1. Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder and the most common type of dementia (60–80% of the cases). The sporadic form of Alzheimer’s disease with late onset (sAD), where symptoms occur after the age of 65 years, affects over 95% of AD patients. sAD has a complex etiology that is still not fully understood. It is believed that sAD is not hereditary and its development is related to interactions between genetic, environmental and metabolic factors [
1]. The most common diagnostic marker in AD is episodic memory impairment; however, the decline in this memory system correlates with age, and is also a symptom of other types of dementia with a different etiology (e.g., frontotemporal dementia, vascular dementia), which significantly complicates and delays diagnosis [
2,
3]. Recent studies suggest that the impairment of spatial memory, including spatial navigation and orientation, may be a more specific feature of AD (even in the early stages of the disease) that distinguishes AD from other types of age-related dementia and memory deficits [
4,
5].
Disturbances in adult neurogenesis have been proposed as an important mechanism underlying cognitive decline (including memory impairment) observed in age-related neurodegenerative disorders such as AD and, to a limited extent, in normal aging [
6,
7]. In the brain of adult mammals, there are only two regions where new neurons are generated. The subgranular zone (SGZ) of the dentate gyrus (DG) is the neurogenic niche for hippocampal neurogenesis, and the subventricular zone (SVZ) is a region where new neurons are formed and then migrate through rostral migratory stream to the olfactory bulb (OB). Newly generated neurons are incorporated into existing neural circuits of the hippocampus (in the DG and hippocampal subregions CA1–CA3) and play an important role in hippocampus-dependent learning and memory (e.g., spatial navigation, spatial memory and orientation). Moreover, adult hippocampal neurogenesis ensures and maintains the functional integrity of this structure, especially in aged individuals, which allows for the formation of new memories, thereby increasing the efficiency of contextual memory, and facilitates learning [
6,
8]. By contrast, new neurons in the OB play a critical role in perception and memory processes related to odours, which in adults may be associated with behavioural adaptive mechanisms in response to environmental changes. It has been shown that patients with mild cognitive impairment (MCI) and olfactory deficits are more likely to develop AD [
9,
10]. The adult neurogenesis can be regulated by many intrinsic and extrinsic factors. Growth factors and neurotrophins, such as brain-derived neurotrophic factor (BDNF), stimulate neurogenesis, increasing the proliferation, differentiation, maturation and survival of newly formed cells [
11]. On the other hand, oxidative stress, neuroinflammation, insulin signalling disorders and age-related changes have been shown to inhibit neurogenesis by reducing neural stem cell proliferation and neuronal differentiation, as well as by delaying maturation and reducing the survival of new neurons [
12,
13,
14].
Currently, there are only six drugs for the treatment of AD that have been approved by the US Food and Drug Administration (FDA) [
15]. The statistics show that over 90% of the tested substances with the potential to alleviate the symptoms of AD in animal models have not completed clinical trials (or have not been approved at all). One of the reasons for such a high failure rate in clinical trials may be the selection of inappropriate animal models for preclinical studies, mainly reflecting the familial, hereditary form of Alzheimer’s disease (fAD). The animal model of an intracerebroventricular injection of streptozotocin (STZ-ICV) is one of the best characterised and clinically applicable methods to induce sAD-like pathology. Behavioural and neurochemical changes occurring in patients with sAD (e.g., cognitive impairments, insulin signalling disorders, oxidative stress, neuroinflammation, neurodegeneration, Aβ and hyperphosphorylated tau pathology) have also been found with the STZ-ICV model. Moreover, in STZ-ICV animals, the severity of cognitive and neurochemical impairments correlates with age, and such a relationship is also common in human sAD [
1,
16,
17,
18,
19].
None of the pharmacological treatments available today for AD are able to slow or stop the neurochemical changes leading to neurodegeneration that causes AD symptoms and makes the disease fatal. Moreover, they are often associated with decreasing efficacy over time and undesirable side effects. Unfortunately, the discovery of new drugs (design, in vitro studies, animal model testing, clinical trials) requires an average of 15 years and huge financial resources before a therapeutic agent is available to patients. Furthermore, many of the new drugs do not even reach the market, despite all the time and cost invested in research and development. Therefore, the application of established drug compounds to new therapeutic interventions (drug repurposing) may be a solution to this problem.
Dimethyl fumarate (DMF) has antioxidant, anti-inflammatory and neuroprotective properties. In 2013, an oral drug Tecfidera, in which DMF is an active compound, was registered as the primary therapy for patients with relapsing–remitting multiple sclerosis (RRMS) due to its high neuroprotective and immunomodulatory efficacy, a satisfactory level of safety in prolonged treatment and a positive balance of possible benefits and side effects [
20,
21]. One of the main mechanisms responsible for the antioxidant properties of DMF is its influence on the nuclear factor erythroid 2-related factor 2 (Nrf2) signalling pathway. The Nrf-2 target genes control the cell’s redox homeostasis, energy metabolism, DNA repair processes, cell survival and proliferation. The anti-inflammatory and immunosuppressive actions of DMF occur mainly through the inhibition of the nuclear factor kappa-light-chain-enhancer of the activated B cells (NF-κB) pathway [
22]. Recently, the ability of DMF to increase BDNF production has also been confirmed in several studies: after spinal cord injury [
23], in a hypothyroidism rat model [
24] and in a D-galactose/ovariectomy-induced rat model of AD [
25].
In our previous studies, we found that DMF therapy prevented the STZ-ICV-induced disruption of spatial memory, loss of Ch1 cholinergic cells, hippocampal interleukins 6 and 10 induction and neurodegeneration in 4-month-old rats [
17]. We also showed that the STZ-ICV acts age dependently, as the aged animals (22-month-old rats) had greater spatial memory impairments, neurodegeneration, microglia activation and oxidative stress in the hippocampus. We confirmed that DMF therapy reduces age-dependent microglia activation and consequently limits oxidative stress and neurodegeneration in STZ-ICV rats [
18].
The present study aims to determine whether DMF therapy attenuates the disruption of adult neurogenesis in two neurogenic regions—the DG and OB in the STZ-ICV rat model of sAD. The influence of DMF on adult neurogenesis has not been studied so far using an in vivo model of sAD. Furthermore, the present study examines the level of neuroprotection associated with BDNF, which is known to promote neurogenesis and the survival of neurons. Moreover, the present study aims to determine whether the effect of both the STZ-ICV and DMF treatment on the assessed parameters is age dependent, considering the fact that age is one of the most important risk factors for developing AD. For this purpose, cognitive processes, adult neurogenesis and BDNF-related neuroprotection were assessed in two age groups of rats: young (3-month-old) and aged (22-month-old) animals.
3. Discussion
In this study, for the first time, the effect of DMF therapy on neural proliferation, neuronal differentiation and the number of newly formed immature neurons in the DG and OB was tested in the STZ-ICV-induced rat model of sAD. The obtained results suggest that this compound has the ability to reduce disturbances in adult neurogenesis evoked by the STZ-ICV in both examined neurogenic regions of young and aged rats. In studies with animal models of neurodegenerative diseases, the dose of DMF considered effective is in a range of 30 mg/kg to 100 mg/kg, as reviewed by Majkutewicz et al. [
22]. We demonstrated that the average daily DMF dose in our study was 44 mg/kg for all the animals receiving chow with DMF (
Table 1); thus, it was in line with the data reported in the literature.
Most studies involving STZ-ICV-induced animal models of sAD have reported a disruption of adult hippocampal neurogenesis, although there are few studies that have shown no such effect. Guo et al. [
26] did not observe a significant effect of the STZ-ICV on the immature neurons (DCX-positive cells) in the hippocampus of young rats 2 months after the STZ-ICV. Sun et al. [
27] compared the effects of the STZ-ICV on the adult hippocampal neurogenesis of young rats in short-term (1 month after the STZ-ICV) and long-term (3 month after the STZ-ICV) paradigms. No significant influence of the STZ-ICV on adult neurogenesis was found in the short-term paradigm, but in the long-term paradigm, the STZ-ICV reduced the number of mature (NeuN
+) and immature neurons, including mitotic active neuronal cells (NeuroD
+) and postmitotic neuronal cells (DCX
+).
The results obtained by our team suggest that the STZ-ICV led to adult hippocampal abnormalities in the young and aged rats, which occurred 22 days after the last STZ-ICV injection, in contrast to the studies cited above. One explanation for these differences may be a specific time pattern in the severity of the STZ-ICV-induced deficits. It has been shown that dysfunctions evoked by the STZ-ICV are associated with time after the treatment, which includes three phases: (1) acute—developing within one month after the STZ-ICV; (2) a temporary improvement (complete or partial normalisation to the pre-STZ-ICV level) between 1 and 3 months after the treatment; and (3) decompensation with slow and chronic progression in the severity of the disruptions from 6 months after the STZ-ICV. This pattern of the development of cognitive and neurochemical impairment is very similar to the human AD pathology and has also not been found in transgenic animal AD models [
19,
28,
29]. In studies that confirmed a significant impact of the STZ-ICV on adult hippocampal neurogenesis, the analyses were usually performed earlier than 1 month after the STZ-ICV or at least 3 months after the treatment. The authors demonstrated that the STZ-ICV induced a reduction in the proliferation (Ki-67
+ or BrdU+nestin
+), differentiation (DCX
+), maturation (NeuN
+) and survival (BrdU+NeuN
+) of the neurons in the DG of rodents. These changes were accompanied by increased levels of neuroinflammatory markers and oxidative stress [
13,
30,
31,
32,
33]. In the present study, we also found a lower number of BrdU-containing cells and DCX-containing cells in the DG of young and aged STZ rats, which indicates disturbances in the proliferation and differentiation of newly formed cells into a neuronal phenotype. Moreover, in our previous study, we showed that young and aged STZ animals presented a higher number of activated microglia (CD68
+), increased level of oxidative/nitrative stress and neurodegeneration in the hippocampus compared to control rats [
18].
It is considered that gliosis, a noncancerous proliferation of glial cells, may indicate the intensification of neuroinflammation, which is one of the most important pathological mechanisms in neurodegenerative diseases, including AD [
1,
34,
35]. Another factor strongly involved in the pathogenesis of sAD is oxidative stress, which has been shown to be one of the crucial mechanisms leading to the STZ-ICV-induced disturbances [
1,
19]. The reactive oxygen species (ROS) are accumulated during adult neurogenesis as a physiological mechanism. However, the accumulation of high levels of ROS, usually due to overproduction or inadequate clearance, disrupts adult neurogenesis. ROS overload has been shown in neuroinflammation, neurodegeneration and ageing [
14]. It is worth emphasizing that brain tissue contains high levels of lipids and has relatively low activity of enzymatic and nonenzymatic antioxidant protection; thus, it is susceptible to ROS damage, whose accumulation over time may contribute to AD pathogenesis [
36].
Despite demonstrating a strong correlation of olfactory deficits and AD development [
10,
37], little is still known about the mechanisms underlying these disorders. In adult humans, the number of newly formed neurons in the OB is significantly lower compared to rodents. However, there is some evidence indicating that new neurons are continuously incorporated in the human OB during adulthood [
38,
39]. Therefore, the dysfunction of adult neurogenesis in the OB may contributes to the olfactory symptoms occurring in AD. To our knowledge, only one study evaluated the influence of the STZ-ICV on adult neurogenesis in the OB. Mishra et al. [
40] showed that an STZ-ICV reduced the number of newly formed cells (BrdU
+) in the SVZ as well as the number of immature neurons (DCX
+) in the SVZ and OB of young rats 20 days after the STZ-ICV. Our results are in accordance with cited findings, as we also found that the STZ-ICV evoked severe disturbances in the proliferation, neuronal differentiation and survival of newly formed immature neurons in the OB of young and aged rats.
In this study, we confirmed that DMF therapy alleviates STZ-ICV-induced disturbances in adult neurogenesis in both neurogenic regions—in the DG and OB. Moreover, the DMF treatment led to complete normalisation in all the analysed parameters of neurogenesis in the DG, but only in the young animals. In the OB, complete normalisation in the proliferation (BrdU
+) and survival of new immature neurons (BrdU+DCX
+) was found in young and aged rats, whereas the DMF therapy did not restore the neuronal differentiation level in both age groups of animals. There is no study evaluating DMF’s influence on adult neurogenesis in vivo; however, its efficacy has been confirmed in vitro in the stimulation of the differentiation of oligodendrocyte progenitor cells [
41]. Considering the fact that AD is a multiple and complex disease which results from many pathological processes that have a significant role in neuroinflammation and oxidative stress, it can be assumed that an effective therapeutic agent should exhibit several equally strong mechanisms of action. The antioxidant and anti-inflammatory properties of DMF have been well established, and approved drugs in which DMF is a primary therapeutic compound are currently used in the treatment of psoriasis and RRMS. The therapeutic potential of DMF has also been reported in cellular (in vitro) and rodent models of several neurodegenerative diseases [
22,
42]. In our previous studies, we showed that DMF therapy improved a broad spectrum of disturbances evoked by an STZ-ICV in a rat model of sAD. The DMF treatment ameliorated the loss of cholinergic cells in the medial septum projecting to the hippocampus, level of the neuroinflammatory markers, oxidative/nitrative stress and neurodegeneration in the hippocampus of rats subjected to the STZ-ICV [
17,
18]. Several mechanisms of the DMF action (or its metabolite—monomethyl fumarate) have been described, and they may play an important role in the attenuation of least some of the pathological conditions occurring in AD. The beneficial effect of the DMF treatment on adult neurogenesis confirmed in this study may be associated with a significant impact of this compound on Nrf2-dependent and NF-κB-dependent signalling pathways. DMF, by activating Nrf2, stimulates antioxidant enzyme production in many cells, including neurons [
43], as well as the expression of the genes involved in cell survival and proliferation [
20]. It has also been shown that DMF stimulates signal transduction via the PI3K/Akt pathway, which leads to phosphorylation, and thus the inhibition of GSK-3β kinase. The increased activity of GSK-3β is associated with Aβ plaques formation, tau hyperphosphorylation, the promotion of the inflammatory response and enhanced apoptosis of newly formed mature neurons [
44,
45].
BDNF is one of the most potent factors stimulating neurogenesis [
46]; thus, another aim of our study was to determine whether the DMF treatment improved the dysfunction of BDNF-related neuroprotection. We found that the STZ-ICV led to a reduction in BDNF-containing cells in the hippocampus (DG and CA1–CA3 areas) as well as in the OB of rats in both age groups. The young and aged animals receiving the STZ-ICV and fed with DMF-containing chow had a significantly better level of neuroprotection associated with BDNF, which was similar to the control rats (Sham group) of the same age in almost all examined brain structures. A similar effect was found by Luo et al. [
47], who found a decrease in the BDNF level in the hippocampus 4 weeks after the second injection of STZ (the procedure of the administration of STZ and the dose was identical to that of the present study). Oral therapy with DMF alleviated the STZ-induced deficit of BDNF, as our STZ+DMF rats showed more cells expressing BDNF than STZ rats in the hippocampus and olfactory bulb in both age groups. These results are in accordance with the study of Abd El-Fatah et al. [
25], where the beneficial effect of DMF on the BDNF level in the hippocampus was found in another model of AD, resembling postmenopausal dementia. Female 18-month-old ovariectomised rats injected with D-galactose (ip) were used. Two-month oral therapy with DMF normalised the BDNF level in the hippocampus. Taking into account the above data, we expected an alleviation of the decline in BDNF expression due to DMF therapy in the AD model used. Our results on the number of BDNF-containing cells and neurogenesis are in mutual accordance.
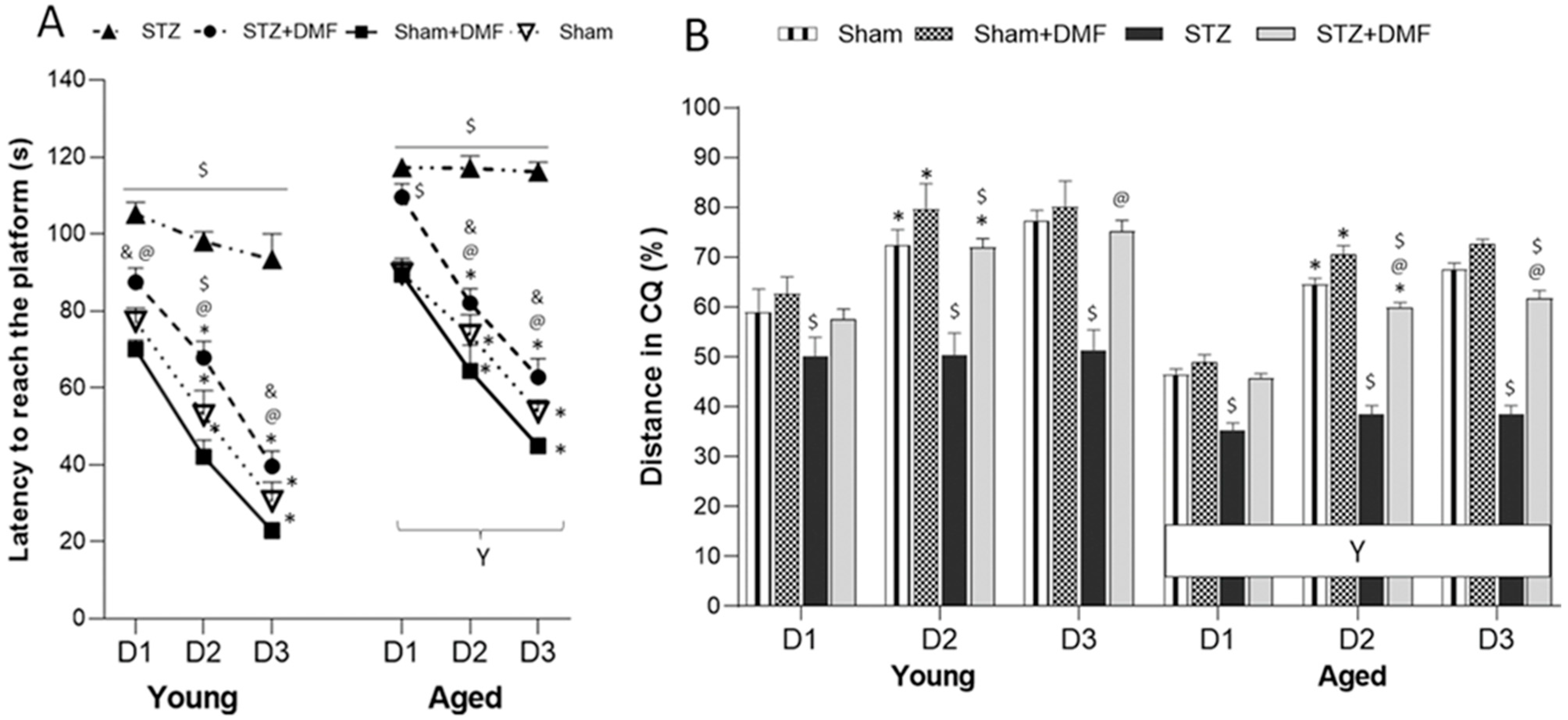
In our study, the STZ-ICV-induced disruption of adult neurogenesis and loss in BDNF-related neuroprotection was accompanied by spatial learning and memory impairment assessed in the MWM test. We showed that the STZ rats presented the most severe cognitive deficits, including the dynamic of spatial learning and working memory performance, and recalling spatial information stored in long-term reference memory. The obtained results indicated that the DMF therapy alleviated these disturbances. Moreover, the DMF treatment led to the normalisation of spatial learning and working memory, as there were only a few significant differences in the measured parameters between the STZ+DMF and control animals (Sham group) in both age groups. The DMF therapy did not fully restore the functionality of long-term reference memory, as the young and aged STZ+DMF rats presented significantly lower levels of the measured parameters compared to the control animals (Sham group) of the same age in the probe test in the MWM.
Our results are in agreement with other studies investigating effect of the STZ-ICV on spatial cognitive processes (e.g., [
33,
48,
49,
50,
51]). The cited data and results obtained in this study may have a great importance in the context of spatial cognitive deficits considered as an early behavioural marker of AD in humans. Spatial navigation is a complex cognitive function that is essential for the independence, quality of life and safety of older adults [
2], and is less influenced by important confounding variables such as educational level, verbal abilities and cultural bias, unlike episodic memory [
52]. The spatial disorientation in AD patients may be related to a decreased density of hippocampal neurons, especially in the CA1 and CA3 areas [
53,
54]. Allison et al. [
55] confirmed that the wayfinding task involving spatial cognitive skills can be used to differentiate healthy older adults from patients with AD. Importantly, spatial cognitive deficits were also found in MCI patients, who are more likely to develop AD [
56]. Our results together with the provided data suggest that spatial cognitive deficits may be a valuable diagnostic tool in the early stage of AD or may be used to predict AD development. The tests assessing spatial memory in humans, e.g., an analogue of the MWM [
57], may enhance the translational value of animal-based research focused on developing new therapeutic strategies.
The results obtained in this study showed that the DMF treatment effectively improved spatial learning and memory deficits in rats subjected to an STZ-ICV. Similar findings were reported by Abd El-Fatah et al. [
25], who found that DMF attenuated memory deficits, hippocampal neurodegeneration and astrocyte activation in ovariectomised D-galactose-injected aged female rats. In another study by Rojo et al. [
58], DMF had a beneficial effect on spatial cognitive disruption in transgenic mouse models of AD with amyloidopathy and tauopathy. These mice exhibited motor deficits and a terminal spinal deformity and died prematurely, at around 14 months of age. Six weeks of therapy with DMF reduced glial and inflammatory markers and improved cognition and motor dysfunctions by stimulating NRF2 activity. Recently, Sun et al. [
59] found that DMF reduced memory impairment and hippocampal atrophy induced by Aβ and ibotenate acid injections into the mice hippocampus, and that it also delayed the progression of AD by an Nrf2-dependent mechanism. So far, the results from only one study are in contrast with the results obtained by our team and the findings cited above. Möhle et al. [
60] demonstrated that the DMF treatment did not ameliorate spatial memory deficits assessed in the MWM and neuroinflammatory processes in the APP/PS1 transgenic mouse model of AD. The authors justified the discrepancies of the results with previous studies by highlighting differences in the models’ characteristics, i.e., blood-brain barrier (BBB) integrity or its disruption, which can facilitate drug penetration into the brain. The STZ-ICV was shown to damage the BBB [
61], whereas, in the model of AD that was similar to that used by Möhle et al. [
60], there was no disruption of the BBB in 6-month-old mice [
62]. However, older APP/PS1 mice (12-month-old) showed an impairment in the BBB [
63]. Therefore, the age of the animals and the age of the onset of symptoms in AD can be the important factor influencing the results of DMF preclinical efficacy in various models of AD. APP/PS1 mice show an early onset of cerebral amyloidosis (between 6–8 weeks of age) [
64], which does not reflect the development of sAD in humans [
19,
29]. The pathogenic role of the Aβ in AD is still controversial, and many researchers believe that the overload of Aβ and plaques formation may represent late events rather than a cause (as reviewed by Alves et al. [
1]). Moreover, it has already been demonstrated that Aβ plaques can be present in older people without cognitive impairment, and the cognitive decline observed in patients with AD or MCI is only weakly related to the change in Aβ deposition [
65,
66]. These findings suggest that animal models of AD focused on Aβ as a main cause of symptom progression may be inadequate. However, further studies with other transgenic and nontransgenic models of AD evaluating DMF’s influence on spatial cognitive deficits and amyloidopathy is undoubtedly required.
Our results demonstrated that the age of the animals was an important factor modifying the effect of the STZ-ICV and DMF therapy on behavioural and neuronal changes. The aspects of age-related differences in the therapeutical effectiveness of DMF in animal models of AD is still poorly known, as most studies use animals of one age.
We confirmed that the aged animals presented worse spatial learning skills, working memory performance and recalling information stored in long-term reference memory compared to the young animals in all experimental groups. The STZ-ICV led to a more severe disruption in spatial cognitive performance, as the aged STZ rats were characterised by the worst scores in every spatial task in the MWM. The DMF treatment restored learning and memory function in both age groups of animals; however, the aged rats coped with the spatial tasks significantly worse compared to the young rats in the STZ+DMF group. Our findings are consistent with the literature data from studies on rodents and humans reviewed by Lester et al. [
67] and Plácido et al. [
68].
The influence of age on adult neurogenesis was also observed in this study. We found that only proliferation (BrdU
+) was more severely disrupted by the STZ-ICV in the aged rats compared to the young STZ animals in the DG, but not in the OB. Age-related differences in neuronal differentiation and the number of newly formed immature neurons in both examined neurogenic regions in the STZ group were not statistically significant. The DMF therapy seemed to be age dependent in both neurogenic regions for all the measured parameters, as the young STZ+DMF rats presented a higher number of labelled cells compared to the aged animals, except for the number of newly formed immature neurons in the DG. This age-dependent pattern was similar to what was found in both control groups (Sham+DMF and Sham). The DMF therapy in the young and aged rats was equally effective at ameliorating adult neurogenesis disturbances evoked by the STZ-ICV in both neurogenic regions. Many studies have confirmed that adult neurogenesis in aged rodents is approximately 3–9 times lower compared to juveniles. The age-related deterioration of adult neurogenesis includes every stage of this process: the proliferation, differentiation, maturation and survival of newly formed neurons [
8,
69,
70,
71]. Age-related decline in adult human neurogenesis remains controversial [
72], although Moreno-Jiménez et al. [
12] showed a negative correlation between the number of immature neurons and age in healthy people. It was found that the number of neural stem cells and neural progenitors, as well as the proportion of astrocytes to neurons in the hippocampus of young and aged rats, remained the same, but significantly fewer cells were actively undergoing mitosis in aged animals. This effect may be caused by a decreased level of proteins stimulating neurogenesis, especially BDNF [
73]. Our results are in agreement with these findings. We found that aged rats showed a lower number of BDNF containing cells in both neurogenic regions. A deficit of BDNF is considered one of most potent factors involved in aging processes in the hippocampus, including the disruption of neurogenesis, changes in hippocampal cytoarchitecture and expression of neurotransmitters [
74,
75]. Interestingly, age-related changes in microglial cells appear much faster in neurogenic zones compared to other regions of the brain, reducing the survival of newly formed neurons [
76]. In our previous study, we found that microglia activation, oxidative/nitrative stress and neurodegeneration in the hippocampus were more severe in the aged rats compared to the young animals, and the DMF treatment was more effective in aged rats [
18]. Considering the fact that age is one of the most important risk factors for developing AD, the results obtained in this study and our previous findings may contribute to a better understanding the role of age-related changes in the STZ-ICV model of sAD, and in the effectiveness of the DMF treatment.