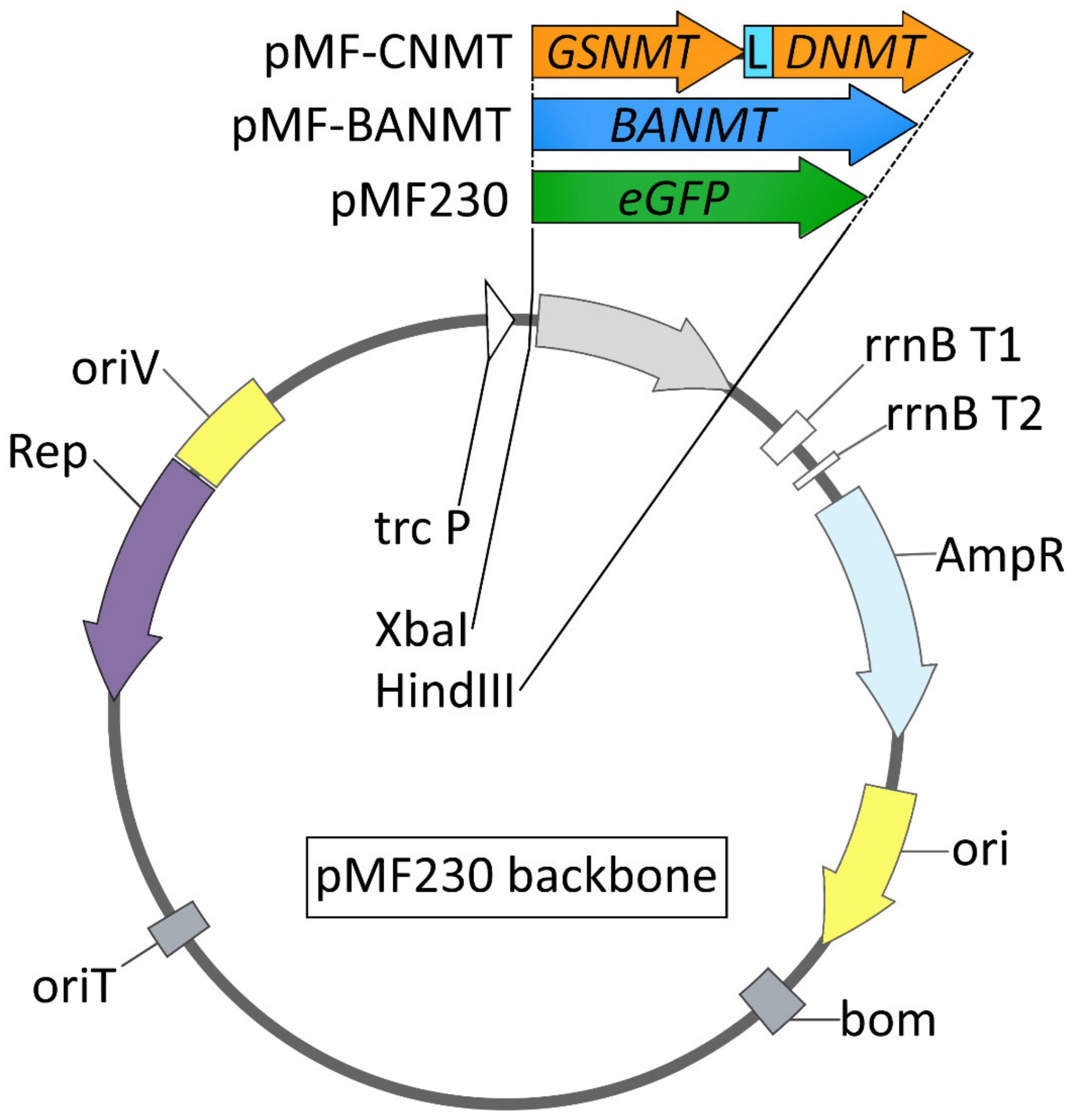
3.2. Construction of Recombinant Plasmids and Gene Transfer by Conjugation
3.3. Mass Spectrometry-Based Identification of β-Alanine Betaine and Glycine Betaine
3.4. Determination of Bacterial Growth Rate under Abiotic Stress Conditions
3.5. Measurement of Vitamin B12 Concentration
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Caspi, R.; Billington, R.; Keseler, I.M.; Kothari, A.; Krummenacker, M.; Midford, P.E.; Ong, W.K.; Paley, S.; Subhraveti, P.; Karp, P.D. The MetaCyc database of metabolic pathways and enzymes—A 2019 update. Nucleic Acids Res. 2020, 48, D445–D453. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.K.; Paal, M.C.; Donohue, T.M., Jr.; Ganesan, M.; Osna, N.A.; Kharbanda, K.K. Beneficial effects of betaine: A comprehensive review. Biology 2021, 10, 456. [Google Scholar] [CrossRef]
- Romano, K.A.; Martinez-Del Campo, A.; Kasahara, K.; Chittim, C.L.; Vivas, E.I.; Amador-Noguez, D.; Balskus, E.P.; Rey, F.E. Metabolic, epigenetic, and transgenerational effects of gut bacterial choline consumption. Cell Host Microbe 2017, 22, 279–290. [Google Scholar] [CrossRef]
- Arias, N.; Arboleya, S.; Allison, J.; Kaliszewska, A.; Higarza, S.G.; Gueimonde, M.; Arias, J.L. The relationship between choline bioavailability from diet, intestinal microbiota composition, and its modulation of human diseases. Nutrients 2020, 12, 2340. [Google Scholar] [CrossRef]
- Rajakovich, L.J.; Fu, B.; Bollenbach, M.; Balskus, E.P. Elucidation of an anaerobic pathway for metabolism of l-carnitine-derived γ-butyrobetaine to trimethylamine in human gut bacteria. Proc. Natl. Acad. Sci. USA 2021, 118, e2101498118. [Google Scholar] [CrossRef] [PubMed]
- Lentine, D.C.; Buzby, G.P. Choline and vitamin B12 deficiencies are interrelated in folate-replete long-term total parenteral nutrition patients. J. Parenter. Enter. Nutr. 2002, 26, 57–62. [Google Scholar]
- Rodionov, D.A.; Arzamasov, A.A.; Khoroshkin, M.S.; Iablokov, S.N.; Leyn, S.A.; Peterson, S.N.; Novichkov, P.S.; Osterman, A.L. Micronutrient Requirements and Sharing Capabilities of the Human Gut Microbiome. Front. Microbiol. 2019, 10, 1316. [Google Scholar] [CrossRef] [PubMed]
- Crespo, A.; Blanco-Cabra, N.; Torrents, E. Aerobic vitamin B12 biosynthesis is essential for Pseudomonas aeruginosa class II ribonucleotide reductase activity during planktonic and biofilm growth. Front. Microbiol. 2018, 9, 986. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, W.H.; Buffa, J.A.; Fu, X.; Britt, E.B.; Koeth, R.A.; Levison, B.S.; Fan, Y.; Wu, Y.; Hazen, S.L. Prognostic value of choline and betaine depends on intestinal microbiota-generated metabolite trimethylamine-N-oxide. Eur. Heart J. 2014, 35, 904–910. [Google Scholar] [CrossRef]
- Yu, Z.-L.; Zhang, L.-Y.; Jiang, X.-M.; Xue, C.-H.; Chi, N.; Zhang, T.; Wang, Y.-M. Effects of dietary choline, betaine, and L-carnitine on the generation of trimethylamine-N-oxide in healthy mice. J. Food Sci. 2020, 85, 2207–2215. [Google Scholar] [CrossRef]
- Wang, Z.J.; Wang, H.Y.; Li, Y.L.; Chua, J.; Huang, M.-Z.; Zhuang, Y.-P.; Zhanga, S.L. Improved vitamin B12 production by step-wise reduction of oxygen uptake rate under dissolved oxygen limiting level during fermentation process. Bioresour. Technol. 2010, 101, 2845–2852. [Google Scholar] [CrossRef]
- Xia, W.; Peng, W.-F.; Chen, W.; Li, K.-T. Interactive performances of betaine on the metabolic processes of Pseudomonas denitrificans. J. Ind. Microbiol. Biotechnol. 2015, 42, 273–278. [Google Scholar] [CrossRef]
- Balabanova, L.; Averianova, L.; Marchenok, M.; Son, O.; Tekutyeva, L. Microbial and genetic resources for cobalamin (vitamin B12) biosynthesis: From ecosystems to industrial biotechnology. Int. J. Mol. Sci. 2021, 22, 4522. [Google Scholar] [CrossRef] [PubMed]
- Li, K.T.; Liu, D.H.; Li, Y.L.; Chu, J.; Wang, Y.H.; Zhuang, Y.P.; Zhang, S.L. Improved large-scale production of vitamin B12 by Pseudomonas denitrificans with betaine feeding. Bioresour. Technol. 2008, 99, 8516–8520. [Google Scholar] [CrossRef] [PubMed]
- Waditee, R.; Tanaka, Y.; Aoki, K.; Hibino, T.; Jikuya, H.; Takano, J.; Takabe, T.; Takabe, T. Isolation and functional characterization of N-methyltransferases that catalyze betaine synthesis from glycine in a halotolerant photosynthetic organism Aphanothece halophytica. J. Biol. Chem. 2003, 278, 4932–4942. [Google Scholar] [CrossRef]
- Raman, S.B.; Rathinasabapathi, B. Beta-alanine N-methyltransferase of Limonium latifolium. cDNA cloning and functional expression of a novel N-methyltransferase implicated in the synthesis of the osmoprotectant beta-alanine betaine. Plant Physiol. 2003, 132, 1642–1651. [Google Scholar] [CrossRef][Green Version]
- Metris, A.; George, S.M.; Mulholland, F.; Carter, A.T.; Baranyi, J. Metabolic shift of Escherichia coli under salt stress in the presence of glycine betaine. Appl. Environ. Microbiol. 2014, 80, 4745–4756. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, Q.; Hao, G.; Liu, X.; Zhao, J.; Chen, Y.; Zhang, H.; Chen, W. The protective role of glycine betaine in Lactobacillus plantarum ST-III against salt stress. Food Control 2014, 44, 208–213. [Google Scholar] [CrossRef]
- Robert, H.; Le Marrec, C.; Blanco, C.; Jebbar, M. Glycine betaine, carnitine, and choline enhance salinity tolerance and prevent the accumulation of sodium to a level inhibiting growth of Tetragenococcus halophila. Appl. Environ. Microbiol. 2000, 66, 509–517. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Q.; Gao, X.; Zhang, Y. Biosynthesis and uptake of glycine betaine as cold-stress response to low temperature in fish pathogen Vibrio anguillarum. J. Microbiol. 2017, 55, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.; Bremer, E. Protection of Bacillus subtilis against cold stress via compatible-solute acquisition. J. Bacteriol. 2011, 193, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Li, K.-T.; Yang, Y.; Cheng, X. Revealing the promoting effect of betaine on vitamin B12 biosynthetic pathway of Pseudomonas denitrificans by using a proteomics analysis. Curr. Pharm. Biotechnol. 2022, 23, 466–475. [Google Scholar] [CrossRef]
- Balabanova, L.; Shkryl, Y.; Slepchenko, L.; Cheraneva, D.; Podvolotskaya, A.; Bakunina, I.; Nedashkovskaya, O.; Son, O.; Tekutyeva, L. Genomic features of a food-derived Pseudomonas aeruginosa strain PAEM and biofilm-associated gene expression under a marine bacterial α-galactosidase. Int. J. Mol. Sci. 2020, 21, 7666. [Google Scholar] [CrossRef] [PubMed]
- Nivens, D.E.; Ohman, D.E.; Williams, J.; Franklin, M.J. Role of alginate and its O acetylation in formation of Pseudomonas aeruginosa microcolonies and biofilms. J. Bacteriol. 2001, 183, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
- Brosius, J.; Erfle, M.; Storella, J. Spacing of the -10 and -35 regions in the tac promoter. J. Biol. Chem. 1985, 260, 3539–3541. [Google Scholar] [CrossRef]
- Franklin, M.J.; Ohman, D.E. Identification of algF in the alginate biosynthetic gene cluster of Pseudomonas aeruginosa which is required for alginate acetylation. J. Bacteriol. 1993, 175, 5057–5065. [Google Scholar] [CrossRef]
- Shkryl, Y.; Yugay, Y.; Avramenko, T.; Grigorchuk, V.; Gorpenchenko, T.; Grischenko, O.; Bulgakov, V. CRISPR/Cas9-mediated knockout of HOS1 reveals its role in the regulation of secondary metabolism in Arabidopsis thaliana. Plants 2021, 10, 104. [Google Scholar] [CrossRef]
- Figurski, D.H.; Helinski, D.R. Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc. Natl. Acad. Sci. USA 1979, 76, 1648–1652. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balabanova, L.; Pentekhina, I.; Nedashkovskaya, O.; Degtyarenko, A.; Grigorchuk, V.; Yugay, Y.; Vasyutkina, E.; Kudinova, O.; Seitkalieva, A.; Slepchenko, L.; et al. Shift of Choline/Betaine Pathway in Recombinant Pseudomonas for Cobalamin Biosynthesis and Abiotic Stress Protection. Int. J. Mol. Sci. 2022, 23, 13934. https://doi.org/10.3390/ijms232213934
Balabanova L, Pentekhina I, Nedashkovskaya O, Degtyarenko A, Grigorchuk V, Yugay Y, Vasyutkina E, Kudinova O, Seitkalieva A, Slepchenko L, et al. Shift of Choline/Betaine Pathway in Recombinant Pseudomonas for Cobalamin Biosynthesis and Abiotic Stress Protection. International Journal of Molecular Sciences. 2022; 23(22):13934. https://doi.org/10.3390/ijms232213934
Chicago/Turabian StyleBalabanova, Larissa, Iuliia Pentekhina, Olga Nedashkovskaya, Anton Degtyarenko, Valeria Grigorchuk, Yulia Yugay, Elena Vasyutkina, Olesya Kudinova, Aleksandra Seitkalieva, Lubov Slepchenko, and et al. 2022. "Shift of Choline/Betaine Pathway in Recombinant Pseudomonas for Cobalamin Biosynthesis and Abiotic Stress Protection" International Journal of Molecular Sciences 23, no. 22: 13934. https://doi.org/10.3390/ijms232213934
APA StyleBalabanova, L., Pentekhina, I., Nedashkovskaya, O., Degtyarenko, A., Grigorchuk, V., Yugay, Y., Vasyutkina, E., Kudinova, O., Seitkalieva, A., Slepchenko, L., Son, O., Tekutyeva, L., & Shkryl, Y. (2022). Shift of Choline/Betaine Pathway in Recombinant Pseudomonas for Cobalamin Biosynthesis and Abiotic Stress Protection. International Journal of Molecular Sciences, 23(22), 13934. https://doi.org/10.3390/ijms232213934




