Transcription Pattern of Neurotrophic Factors and Their Receptors in Adult Zebrafish Spinal Cord
Abstract
:1. Introduction
2. Results
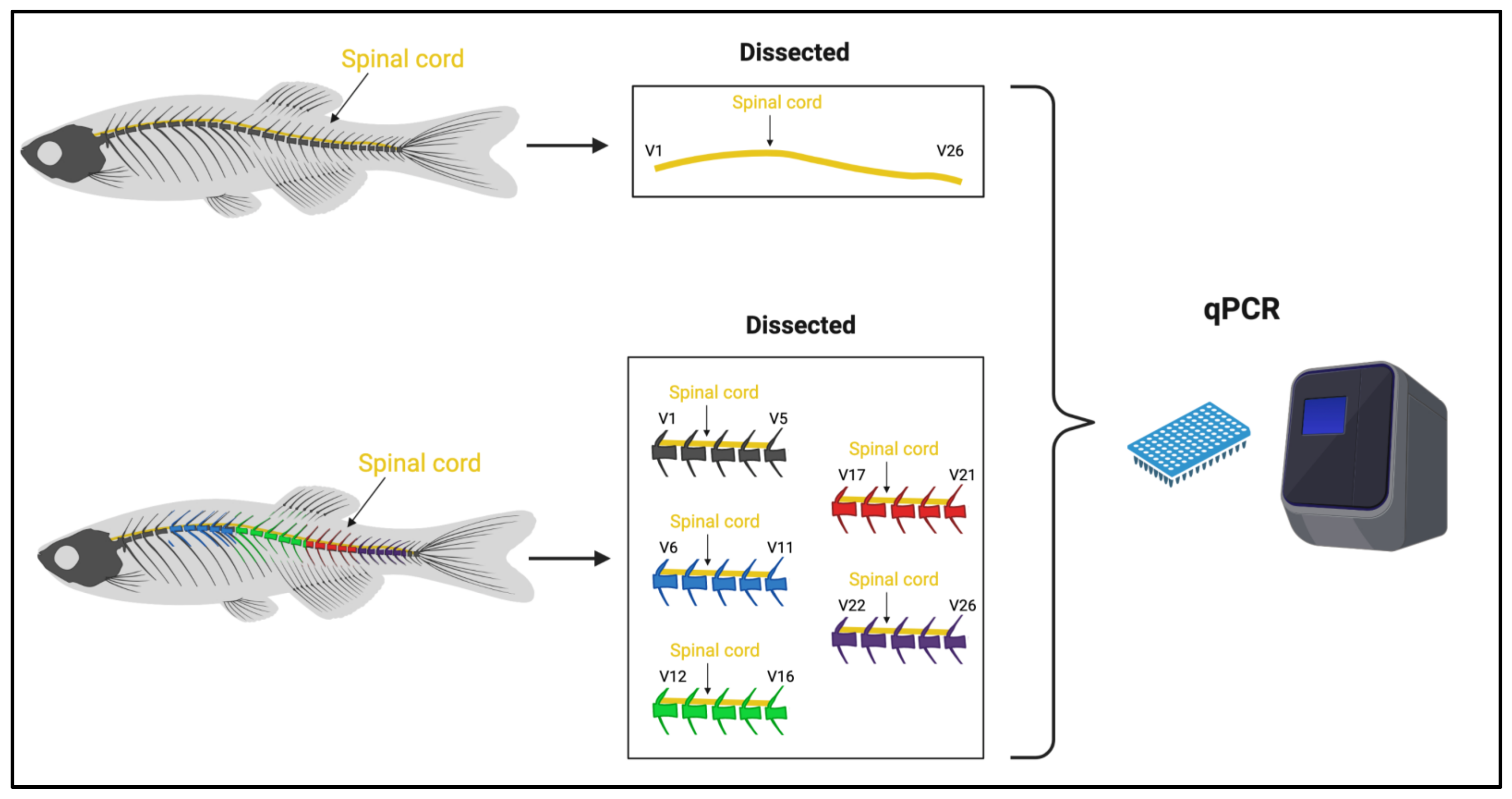
2.1. Experimental Design Outline for qPCR
2.2. Quantitative Analysis of Neurotrophin and Receptor Transcription Levels in Adult Zebrafish Spinal Cord
2.3. Differential Transcription Pattern of Neurotrophins in Adult Zebrafish Spinal Cord
2.4. Distribution of Neurotrophin Receptor Tyrosine Kinase mRNAs in Adult Zebrafish Spinal Cord
3. Discussion
4. Materials and Methods
4.1. Animals and Spinal Cord Dissection
4.2. RNA Extraction
4.3. Reverse Transcriptase PCR
4.4. Quantitative Real-Time PCR
4.5. Statistical Analysis
4.6. Synthesis of Riboprobes for Neurotrophins and Receptors (for In Situ Hybridization)
4.7. In Situ Hybridization
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Cohen, S.; Levi-Montalcini, R.; Hamburger, V. A Nerve Growth-Stimulating Factor Isolated from Sarcom as 37 and 180. Proc. Natl. Acad. Sci. USA 1954, 40, 1014–1018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, S.; Levi-Montalcini, R. A Nerve Growth-Stimulating Factor Isolated from Snake Venom. Proc. Natl. Acad. Sci. USA 1956, 42, 571–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levi-Montalcini, R.; Cohen, S. In Vitro and in Vivo Effects of a Nerve Growth-Stimulating Agent Isolated from Snake Venom. Proc. Natl. Acad. Sci. USA 1956, 42, 695–699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibanez, C.F.; Hallbook, F.; Godeau, F.; Persson, H. Expression of neurotrophin-4 mRNA during oogenesis in Xenopus laevis. Int. J. Dev. Biol. 1992, 36, 239–245. [Google Scholar] [PubMed]
- Berkemeier, L.R.; Winslow, J.W.; Kaplan, D.R.; Nikolics, K.; Goeddel, D.V.; Rosenthal, A. Neurotrophin-5: A novel neurotrophic factor that activates trk and trkB. Neuron 1991, 7, 857–866. [Google Scholar] [CrossRef]
- Koliatsos, V.E.; Cayouette, M.H.; Berkemeier, L.R.; Clatterbuck, R.E.; Price, D.L.; Rosenthal, A. Neurotrophin 4/5 is a trophic factor for mammalian facial motor neurons. Proc. Natl. Acad. Sci. USA 1994, 91, 3304–3308. [Google Scholar] [CrossRef] [Green Version]
- Hallbook, F. Evolution of the vertebrate neurotrophin and Trk receptor gene families. Curr. Opin Neurobiol. 1999, 9, 616–621. [Google Scholar] [CrossRef]
- Spaink, H.P.; Jansen, H.J.; Dirks, R.P. Advances in genomics of bony fish. Brief. Funct. Genom. 2014, 13, 144–156. [Google Scholar] [CrossRef] [Green Version]
- Hallbook, F.; Lundin, L.G.; Kullander, K. Lampetra fluviatilis neurotrophin homolog, descendant of a neurotrophin ancestor, discloses the early molecular evolution of neurotrophins in the vertebrate subphylum. J. Neurosci. 1998, 18, 8700–8711. [Google Scholar] [CrossRef] [Green Version]
- Tettamanti, G.; Cattaneo, A.G.; Gornati, R.; de Eguileor, M.; Bernardini, G.; Binelli, G. Phylogenesis of brain-derived neurotrophic factor (BDNF) in vertebrates. Gene 2010, 450, 85–93. [Google Scholar] [CrossRef]
- De Girolamo, P.; D’Angelo, L. Neurotrophins in the Brain of Teleost Fish: The State of the Art. Adv. Exp. Med. Biol. 2021, 1331, 289–307. [Google Scholar] [CrossRef]
- Dos Santos, S.; Mazan, S.; Venkatesh, B.; Cohen-Tannoudji, J.; Querat, B. Emergence and evolution of the glycoprotein hormone and neurotrophin gene families in vertebrates. BMC Evol. Biol. 2011, 11, 332. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, A.S.; Fainzilber, M.; Falck, P.; Ibanez, C.F. Neurotrophin-7: A novel member of the neurotrophin family from the zebrafish. FEBS Lett. 1998, 424, 285–290. [Google Scholar] [CrossRef] [Green Version]
- Jaillon, O.; Aury, J.M.; Brunet, F.; Petit, J.L.; Stange-Thomann, N.; Mauceli, E.; Bouneau, L.; Fischer, C.; Ozouf-Costaz, C.; Bernot, A.; et al. Genome duplication in the teleost fish Tetraodon nigroviridis reveals the early vertebrate proto-karyotype. Nature 2004, 431, 946–957. [Google Scholar] [CrossRef] [Green Version]
- Martin, S.C.; Sandell, J.H.; Heinrich, G. Zebrafish TrkC1 and TrkC2 receptors define two different cell populations in the nervous system during the period of axonogenesis. Dev. Biol. 1998, 195, 114–130. [Google Scholar] [CrossRef] [Green Version]
- Martin, S.C.; Marazzi, G.; Sandell, J.H.; Heinrich, G. Five Trk receptors in the zebrafish. Dev. Biol. 1995, 169, 745–758. [Google Scholar] [CrossRef] [Green Version]
- Cacialli, P. Neurotrophins Time Point Intervention after Traumatic Brain Injury: From Zebrafish to Human. Int. J. Mol. Sci. 2021, 22, 1585. [Google Scholar] [CrossRef]
- Scarisbrick, I.A.; Isackson, P.J.; Windebank, A.J. Differential expression of brain-derived neurotrophic factor, neurotrophin-3, and neurotrophin-4/5 in the adult rat spinal cord: Regulation by the glutamate receptor agonist kainic acid. J. Neurosci. 1999, 19, 7757–7769. [Google Scholar] [CrossRef] [Green Version]
- Skup, M.; Dwornik, A.; Macias, M.; Sulejczak, D.; Wiater, M.; Czarkowska-Bauch, J. Long-term locomotor training up-regulates TrkB(FL) receptor-like proteins, brain-derived neurotrophic factor, and neurotrophin 4 with different topographies of expression in oligodendroglia and neurons in the spinal cord. Exp. Neurol. 2002, 176, 289–307. [Google Scholar] [CrossRef]
- Rahman, M.M.; Islam, M.R.; Supti, F.A.; Dhar, P.S.; Shohag, S.; Ferdous, J.; Shuvo, S.K.; Akter, A.; Hossain, M.S.; Sharma, R. Exploring the Therapeutic Effect of Neurotrophins and Neuropeptides in Neurodegenerative Diseases: At a Glance. Mol. Neurobiol. 2023, 60, 4206–4231. [Google Scholar] [CrossRef]
- Ricci, S.; Cacialli, P. Stem Cell Research Tools in Human Metabolic Disorders: An Overview. Cells 2021, 10, 2681. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Zhang, X.; Zhai, J.; Xue, J. The roles and applications of neural stem cells in spinal cord injury repair. Front. Bioeng. Biotechnol. 2022, 10, 966866. [Google Scholar] [CrossRef] [PubMed]
- Choi, T.Y.; Choi, T.I.; Lee, Y.R.; Choe, S.K.; Kim, C.H. Zebrafish as an animal model for biomedical research. Exp. Mol. Med. 2021, 53, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Cacialli, P.; Lucini, C. Adult neurogenesis and regeneration in zebrafish brain: Are the neurotrophins involved in? Neural Regen. Res. 2019, 14, 2067–2068. [Google Scholar] [CrossRef] [PubMed]
- El-Daher, F.; Becker, C.G. Neural circuit reorganisation after spinal cord injury in zebrafish. Curr. Opin Genet. Dev. 2020, 64, 44–51. [Google Scholar] [CrossRef]
- Hui, S.P.; Nag, T.C.; Ghosh, S. Neural cells and their progenitors in regenerating zebrafish spinal cord. Int. J. Dev. Biol. 2020, 64, 353–366. [Google Scholar] [CrossRef]
- Tsata, V.; Wehner, D. Know How to Regrow-Axon Regeneration in the Zebrafish Spinal Cord. Cells 2021, 10, 1404. [Google Scholar] [CrossRef]
- Tsata, V.; Kroehne, V.; Wehner, D.; Rost, F.; Lange, C.; Hoppe, C.; Kurth, T.; Reinhardt, S.; Petzold, A.; Dahl, A.; et al. Reactive oligodendrocyte progenitor cells (re-)myelinate the regenerating zebrafish spinal cord. Development 2020, 147, dev193946. [Google Scholar] [CrossRef]
- Shen, W.Y.; Fu, X.H.; Cai, J.; Li, W.C.; Fan, B.Y.; Pang, Y.L.; Zhao, C.X.; Abula, M.; Kong, X.H.; Yao, X.; et al. Identification of key genes involved in recovery from spinal cord injury in adult zebrafish. Neural Regen. Res. 2022, 17, 1334–1342. [Google Scholar] [CrossRef]
- Menke, A.L.; Spitsbergen, J.M.; Wolterbeek, A.P.; Woutersen, R.A. Normal anatomy and histology of the adult zebrafish. Toxicol. Pathol. 2011, 39, 759–775. [Google Scholar] [CrossRef]
- Zeng, C.W.; Sheu, J.C.; Tsai, H.J. The Neuronal Regeneration of Adult Zebrafish after Spinal Cord Injury Is Enhanced by Transplanting Optimized Number of Neural Progenitor Cells. Cell Transplant. 2020, 29, 963689720903679. [Google Scholar] [CrossRef] [Green Version]
- Aragona, M.; Porcino, C.; Guerrera, M.C.; Montalbano, G.; Laura, R.; Cometa, M.; Levanti, M.; Abbate, F.; Cobo, T.; Capitelli, G.; et al. The BDNF/TrkB Neurotrophin System in the Sensory Organs of Zebrafish. Int. J. Mol. Sci. 2022, 23, 10411. [Google Scholar] [CrossRef]
- Aragona, M.; Porcino, C.; Guerrera, M.C.; Montalbano, G.; Levanti, M.; Abbate, F.; Laura, R.; Germana, A. Localization of Neurotrophin Specific Trk Receptors in Mechanosensory Systems of Killifish (Nothobranchius guentheri). Int. J. Mol. Sci. 2021, 22, 10411. [Google Scholar] [CrossRef]
- Cacialli, P.; Gueguen, M.M.; Coumailleau, P.; D’Angelo, L.; Kah, O.; Lucini, C.; Pellegrini, E. BDNF Expression in Larval and Adult Zebrafish Brain: Distribution and Cell Identification. PLoS ONE 2016, 11, e0158057. [Google Scholar] [CrossRef] [Green Version]
- Cacialli, P.; D’Angelo, L.; de Girolamo, P.; Avallone, L.; Lucini, C.; Pellegrini, E.; Castaldo, L. Morpho-Functional Features of the Gonads of Danio rerio: The Role of Brain-Derived Neurotrophic Factor. Anat. Rec. 2018, 301, 140–147. [Google Scholar] [CrossRef] [Green Version]
- Cacialli, P. Expression of Nerve Growth Factor and Its Receptor TrkA in the Reproductive System of Adult Zebrafish. Vet. Sci. 2022, 9, 225. [Google Scholar] [CrossRef]
- Cacialli, P.; Gatta, C.; D’Angelo, L.; Leggieri, A.; Palladino, A.; de Girolamo, P.; Pellegrini, E.; Lucini, C. Nerve growth factor is expressed and stored in central neurons of adult zebrafish. J. Anat. 2019, 235, 167–179. [Google Scholar] [CrossRef]
- Gatta, C.; Schiano, V.; Attanasio, C.; Lucini, C.; Palladino, A. Neurotrophins in Zebrafish Taste Buds. Animals 2022, 12, 1613. [Google Scholar] [CrossRef]
- Germana, A.; Gonzalez-Martinez, T.; Catania, S.; Laura, R.; Cobo, J.; Ciriaco, E.; Vega, J.A. Neurotrophin receptors in taste buds of adult zebrafish (Danio rerio). Neurosci. Lett. 2004, 354, 189–192. [Google Scholar] [CrossRef]
- Cacialli, P.; Lucini, C. Analysis of the Expression of Neurotrophins and Their Receptors in Adult Zebrafish Kidney. Vet. Sci. 2022, 9, 296. [Google Scholar] [CrossRef]
- Gatta, C.; Altamura, G.; Avallone, L.; Castaldo, L.; Corteggio, A.; D’Angelo, L.; de Girolamo, P.; Lucini, C. Neurotrophins and their Trk-receptors in the cerebellum of zebrafish. J. Morphol. 2016, 277, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Mollmert, S.; Kharlamova, M.A.; Hoche, T.; Taubenberger, A.V.; Abuhattum, S.; Kuscha, V.; Kurth, T.; Brand, M.; Guck, J. Zebrafish Spinal Cord Repair Is Accompanied by Transient Tissue Stiffening. Biophys. J. 2020, 118, 448–463. [Google Scholar] [CrossRef] [PubMed]
- Stil, A.; Drapeau, P. Neuronal labeling patterns in the spinal cord of adult transgenic Zebrafish. Dev. Neurobiol. 2016, 76, 642–660. [Google Scholar] [CrossRef] [PubMed]
- Large, T.H.; Weskamp, G.; Helder, J.C.; Radeke, M.J.; Misko, T.P.; Shooter, E.M.; Reichardt, L.F. Structure and developmental expression of the nerve growth factor receptor in the chicken central nervous system. Neuron 1989, 2, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Ip, F.C.; Cheung, J.; Ip, N.Y. The expression profiles of neurotrophins and their receptors in rat and chicken tissues during development. Neurosci. Lett. 2001, 301, 107–110. [Google Scholar] [CrossRef]
- Averill, S.; McMahon, S.B.; Clary, D.O.; Reichardt, L.F.; Priestley, J.V. Immunocytochemical localization of trkA receptors in chemically identified subgroups of adult rat sensory neurons. Eur. J. Neurosci. 1995, 7, 1484–1494. [Google Scholar] [CrossRef]
- Mu, X.; Silos-Santiago, I.; Carroll, S.L.; Snider, W.D. Neurotrophin receptor genes are expressed in distinct patterns in developing dorsal root ganglia. J. Neurosci. 1993, 13, 4029–4041. [Google Scholar] [CrossRef] [Green Version]
- Josephson, A.; Widenfalk, J.; Trifunovski, A.; Widmer, H.R.; Olson, L.; Spenger, C. GDNF and NGF family members and receptors in human fetal and adult spinal cord and dorsal root ganglia. J. Comp. Neurol. 2001, 440, 204–217. [Google Scholar] [CrossRef]
- Funakoshi, H.; Frisen, J.; Barbany, G.; Timmusk, T.; Zachrisson, O.; Verge, V.M.; Persson, H. Differential expression of mRNAs for neurotrophins and their receptors after axotomy of the sciatic nerve. J. Cell Biol. 1993, 123, 455–465. [Google Scholar] [CrossRef]
- Hajebrahimi, Z.; Mowla, S.J.; Movahedin, M.; Tavallaei, M. Gene expression alterations of neurotrophins, their receptors and prohormone convertases in a rat model of spinal cord contusion. Neurosci. Lett. 2008, 441, 261–266. [Google Scholar] [CrossRef]
- Ramer, M.S.; Priestley, J.V.; McMahon, S.B. Functional regeneration of sensory axons into the adult spinal cord. Nature 2000, 403, 312–316. [Google Scholar] [CrossRef]
- Lever, I.J.; Bradbury, E.J.; Cunningham, J.R.; Adelson, D.W.; Jones, M.G.; McMahon, S.B.; Marvizon, J.C.; Malcangio, M. Brain-derived neurotrophic factor is released in the dorsal horn by distinctive patterns of afferent fiber stimulation. J. Neurosci. 2001, 21, 4469–4477. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, N.P.; Aldskogius, H.; Grant, G.; Lindsay, R.M.; Rivero-Melian, C. Effects of nerve growth factor, brain-derived neurotrophic factor and neurotrophin-3 on the laminar distribution of transganglionically fransported choleragenoid in the spinal cord dorsal horn following transection of the sciatic nerve in the adult rat. Neuroscience 1997, 78, 863–872. [Google Scholar] [CrossRef]
- Dalton, V.S.; Roberts, B.L.; Borich, S.M. Brain derived neurotrophic factor and trk B mRNA expression in the brain of a brain stem-spinal cord regenerating model, the European eel, after spinal cord injury. Neurosci. Lett. 2009, 461, 275–279. [Google Scholar] [CrossRef]
- Coulibaly, A.P.; Deer, M.R.; Isaacson, L.G. Distribution and phenotype of TrkB oligodendrocyte lineage cells in the adult rat spinal cord. Brain Res. 2014, 1582, 21–33. [Google Scholar] [CrossRef] [Green Version]
- Bao, S.S.; Zhao, C.; Chen, H.W.; Feng, T.; Guo, X.J.; Xu, M.; Rao, J.S. NT3 treatment alters spinal cord injury-induced changes in the gray matter volume of rhesus monkey cortex. Sci. Rep. 2022, 12, 5919. [Google Scholar] [CrossRef]
- Zhao, C.; Rao, J.S.; Duan, H.; Hao, P.; Shang, J.; Fan, Y.; Zhao, W.; Gao, Y.; Yang, Z.; Sun, Y.E.; et al. Chronic spinal cord injury repair by NT3-chitosan only occurs after clearance of the lesion scar. Signal Transduct. Target Ther. 2022, 7, 184. [Google Scholar] [CrossRef]
- Keefe, K.M.; Sheikh, I.S.; Smith, G.M. Targeting Neurotrophins to Specific Populations of Neurons: NGF, BDNF, and NT-3 and Their Relevance for Treatment of Spinal Cord Injury. Int. J. Mol. Sci. 2017, 18, 548. [Google Scholar] [CrossRef] [Green Version]
- Sakuma, K.; Watanabe, K.; Sano, M.; Uramoto, I.; Nakano, H.; Li, Y.J.; Kaneda, S.; Sorimachi, Y.; Yoshimoto, K.; Yasuhara, M.; et al. A possible role for BDNF, NT-4 and TrkB in the spinal cord and muscle of rat subjected to mechanical overload, bupivacaine injection and axotomy. Brain Res. 2001, 907, 1–19. [Google Scholar] [CrossRef]
- Long, S.L.; Liu, F.; Wang, T.H.; Xu, X.Y.; Guang, Y.G.; Wang, T.W.; Ke, Q.; Yuan, Y. The changes of NT-4 expression in spared root ganglion and spinal cord following partial dorsal root rhizotomy. Sichuan Da Xue Xue Bao Yi Xue Ban 2005, 36, 35–38. [Google Scholar]
- Nittoli, V.; Sepe, R.M.; Coppola, U.; D’Agostino, Y.; De Felice, E.; Palladino, A.; Vassalli, Q.A.; Locascio, A.; Ristoratore, F.; Spagnuolo, A.; et al. A comprehensive analysis of neurotrophins and neurotrophin tyrosine kinase receptors expression during development of zebrafish. J. Comp. Neurol. 2018, 526, 1057–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]








| bdnf | F: CGAGGAATAGACAAGCGGCA; | R: ATCCGTATAAACCGCCAGCC |
| ngf | F: GAGAAGACTACAAGCGAAT; | R: CGACAACAATAAGGAGGAT |
| nt3 | F: CCCATCAGTGCGCTCATC; | R: TCCGAACTGTCCACCATG |
| nt4/5 | F: GCTCCTCCTAGAACAGAG; | R: CGTCCTGGATGCATCTTCT |
| nt6/7 | F: GCATTTACAATGGCAGCCAG; | R: CTTCTTGAGTGGTCACTGTC |
| gapdh | F: GCTGGCATCTCCCTCAA | R: TCAGCAACACGATGGCTG |
| trka | F: GCATTTACAATGGCAGCCAG; | R: CTTCTTGAGTGGTCACTGTC |
| trkb1 | F: TCACCTATGGCAAGCAACCC | R: CTTTGGGGCAAGTACGAGGT |
| trkb2 | F: GAAGTTCTACTCGAATCTCAGG | R: CCAGATGTTCTCACATGCAC |
| trkc1 | F: CGGAAGTGGATTGGACAGTT; | R: CATGAAGCCGTTATCGTCC |
| trkc2 | F: CTCAAGCATCTTCCAGGGT | R: GATCTGCCGTAGATTGCAG |
| gapdh | F: GCTGGCATCTCCCTCAA | R: TCAGCAACACGATGGCTG |
| bdnf | F: ATAGTAACGAACAGGATGG | R: GCTCAGTCATGGGAGTCC |
| ngf | F: CACAGGAGATCTACGC | R: CGTGGAAAAACCCAACTC |
| nt3 | F: TGGTTACCTTTATTACGATC | R: CCACCATTTTTCACGTCC |
| nt4/5 | F: CAGAGAAGATGCATCCAGG | R: CGTTTCCTGACACGCG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cacialli, P.; Ricci, S.; Lazzari, M.; Milani, L.; Franceschini, V. Transcription Pattern of Neurotrophic Factors and Their Receptors in Adult Zebrafish Spinal Cord. Int. J. Mol. Sci. 2023, 24, 10953. https://doi.org/10.3390/ijms241310953
Cacialli P, Ricci S, Lazzari M, Milani L, Franceschini V. Transcription Pattern of Neurotrophic Factors and Their Receptors in Adult Zebrafish Spinal Cord. International Journal of Molecular Sciences. 2023; 24(13):10953. https://doi.org/10.3390/ijms241310953
Chicago/Turabian StyleCacialli, Pietro, Serena Ricci, Maurizio Lazzari, Liliana Milani, and Valeria Franceschini. 2023. "Transcription Pattern of Neurotrophic Factors and Their Receptors in Adult Zebrafish Spinal Cord" International Journal of Molecular Sciences 24, no. 13: 10953. https://doi.org/10.3390/ijms241310953






