Abstract
Dientamoeba fragilis is a cosmopolitan intestinal protist colonizing the human gut with varying prevalence depending on the cohort studied and the diagnostic methods used. Its role in human health remains unclear mainly due to the very sporadic number of cross-sectional studies in gut-healthy populations. The main objective of this study was to expand knowledge of the epidemiology of D. fragilis in gut-healthy humans and their animals. A total of 296 stool samples from humans and 135 samples from 18 animal species were analyzed. Using qPCR, a prevalence of 24% was found in humans in contrast to conventional PCR (7%). In humans, several factors were found to influence the prevalence of D. fragilis. A more frequent occurrence of D. fragilis was associated with living in a village, traveling outside Europe and contact with farm animals. In addition, co-infection with Blastocystis spp. was observed in nearly half of the colonized humans. In animals, D. fragilis was detected in 13% of samples from eight species using qPCR. Our molecular phylogenies demonstrate a more frequent occurrence of Genotype 1 in gut-healthy humans and also revealed a likely a new protist species/lineage in rabbits related to D. fragilis and other related organisms.
1. Introduction
Dientamoeba fragilis is a human intestinal protist with cosmopolitan distribution and prevalence ranging from 0.2% to 82% (reviewed in [1,2,3]). Most studies addressing its occurrence are from high-income countries and surprisingly, less is known from low-income countries [1,2,4]. This protist was first described more than a century ago and was originally classified as a binucleate intestinal amoeba due to the absence of flagella (summarized in [5]). However, later morphological and molecular-phylogenetic analyzes revealed that D. fragilis is related to the flagellating trichomonads [2,5]. Nevertheless, some aspects of the life cycle of D. fragilis are still unclear. While colonization of the host hindgut by the trophozoite stage has been described in detail [2], the question of the mode of human-to-human transmission remains open. One hypothesis assumes involvement of the helminth vector with Enterobius vermicularis being the main candidate [6,7]. This is supported by observations such as (i) the colonization of a volunteer with D. fragilis after ingestion of E. vermicularis eggs derived from a donor colonized with both parasites [8], (ii) the molecular detection of D. fragilis DNA in E. vermicularis eggs [6,7], and (iii) also the frequent observation of co-infections of D. fragilis and E. vermicularis in children [9,10,11]. Another possibility of D. fragilis transmission has recently been discussed based on the description of a possible cyst stage [12,13], but this is considered controversial [14].
The role of D. fragilis in human health is currently debated in the medical and scientific community. In the past, this protist was neglected because of its apparent minor clinical significance [5], but some evidence of its possible pathogenic potential has accumulated over the last two decades [4]. Colonization with D. fragilis is often associated with gastrointestinal problems (e.g., [15,16,17]) and also with functional or inflammation-associated bowel diseases (Inflammatory Bowel Disease—IBD, Inflammatory Bowel Syndrome—IBS) [18,19,20,21]. That D. fragilis is a cause of some gastrointestinal diseases is often assumed due to the improvement of patients’ condition by its elimination after treatment [2]. However, these facts are not conclusive because, on the contrary, some studies reported that the elimination of D. fragilis had no effect on the condition of patients with gastrointestinal symptoms [22,23]. Unalan-Altintop et al. [24] stated that they found no association between the presence of D. fragilis and the development or exacerbation of ulcerative colitis or IBS. Another fact that may indicate a rather commensal role of D. fragilis is its frequent presence in the intestines of healthy individuals, based on the results of comparable or higher prevalence in healthy subjects in surveillance studies [25,26,27,28,29,30]. In addition, D. fragilis, along with another intestinal protist Blastocystis spp., is associated with higher diversity of the intestinal bacteriome, a marker of a healthy intestinal microbiome [31].
Host specificity and genetic diversity are among the other biological aspects of D. fragilis that remain to be elucidated. Regarding the host range, D. fragilis has only been detected in a few non-human species, most likely due to the lack of molecular epidemiological studies and the small number of animal species studied. To date, D. fragilis has only been detected in domestic pigs [32,33], non-human primates [34,35,36,37], cats and dogs [38], rats [39], and more recently in cattle [40] and pet budgerigars [41]. As part of the search for suitable experimental models for D. fragilis, laboratory mice also appear to be a susceptible host [12,42].
Genetic diversity of D. fragilis appears to be generally low. So far, only two genotypes have been distinguished, specifically Genotype 1 and Genotype 2 (reviewed in [1]). Moreover, currently available sources also show a low level of genetic polymorphism (i.e., about 3%) in the most studied D. fragilis genes to date, such as SSU rRNA, EF1α, and actin. This low genetic variability has been demonstrated for the above genes using phylogenetic approaches, analyzed both individually [1] and in concatenated form in the case of EF1α and actin [43]. Of the two known genotypes, Genotype 1 is the most common in the human population based on the available D. fragilis isolates from different regions around the globe [11,32,44]. In these surveys, Genotype 1 has been associated with both asymptomatic and symptomatic cases in individuals of different ages [4,11,45,46] and in patients suffering from IBS [24]. In contrast, Genotype 2 appears to be very rare in humans, resulting in limited data on this type [43,44]. This suggests that humans are probably not a natural host for Genotype 2 and the animal reservoir has not yet been discovered, due to the very limited number of studies on the occurrence of D. fragilis in animal hosts. To date, the possible zoonotic transmission of D. fragilis has been considered only between domestic pigs and their keepers originating from the same farm, based on molecular-sequence analyses of SSU rRNA and 5.8S rRNA gene fragments [32]. The sequences obtained showed high concordance with the defined Genotype 1 sequence from the GenBank database.
Since epidemiological surveys of D. fragilis in the gut-healthy human population are very rare, the main objective of the present study was to expand knowledge about the occurrence and prevalence of D. fragilis in such subjects in the Czech Republic, i.e., in a high-income country. We also investigated the presence of D. fragilis in animals with which these subjects were in close contact to evaluate potential zoonotic transmission. In humans, we then examined the association between the presence of D. fragilis and various factors such as lifestyle (traveling, living in urban versus rural areas), contact with animals, age, and gender. In addition, using the data from our previous study, Lhotská et al. [47], we were able to investigate the frequency of co-infection of D. fragilis and Blastocystis spp.
2. Results
We obtained a total 431 samples of which 296 samples were from humans and 135 samples were from animals (Table 1). Most of the non-human samples were from dogs (54/135), followed by cats (19/135), horses (15/135), rabbits (13/135), ruminants (12/135), chickens (8/135) and pigs (3/135), as well as some other animal species (11/135) (summary in Table 1).

Table 1.
List of human and animal species included in this study and the number of positive Dientamoeba fragilis samples for each species. (N—number of samples; cPCR—conventional PCR; qPCR—real-time PCR).
Human samples were divided into eight consecutive age categories (see Table 2 for details); most samples were from individuals older than 18 years (244/296). We obtained 124 samples from men (42%; 124/296) and 172 from women (58%; 172/296) from 14 regions of the Czech Republic. All subjects confirmed that they do not suffer from any gastrointestinal symptoms or Inflammatory Bowel Disease (IBD). Regarding travel activity, 52 subjects reported not travelling outside the Czech Republic (18%; 52/296), whereas 140 subjects reported travelling within Europe (47%; 140/296), and 104 participants reported traveling outside Europe (35%; 104/296). Eighty-three were villagers (28%; 83/296) and the other 213 lived in town/city (72%; 213/296). In addition, 252 people (85 %; 252/296) reported recent contact with animals, of which 180 (71%; 180/252) had contact with pets only and 72 had contact with farm animals (29%; 72/252).

Table 2.
Descriptive summary of incidence and proportion of colonized individuals in all age categories detected by qPCR. (N—number of samples obtained; N positive—number of positive samples in qPCR; SD—standard deviation).
2.1. Prevalence of Dientamoeba fragilis Based on Two Molecular Approaches
Conventional PCR (cPCR) detected D. fragilis in 32 samples (31 from humans and one from animal) out of total 431 samples. However, Sanger sequencing confirmed its presence in only 23 samples, 22 of which were from humans and one from the animal host (rabbit). Only these verified samples were considered positive based on cPCR (Table 1). According to this, the overall prevalence was 7% (22/296) in humans and 0.7% in non-human hosts (1/135). We were unable to obtain sequences from the remaining nine samples, so they were classified as negative.
Real-time PCR (qPCR) detected D. fragilis in 72 human samples with a prevalence of 24% (72/296) and in 15 samples from animals (11%; 15/135). Positive animals included dogs (Canis lupus familiaris), cat (Felis silvestris catus), guinea pig (Cavia aperea porcellus), rabbits (Oryctolagus cuniculus domesticus), goat (Capra aegagrus hircus), sheep (Ovis aries), and horses (Equus caballus) (see Table 1 for details). All 23 positive samples detected by cPCR (confirmed by Sanger sequencing) were also positive in qPCR (see Table 3 for details). In addition, we verified the presence of D. fragilis in the qPCR amplicons of all qPCR-positive samples by Sanger sequencing. Because we did not have a culture of D. fragilis to generate a quantification curve in qPCR, only Ct values are shown for each positive sample (Table 3).

Table 3.
Summary of qPCR-positive human and animal samples (N = 90) for Dientamoeba fragilis with Ct values and comparison with conventional PCR (cPCR) results.
2.2. Comparison of the Sensitivity of cPCR and qPCR for Dientamoeba fragilis Detection
We compared the sensitivity of these two methods on a set of human samples only (N = 296). The McNemar test showed that qPCR was a significantly more sensitive method for diagnostics of D. fragilis (p < 0.001; χ2 = 49; Supplementary Data S1). In addition, qPCR was also more successful in detecting D. fragilis in animal samples (Table 3), but these were not included in the statistical analyses.
2.3. Influence of the Specific Factors on the Occurrence of Dientamoeba fragilis
An effect of the specific factors on the distribution on D. fragilis was tested on the set of human samples only (N = 296) and statistically evaluated (for details see Section 4.7).
2.3.1. Lifestyle
All 296 human samples were tested for the effect of lifestyle on D. fragilis colonization. Two factors were selected for this investigation: (i) urban life (town/city) versus village life, and (ii) impact of traveling (Table 4). For the latter factor, we distinguished three categories of individuals: (a) travelers outside Europe (104/296), (b) travelers only in Europe (104/296), and (c) non-travelers (52/296; Table 4). Regarding the living locality, a higher prevalence was observed in individuals living in village areas (29%, 24/83), compared to individuals from urban areas (23%, 48/213; Table 4), but this difference was not statistically significant. We observed a higher prevalence of D. fragilis in travelers. The prevalence among travelers within Europe was 24% (34/140), while 29% (30/104) was in travelers outside Europe (Table 4). However, the prevalence in non-travelers was significantly lower (15%, 8/52; p = 0.0324; Table 4).

Table 4.
Prevalence of Dientamoeba fragilis in human samples according to the specific categories such as lifestyle (village versus city life, traveling) and contact with animals (pets, farm animals). (Samples N—number of samples obtained in each category out of the total number of samples). * Total number of volunteers in contact with animals. # Statistically significant differences, for details see Section 2.3.1 and Section 2.3.2.
2.3.2. Contact with Animals
The overall prevalence of D. fragilis in the group of volunteers who reported contact with animals was 25% (62/252; Table 4). Among those without animal contact, the prevalence was 23% (10/44, Table 4). Based on the information from questionnaires, we divided the group of subjects with animal contact into (i) those with contact with pets (180/252) and (ii) those with contact with farm animals (72/252; Table 4). We found a significant difference in the incidence of D. fragilis between these two groups (p = 0.004; Table 4), specifically between 36% prevalence (26/72) in the group with contact with farm animals and 20% prevalence (36/180) in the group with contact with pets.
2.3.3. Age and Gender
We obtained samples in eight age categories, specifically 0–3, 4–6, 7–12, 13–17, 18–30, 31–49, 50–60, >60 years (Table 2). The highest prevalence of D. fragilis was observed in the 4–6 years group (50%, 7/14) and the lowest number of positive individuals was found in children under three years of age (11%, 2/18; Table 2). However, no significant differences were found between age categories (Fisher’s exact test; p = 0.91). The same conclusion was reached when the effect of age (0–79 years) was modelled as a continuous variable (Generalized linear model; χ2 = 0.30, p = 0.59). Pairwise comparisons of proportions between all age categories also revealed no significant differences using the binominal exact test with p-values adjusted by the Holm method (all p > 0.34; Table 2). Regarding gender effects there was no difference in the prevalence of D. fragilis between men (24%, 30/124) and women (24%, 41/172).
2.4. Occurrence of Dientamoeba fragilis in the Family Environment
We obtained samples from several family members from a total of 69 families. Colonization of D. fragilis was detected in 34 families (i.e., 49%; Table 5). In addition, D. fragilis was detected in some families whose members were in close contact with a pet or farm animal that was positive for D. fragilis in qPCR (i.e., rabbits, cat, horses, dogs, and small ruminants; Table 5).

Table 5.
Overview of families including individuals colonized by Dientamoeba fragilis as well as D. fragilis-positive animals with which these families were in close or frequent contact (N family members—total number of family members; N positive members—number of family members colonized with D. fragilis; contact animal—animals colonized by D. fragilis which were in close contact with family members).
2.5. Coinfection of Dientamoeba fragilis with Blastocystis spp.
Within the human samples, we detected 33 cases of co-infection of D. fragilis with Blastocystis spp. (based on data published in [47]) out of 72 human samples positive for D. fragilis (46%; 33/72). Eleven of these subjects were men and 22 were women. All of these volunteers reported frequent contact with animals. In addition, ten subjects reported traveling within Europe (30%), 18 outside Europe (55%), and only five (15%) did not travel abroad at all. Twelve subjects lived in the village, 21 in the city. Most positive samples for both protists fell into the 31–49 age category (33%; 11/33). This was followed by the 18–30 age category (21%; 7/33). Among Blastocystis subtypes, the presence of D. fragilis was most frequently associated with ST1 (10/33), ST2 (6/33), ST3 (5/33) and ST4 (4/33) subtypes. In addition, some co-infections with ST5 (1/33), ST6 (3/33), and ST7 (1/33) subtypes were detected. The Blastocystis subtypes was not identified in the remaining three samples.
2.6. Genetic Diversity of Dientamoeba fragilis Based on Molecular Phylogenies
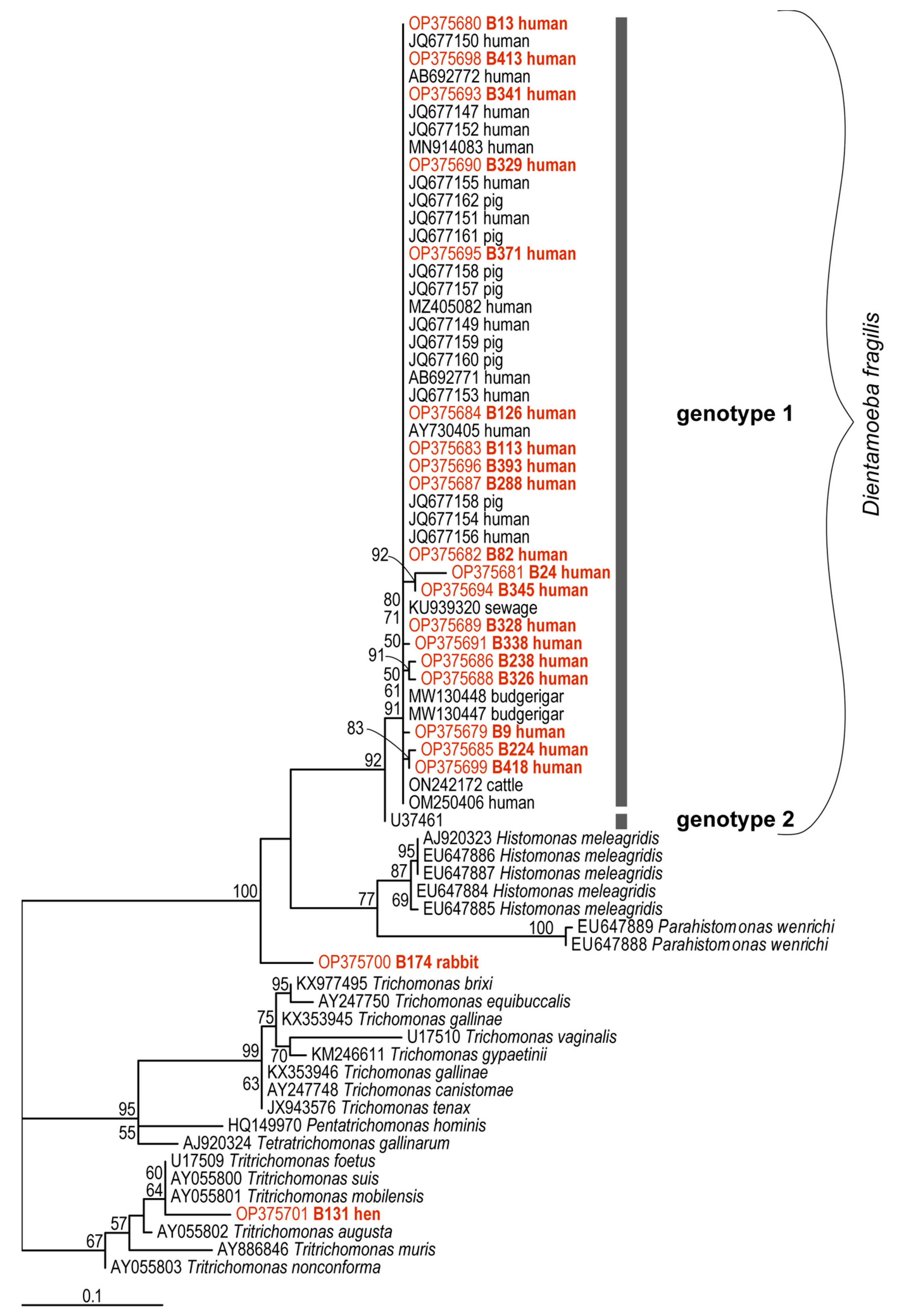
For the phylogenetic analyses, a total of twenty partial sequences of the SSU rDNA of D. fragilis were examined, specifically 19 sequences from colonized humans and one from a rabbit (B174). In addition, the Trichomonas sp. sequence (B131) derived from a chicken was included in the final dataset. This dataset included ten contigs (B9, B13, B126, B224, B238, B288, B329, B338, B174, B131), eight forward sequences (B113, B326, B328, B341, B371, B393, B413, B418), and three reverse sequences (B24, B82, B345). All our sequences were cloned to avoid unclear multiple signals or chimeras. The DNA sequences obtained in this study were deposited in the GenBankTM under accession numbers OP375680—OP375701.
The final dataset for phylogenetic analyzes was generated to cover the diversity of D. fragilis from different hosts and all sequences obtained were compared with other D. fragilis sequences obtained from GenBankTM (AB692771-73, AY730405, JQ677148-162, KU939320, MN91483, MW130447-8, MZ405082, OM250406, ON242172, U37461) and sequences from related organisms such as Histomonas meleagridis (EU647884-7, AJ920323), Parahistomonas wenrichi (EU647888,-89) and various trichomonads (AJ920324, AY247748/-50, AY886846, AY055800-3, JX943576, HQ149970, KM246611, KX353945-46, KX977495, U17509-10). This tree was rooted with B131 “Trichomonas” (OP375701) as an outgroup.
The maximum likelihood tree (TVMe+G4+F, IQ-TREE) (Figure 1) shows a large clade containing D. fragilis-, H. meleagridis- and P. wenrichi-subclades, and then two trichomonad clades, one of which contains our sequence of Trichomonas sp. from chicken (B131/OP375701). The D. fragilis subclade includes our human sequences (OP375679-99), sequences defined as Genotype 1 from GenBankTM, and also contains only one Genotype 2 sequence (U37461) that is currently available in the GenBank database and it is represented by a separate branch (Figure 1). All our sequences from humans were unambiguously assigned to the Genotype 1 group, which has some internal polytomy (identical sequences) and three stable subgroups (one containing sequences OP375681, OP375694; the second containing sequences OP375686, OP375688; and the third with OP375685, OP375699). A very interesting result is the phylogenetic position of the sequence from rabbit (B174/OP375700). Although this sequence has a high identity with D. fragilis and H. meleagridis in the BlastNt database (concordance with D. fragilis: query coverage—70–76%, e-value—0.0, percent identity—89–92%; concordance with H. meleagridis: query coverage—75%, e-value—0.0, percent identity—89%), it forms a separate, strongly supported branch representing a new lineage of organisms closely related to D. fragilis, H. meleagridis and P. wenrichi.
Figure 1.
SSU rDNA tree from SSU rDNA sequences of Dientamoeba fragilis computed by IQTree. The numbers of the branches above represent maximum likelihood bootstrap supports as computed from 10,000 UFBoot. The scale bar represents 10 substitutions per 100 positions. New sequences are in red color.
3. Discussion
To our knowledge, this is the first cross-sectional study addressing the occurrence of Dientamoeba fragilis in the Czech Republic in a set of stool and fecal samples obtained from gut-healthy volunteers and their animals of different species (see Table 1 for details). In humans, using real-time diagnostics (qPCR), we found an overall prevalence of 24% in a group of 296 humans. We also investigated the difference in sensitivity for detection of D. fragilis between qPCR and conventional PCR (cPCR) in human samples. Real-time PCR identified 50 more positive samples than cPCR, making it more sensitive for the detection of D. fragilis. The prevalence based on cPCR was 7%. Our observation is consistent with other studies comparing the sensitivity of molecular methods for the detection of other intestinal protists (e.g., [48]). In general, the diagnostics of D. fragilis are challenging and the sensitivity of different diagnostic methods can vary widely [49]. For example, Sharzhanov et al. [50] showed that the sensitivity of traditional microscopic methods for the detection of D. fragilis is lower compared to qPCR. Thus, qPCR appears to be the gold standard for D. fragilis diagnostics, and some recent studies already use this method routinely (reviewed in [1]).
Current data on the occurrence and prevalence of D. fragilis in asymptomatic individuals (i.e., subjects without gastrointestinal symptoms or disease) are generally very sporadic and come primarily from control groups of healthy subjects included in studies that focused on patients with various gastrointestinal problems (acute or chronic diarrhea, Inflammatory Bowel disease—IBD, or Inflammatory Bowel Syndrome—IBS). Because epidemiologic studies of D. fragilis use a variety of diagnostic approaches that may differ in specificity and sensitivity [1], we compared our data only with studies using qPCR.
The available literature shows that the prevalence of D. fragilis in control groups of healthy individuals ranges from 36% to 71% [25,27,29,30,51,52]. For example, Heusinkveld et al. [52] conducted a large-scale cross-sectional survey to investigate the association between the occurrence of a number of enteropathogens (including selected protists) and acute gastroenteritis, and included more than 8500 pairs of a preschool-aged child and a parent living in the same household. They found that the prevalence of D. fragilis ranged from 36% to 39% in individuals without acute intestinal problems. Our results show a prevalence of 24% in gut-healthy people in different age groups, which is slightly lower than in the above-mentioned studies. Interestingly, a ten-year study conducted at the University Hospital of Parma demonstrated an almost comparable prevalence of D. fragilis (i.e., 20%) in patients with gastrointestinal symptoms [3]. Following data from patient cohorts and healthy subjects, the incidence of D. fragilis seems to be more or less comparable. Therefore, it is not possible to decide whether it is rather a pathogenic or a commensal organism. Determination of the so-called fecal protist load [48] could shed more light on deciphering the role of D. fragilis in human health. Such quantitative data could subsequently be compared between asymptomatic and symptomatic individuals, and in patients with IBD. Unfortunately, we were not able to quantify the fecal Dientamoeba load in our study because we did not have a monoxenic culture of D. fragilis to construct the quantification curve as was done in case of Blastocystis spp. in the study by Šloufová et al. [48].
It is important to mention that the prevalence of D. fragilis may be influenced by various factors, for example, age group selection, geographic location, or socioeconomic status [1,2]. The prevalence observed in our study may be affected by the wide age range in the group of volunteers because it ranged from 0 months to >60 years.
Our set of human samples allowed us to observe the frequency of co-infections of D. fragilis with another intestinal protist Blastocystis spp. which was the subject of our previous study [47]. Based on the results of both studies, we found that almost half of the samples (i.e., 46%) from individuals colonized with D. fragilis had co-infection with Blastocystis spp. Interestingly, these cases of co-infections were associated with factors such as frequent contact with animals and traveling. Co-infection with these protists was also recorded in other studies that focused primarily on patients with gastrointestinal disease or symptoms, and the rate of co-infection rate varied widely. While several studies found co-infection rates ranging from 17.6% to 33.6% in patients with gastrointestinal symptoms [17,21,50,53], Yakoob et al. [20] detected co-infections in only 3% of IBS patients but found nothing comparable in a group of healthy subjects. In contrast, our results show a higher rate of co-infection in gut-healthy volunteers. Similarly, a higher co-infection rate of D. fragilis with Blastocystis spp. (i.e., 66.2%) was demonstrated in mostly asymptomatic children [54]. The authors of the latter study hypothesize that co-infection of D. fragilis and Blastocystis spp. is associated with healthy gut microbiome, as was the case in the study by de Boer et al. [26]. In addition, our data are unique because we were able to assess the presence D. fragilis and Blastocystis subtypes. Dientamoeba was most frequently detected in co-infection with ST1, ST2, ST3, and ST4 subtypes.
As far as we know, only one larger epidemiological study has been conducted for D. fragilis, covering a wider range of animal species [38]. However, D. fragilis was detected in only two fecal samples out of 420 samples and in two animal species (dog and cat) out of a total of 37 species. In our study, we found 13% of animals in close contact with human volunteers (135 samples obtained from 18 species) that were positive for D. fragilis using qPCR method. Dientamoeba was detected in eight animal species, several of which have already been described as potential hosts, specifically dogs (4/55), cat (1/19), and sheep (1/6). In dogs and cats, D. fragilis has been previously detected by PCR, whereas in sheep it was detected by traditional coproscopic methods [38,40,55]. In addition, we found D. fragilis in rabbits (5/13), horses (2/15), goat (1/4), and guinea pig (1/2). Surprisingly, we found no evidence of D. fragilis presence in domestic pigs, although a high prevalence (44–63%) has been recorded in the past [32,33].
Regarding the specific factors that might affect the distribution of D. fragilis in the gut-healthy human population, we examined the influence of living locality (urban versus rural), contact with animals, traveling (within and outside Europe, no traveling), age, and gender. Our results showed that prevalence was higher in people living in villages, and in travelers, especially outside Europe. Traveling is also considered an important predisposing factor for D. fragilis colonization in several other studies that focused on patients with diarrhea [15,16] or in a cross-sectional study in children under six years of age [29]. Interestingly, in a study by Norberg et al. [15], there was recorded a trend toward a higher incidence of D. fragilis in more than half of the patients with a history of traveling, and almost half of them had traveled outside Europe.
Not much is known about the effect of living locality; only Heusinkveld et al. [52] found a correlation between more frequent occurrence of D. fragilis and less urbanized areas. They also described a correlation between the occurrence of D. fragilis and the ownership of dogs. In contrast, Jokelainen et al. [29] concluded that contact with animals, especially those species considered potential hosts, had no effect on the spread of D. fragilis. Similarly, in our study, we found no significant effect of general contact with animals on the spread of D. fragilis in humans, but we showed a possible effect of keeping some farm animals, as more than one third of their breeders were colonized. Unfortunately, our data do not allow us to assess zoonotic transmission because we confirmed the presence of D. fragilis in animals only by qPCR without any subsequent sequence information (detailed information further). So far, the only case of possible zoonotic transmission was described in the study by Cacciò et al. [32], in which Genotype 1 was confirmed in pigs and their keepers based on molecular analyses of partial SSU- and 5.8S-rDNA sequences within one farm.
Age is generally assumed to have a significant impact on the distribution of D. fragilis in humans [1], but this has not always been confirmed [4,30]. In general, a higher prevalence is thought to occur in children up to about 15 years of age [29,51,56]. Although our results showed a more frequent occurrence of D. fragilis in the group of children younger than 12 years, these differences were not statistically significant, as in other studies [17,49,57]. Here, this trend could be due to an imbalanced number of samples collected from children under 18 years of age. The occurrence of D. fragilis in the other age groups was very similar. Regarding gender, we did not observe a difference in the incidence of D. fragilis between men and women, as was the case in the recent study by Sarzhanov et al. [50]. Nevertheless, findings of higher D. fragilis prevalence in women of childbearing age are frequently described [4,58].
A total of 69 families provided us with samples from all or several of their members so that we could track the occurrence of D. fragilis within the family. In almost half of the families (49%), the presence of D. fragilis was detected in at least one member, but usually in several members. Our results suggest that D. fragilis appears to circulate among family members or that positive family members may have been colonized from the same source. In six families, we also detected the presence of D. fragilis in their animals. However, based on our data, we were unable confirm zoonotic transmission.
The genetic diversity of D. fragilis appears to be low, and so far, only two genotypes have been proposed based on the variability of three genes (SSU rRNA, EF1, actin) [16,43,59,60]. However, most studies dealing with molecular or phylogenetic identification of D. fragilis isolates describe a predominant occurrence of Genotype 1 in different geographical areas, in different human cohorts (symptomatic versus asymptomatic individuals) and in animals [16,24,40,41]. Our nineteen SSU rDNA sequences of D. fragilis obtained from human samples also belong to Genotype 1 according to our phylogenetic analyses, and the sequences appeared in the phylogenetic tree together with Genotype 1 sequences from GenBank. Nevertheless, this Genotype 1 group exhibits some small genetic variability. In addition to the identical sequences forming a polytomy, we identified three highly supported subgroups. Analysis of the entire SSU rDNA sequence or another gene—as suggested by Simone M. Cacciò [1]—would perhaps provide more insight into the genetic diversity of D. fragilis.
In contrast to the human isolates, we were not able to obtain SSU rDNA sequences of D. fragilis by cPCR from most of the positive animals (with the exception of one sequence from a rabbit), apparently due to the low intensity of colonization, considering the Ct values obtained in qPCR (for details, see Table 3). Therefore, the presence of D. fragilis in these samples was confirmed only by qPCR followed by sequencing of very short qPCR amplicons (∼97 bp). Therefore, we were not able to determine whether it was indeed D. fragilis. To determine the genotypes of D. fragilis in animals, the partial SSU rDNA sequences obtained by cPCR (∼850 bp) are required. So far, only the occurrence of Genotype 1 has been described in pigs, cattle and budgerigars [40,41,44].
The fact that these 850 bp-SSU rDNA sequences are absolutely essential for the definitive genetic identification of D. fragilis in animals is also underlined by our finding of an apparently new protist in a rabbit, that appears to be closely related to D. fragilis. This SSU rDNA sequence was assigned to D. fragilis using the BlastNt database, but our phylogenetic analyzes indicated that it is the strongly supported separate branch next to D. fragilis, Histomonas meleagridis, and Parahistomonas wenrichi within one clade. These results suggest that it may be a new lineage or genus of a related protist that has not been described previously. However, further studies focusing on detailed morphological and phylogenetic-molecular identification is required for a taxonomic description. Based on all these facts, there is also a need to optimize or develop a new diagnostic qPCR protocol that is highly specific for D. fragilis.
4. Materials and Methods
4.1. Sample Collection and Ethical Approval
The present study was conducted between 2017 and 2019 in the Czech Republic and follows the previous study on Blastocystis spp. [47] with identical sampling strategy and sample dataset. Briefly, stool samples were collected from healthy volunteers, none of whom had gastrointestinal symptoms or intestinal inflammatory diseases (inflammatory Bowel Disease-IBD, or Inflammatory Bowel Syndrome—IBS) at the time of sampling. To investigate possible zoonotic transmission, fecal samples were also collected from animals, with which the volunteers were in a close contact. All participants completed a questionnaire that included information on living locality (city/village), traveling (non-travel/travel in Europe/travel outside Europe), contact with animals (no contact/contact with pets only/contact with pets and livestock), gender and age (age categories: 0–3, 4–6, 7–12, 13–17, 18–30, 31–49, 50–60, >60) (for more details see [47]).
Each participant signed an informed consent declaration to participate in the study. The procedure, conditions, and ethical rules of this study are in accordance with the Declaration of Helsinki 2013 (World Medical Association). All data were strictly anonymized and processed in accordance with applicable laws of the Czech Republic (e.g., Act No 101/2000 Coll and subsequent regulations). The study was approved by the Ethics Committee of the Biology Centre of the Czech Academy of Sciences (reference number: 1/2017).
4.2. DNA Extraction and Samples Processing
Total DNA was extracted directly from stool/fecal samples using the commercial kit PSP Spin Stool DNA Kit (Stratec, Germany) according to the manufacturer’s protocol. All collected samples were subjected to two molecular diagnostic procedures such as conventional PCR (cPCR) [16] and real-time PCR (qPCR) [61]. As a positive control, we used DNA from the xenic culture of D. fragilis. This DNA was provided to us by our colleague Prof. J. Kulda from Charles University (Prague, Czech Republic).
4.3. Conventional PCR
Conventional PCR for detection of D. fragilis was performed using primers DF400 (5′–TATCGGAGGTGGTAATGACC–3′) and DF1250 (5′–CATCTTCCTCCTGCTTAGAC–3′) which amplify the ∼850 bp region of SSU rDNA [16]. PCR was performed in the T100TM Thermal Cycler (Biorad, Hercules, CA, USA) under the following conditions: 95 °C/5 min, 34 × (94 °C/1 min; 58 °C/1,5 min; 72 °C/2 min), and 72 °C/10 min. All PCR reactions were prepared in a final volume of 10 μL containing 5 μL of commercially produced 2× concentrated Master Mix (AccuPowerR Taq PCR PreMix; Bioneer, Daejeon, Republic of Korea), 1 μL of each primer, 1 μL of MiliQ water and 2 μL of template DNA. PCR products were visualized by electrophoresis, loading 8 μL of PCR product on a 1% agarose gel with ethidium bromide using the Thermo Fisher Scientific electrophoresis system (Waltham, MA, USA).
4.4. Real-Time Diagnostics
Real-time PCR was performed using LightCycler LC 480 I (Roche, Basel, Switzerland) with a 96-well block under following conditions—95 °C/12 min; 50 × (95 °C/15 s; 60 °C/30 s; 72 °C/30 s) [61], using Master Mix (HOT FIREPol® Probe qPCR Mix Plus Rox, Solis BioDyne, Tartu, Estonia). Dientamoeba-specific primers and probe set for part of SSU rRNA gene consisted of forward primer Df-124F (5′–CAACGGATGTCTTGGCTCTTTA–3′) and reverse primer Df-221R (5′–TGCATTCAAAGATCGAACTTATCAC–3′), amplifying the ∼97 bp region and Taqman probe Df-172 (FAM-5′–CAATTCTAGCCGCTTAT-3′-MGB) (Generi Biotech, Hradec Králové, Czech Republic). All negative samples were tested for PCR inhibition by addition of foreign DNA (obtained from experimental rat tissues) and a specific qPCR protocol (commercial primers and Taqman probe for detection of the rat gene for beta-2 microglobulin; Thermofisher Scientific).
4.5. Cloning and Sequencing of Positive Samples
Sequences were always obtained from positive samples using cloning using pGEM®-T Easy Vector System I (Promega, Madison, WI, USA) followed by Sanger sequencing by a commercial company (Eurofins GATC Biotech, Konstanz, Germany). PCR amplicons of the appropriate size were purified using the GenEluteTM Gel Extraction Kit (Sigma-Aldrich, St. Louis, MO, USA) and sequenced in both directions using PCR primers. All sequences were then processed using the Geneious Prime 2019.0.4. software and subsequently compared with sequences in the GenBankTM database (National Centre for Biotechnology Information) using BLASTn.
4.6. Phylogenetic Analysis
The newly obtained SSU rRNA gene sequences were aligned to the available homologs from GenBank using the Clustal Omega algorithm in SeaView [62]. Alignment was manually edited in BioEdit [63] removing gaps, ambiguously aligned and hypervariable positions. Maximum likelihood trees were constructed using a TVMe+G4+F evolutionary model (selected by ModelFinder [64]) implemented in IQ-TREE [65]. Branch support was estimated from 10,000 UFBoot (ultrafast bootstrap approximation) [66] using the above software and conditions.
4.7. Statistical Analysis
Difference in sensitivity of cPCR and qPCR for detection of D. fragilis was evaluated using the McNemar test with Yates’s correction. We employed Fisher’s Exact Test to examine differences between age categories in the incidence of D. fragilis in humans detected by qPCR. In addition, we used a generalized linear model to examine the effects of age as a continuous variable. The chi-square test was used to statistically evaluate the significance of the difference in D. fragilis prevalence between groups of humans within a single observed factor, such as lifestyle (urban versus rural), traveling (within and out of Europe), contact with animals (with and without), and gender. These analyzes were performed using GraphPad Prism 5.0 software and R Studio version 3.6.2. A p-value of less than 0.05 was considered to indicate statistical significance.
5. Conclusions
The prevalence of D. fragilis found in our study in the gut-healthy individuals suggests that the mere presence of this protist should not generally lead to efforts to eliminate it from the intestine, especially in asymptomatic individuals. This fact has been also considered for other intestinal protists, for example Blastocystis spp. [67]. Moreover, it is very difficult to decide whether D. fragilis is more of a commensal or a pathogen, since the frequency of its occurrence in patients with gastrointestinal symptoms is more or less comparable to healthy cohorts. It seems that the information about the presence of D. fragilis is not sufficient to assess its role in human health and that a quantitative indicator, the so-called fecal protist load, should be introduced into its diagnostics [48]. Such quantitative data on D. fragilis colonization and their comparison between different cohorts could facilitate understanding of its role in human health and disease. In addition, knowledge about D. fragilis in gut healthy individuals in high-income countries and in the human populations with traditional lifestyles needs to be expanded, including also knowledge about correlations with intestinal bacterial diversity. Worth mentioning is recent evidence of regulatory capabilities of some intestinal eukaryotes, including protists, which may be critical to the host intestinal ecosystem and immune system [68,69,70]. Dientamoeba fragilis may have a similar regulatory function, either alone or in co-infection with Blastocystis spp. but this remains to be tested experimentally. Finally, it is important to emphasize that to understand the zoonotic transmission of D. fragilis, it is necessary to monitor its occurrence in a range of animal species and also to decipher its genetic diversity.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms232315407/s1.
Author Contributions
Field work: M.J., A.K., Z.L. and O.K.; Conceptualization: M.J., Z.L., C.R.S., K.J.P. and K.J.; Methodology and investigation: M.J., A.K., Z.L., K.B. and M.O.; Software: M.J., P.S., M.O. and K.J.P.; Validation: M.J., A.K., K.J.P., P.S., M.O., C.R.S. and K.J.; Formal analyses: M.J., K.J.P., M.O., C.R.S. and K.J.; Data curation: M.J., K.J.P., P.S. and M.O.; Writing—original draft preparation: M.J., M.O., K.J.P., C.R.S. and K.J.; Visualization: M.J., M.O., K.J.P. and K.J.; Supervision: M.J. and K.J.; Funding acquisition and project administration: A.K. and K.J.; Draft writing: M.J., A.K., Z.L., M.O., C.R.S., K.J.P. and K.J. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the grant from the Czech Science Foundation (22-04837S) to K.J. ERDF/ESF Centre for research of pathogenicity and virulence of parasites (No.CZ.02.1.01/0.0/0.0/16_019/0000759) to M.O. and by the grant from the Student Grant Agency (SGA) at the Faculty of Science of the University of South Bohemia to A.K. (in 2019).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the Biology Centre of the Czech Academy of Sciences (protocol code 1/2017 and 25 January 2017).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Publicly available datasets were analyzed in this study. This data can be found in the GenBankTM database under these accession numbers [OP375679-OP375701].
Acknowledgments
We thank Martina Šloufová, Dagmar Jirsová, and Tomáš Skalický for laboratory and software assistance. We also thank the reviewers for their very valuable comments to improve this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cacciò, S.M. Molecular Epidemiology of Dientamoeba fragilis. Acta Trop. 2018, 184, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.; Barratt, J.; Chan, D.; Ellis, J.T. Dientamoeba fragilis, the Neglected Trichomonad of the Human Bowel. Clin. Microbiol. Rev. 2016, 29, 553–580. [Google Scholar] [CrossRef] [PubMed]
- Calderaro, A.; Buttrini, M.; Montecchini, S.; Rossi, S.; Farina, B.; Arcangeletti, M.C.; De Conto, F.; Chezzi, C. Prevalence of Intestinal Parasitoses in a Non-Endemic Setting during a 10-Year Period (2011–2020): A Focus on Dientamoeba fragilis. Microorganisms 2022, 10, 426. [Google Scholar] [CrossRef] [PubMed]
- Barratt, J.L.N.; Harkness, J.; Marriott, D.; Ellis, J.T.; Stark, D. A Review of Dientamoeba fragilis Carriage in Humans: Several Reasons Why This Organism Should Be Considered in the Diagnosis of Gastrointestinal Illness. Gut Microbes 2011, 2, 3–12. [Google Scholar] [CrossRef]
- Johnson, E.H.; Windsor, J.J.; Clark, C.G. Emerging from Obscurity: Biological, Clinical, and Diagnostic Aspects of Dientamoeba fragilis. Clin. Microbiol. Rev. 2004, 17, 553–570. [Google Scholar] [CrossRef] [PubMed]
- Ögren, J.; Dienus, O.; Löfgren, S.; Iveroth, P.; Matussek, A. Dientamoeba fragilis DNA Detection in Enterobius Vermicularis Eggs. Pathog. Dis. 2013, 69, 157–158. [Google Scholar] [CrossRef] [PubMed]
- Röser, D.; Nejsum, P.; Carlsgart, A.J.; Nielsen, H.V.; Stensvold, C.R. DNA of Dientamoeba fragilis Detected within Surface-Sterilized Eggs of Enterobius vermicularis. Exp. Parasitol. 2013, 133, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Ockert, G. Symptomatology, Pathology, Epidemiology, and Diagnosis of Dientamoeba fragilis. In Trichomonads Parasitic in Humans; Honigberg, B.M., Ed.; Springer: New York, NY, USA, 1990; pp. 394–410. ISBN 978-1-4612-3224-7. [Google Scholar]
- Girginkardeşler, N.; Kurt, Ö.; Kilimcioǧlu, A.A.; Ok, Ü.Z. Transmission of Dientamoeba fragilis: Evaluation of the Role of Enterobius Vermicularis. Parasitol. Int. 2008, 57, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Ögren, J.; Dienus, O.; Löfgren, S.; Einemo, I.M.; Iveroth, P.; Matussek, A. Dientamoeba fragilis Prevalence Coincides with Gastrointestinal Symptoms in Children Less than 11 Years Old in Sweden. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1995–1998. [Google Scholar] [CrossRef]
- Yıldız, İ.; Ertuğ, S.; Tileklioğlu, E.; Malatyalı, E.; Güçlü, Ö.; Ertabaklar, H. Investigation of Dientamoeba fragilis Frequency in Faecal Samples of Patients with Enterobius vermicularis Infection by Polymerase Chain Reaction. Turk. Parazitolojii Derg. 2021, 45, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Munasinghe, V.S.; Vella, N.G.F.; Ellis, J.T.; Windsor, P.A.; Stark, D. Cyst Formation and Faecal-Oral Transmission of Dientamoeba fragilis—The Missing Link in the Life Cycle of an Emerging Pathogen. Int. J. Parasitol. 2013, 43, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.; Garcia, L.S.; Barratt, J.L.N.; Phillips, O.; Roberts, T.; Marriott, D.; Harkness, J.; Ellis, J.T. Description of Dientamoeba fragilis Cyst and Precystic Forms from Human Samples. J. Clin. Microbiol. 2014, 52, 2680–2683. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.G.; Röser, D.; Stensvold, C.R. Transmission of Dientamoeba fragilis: Pinworm or Cysts? Trends Parasitol. 2014, 30, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Norberg, A.; Nord, C.E.; Evengård, B. Dientamoeba fragilis—A Protozoal Infection Which May Cause Severe Bowel Distress. Clin. Microbiol. Infect. 2003, 9, 65–68. [Google Scholar] [CrossRef]
- Stark, D.; Beebe, N.; Marriott, D.; Ellis, J.; Harkness, J. Prospective Study of the Prevalence, Genotyping, and Clinical Relevance of Dientamoeba fragilis Infections in an Australian Population. J. Clin. Microbiol. 2005, 43, 2718–2723. [Google Scholar] [CrossRef] [PubMed]
- Aykur, M.; Calıskan Kurt, C.; Dirim Erdogan, D.; Biray Avcı, C.; Vardar, R.; Aydemir, S.; Girginkardeşler, N.; Gündüz, C.; Dagci, H. Investigation of Dientamoeba fragilis Prevalence and Evaluation of Sociodemographic and Clinical Features in Patients with Gastrointestinal Symptoms. Acta Parasitol. 2019, 64, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. Prevalence, Incidence, and Risk Factors of Intestinal Parasites in Danish Primary Care Patients with Irritable Bowel Syndrome. Scand. J. Infect. Dis. 2014, 46, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.; van Hal, S.; Marriott, D.; Ellis, J.; Harkness, J. Irritable Bowel Syndrome: A Review on the Role of Intestinal Protozoa and the Importance of Their Detection and Diagnosis. Int. J. Parasitol. 2007, 37, 11–20. [Google Scholar] [CrossRef]
- Yakoob, J.; Jafri, W.; Beg, A.; Abbas, Z.; Naz, S.; Islam, M.; Khan, R. Blastocystis hominis and Dientamoeba fragilis in Patients Fulfilling Irritable Bowel Syndrome Criteria. Parasitol. Res. 2010, 107, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Piubelli, C.; Soleymanpoor, H.; Giorli, G.; Formenti, F.; Buonfrate, D.; Bisoffi, Z.; Perandin, F. Blastocystis Prevalence and Subtypes in Autochthonous and Immigrant Patients in a Referral Centre for Parasitic Infections in Italy. PLoS ONE 2019, 14, e0210171. [Google Scholar] [CrossRef] [PubMed]
- Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. Treatment of Dientamoeba fragilis in Patients with Irritable Bowel Syndrome. Am. J. Trop. Med. Hyg. 2012, 87, 1046–1052. [Google Scholar] [CrossRef] [PubMed]
- Röser, D.; Simonsen, J.; Stensvold, C.R.; Olsen, K.E.P.; Bytzer, P.; Nielsen, H.V.; Mølbak, K. Metronidazole Therapy for Treating Dientamoebiasis in Children Is Not Associated with Better Clinical Outcomes: A Randomized, Double-Blinded and Placebo-Controlled Clinical Trial. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2014, 58, 1692–1699. [Google Scholar] [CrossRef] [PubMed]
- Unalan-Altintop, T.; Vahabov, C.; Ergunay, K.; Kurt, O.; Kav, T.; Akyon, Y.; Erguven, S. Investigation of Dientamoeba fragilis and Blastocystis in Patients from Turkey with Ulcerative Colitis and Irritable Bowel Syndrome: Any Relation with Genotypes? Acta Trop. 2022, 231, 106451. [Google Scholar] [CrossRef] [PubMed]
- Brands, M.R.; Van de Vijver, E.; Haisma, S.M.; Heida, A.; van Rheenen, P.F. No Association between Abdominal Pain and Dientamoeba in Dutch and Belgian Children. Arch. Dis. Child. 2019, 104, 686–689. [Google Scholar] [CrossRef] [PubMed]
- de Boer, M.D.; Schuurs, T.A.; Vermeer, M.; Ruijs, G.J.H.M.; van der Zanden, A.G.M.; Weel, J.F.; Bruijnesteijn van Coppenraet, L.E.S. Distribution and Relevance of Dientamoeba fragilis and Blastocystis Species in Gastroenteritis: Results from a Case-Control Study. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 197–203. [Google Scholar] [CrossRef] [PubMed]
- De Jong, M.J.; Korterink, J.J.; Benninga, M.A.; Hilbink, M.; Widdershoven, J.; Deckers-Kocken, J.M. Dientamoeba fragilis and Chronic Abdominal Pain in Children: A Case-Control Study. Archives of Disease in Childhood: Educ. Pract. Ed. 2014, 99, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Holtman, G.A.; Kranenberg, J.J.; Blanker, M.H.; Ott, A.; Lisman-van Leeuwen, Y.; Berger, M.Y. Dientamoeba fragilis Colonization Is Not Associated with Gastrointestinal Symptoms in Children at Primary Care Level. Fam. Pract. 2017, 34, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Jokelainen, P.; Jensen, B.H.; Andreassen, B.U.; Petersen, A.M.; Röser, D.; Krogfelt, K.A.; Nielsen, H.V.; Stensvold, C.R. Dientamoeba fragilis, a Commensal in Children in Danish Day Care Centers. J. Clin. Microbiol. 2017, 55, 1707–1713. [Google Scholar] [CrossRef] [PubMed]
- Krogsgaard, L.R.; Engsbro, A.L.; Stensvold, C.R.; Nielsen, H.V.; Bytzer, P. The Prevalence of Intestinal Parasites Is Not Greater among Individuals with Irritable Bowel Syndrome: A Population-Based Case-Control Study. Clin. Gastroenterol. Hepatol. 2015, 13, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; van der Giezen, M. Associations between Gut Microbiota and Common Luminal Intestinal Parasites. Trends Parasitol. 2018, 34, 369–377. [Google Scholar] [CrossRef]
- Cacciò, S.M.; Sannella, A.R.; Manuali, E.; Tosini, F.; Sensi, M.; Crotti, D.; Pozio, E. Pigs as Natural Hosts of Dientamoeba fragilis Genotypes Found in Humans. Emerg. Infect. Dis. 2012, 18, 838–841. [Google Scholar] [CrossRef] [PubMed]
- Crotti, D.; Sensi, M.; Crotti, S.; Grelloni, V.; Manuali, E. Dientamoeba fragilis in Swine Population: A Preliminary Investigation. Vet. Parasitol. 2007, 145, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.; Phillips, O.; Peckett, D.; Munro, U.; Marriott, D.; Harkness, J.; Ellis, J. Gorillas Are a Host for Dientamoeba fragilis: An Update on the Life Cycle and Host Distribution. Vet. Parasitol. 2008, 151, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Lankester, F.; Kiyang, J.A.; Bailey, W.; Unwin, S. Dientamoeba fragilis: Initial Evidence of Pathogenicity in the Western Lowland Gorilla (Gorilla Gorilla Gorilla). J. Zoo Wildl. Med. 2010, 41, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Helenbrook, W.D.; Wade, S.E.; Shields, W.M.; Stehman, S.V.; Whipps, C.M. Gastrointestinal Parasites of Ecuadorian Mantled Howler Monkeys (Alouatta Palliata Aequatorialis) Based on Fecal Analysis. J. Parasitol. 2015, 101, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Menu, E.; Davoust, B.; Mediannikov, O.; Akiana, J.; Mulot, B.; Diatta, G.; Levasseur, A.; Ranque, S.; Raoult, D.; Bittar, F. Occurrence of Ten Protozoan Enteric Pathogens in Three Non-Human Primate Populations. Pathogens 2021, 10, 280. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.; Barratt, J.; Roberts, T.; Phillips, O.; Šlapeta, J.; Ryan, U.; Marriott, D.; Harkness, J.; Ellis, J.; Stark, D. Detection of Dientamoeba fragilis in Animal Faeces Using Species-Specific Real-Time PCR Assay. Vet. Parasitol. 2016, 227, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Ogunniyi, T.; Balogun, H.; Shasanya, B. Ectoparasites and Endoparasites of Peridomestic House-Rats in Ile-Ife, Nigeria and Implication on Human Health. Iran. J. Parasitol. 2014, 9, 134–140. [Google Scholar] [PubMed]
- Yildiz, I.; Aynur, Z.E. First Detection and Molecular Characterization of Dientamoeba fragilis in Cattle. Zoonoses Public Health 2022, 69, 897–903. [Google Scholar] [CrossRef]
- Yetismis, G.; Yildrim, A.; Pekmezci, D.; Duzlu, O.; Ciloglu, A.; Onder, Z.; Simsek, E.; Ercan, N.; Pekmezci, G.Z.; Inci, A. First Report and Genotyping of Dientamoeba fragilis in Pet Budgerigars (Melopsattacus undulatus), with Zoonotic Importance. Zoonoses Public Health 2022, 69, 572–578. [Google Scholar] [CrossRef] [PubMed]
- El-Gayar, E.K.; Mokhtar, A.B.; Hassan, W.A. Study of the Pathogenic Potential of Dientamoeba fragilis in Experimentally Infected Mice. Parasite Epidemiol. Control 2016, 1, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Clark, C.G.; Röser, D. Limited Intra-Genetic Diversity in Dientamoeba fragilis Housekeeping Genes. Infect. Genet. Evol. 2013, 18, 284–286. [Google Scholar] [CrossRef] [PubMed]
- Cacciò, S.M.; Sannella, A.R.; Bruno, A.; Stensvold, C.R.; David, E.B.; Guimarães, S.; Manuali, E.; Magistrali, C.; Mahdad, K.; Beaman, M.; et al. Multilocus Sequence Typing of Dientamoeba fragilis Identified a Major Clone with Widespread Geographical Distribution. Int. J. Parasitol. 2016, 46, 793–798. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stark, D.J.; Beebe, N.; Marriott, D.; Ellis, J.T.; Harkness, J. Dientamoebiasis: Clinical Importance and Recent Advances. Trends Parasitol. 2006, 22, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, O.; Peek, R.; Souayah, H.; Dediste, A.; Buset, M.; Scheen, R.; Retore, P.; Zissis, G.; van Gool, T. Clinical and Microbiological Features of Dientamoebiasis in Patients Suspected of Suffering from a Parasitic Gastrointestinal Illness: A Comparison of Dientamoeba fragilis and Giardia lamblia Infections. Int. J. Infect. Dis. 2006, 10, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Lhotská, Z.; Jirků, M.; Hložková, O.; Brožová, K.; Jirsová, D.; Stensvold, C.R.; Kolísko, M.; Jirků-Pomajbíková, K. A Study on the Prevalence and Subtype Diversity of the Intestinal Protist Blastocystis sp. in a Gut-Healthy Human Population in the Czech Republic. Front. Cell. Infect. Microbiol. 2020, 10, 544335. [Google Scholar] [CrossRef] [PubMed]
- Šloufová, M.; Lhotská, Z.; Jirků, M.; Petrželková, K.J.; Stensvold, C.R.; Cinek, O.; Jirků-Pomajbíková, K. Comparison of Molecular Diagnostic Approaches for the Detection and Differentiation of the Intestinal Protist Blastocystis sp. in Humans. Parasite 2022, 29, 30. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Arendrup, M.C.; Mølbak, K.; Nielsen, H.V. The Prevalence of Dientamoeba fragilis in Patients with Suspected Enteroparasitic Disease in a Metropolitan Area in Denmark. Clin. Microbiol. Infect. 2007, 13, 839–842. [Google Scholar] [CrossRef]
- Sarzhanov, F.; Dogruman-Al, F.; Santin, M.; Maloney, J.G.; Gureser, A.S.; Karasartova, D.; Taylan-Ozkan, A. Investigation of Neglected Protists Blastocystis sp. And Dientamoeba fragilis in Immunocompetent and Immunodeficient Diarrheal Patients Using Both Conventional and Molecular Methods. PLoS Negl. Trop. Dis. 2021, 15, e0009779. [Google Scholar] [CrossRef]
- Bruijnesteijn van Coppenraet, L.E.S.; Dullaert-de Boer, M.; Ruijs, G.J.H.M.; van der Reijden, W.A.; van der Zanden, A.G.M.; Weel, J.F.L.; Schuurs, T.A. Case-Control Comparison of Bacterial and Protozoan Microorganisms Associated with Gastroenteritis: Application of Molecular Detection. Clin. Microbiol. Infect. 2015, 21, 592.e9–592.e19. [Google Scholar] [CrossRef]
- Heusinkveld, M.; Mughini-Gras, L.; Pijnacker, R.; Vennema, H.; Scholts, R.; van Huisstede-Vlaanderen, K.W.; Kortbeek, T.; Kooistra-Smid, M.; van Pelt, W. Potential Causative Agents of Acute Gastroenteritis in Households with Preschool Children: Prevalence, Risk Factors, Clinical Relevance and Household Transmission. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Burgaña, A.; Abellana, R.; Yordanov, S.Z.; Kazan, R.; Pérez Ortiz, A.M.; Ramos, C.C.; Hernández, C.G.; Rivero, M.M.; Gonçalves, A.Q.; Padilla, E.; et al. Paromomycin Is Superior to Metronidazole in Dientamoeba fragilis Treatment. Int. J. Parasitol. Drugs Drug Resist. 2019, 11, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Venturini, E.; Scarso, S.; Prelazzi, G.A.; Niccolai, C.; Bianchi, L.; Montagnani, C.; Lapini, M.; Chiappini, E.; Antonelli, A.; Rossolini, G.M.; et al. Epidemiology and Clinical Features of Intestinal Protozoan Infections Detected by Real-Time PCR in Non-Native Children within an Italian Tertiary Care Children’s Hospital: A Cross-Sectional Study. Travel Med. Infect. Dis. 2021, 43, 102107. [Google Scholar] [CrossRef] [PubMed]
- Noble, G.A.; Noble, E.R. Entamoebae in Farm Mammals. J. Parasitol. 1952, 38, 571–595. [Google Scholar] [CrossRef]
- Fletcher, S.; Caprarelli, G.; Merif, J.; Andresen, D.; Van Hal, S.; Stark, D.; Ellis, J. Epidemiology and Geographical Distribution of Enteric Protozoan Infections in Sydney, Australia. J. Public Health Res. 2014, 3, 83–91. [Google Scholar] [CrossRef]
- Lagacé-Wiens, P.R.; Van Caeseele, P.G.; Koschik, C. Dientamoeba fragilis: An Emerging Role in Intestinal Disease. Can. Med. Assoc. J. 2006, 175, 468–469. [Google Scholar] [CrossRef]
- Röser, D.; Simonsen, J.; Nielsen, H.V.; Stensvold, C.R.; Mølbak, K. Dientamoeba fragilis in Denmark: Epidemiological Experience Derived from Four Years of Routine Real-Time PCR. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1303–1310. [Google Scholar] [CrossRef]
- Johnson, J.A.; Clark, C.G. Cryptic Genetic Diversity in Dientamoeba fragilis. J. Clin. Microbiol. 2000, 38, 4653–4654. [Google Scholar] [CrossRef]
- Peek, R.; Reedeker, F.R.; van Gool, T. Direct Amplification and Genotyping of Dientamoeba fragilis From Human Stool Specimens. J. Clin. Microbiol. 2004, 42, 631–635. [Google Scholar] [CrossRef]
- Verweij, J.J.; Mulder, B.; Poell, B.; van Middelkoop, D.; Brienen, E.A.T.T.; van Lieshout, L. Real-Time PCR for the Detection of Dientamoeba fragilis in Fecal Samples. Mol. Cell. Probes 2007, 21, 400–404. [Google Scholar] [CrossRef]
- Gouy, M.; Guindon, S.; Gascuel, O. SeaView Version 4: A Multiplatform Graphical User Interface for Sequence Alignment and Phylogenetic Tree Building. Mol. Biol. Evol. 2010, 27, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast Model Selection for Accurate Phylogenetic Estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Kurt, Ö.; Dogruman, F.; Tanyüsel, M. Eradication of Blastocystis in Humans: Really Necessary for All? Parasitol. Int. 2016, 65, 797–801. [Google Scholar] [CrossRef]
- Billy, V.; Lhotská, Z.; Jirků, M.; Kadlecová, O.; Frgelecová, L.; Wegener Parfrey, L.; Jirků, P. Blastocystis Colonization Alters the Gut Microbiome and, In Some Cases, Promotes Faster Recovery from Induced Colitis. Front. Microbiol. 2021, 12, 641483. [Google Scholar]
- Sobotková, K.; Parker, W.; Levá, J.; Růžková., J.; Lukeš, J.; Jirků, P. Helminth Therapy—From the Parasite Perspective. Trends Parasitol. 2019, 35, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Wojciech, L.; Gascoigne, N.R.J.; Peng, G.; Tan, K.S.W. New Insights into the Interactions between Blastocystis, the Gut Microbiota, and Host Immunity. PLoS Pathog. 2021, 17, e1009253. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).