Abstract
Zinc (Zn2+) is released by platelets during a hemostatic response to injury. Extracellular zinc ([Zn2+]o) initiates platelet activation following influx into the platelet cytosol. However, the mechanisms that permit Zn2+ influx are unknown. Fluctuations in intracellular zinc ([Zn2+]i) were measured in fluozin-3-loaded platelets using fluorometry and flow cytometry. Platelet activation was assessed using light transmission aggregometry. The detection of phosphoproteins was performed by Western blotting. [Zn2+]o influx and subsequent platelet activation were abrogated by blocking the sodium/calcium exchanged, TRP channels, and ZIP7. Cation store depletion regulated Zn2+ influx. [Zn2+]o stimulation resulted in the phosphorylation of PKC substates, MLC, and β3 integrin. Platelet activation via GPVI or Zn2+ resulted in ZIP7 phosphorylation in a casein kinase 2-dependent manner and initiated elevations of [Zn2+]i that were sensitive to the inhibition of Orai1, ZIP7, or IP3R-mediated pathways. These data indicate that platelets detect and respond to changes in [Zn2+]o via influx into the cytosol through TRP channels and the NCX exchanger. Platelet activation results in the externalization of ZIP7, which further regulates Zn2+ influx. Increases in [Zn2+]i contribute to the activation of cation-dependent enzymes. Sensitivity of Zn2+ influx to thapsigargin indicates a store-operated pathway that we term store-operated Zn2+ entry (SOZE). These mechanisms may affect platelet behavior during thrombosis and hemostasis.
1. Introduction
Dietary zinc (Zn2+) deficiency results in prolonged bleeding phenotypes in both rodents [1,2,3] and humans [4,5,6] that can be reversed by dietary supplementation. Plasma Zn2+ concentrations range from 10 to 20 µM; however, binding to plasma proteins (i.e., albumin and α2 microglobulin) reduces the free (labile) concentration to around 0.5 µM [7,8,9]. Zn2+ concentrations are elevated within atherosclerotic plaques and at sites of vascular injury as a result of a release from platelet granules, and are likely to be much higher than the average plasma concentration [10,11].
At concentrations of 100 μM, Zn2+ acts as a platelet agonist, regulating intracellular signaling resulting in granule secretion, the tyrosine phosphorylation of protein kinase C (PKC), integrin αIIbβ3 activation, and platelet aggregation [12,13,14]. At low concentrations, Zn2+ is a coactivator, potentiating platelet activation to threshold concentrations of conventional agonists [12,15]. Previous research has demonstrated changes in platelet intracellular Zn2+ concentrations [Zn2+]i following agonist stimulation, indicating a release from intracellular stores, which is consistent the a role of Zn2+ as an intracellular secondary messenger [14]. Agonist-evoked increases in [Zn2+]i in the absence of [Zn2+]o result in platelet activation, shape change, degranulation, and phosphatidyl-serine exposure. Similarly, Zn2+ regulates Ca2+ release from stores and may therefore be a central factor in regulating platelet activation [16,17].
In nucleated cells, Zn2+ permeability across the cell membrane is regulated by Zn2+ transporting proteins, including non-selective cation channels, transporters, and exchangers [18,19,20]. Cellular Zn2+ homeostasis is regulated by Zn2+ transporters (ZnTs) and Zrt-Irt-like proteins (ZIPs), of which transcripts are detected in megakaryocytes [20,21,22]. Although the Na2+/Ca2+ exchanger (NCX), operating in reverse mode, and transient receptor potential (TRP) channels have been reported to facilitate Zn2+ movement in nucleated cells, their role in platelet Zn2+ homeostasis has yet to be investigated [23,24]. [Zn2+]i buffering systems are important mechanisms for regulating Zn2+ bioavailability [25]. In platelets, agonist-evoked increases in [Zn2+]i are regulated by changes in the platelet redox state, suggestive of a role for redox-sensitive proteins, such as metallothioneins [26].
Intracellular platelet calcium concentrations ([Ca2+]i) are maintained by store-operated Ca2+ entry (SOCE) and receptor-operated Ca2+ entry (ROCE) pathways, where the dense tubular system (DTS) is the primary Ca2+ store [27,28,29]. SOCE is induced via the activation of cation-selective CRAC channels [30,31,32,33]. The resting platelet Ca2+ concentration ([Ca2+]rest) is maintained at approximately 100 nM by the action of channels and exchangers expressed along the plasma membrane and the surface of the DTS [34,35]. Interestingly, ryanodine receptors, expressed along the cardiomyocyte sarcoplasmic reticulum, have been shown to be regulated by the elevation of [Zn2+]i; although, it is not clear whether the equivalent IP3R in platelets is modulated in a similar manner [36,37].
We have previously shown that the exposure of platelets to [Zn2+]o leads to its rapid and sustained accumulation within the platelet cytosol [14,26]. In this study, we investigate the mechanisms underpinning platelet Zn2+ influx, and the storage mechanisms that contribute to Zn2+-induced platelet activation (ZIPA).
2. Results
2.1. TRP Channels and Reverse-Mode NCX Regulate Zn2+ Influx during Zn2+-Induced Platelet Activation
Zn2+ acts in a dual manner to regulate platelet activation. When applied extracellularly, Zn2+ gains access to the platelet cytosol, whilst, in the absence of extracellular Zn2+, platelet activation via canonical receptors, such as GPVI, results in increases in intracellular Zn2+ as a result of a release from stores [12,14,26]. Increases in [Zn2+]i via either pathway results in ZIPA, which is sensitive to a range of inhibitors and metal ion chelators [12]. However, the molecular identity and the contribution of cation channels underlying Zn2+ influx in platelets have not been investigated. TRP proteins are non-selective cation channel of which, TRPC6, TRPC1, TRPV1, and TRPC5 are expressed in platelets or megakaryocytes, and have been reported to facilitate cellular Zn2+ uptake in nucleated cells [24,38,39,40]. Additionally, the Na+/Ca2+ exchanger (NCX) substitutes Ca2+ for Zn2+ in order to mediate Zn2+ influx in intestinal epithelial cells [18,41].
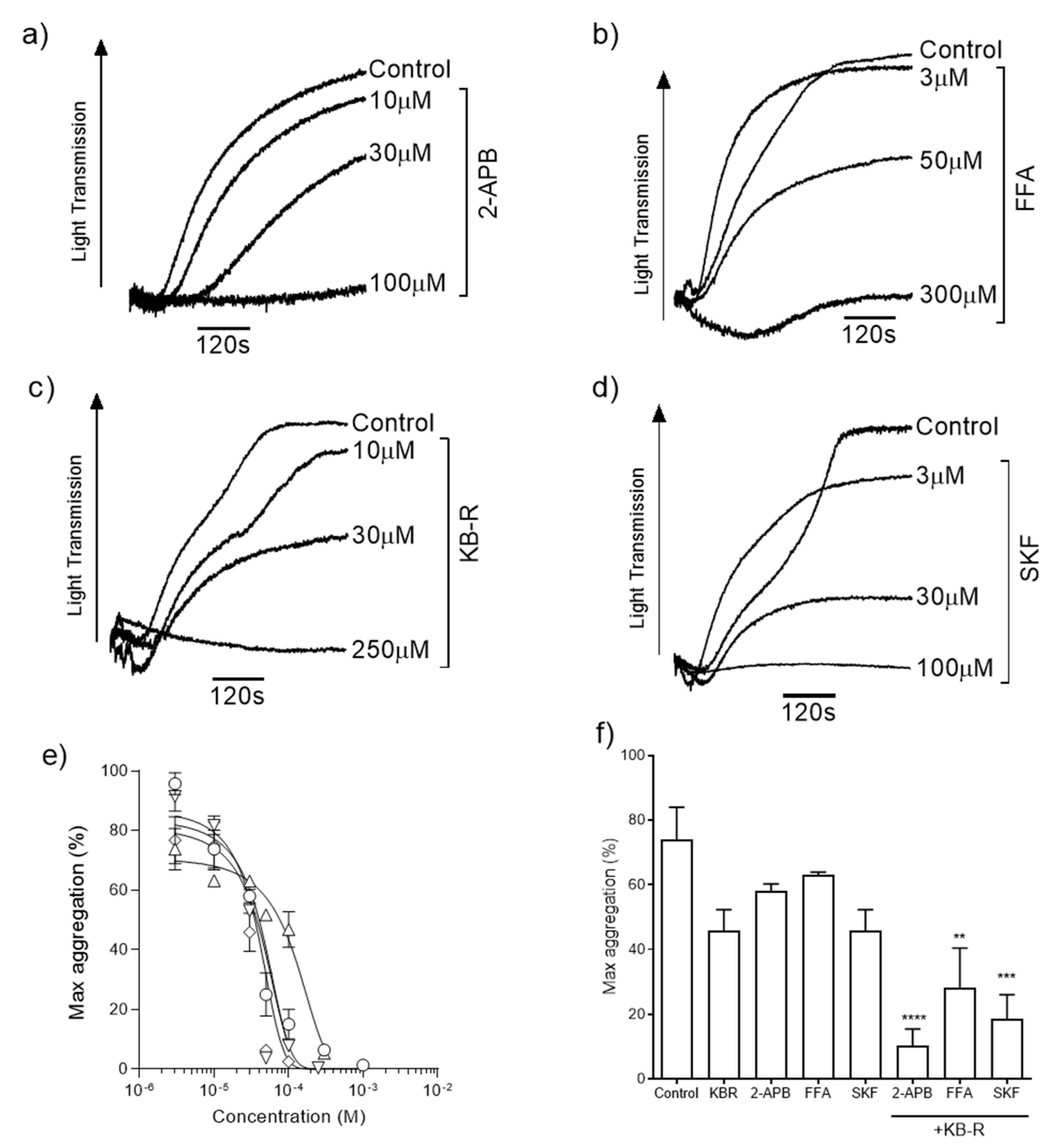
We evaluated the roles of TRP channels and NCX during ZIPA. Platelets were stimulated by exposure to 100 µM ZnSO4 in the presence of increasing concentrations of TRP channel blockers 2-APB (Figure 1a), FFA (Figure 1b), and SKF (Figure 1d), or KB-R7943 mesylate, which is a reverse-mode NCX inhibitor and has also been shown to be a potent blocker of TRP channels (KB-R, Figure 1c) [42]. ZIPA was reduced in a concentration-dependent manner following treatment with each inhibitor. Calculated IC50 values (Figure 1e, Table 1) were comparable for 2-APB, KB-R, and SKF, whilst the curve for FFA was right-shifted. Platelet aggregation responses to 100 µM Zn2+ were reduced from 73.9 ± 12.1% in the presence of KB-R alone, and to 10.4 ± 5.2% by the combined blocking of KB-R and 2-APB (Figure 1f, p < 0.01). Residual ZIPA responses suggested that multiple Zn2+-permeable channels, and potentially Zn2+ transporters, contributed to platelet responses to Zn2+.
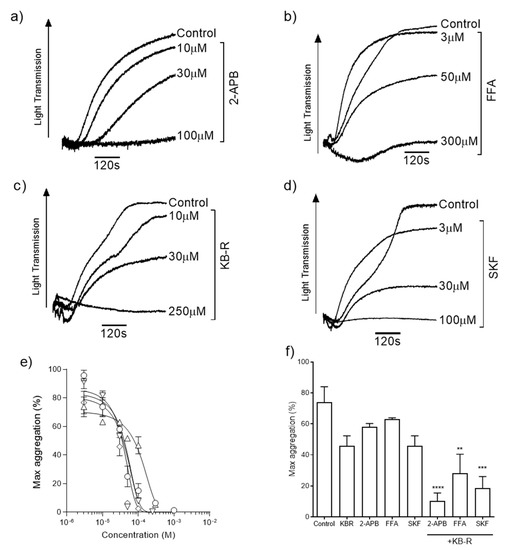
Figure 1.
Inhibition of Zn2+-induced aggregation by TRP channel and NCX blockers. Representative aggregation traces for washed platelets stimulated with Zn2+ in the presence of ion channel inhibitors 30 µM 2-APB (a), 30 µM FFA (b), 30 µM KB-R (c), and 30 µM SKF (d). Concentration–response relationships were calculated for each reagent (e) and IC50 values are reported in Table 1. Co-application of 30 µM KB-R plus 30 µM 2-APB, 30 µM FFA, or 30 µM SKF caused a further inhibition of platelet aggregation relative to the vehicle control (f). Experiments were performed in the absence of [Ca2+]o. Data are the means ± SEM of independent experiments. p < 0.0001 (****), p < 0.001 (***), and p < 0.01 (**) are indicated.

Table 1.
IC50 values for the inhibition of 100 µM Zn2+-induced platelet aggregation.
The stimulation of platelets with [Zn2+]o leads to a concentration-dependent increase in [Zn2+]i; however, the nature of the Zn2+ influx pathway or the relative contribution of the release of [Zn2+]i stores remains unknown [12]. We assessed the role of NCX and TRP channels in platelet Zn2+ influx using FZ-3-loaded platelets. Platelets were stimulated by 100 µM Zn2+ following treatment with inhibitors, KB-R or SN-6, or 2-APB and the resultant Zn2+ signals were quantified using flow cytometry (Figure 2a). An inhibition of Zn2+ influx was observed upon pre-incubation with 30 µM KB-R (to 2611 ± 516 a.u.) or 0.5 µM SN-6, (to 2537 ± 204 a.u.) compared with 4882 ± 531 a.u. for the vehicle control, representing decreases of 53.4 ± 3.2% and 51.9 ± 1.8%, respectively (Figure 2a, p < 0.01). The pre-incubation of FZ-3-loaded platelets with 2-APB reduced the peak Zn2+ influx to 2035 ± 322 a.u. (41.7 ± 3.6% of vehicle control, Figure 2a, p < 0.001), and to 2662 + 420 a.u. following co-treatment with 30 µM KB-R and 2-APB, as observed in the platelet aggregation (54.5 ± 3.6% of vehicle control, Figure 2a, p < 0.001). These data are suggestive of a constitutively active Zn2+ entry pathway that allows platelets to sense changes in local Zn2+ concentrations.
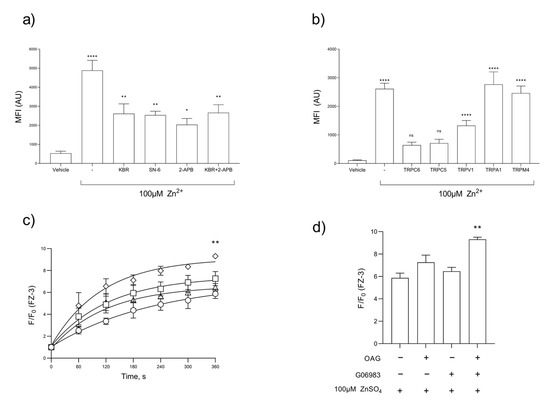
Figure 2.
Platelet Zn2+ entry is mediated by TRP channel and NCX-mediated pathways. FZ-3-loaded washed platelet suspensions were stimulated by 100 µM Zn2+ and changes in fluorescence in response to the inhibition of NCX-mediated pathways were observed by flow cytometry (a,b) or fluorometry (c,d) and (b,d). (a) Inhibitors included 2-APB (30 µM), KB-R (30 µM), SN6 (0.5 µM), or a combination of 2-APB and KB-R (30 µM each), in comparison to the vehicle control or unstimulated platelets. (b) Inhibition of TRPC6 (SAR7334, 15 µM), TRPC5 (AC1903, 15 µM), TRPV1 (AMG 9810, 2.5 µM), TRPA1 (AM0902, 15 µM), and TRPM4 (9-phenanthrol, 15 µM), compared to the vehicle control or unstimulated platelets. (c) FZ-3-loaded platelet suspensions were stimulated with Zn2+ (○, 100 μM), following pre-treatment with OAG (□, TrpC6 stimulator, 100 μM), or GO6983 (△, PKC inhibitor that enhances TrpC6 activity, 300 nM) or both OAG and GO6983 in combination (♢). Maximum FZ-3 fluorescence was calculated (d). Data are the means ± SEM of a minimum of 4 independent experiments. p < 0.0001 (****), p < 0.01 (**), p < 0.05 (*), and p > 0.05 (ns) are indicated.
As KB-R is an inhibitor of both NCX and TRP channels, we further investigated the roles of specific TRP channels in Zn2+ influx [42]. A range of TRP inhibitors were screened for their influence on the stimulation of FZ-3-loaded platelet suspensions with 100 μM Zn2+. Peak Zn2+ influx was reduced from 2612 ± 198 a.u (for the vehicle control) to 643 ± 103.9 a.u. in platelets treated with the TRPC6 inhibitor SAR7334 (Figure 2b, p < 0.001). Similarly, treatment with TRPC5 or TRPV1 blockers (AC1903 and AMG9810, respectively) reduced Zn2+ influx to 711 ± 136 and 1328 ± 173 a.u., respectively (Figure 2b, p < 0.001). The inhibition of TRPs that were not expressed on platelets (TRPM4 and TRPA1) [43,44] with AM0902 or 9-Phenanthrol, respectively, had no effect on Zn2+ influx (mean fluorescence intensities were 2462 ± 248 and 2766 ± 439 a.u. respectively; Figure 2b, ns). These data indicate that a component of observed [Zn2+]o influx was insensitive to NCX and TRP blockers, suggesting that other Zn2+ entry pathways may also contribute to Zn2+ influx. Such pathways can include Zn2+ transporters or the release of Zn2+ into the cytosol from internal stores.
Further experiments were performed to further confirm the role of TRP channels in [Zn2+]o influx. OAG and the PKC inhibitor, GO6983, have previously been used to potentiate TRPC6 activity in HEK293 and smooth muscle cells [45,46]. FZ-3-loaded platelets were pre-treated with OAG, GO6983, or a combination of the two prior to stimulation by Zn2+. Zn2+ influx was increased in dual-treated platelets (F/F0 for dual-treated platelets was 9.3 ± 0.2 a.u., compared to untreated platelets: 5.9 ± 0.4 a.u.; Figure 2c,d, p < 0.01.), supporting the evidence that TRP channels are a route for Zn2+ influx.
2.2. Zn2+-Induced Platelet Activation Is Associated with the Phosphorylation of Substrates of Ca2+-Dependent Enzymes
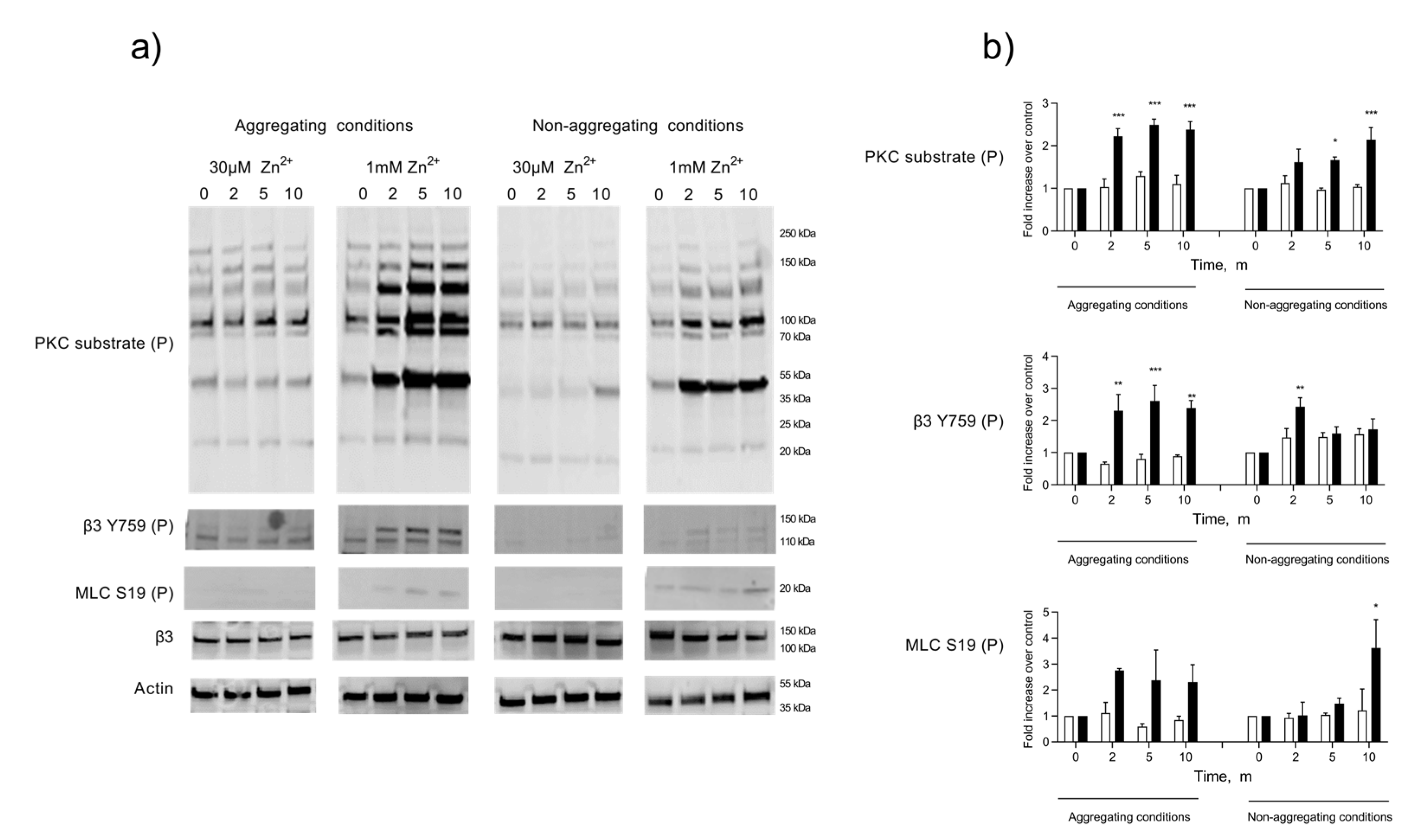
Whilst ZIPA produces a distinct pattern of tyrosine phosphorylation, the effect on other phosphoproteins has not been studied [14]. It has been reported that Zn2+ can substitute for Ca2+ at binding sites on calmodulin and other Ca2+-dependent enzymes [47,48]. Therefore, we investigated the association of ZIPA with the phosphorylation and activation of key activatory proteins, including the Ca2+-dependent myosin light chain kinase (MLCK) and PKC. Previous work using PKC inhibitors highlighted a role for MLCK and PKC during ZIPA [14], with Zn2+ possibly substituting for Ca2+ at the active site of these enzymes, leading to their activation [17]. Platelet lysates were prepared at 0, 2, 5, and 10 min intervals following stimulation by activatory (1 mM) or sub-activatory (30 µM) concentrations of Zn2+ (Figure 3). Increases in the protein phosphorylation of PKC and MLCK substrates alongside the phosphorylation of the integrin β3 chain occurred in a temporal manner and peaked within 5 min of stimulation with 1 mM Zn2+ (Figure 3), consistent with the time course for ZIPA. The phosphorylation of these substrates was observed in platelet populations treated with the integrin αIIbβ3. antagonist integrilin, demonstrating that phosphorylation was not attributable to outside–in signaling through αIIbβ3.
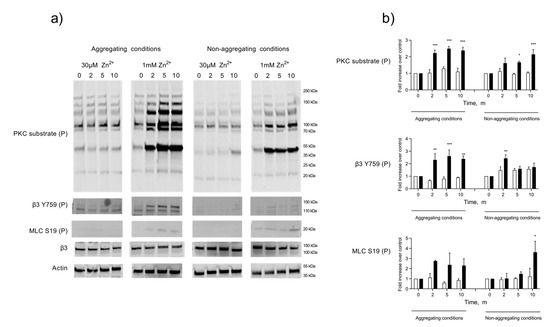
Figure 3.
Zn2+-mediated platelet activation induces the phosphorylation of Ca2+-dependent substrates. Washed platelets were stimulated by 30 µM or 1 mM Zn2+ in an aggregometer under stirring conditions, following pre-treatment with integrilin (5 µM, non-aggregating conditions), or untreated (aggregating conditions). Whole-cell lysates were generated at 0, 2, 5, and 10 min. Phosphorylations of PKC substrates, MLC, and β3 were analyzed by Western blotting (a). Samples were probed for β3 or actin to assess equal protein loading. Densitometric analyses of selected bands (MLC S19P and β3 Y759P) or whole lanes (PKC substrate) were performed to quantify changes in phosphorylation relative to the control at each time point (b). White bars: 30 µM Zn2+, black bars: 1 mM Zn2+. Data are the means ± SEM of a minimum of four independent experiments. p < 0.001 (***), p < 0.01 (**), and p < 0.05 (*) are indicated.
2.3. Zn2+ Influx Is Mediated by Ca2+ Store Depletion
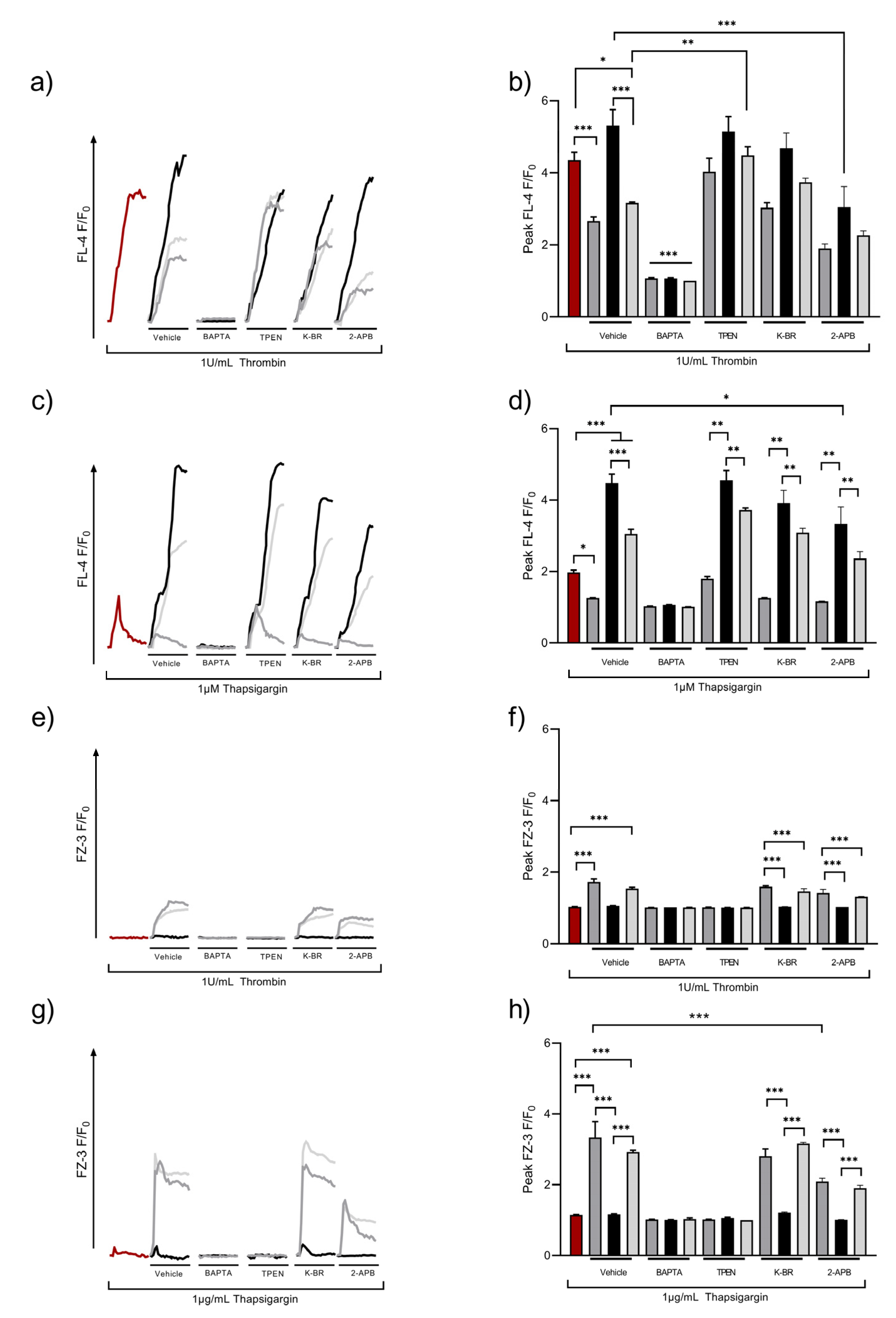
Upon receptor-mediated activation, platelets generate IP3 that activates IP3R leading to the release of Ca2+ from intracellular stores into the cytosol. This process is supported by casein kinase 2 (CK2), which has also been shown to be involved in ZIP7 phosphorylation in nucleated cells [49,50]. SOCE or ROCE both act to increase [Ca2+]o uptake, and we hypothesized that these mechanisms might also be responsible for Zn2+ influx. Washed platelet suspensions were loaded with FZ-3 or Fluo-4, and thrombin- or thapsigargin (TG)-induced fluctuations of [Ca2+]i or [Zn2+]i were investigated using flow cytometry (Figure 4). As shown previously, thrombin caused a rapid increase in [Ca2+]i, which was enhanced in the presence of 2 mM [Ca2+]o, consistent with SOCE (F/F0 peaked at 5.3 ± 0.4 a.u. compared to 4.4 ± 0.2 a.u for thrombin alone, Figure 4a,b). In the presence of subactivatory concentrations (30 μM) of [Zn2+]o, thrombin-mediated [Ca2+]i increases were significantly reduced (to 2.7 ± 0.1 a.u., p < 0.001). This effect was partially restored with the addition of 2 mM [Ca2+]o (3.2 ± 0.0 a.u., p < 0.001, Figure 4a,b), indicating that [Zn2+]o has a negative effect on Ca2+ influx. Fluctuations in Ca2+ were sensitive to pre-treatment with the non-specific cation chelator BAPTA-AM (10 µM), confirming the nature of the [Ca2+]i response. Pre-treatment with the intracellular Zn2+ chelator, TPEN (50 µM), had no significant effect on thrombin-induced [Ca2+]i signals, but abolished the reductive effect of [Zn2+]o on [Ca2+]i signals (F/F0 was 4.0 ± 0.1 a.u compared to 4.4 ± 0.2 a.u; Figure 4a,b, ns), further confirming the role of Zn2+ in regulating platelet [Ca2+]i responses.
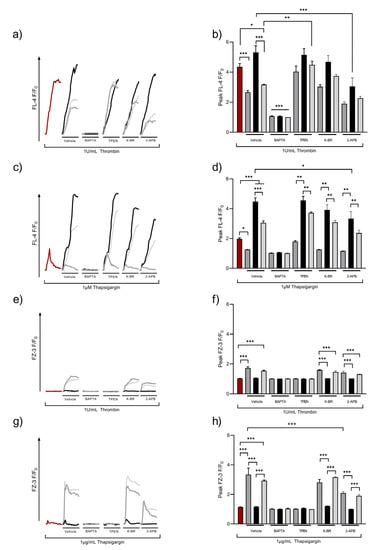
Figure 4.
Zn2+ influx is regulated by agonist- and store-operated mechanisms. Fluo-4 (FL-4)- or FZ-3-loaded platelet suspensions were stimulated after 30 s by 1 U/mL thrombin (a,b,e,f) or 1 μM thapsigargin (c,d,g,h), either in the absence of external cations (red line) or in the presence of 30 µM Zn2+ (dark gray), 2 mM Ca2+ (black), or both 2 mM Ca2+/30 μM Zn2+ (light gray), and changes in fluorescence were observed by fluorometry. Representative traces (a,c) following thrombin or thapsigargin stimulations under control conditions (no cation applied), in addition to pre-treatment with the vehicle control (dH2O), BAPTA (10 μM), TPEN (25 μM), 2-APB (30 μM), or KB-R (30 μM) to assess the mechanisms involved in mediating Zn2+ influx upon activation are shown. Peak responses are also shown (b,d–f). Data are the means ± SEM of a minimum of 4 independent experiments. p < 0.001 (***), p < 0.01 (**), and p < 0.05 (*) are indicated.
As 2-APB and KB-R were implicated in Zn2+ influx (Figure 1), these pathways were assessed to further explore the mechanisms that contributed to the Zn2+ regulation of SOCE. In 2 mM [Ca2+]o, 2-APB caused a significant decrease in thrombin-evoked [Ca2+]i responses (from 5.3 ± 0.4 a.u to 3.0 ± 0.3, p < 0.01; Figure 4a,b). As 2-APB affected both TRP channels and IP3R, these findings implicate Zn2+ in the regulation of cation entry via TRP channels or Ca2+ or Zn2+ release via DTS. KB-R pre-treatment had no significant effect on thrombin-evoked [Ca2+]i fluctuations, irrespective of the cation conditions tested. Interestingly, in 2-APB-treated platelets, thrombin-evoked Ca2+ signals in the presence of [Zn2+]o did not differ to the vehicle controls. Thus, 2-APB-sensitive pathways were not involved in the Zn2+-mediated regulation of [Ca2+]i.
To investigate whether [Zn2+]o influenced Ca2+ influx following store depletion, Fluo-4-loaded platelets were stimulated by TG in the presence and absence of [Zn2+]o, and [Ca2+]I fluctuations were quantified using flow cytometry. TG (1 µM) stimulation elevated [Ca2+]i to 2.0 ± 0.1 a.u (Figure 4c,d), reflecting Ca2+ release from intracellular stores. This was increased significantly in the presence of [Ca2+]o, consistent with SOCE (peak Fluo-4 fluorescence was 4.5 + 0.5 a.u.; Figure 4c,d, p < 0.05). In the presence of [Zn2+]o, Ca2+ influx was reduced by 72% (to 1.3 + 0.1; Figure 4c,d, p < 0.05). In the presence of both [Ca2+]o and [Zn2+]o, peak TG-induced [Ca2+]i signals were reduced to 3.0 + 0.3 a.u. (Figure 4c,d, p < 0.01) suggesting a competition between Zn2+ and Ca2+ for cation entry mechanisms. As expected, BAPTA pre-treatment abolished Ca2+ responses to TG treatment, irrespective of extracellular cation status (Figure 4c,d, p < 0.05). The peak signal evoked by 1 μM TG upon TPEN (25 μM) pre-treatment was 1.8 ± 0.1 a.u, compared to the vehicle control (2.0 ± 0.1 a.u; Figure 4c,d, ns) indicating that increases in [Zn2+]i, either as a result of its release from stores or via Zn2+ entry, did not affect SOCE. KB-R pre-treatment did not affect Ca2+ influx, whilst 2-APB pre-treatment significantly reduced Ca2+ influx in the presence of 2 mM [Ca2+]o (peak fluorescence was 3.3 + 0.4 following 2-APB treatment, compared to 4.4 + 0.3 a.u for untreated platelets; Figure 4c,d, p < 0.05). These findings implicate Zn2+ in the regulation of cation influx.
We hypothesized that cation channels involved in SOCE could also facilitate [Zn2+]o influx. To test this, platelets were loaded with FZ-3 and stimulated with thrombin or TG, and Zn2+ influx was quantified using flow cytometry. In the absence of [Zn2+]o, thrombin stimulation did not affect FZ-3 fluorescence. However, with 30 μM [Zn2+]o, thrombin stimulation led to an increase in FZ-3 fluorescence (to 1.7 ± 0.1 a.u for [Zn2+]o and 1.5 ± 0.1 a.u for [Zn2+]o/[Ca2]o; Figure 4e,f, p < 0.0001) demonstrating an activation-dependent [Zn2+]o influx. FZ-3 fluorescence was unaffected by [Ca2+]o. Further experiments employing the cation chelators BAPTA and TPEN abrogated increases in FZ-3 fluorescence, confirming that Zn2+ was responsible for the FZ-3 signal. The pre-treatment of platelets with KB-R or 2-APB had no significant effect on the [Zn2+]i signal evoked in the presence of [Zn2+]o or [Ca2+]o, or a combination of both cations (Figure 4e,f).
TG was used to deplete internal stores, and Zn2+ influx into FZ-3-loaded platelets quantified using flow cytometry. In the absence of [Zn2+]o, TG stimulation did not result in FZ-3 fluorescence in the absence of external cations (Figure 4g,h), and [Ca2+]o did not effect FZ-3 responses (1.2 ± 0.1 a.u; Figure 4g,h, ns). However, peak FZ-3 fluorescence increased in response to TG stimulation. F/F0 was 3.3 + 0.9 a.u or 2.9 + 0.1 a.u. in the presence of [Zn2+]o or [Zn2+]o/[Ca2+]o, respectively (Figure 4g,h, p < 0.001). TPEN or BAPTA abolished TG-evoked increases in FZ-3 fluorescence (Figure 4g,h). TG-mediated F/F0 signals did not differ upon pre-treatment with 30 μM KB-R in comparison to the vehicle control (Figure 4e,f, ns). However, in 2-APB-treated platelets, the Zn2+ signal was significantly reduced in the presence of [Zn2+]o (2.1 ± 0.1 a.u) compared to the vehicle control (3.3 ± 0.1 a.u; Figure 4g,h, p < 0.0001). This was also the case for [Zn2+]o/Ca2+]o, where the peak signal was 1.9 ± 0.1 a.u (Figure 4g,h; p < 0.001).
These data indicate a mechanism where the depletion of cation stores in platelets following SERCA inhibition results in increased [Zn2+]i as a result of Zn2+ influx from the extracellular medium in a similar manner to SOCE. We described this mechanism as store-operated Zn2+ entry (SOZE).
2.4. [Zn2+]o Regulates Intracellular Signaling via the Regulation of Zn2+ Stores
Zn2+ signaling may play a regulatory role in canonical Ca2+ signaling responses upon platelet activation. Zn2+ regulates Ca2+ efflux from platelet stores and it is likely that distinct intracellular Zn2+ stores contribute to Ca2+-dependent platelet responses [16,17]. In nucleated cells, PLC-dependent generation of IP3 causes cation store depletion resulting in STIM1 and Orai1 clustering with a resultant extracellular Ca2+ influx by SOCE-mediated activation [29,51]. Exogenous Zn2+ induces ZIP7-mediated Zn2+ influx leading to tyrosine kinase activation in a process that is regulated by CK2 [50]. We investigated the role of the Zn2+ transporter, ZIP7, in elevating [Zn2+]i by facilitating Zn2+ entry in platelets.
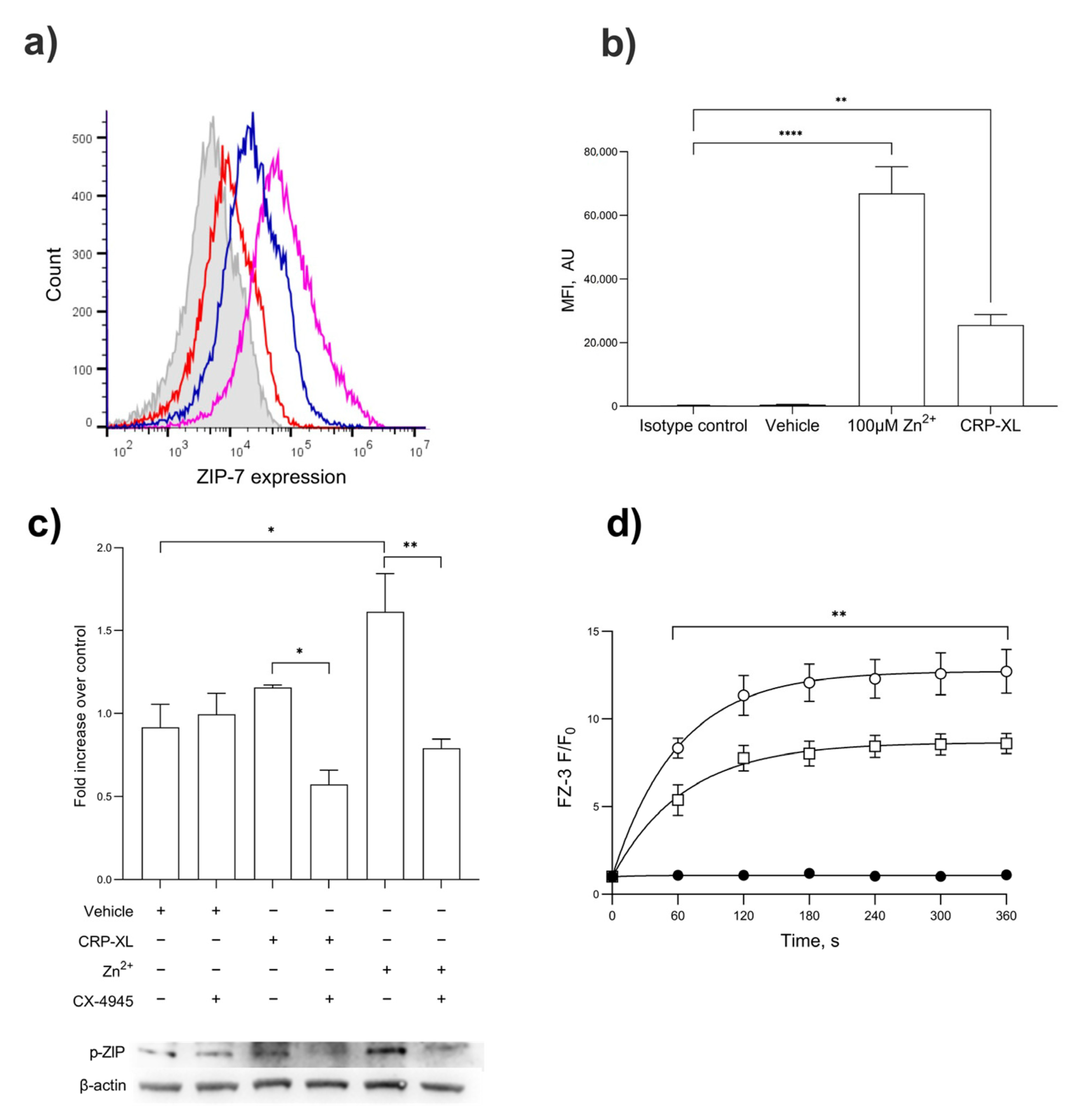
The activation of platelets with CRP-XL (1 µg/mL) or Zn2+ (100 µM) led to an increased expression of ZIP7 on the platelet surface demonstrated by flow cytometry (Figure 5a,b; p < 0.001). The stimulation of platelets with CRP-XL, but not the TP agonist U46619, resulted in increases in ZIP7 phosphorylation consistent with the role of GPVI in Zn2+ signaling (Figure 5c; p < 0.001) [14]. CRP-XL- and Zn2+-mediated ZIP7 phosphorylation was abrogated by pre-treatment with the CK2 inhibitor CX-4945 (Figure 5c), consistent with the roles of CK2 and ZIP7 in Zn2+ release from stores. The role of ZIP7 in Zn2+ influx was further investigated using the ZIP7 inhibitor NVS-ZP7-4. The stimulation of FZ-3-loaded platelets with 100 µM Zn2+ following NVS-ZP7-4 inhibition (5 μM) resulted in reduced FZ-3 fluorescence in a concentration-dependent manner (Figure 5d, p < 0.001). These findings demonstrate the role of ZIP7 in transiently elevating [Zn2+]i in response to extracellular signals, and the role of ZIP7 phosphorylation involving CK2.
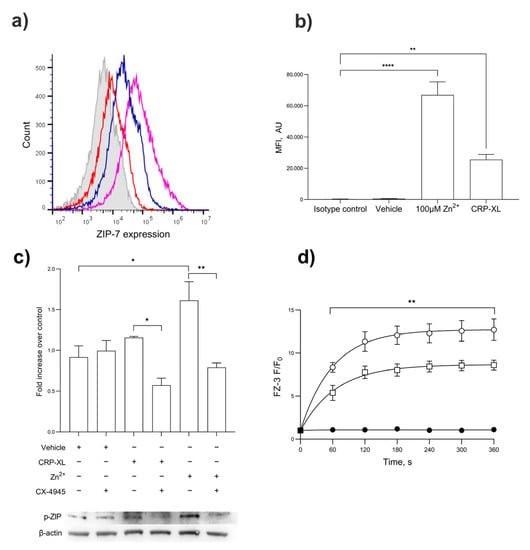
Figure 5.
ZIP7 is externalized upon platelet activation, where it contributes to Zn2+ influx. (a) Flow cytometry demonstrates the presence of ZIP7 on the surface of platelets. ZIP7 expression was demonstrated using an anti-ZIP7 antibody in unstimulated platelets (red) or in platelet suspensions stimulated by CRP-XL (1 μg/mL, blue) or Zn2+ (100 μM, purple), compared to the isotype control (gray). (b) Quantitation of mean fluorescent intensity of ZIP7 expression on platelets treated with Zn2+ (100 µM) or CRP-XL (1 µg/mL) compared to unstimulated or isotype controls. (c) Western blotting for phospho-ZIP7 in platelets stimulated with CRP-XL (1 µg/mL) or ZnSO4 100 µM) following pre-treatment with the casein kinase II inhibitor CX-4945. (d) Fluorescence in FZ-3-loaded platelets was measured in response to 100 µM Zn2+ by fluorimetry, following pre-treatment with NVS-ZP7-4 (10 µM, □) or vehicle control (DMSO, ○), or unstimulated (●) Data are representative of a minimum of 4–6 independent experiments. p < 0.01 (**), p < 0.05 (*), and p < 0.0001 (****), are indicated. Error bars indicate the standard error of the mean (SEM).
The relative contributions of [Zn2+]o influx or [Zn2+]i release from stores to [Zn2+]i signals remains unclear. Therefore, we utilized flow cytometry to investigate the influence of Ca2+- or Zn2+-release mechanisms on activation-dependent [Zn2+]i signals in the presence and absence of [Zn2+]o, to isolate the contribution of [Zn2+]i to intracellular stores.
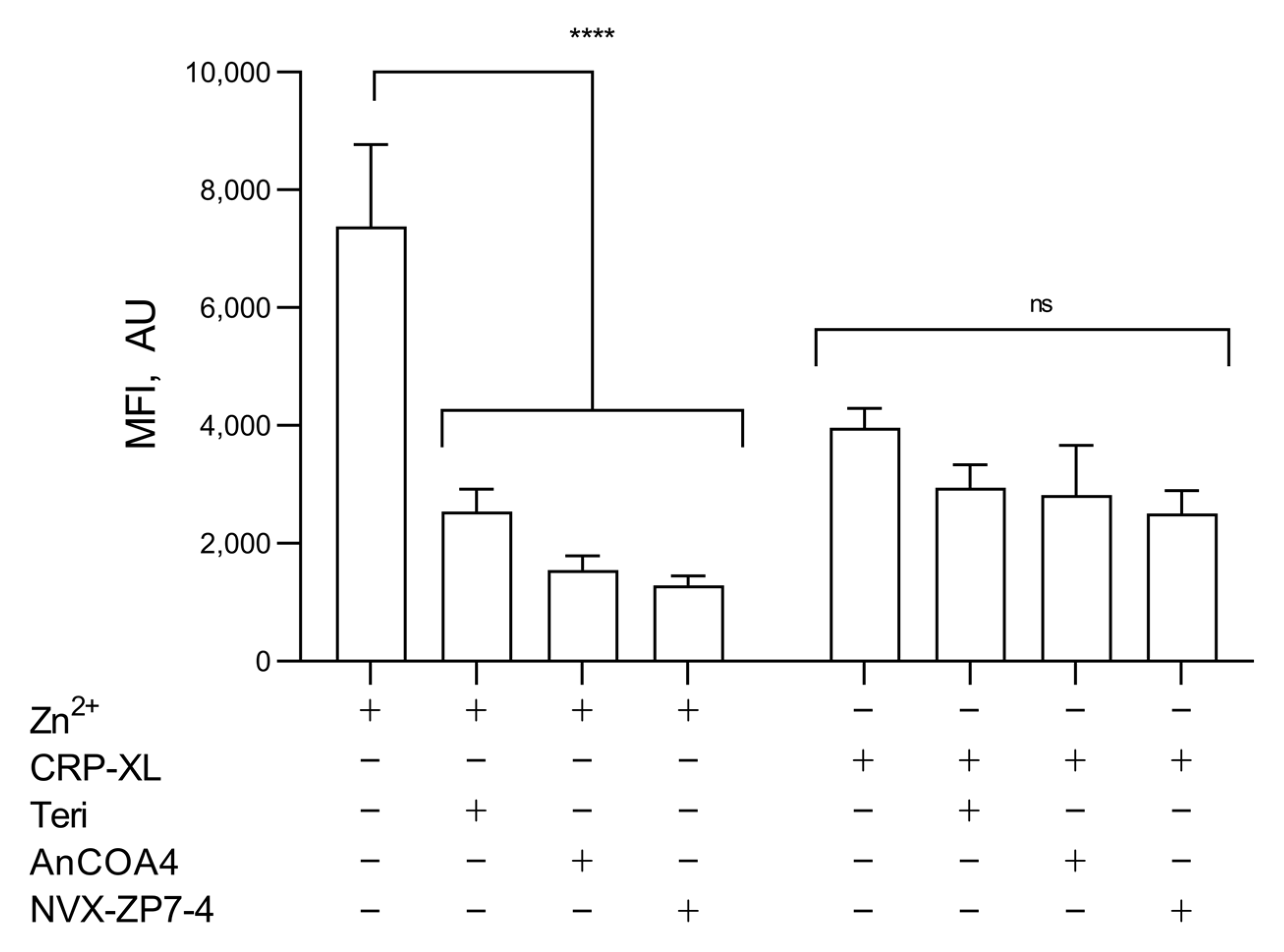
As expected, a higher FZ-3 signal was observed following [Zn2+]o stimulation compared to CRP-XL (7375.6 ± 1394.2 a.u. and 3962 ± 328.6, respectively; Figure 6), consistent with contributions to [Zn2+]i from both extracellular and intracellular sources, but with a greater relative contribution from [Zn2+]o. The effects of the inhibitions of IP3R (with Teriflunomide, 100 μM) [39], Orai1 (AnCOA4, 5 μM) [52], or ZIP7 (NVS-ZP7-4, 5 μM) were assessed. The inhibition of IP3R, Orai1, or ZIP7 reduced Zn2+-mediated FZ-3 increases to 2533.4 ± 385.2 a.u., 1549 ± 240.5 a.u., and 1289.4 ± 155.6 a.u. respectively, compared to the vehicle control (7375.6 ± 1394.2 a.u.; Figure 6, p < 0.001). CRP-XL stimulation (100 μg/mL) in the absence of extracellular [Zn2+]o resulted in FZ-3 signals that were insensitive to the inhibition of IP3R, ZIP7, or Orai1 (Figure 6, ns) indicating that Zn2+ was not being released from intracellular stores through IP3R (as is the case with Ca2+ signaling) or ZIP7.
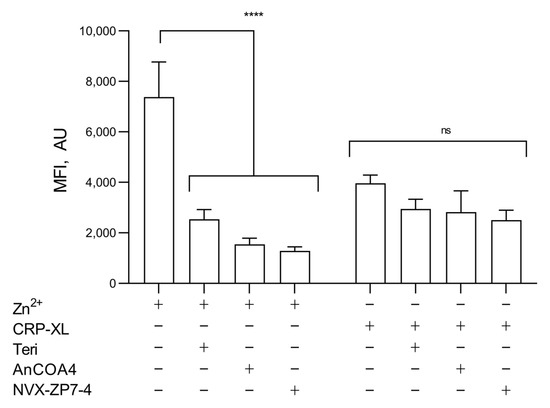
Figure 6.
Both Zn2+ influx and agonist-dependent release from stores contribute to increases in [Zn2+]i. FZ-3-loaded platelets were stimulated with CRP-XL (1 µg/mL) or [Zn2+]o (100 µM) following pre-treatment with inhibitors of IP3R: Teriflunomide (Teri, 100 µM), Orai1: AnCOA4 (5 μM), and ZIP7: NVS-ZP7 (5 μM), and fluctuations in fluorescence were recorded using fluorometry. Data are means ± SEM of a minimum of 4–6 independent experiments. p < 0.0001 (****), p > 0.05 (ns) are indicated.
3. Discussion
Here, we demonstrate for the first time that Zn2+ influx into platelets and subsequent platelet activation are partially blocked by the conventional TRP channel and NCX blockers and an inhibitor of ZIP-7, indicating multiple pathways for Zn2+ entry into platelets.
In nucleated cells, [Zn2+]o uptake and the elevation of [Zn2+]i are mediated by the involvement of Zn2+-permeable ion channels, exchangers, and Zn2+ transporters, including NCX and TRP channels [20,39,53]. Our data show that KB-R (a reverse-mode NCX inhibitor) caused substantial reductions in ZIPA (Figure 1c), suggesting a distinct role for these exchangers in Zn2+ influx into platelets. Reductions in ZIPA were associated with the reduced fluorescence of FZ-3-loaded platelets, consistent with a reduction in reduced [Zn2+]o-induced Zn2+ influx (Figure 2a,b). These observations are consistent with a previous study that shows NCX’s role in mediated Zn2+ influx in neuronal cells and that Zn2+ allosterically inhibits Na2+-independent Ca2+ release by inhibiting NCX activity [18,41].
Inhibition of both Zn2+ influx and ZIPA were observed following pre-treatment with 2-APB, SKF, or FFA, which suggests the involvement of TRP channels (Figure 2a,b,d). The lack of the full inhibition of Zn2+ influx as a result of NCX or TRP inhibitions indicates the existence of alternative Zn2+ entry pathways. In addition to being a TRP channel inhibitor, 2-APB also inhibits IP3R, and therefore could potentially have inhibited [Ca2+]i release from IP3-sensitive stores. Among the TRP channels, TRPC1, TRPC3, TRPC4, TRPC5, TRPC6, and TRPV1 are expressed on the platelets [54,55]. Specific inhibition identified roles for TRPC6, TRPC5, and TRPV1 in mediating Zn2+ influx (Figure 2b), which is consistent with previous studies that show TRPC6 to be involved in Zn2+ influx in nucleated cells [24,56]. Further evidence for the involvement of TRP channels comes from the observation that OAG and GO6983 treatments (both TRP channel activators) resulted in increased Zn2+ influx (Figure 2c,d).
We observed Zn2+-induced phosphorylation of substrates of Ca2+-dependent kinases, PKC, and MLCK (Figure 3). The activation of MLCK requires interactions with the Ca2+-binding protein calmodulin (CaM), whilst PKC has a Ca2+-binding domain [57,58]. Zn2+ was shown to substitute for Ca2+ at the EF hand domains of CaM, which may lead to the conformational change and activation of CaM-dependent substrates [47,59]. Additionally, Zn2+ binds to and activates PKC isoforms [13,60]. Thus, Zn2+ may influence platelet activation by the direct activation of Ca2+-dependent proteins. This process may act as a mechanism for priming the platelet for activation in response to low concentrations of conventional agonists [14].
Depletion of Ca2+ stores leads to the opening of membrane Ca2+ channels in a variety of cell types, including platelets, in a process known as store-dependent Ca2+ entry (SOCE). In platelets, the site of the Ca2+ store is the DTS. Here, we demonstrate that thrombin stimulation or the depletion of intracellular cation stores using thapsigargin results in increases in [Zn2+]i (Figure 4). As this signal was not observed in the absence of subactivatory (30 μM) [Zn2+]o, we conclude that this signal is due to Zn2+ influx, in a process we describe as store-operated Zn2+ entry (SOZE). Conversely, thrombin or thapsigargin stimulation results in increases in [Ca2+]i in the absence of any extracellular cation, consistent with Ca2+ from intracellular stores. Therefore, SOZE does not result in the liberation of Zn2+ from intracellular stores.
SOZE, but not SOCE, was sensitive to TPEN pre-treatment, both confirming the nature of the Zn2+ signal and providing evidence that SOCE is not dependent on Zn2+. As neither SOCE nor SOZE were affected by KB-R, NCX is not involved in these processes. This is consistent with the previous data which indicate that NCX is not involved in the regulation of [Ca2+]i in neuronal cells [41]. Interestingly, thapsigargin-, but not thrombin-mediated cation influx, was influenced by 2-APB. As 2-APB is also known to inhibit IP3R, this indicates the absence of IP3R activity in SOCE [61]. The extent of Ca2+ influx in SOCE was reduced in the presence of [Zn2+]o, consistent with competition for cation entry mechanisms. Conversely, the rate and extent of Zn2+ influx were unaffected by [Ca2+]o, suggesting that Zn2+ entry mechanisms, whilst being cation non-specific, favored Zn2+. Competition between Ca2+ and Zn2+ during intestinal absorption has been previously described where the two ions share a common transport pathway [62]. Hence, we suggest the possibility of competition between the two ions occurring via Zn2+ entry mechanisms, including TRP channels and NCX. Additionally, inhibition by 2-APB did not affect TG-induced Zn2+ influx in the presence of exogenous 2 mM/30 μM [Ca2+]o/[Zn2+]o (Figure 4), suggesting that NCX mediates Zn2+ influx, but does not influence store-operated Ca2+ release.
SOCE involves the detection of store Ca2+ levels by Orai, with subsequent signaling to the membrane cation, STIM1. Whether SOZE is regulated by the detection of Ca2+ or Zn2+ depletion is not known. Platelets have a Zn2+ store that is released into the cytosol following agonist stimulation, in a manner that is consistent with a secondary messenger [14,26]. However, the nature of the Zn2+ store and the mechanism by which Zn2+ exerts its actions has received little attention. Previous studies indicate that the store is sensitive to platelet stimulation via GPVI and TP, but not PARs, and is sensitive to the redox state [14]. A further mechanism by which cells regulate Zn2+ levels is through Zn2+ transporters, of which ZIP7 is present in the platelet proteome [63]. In nucleated cells, the activation of tyrosine kinase pathways depend on the release of Zn2+ from the ER into the cytosol, mediated by the Zn2+ transporter, ZIP7 [64,65,66]. We confirm that ZIP7 is present on the platelet surface (Figure 5) and that platelet activation via Zn2+ or CRP-XL leads to increases in surface expression. This is consistent with the presence of ZIP7 on intracellular membranes of the open canalicular system, which becomes externalized upon platelet activation. Platelet activation using Zn2+ results in an increase in ZIP7 phosphorylation, which is sensitive to inhibition of CK2, indicating an activatory pathway. These results complement the findings on nucleated cells, where ZIP7 is regulated via CK2-mediated phosphorylation in MCF-7 cells. In this work, Zn2+ stimulation resulted in an increase in [Zn2+]I, which was inhibited upon ZIP7 knockdown via siRNA [65].
Previous work has demonstrated increases in [Zn2+]i, both as a result of Zn2+ influx and release from stores [12,14,26]. To determine the relative contributions of each pathway to [Zn2+]i increases, we stimulated FZ-3-loaded platelets with [Zn2+]o or CRP-XL (in the absence of [Zn2+]o, precluding Zn2+ influx). Both pathways result in increases in [Zn2+]i with Zn2+ stimulation generating a stronger signal, consistent with a greater contribution from Zn2+ influx. Other published works have implicated IP3R in the persistent, rapid [Zn2+]I increases in cortical neurons [67]. To investigate the influence of IP3R on Zn2+ store release, we inhibited IP3R and observed reduced [Zn2+]i increases in the response to [Zn2+]o, but not CRP-XL. This profile is consistent with the roles of IP3R, ZIP7, and Orai1 in Zn2+ influx, but not for agonist-dependent Zn2+ release in the absence of [Zn2+]o. Therefore, Zn2+ is not stored and released in a similar manner to Ca2+, and ZIP7 was only effective in Zn2+ influx following activation-mediated externalization. This is consistent with the observation that [Zn2+]i release, but not Ca2+ release, is sensitive to the platelet redox state [14,26] and is supportive of a role for redox-sensitive cation storage proteins, such as metallothioneins. Further experiments are required to further elucidate the nature of the Zn2+ store in platelets. The reason why IP3R inhibition reduces [Zn2+]o influx is unclear; however, it is suggestive of a role for Ca2+ in regulating Zn2+ entry. This is consistent with the observation that both TG- and thrombin-mediated increases in [Ca2+]i were reduced in the presence of [Zn2+]o (Figure 4). This observation may suggest cross-talk between Zn2+- and Ca2+-handling mechanisms during platelet activation.
In conclusion, this study provides evidence that platelets are able to detect and respond to increases in [Zn2+]o via influx through TRP channels, the NCX exchanger, and ZIP7 (Figure 7). Store depletion facilitates Zn2+ entry via Orai1, which we have termed store-operated Zn2+ entry (SOZE). ZIP7 is externalized following platelet activation. Both influx and Zn2+ release from stores both contribute to increases in [Zn2+]i. It is likely that other cell systems employ similar mechanisms to detect and respond to changes in [Zn2+]o.
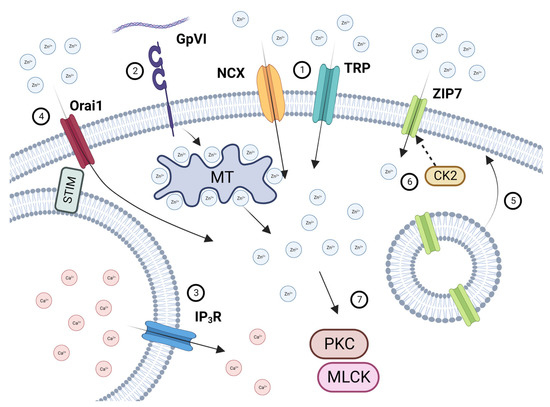
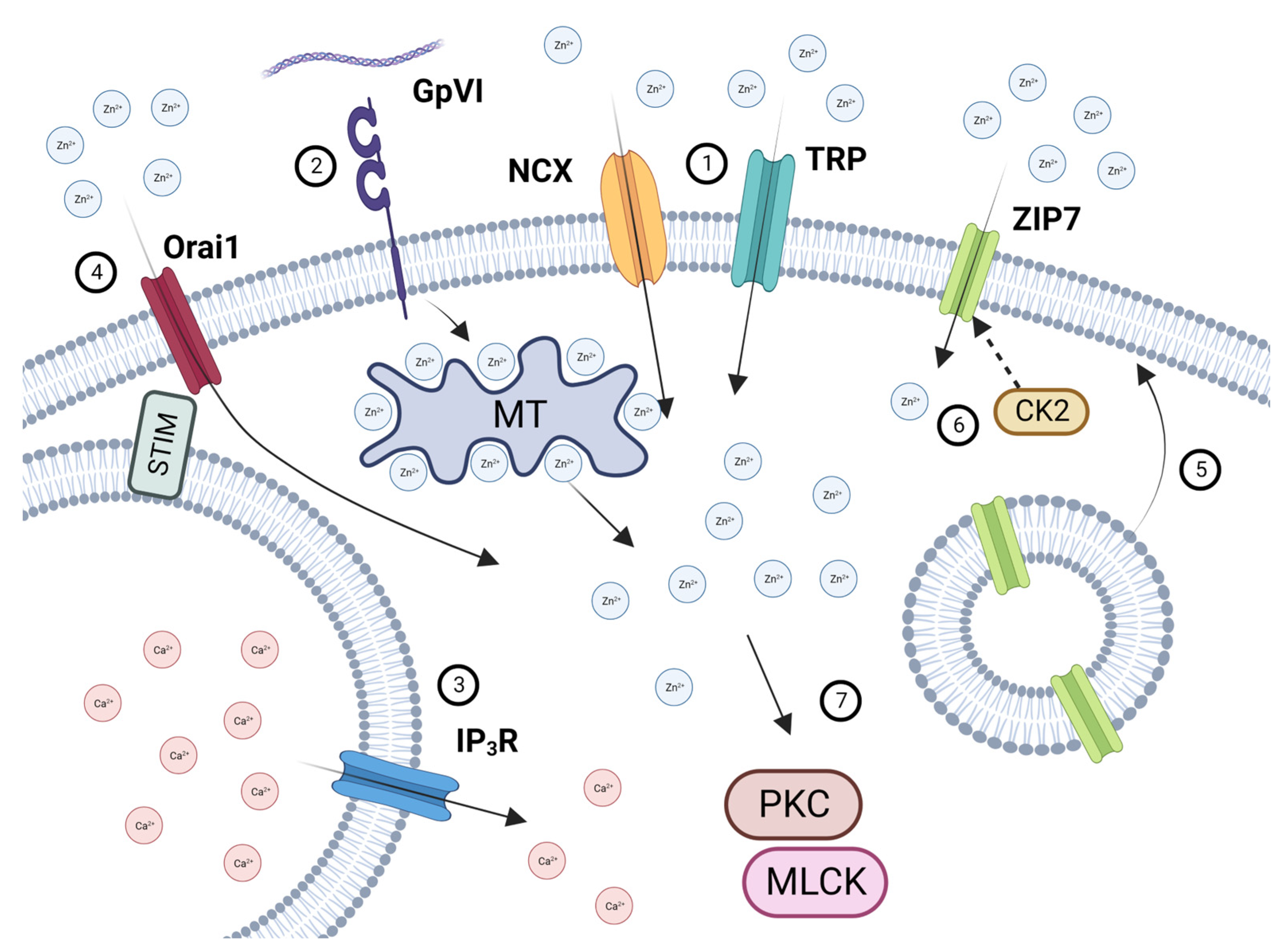
Figure 7.
Schematic illustration of [Zn2+]o and [Zn2+]I handling in platelets. Increases in [Zn2+]o following endothelial damage, atherosclerotic plaque rupture, or platelet degranulation results in Zn2+ influx via NCX and TRP channels (1). Platelet activation by GPVI (2) results in the liberation of [Zn2+]i from intracellular stores likely to be redox-sensitive proteins, such as metallothioneins). Platelet activation also results in Ca2+ store depletion (3), which is detected by STIM, leading to [Zn2+]o influx via Orai1 (4). Activation-dependent degranulation leads to the externalization of ZIP7 on the platelet surface (5), which is phosphorylated by CK2, further facilitating Zn2+ influx (6). Increases in [Zn2+]i results in the activation of PKC and MLCK, contributing to downstream platelet activatory responses (7). Created with BioRender.com.
4. Materials and Methods
Materials: 2-Aminoethoxydiphenyl borate (2-APB), SKF 96365 hydrochloride (SKF; 1-[2-(4-Methoxyphenyl)-2-[3-(4-methoxyphenyl)propoxy]ethyl-1H-imidazole hydrochloride), KB-R7943 mesylate (KB-R; 2-[2-[4-(4-Nitrobenzyloxy)phenyl]ethyl]isothiourea mesylate), SN-6 (2-[[4-[(4-Nitrophenyl)methoxy]phenyl]methyl]-4-thiazolidinecarboxylic acid ethyl ester), SAR7334 (4-[[(1R,2R)-2-[(3R)-3-Amino-1-piperidinyl]-2,3-dihydro-1H-inden-1-yl] oxy]-3-chlorobenzonitrile dihydrochloride), 9-Phenanthrol, AM0902 (1-[[3-[2-(4-Chlorophenyl)ethyl]-1,2,4-oxadiazol-5-yl]methyl]-1,7-dihydro-7-methyl-6H-purin-6-one), Teriflunomide (2-Cyano-3-hydroxy-N-[4-(trifluoromethyl)phenyl]-2-butenamide), and integrilin were from Tocris (Bristol, UK). Flufenamic acid (FFA), 1-Oleoyl-2-acetyl-sn-glycerol (OAG), thrombin, calcium chloride, and zinc sulphate were supplied by Sigma (Poole, UK), and thapsigargin (TG) was from Calbiochem (Nottingham, UK). AC1903 and NVS-ZP7-4 were from MedChem express (Monmouth Junction, NJ, USA); AMG9810 ((2E)-N-(2,3-Dihydro-1,4-benzodioxin-6-yl)-3-[4-(1,1-dimethylethyl)phenyl]-2-propenamide) was from Abcam (Cambridge, UK). AnCoA4 was from Merck (Watford, UK). CX-4945 was from Stratetech, Ely, UK). Unless stated, all other reagents were from Sigma Aldrich.
Preparation of washed human platelets: ethical approval for this study was obtained from the Faculty Research Ethics Committee at Anglia Ruskin University. Human blood was collected from healthy volunteers who had not taken medication for two weeks, following informed consent in accordance with the Declaration of Helsinki. Blood was collected in 11 mM of sodium citrate and washed platelets were prepared as described previously [12]. For studies of Ca2+ or Zn2+ mobilizations, aliquots of platelet-rich plasma (PRP) were retained. Platelets were resuspended using a calcium-free Tyrodes buffer (CFT) containing, in mM, 140 NaCl, 5 KCl, 10 HEPES, 5 Glucose, 0.42 NaH2PO4, and 12 NaHCO3, titrated to pH 7.4 with NaOH.
Light transmission aggregometry: platelet aggregation was monitored as described previously using an AggRam light transmission aggregometer (Helena Biosciences, Gateshead, UK) [12]. Washed platelet suspensions in CFT were stimulated under stirring conditions at 37 °C and aggregation traces were acquired digitally using proprietary software (HemoRam v1.3, Helena Biosciences). Platelets were stimulated by 100 µM ZnSO4 following pre-incubation (up to 15 min) with specified channel blockers or vehicle control (0.1% DMSO).
Fluorometry: platelets in PRP were loaded with Fluozin-3 (FZ-3, 1 µM, Invitrogen, Paisley, UK) or Fluo-4 (1 µM, Invitrogen) for 30 min, at 37 °C. Platelets were collected by centrifugation (350× g, 15 min), resuspended in CFT, and rested at 37 °C for 30 min prior to use. FZ-3 fluorescence data were acquired under 494 nm excitation and 516 nm emission using a Fluoroskan Ascent Fluorometer (ThermoScientific, Oxford, UK).
Flow cytometry: PRP was loaded with Fluo-4 or FZ-3 (1 µM) and elevations of [Ca2+]i or [Zn2+]i were monitored over a 4 min period using an Accuri C6 flow cytometer (BD Biosciences, Oxford, UK). Platelets were stimulated by thrombin (1 U/mL) in the presence of 2 mM [Ca2+]o, 30 μM [Zn2+]o, or a combination of both. Where stated, platelets were incubated with 1 µM TG, a SERCA pump inhibitor, for 5 min in the absence of [Ca2+]o to assess Ca2+ release from the DTS. For experiments assessing the role of NCX or TRP channels, platelets were incubated with inhibitors for 30 min at 37 °C, before stimulation with 100 µM Zn2+, and the FZ-3 signal was measured for 5 min. For experiments assessing [Zn2+] release from internal stores, platelets were pre-treated with inhibitors against Orai1, IP3R, or ZIP7 for 30 min at 37 °C, prior to stimulation with 100 µM Zn2+ or 1 μg/mL CRP-XL (Cambcol, Ely, UK).
Western blotting: Western blotting was performed as described previously [12]. Briefly, washed platelet suspensions were pre-treated with the αIIbβ3 inhibitor integrilin (5 μM) for 5 min prior to stimulation with 30 μM or 1 mM Zn2+ for the given time periods, where platelets were then lysed with RIPA buffer (150 mM NaCl, 50 mM Tris-HCL, pH 8.0, 1% nonidet P-40, 0.5% Na deoxycholate, 0.15 SDS). Lysates were separated on 8% SDS-PAGE or 4–12% Gradient NuPAGE Bis-Tris (BioRad) gels and transferred to PVDF membranes. Zn2+-induced protein phosphorylation was assessed using antibodies against phospho-β3 and Y759 (AbCam, Cambridge, UK), PKC substrate (AbCam), or p-MLC S19 (Cell Signalling Technology, Danvers, MA, USA), or anti-Phospho-ZIP7 antibodies (Cambridge Bioscience, Cambridge, UK), followed by anti-mouse, anti-rabbit, or anti-goat HRP-conjugated secondary antibodies (AbCam. Antibodies against total β3 or β-actin (Santa Cruz, Heidelberg, Germany) were included as loading controls. Blots were representative of 3–4 platelet preparations.
Data analysis and statistics: maximum aggregation and mean fluorescence values were calculated using Microsoft Excel 365 (Microsoft, Redmond, WA, USA). Representative traces of FZ-3 or Fluo-4 fluorescence responses were generated using Flow Jo (V10.2, Ashland, OR, USA). Western blots were analyzed using ImageJ (v1.45, NIH, Bethesda, MD, USA). Data were analyzed in GraphPad Prism by a two-way ANOVA of Student’s t-test, where p < 0.0001 (****), p < 0.001 (***), p < 0.01 (**), p < 0.05 (*), and p > 0.05 (ns) were indicated.
Author Contributions
S.J.K., N.S.A., K.A.T., E.M.C., A.J.U. and A.B. conceived and performed experiments, analyzed, interpreted the data, and co-wrote the manuscript. N.P. and J.M.G. conceived, organized, and conducted the study, including the analysis and interpretation of the data and critique of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by British Heart Foundation project grants (PG/14/47/30912, PG/18/64/33922, and FS/PhD/22/29317.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Research Ethics Committee at Anglia Ruskin University (Protocol code: FST FREP13/363, approved 4 December 2019, and ETH2022-0111, approved 11 January 2023).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are openly available in FigShare at https://doi.org/10.25411/aru.23703288.v1.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Apgar, J. Effect of zinc deficiency on parturition in the rat. Am. J. Physiol. 1968, 215, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Gordon, P.R.; O’Dell, B.L. Short-term zinc deficiency and hemostasis in the rat. Proc. Soc. Exp. Biol. Med. 1980, 163, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Gordon, P.R.; O’Dell, B.L. Rat platelet aggregation impaired by short-term zinc deficiency. J. Nutr. 1980, 110, 2125–2129. [Google Scholar] [CrossRef]
- Gordon, P.R.; Woodruff, C.W.; Anderson, H.L.; O’Dell, B.L. Effect of acute zinc deprivation on plasma zinc and platelet aggregation in adult males. Am. J. Clin. Nutr. 1982, 35, 113–119. [Google Scholar] [CrossRef]
- Stefanini, M. Cutaneous bleeding related to zinc deficiency in two cases of advanced cancer. Cancer 1999, 86, 866–870. [Google Scholar] [CrossRef]
- Taylor, K.A.; Pugh, N. The contribution of zinc to platelet behaviour during haemostasis and thrombosis. Metallomics 2016, 8, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Stewart, A.J.; Sadler, P.J.; Pinheiro, T.J.T.; Blindauer, C.A. Albumin as a zinc carrier: Properties of its high-affinity zinc-binding site. Biochem. Soc. Trans. 2008, 36, 1317–1321. [Google Scholar] [CrossRef]
- Chilvers, D.C.; Dawson, J.B.; Bahreyni-Toosi, M.H.; Hodgkinson, A. Identification and determination of copper--and zinc--protein complexes in blood plasma after chromatographic separation on DEAE-Sepharose CL-6B. Analyst 1984, 109, 871–876. [Google Scholar] [CrossRef]
- Foote, J.W.; Delves, H.T. Distribution of zinc amongst human serum proteins determined by affinity chromatography and atomic-absorption spectrophotometry. Analyst 1983, 108, 492–504. [Google Scholar] [CrossRef]
- Stadler, N.; Stanley, N.; Heeneman, S.; Vacata, V.; Daemen, M.J.; Bannon, P.G.; Waltenberger, J.; Davies, M.J. Accumulation of zinc in human atherosclerotic lesions correlates with calcium levels but does not protect against protein oxidation. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1024–1030. [Google Scholar] [CrossRef]
- Marx, G.; Korner, G.; Mou, X.; Gorodetsky, R. Packaging zinc, fibrinogen, and factor XIII in platelet alpha-granules. J. Cell. Physiol. 1993, 156, 437–442. [Google Scholar] [CrossRef]
- Watson, B.; White, N.; Taylor, K.; Howes, J.-M.; Malcor, J.-D.; Bihan, D.; Sage, S.O.; Farndale, R.W.; Pugh, N. Zinc is a Transmembrane Agonist that Induces Platelet Activation in a Tyrosine Phosphorylation-Dependent Manner. Metallomics 2016, 8, 91–100. [Google Scholar] [CrossRef]
- Forbes, I.J.; Zalewski, P.D.; Giannakis, C.; Petkoff, H.S.; Cowled, P.A. Interaction between protein kinase C and regulatory ligand is enhanced by a chelatable pool of cellular zinc. Biochim. Biophys. Acta 1990, 1053, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.S.; Lopes Pires, M.E.; Taylor, K.A.; Pugh, N. Agonist-Evoked Increases in Intra-Platelet Zinc Couple to Functional Responses. Thromb. Haemost. 2019, 119, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Heyns Adu, P.; Eldor, A.; Yarom, R.; Marx, G. Zinc-induced platelet aggregation is mediated by the fibrinogen receptor and is not accompanied by release or by thromboxane synthesis. Blood 1985, 66, 213–219. [Google Scholar] [CrossRef]
- O’Dell, B.L.; Emery, M. Compromised zinc status in rats adversely affects calcium metabolism in platelets. J. Nutr. 1991, 121, 1763–1768. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; O’Dell, B.L. Zinc deficiency in rats decreases thrombin-stimulated platelet aggregation by lowering protein kinase C activity secondary to impaired calcium uptake. J. Nutr. Biochem. 1995, 6, 661–666. [Google Scholar] [CrossRef]
- Sensi, S.L.; Canzoniero, L.M.; Yu, S.P.; Ying, H.S.; Koh, J.Y.; Kerchner, G.A.; Choi, D.W. Measurement of intracellular free zinc in living cortical neurons: Routes of entry. J. Neurosci. 1997, 17, 9554–9564. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Haragopal, H.; Slepchenko, K.G.; Stork, C.; Li, Y.V. Intracellular zinc distribution in mitochondria, ER and the Golgi apparatus. Int. J. Physiol. Pathophysiol. Pharmacol. 2016, 8, 35–43. [Google Scholar] [PubMed]
- Kambe, T.; Taylor, K.M.; Fu, D. Zinc transporters and their functional integration in mammalian cells. J. Biol. Chem. 2021, 296, 100320. [Google Scholar] [CrossRef]
- Bin, B.-H.; Seo, J.; Kim, S.T. Function, Structure, and Transport Aspects of ZIP and ZnT Zinc Transporters in Immune Cells. J. Immunol. Res. 2018, 2018, 9365747. [Google Scholar] [CrossRef] [PubMed]
- Cousins, R.J.; Liuzzi, J.P.; Lichten, L.A. Mammalian zinc transport, trafficking, and signals. J. Biol. Chem. 2006, 281, 24085–24089. [Google Scholar] [CrossRef]
- Monteilh-Zoller, M.K.; Hermosura, M.C.; Nadler, M.J.S.; Scharenberg, A.M.; Penner, R.; Fleig, A. TRPM7 Provides an Ion Channel Mechanism for Cellular Entry of Trace Metal Ions. J. Gen. Physiol. 2003, 121, 49–60. [Google Scholar] [CrossRef]
- Chevallet, M.; Jarvis, L.; Harel, A.; Luche, S.; Degot, S.; Chapuis, V.; Boulay, G.; Rabilloud, T.; Bouron, A. Functional consequences of the over-expression of TRPC6 channels in HEK cells: Impact on the homeostasis of zinc. Met. Integr. Biometal. Sci. 2014, 6, 1269–1276. [Google Scholar] [CrossRef] [PubMed]
- Maret, W. The redox biology of redox-inert zinc ions. Free Radic. Biol. Med. 2019, 134, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Pires, M.E.; Ahmed, N.S.; Vara, D.; Gibbins, J.M.; Pula, G.; Pugh, N. Zinc regulates reactive oxygen species generation in platelets. Platelets 2020, 32, 368–377. [Google Scholar] [CrossRef]
- Gombedza, F.C.; Shin, S.; Kanaras, Y.L.; Bandyopadhyay, B.C. Abrogation of store-operated Ca2+ entry protects against crystal-induced ER stress in human proximal tubular cells. Cell Death Discov. 2019, 5, 124. [Google Scholar] [CrossRef]
- Tolstykh, G.P.; Cantu, J.C.; Tarango, M.; Ibey, B.L. Receptor- and store-operated mechanisms of calcium entry during the nanosecond electric pulse-induced cellular response. Biochim. Biophys. Acta Biomembr. 2019, 1861, 685–696. [Google Scholar] [CrossRef]
- Prakriya, M.; Lewis, R.S. Store-Operated Calcium Channels. Physiol. Rev. 2015, 95, 1383–1436. [Google Scholar] [CrossRef]
- Harper, M.T.; Poole, A.W. Store-operated calcium entry and non-capacitative calcium entry have distinct roles in thrombin-induced calcium signalling in human platelets. Cell Calcium 2011, 50, 351–358. [Google Scholar] [CrossRef]
- Varga-Szabo, D.; Braun, A.; Nieswandt, B. STIM and Orai in platelet function. Cell Calcium 2011, 50, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Varga-Szabo, D.; Authi, K.S.; Braun, A.; Bender, M.; Ambily, A.; Hassock, S.R.; Gudermann, T.; Dietrich, A.; Nieswandt, B. Store-operated Ca2+ entry in platelets occurs independently of transient receptor potential (TRP) C1. Pflugers Arch. 2008, 457, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Gilio, K.; van Kruchten, R.; Braun, A.; Berna-Erro, A.; Feijge, M.A.H.; Stegner, D.; van der Meijden, P.E.J.; Kuijpers, M.J.E.; Varga-Szabo, D.; Heemskerk, J.W.M.; et al. Roles of platelet STIM1 and Orai1 in glycoprotein VI- and thrombin-dependent procoagulant activity and thrombus formation. J. Biol. Chem. 2010, 285, 23629–23638. [Google Scholar] [CrossRef] [PubMed]
- Varga-Szabo, D.; Braun, A.; Nieswandt, B. Calcium signaling in platelets. J. Thromb. Haemost. 2009, 7, 1057–1066. [Google Scholar] [CrossRef]
- Pugh, N.; Bihan, D.; Perry, D.J.; Farndale, R.W. Dynamic analysis of platelet deposition to resolve platelet adhesion receptor activity in whole blood at arterial shear rate. Platelets 2015, 26, 216–219. [Google Scholar] [CrossRef]
- Gaburjakova, J.; Gaburjakova, M. The Cardiac Ryanodine Receptor Provides a Suitable Pathway for the Rapid Transport of Zinc (Zn2+). Cells 2022, 11, 868. [Google Scholar] [CrossRef]
- Woodier, J.; Rainbow, R.D.; Stewart, A.J.; Pitt, S.J. Intracellular Zinc Modulates Cardiac Ryanodine Receptor-mediated Calcium Release. J. Biol. Chem. 2015, 290, 17599–17610. [Google Scholar] [CrossRef]
- Harper, A.G.S.; Brownlow, S.L.; Sage, S.O. A role for TRPV1 in agonist-evoked activation of human platelets. J. Thromb. Haemost. JTH 2009, 7, 330–338. [Google Scholar] [CrossRef]
- Brownlow, S.L.; Sage, S.O. Transient receptor potential protein subunit assembly and membrane distribution in human platelets. Thromb. Haemost. 2005, 94, 839–845. [Google Scholar] [CrossRef]
- Zheng, J. Molecular mechanism of TRP channels. Compr. Physiol. 2013, 3, 221–242. [Google Scholar] [CrossRef]
- Colvin, R.A. Zinc inhibits Ca2+ transport by rat brain NA+/Ca2+ exchanger. Neuroreport 1998, 9, 3091–3096. [Google Scholar] [CrossRef] [PubMed]
- Kraft, R. The Na+/Ca2+ exchange inhibitor KB-R7943 potently blocks TRPC channels. Biochem. Biophys. Res. Commun. 2007, 361, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Albarrán, L.; Lopez, J.J.; Dionisio, N.; Smani, T.; Salido, G.M.; Rosado, J.A. Transient receptor potential ankyrin-1 (TRPA1) modulates store-operated Ca2+ entry by regulation of STIM1-Orai1 association. Biochim. Biophys. Acta 2013, 1833, 3025–3034. [Google Scholar] [CrossRef] [PubMed]
- Kazandzhieva, K.; Mammadova-Bach, E.; Dietrich, A.; Gudermann, T.; Braun, A. TRP channel function in platelets and megakaryocytes: Basic mechanisms and pathophysiological impact. Pharmacol. Ther. 2022, 237, 108164. [Google Scholar] [CrossRef] [PubMed]
- Estacion, M.; Li, S.; Sinkins, W.G.; Gosling, M.; Bahra, P.; Poll, C.; Westwick, J.; Schilling, W.P. Activation of human TRPC6 channels by receptor stimulation. J. Biol. Chem. 2004, 279, 22047–22056. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, S.M.; Monet, M.; Boulay, G. Protein kinase C-dependent phosphorylation of transient receptor potential canonical 6 (TRPC6) on serine 448 causes channel inhibition. J. Biol. Chem. 2010, 285, 40534–40543. [Google Scholar] [CrossRef]
- Warren, J.T.; Guo, Q.; Tang, W.-J. A 1.3-A structure of zinc-bound N-terminal domain of calmodulin elucidates potential early ion-binding step. J. Mol. Biol. 2007, 374, 517–527. [Google Scholar] [CrossRef]
- Mills, J.S.; Johnson, J.D. Metal ions as allosteric regulators of calmodulin. J. Biol. Chem. 1985, 260, 15100–15105. [Google Scholar] [CrossRef]
- Ziliotto, S.; Gee, J.M.W.; Ellis, I.O.; Green, A.R.; Finlay, P.; Gobbato, A.; Taylor, K.M. Activated zinc transporter ZIP7 as an indicator of anti-hormone resistance in breast cancer. Met. Integr. Biometal Sci. 2019, 11, 1579–1592. [Google Scholar] [CrossRef]
- Taylor, K.M.; Hiscox, S.; Nicholson, R.I.; Hogstrand, C.; Kille, P. Protein kinase CK2 triggers cytosolic zinc signaling pathways by phosphorylation of zinc channel ZIP7. Sci. Signal. 2012, 5, ra11. [Google Scholar] [CrossRef]
- Calloway, N.; Vig, M.; Kinet, J.-P.; Holowka, D.; Baird, B. Molecular clustering of STIM1 with Orai1/CRACM1 at the plasma membrane depends dynamically on depletion of Ca2+ stores and on electrostatic interactions. Mol. Biol. Cell 2009, 20, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Sadaghiani, A.M.; Lee, S.M.; Odegaard, J.I.; Leveson-Gower, D.B.; McPherson, O.M.; Novick, P.; Kim, M.R.; Koehler, A.N.; Negrin, R.; Dolmetsch, R.E.; et al. Identification of Orai1 channel inhibitors by using minimal functional domains to screen small molecule microarrays. Chem. Biol. 2014, 21, 1278–1292. [Google Scholar] [CrossRef] [PubMed]
- Hojyo, S.; Fukada, T. Zinc transporters and signaling in physiology and pathogenesis. Arch. Biochem. Biophys. 2016, 611, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Authi, K.S. TRP channels in platelet function. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2007; pp. 425–443. [Google Scholar] [CrossRef]
- Dionisio, N.; Redondo, P.C.; Jardin, I.; Rosado, J.A. Transient receptor potential channels in human platelets: Expression and functional role. Curr. Mol. Med. 2012, 12, 1319–1328. [Google Scholar] [CrossRef]
- Ramanathan, G.; Gupta, S.; Thielmann, I.; Pleines, I.; Varga-Szabo, D.; May, F.; Mannhalter, C.; Dietrich, A.; Nieswandt, B.; Braun, A. Defective diacylglycerol-induced Ca2+ entry but normal agonist-induced activation responses in TRPC6-deficient mouse platelets. J. Thromb. Haemost. 2012, 10, 419–429. [Google Scholar] [CrossRef]
- Fajmut, A.; Brumen, M.; Schuster, S. Theoretical model of the interactions between Ca2+, calmodulin and myosin light chain kinase. FEBS Lett. 2005, 579, 4361–4366. [Google Scholar] [CrossRef]
- Kamm, K.E.; Stull, J.T. Dedicated myosin light chain kinases with diverse cellular functions. J. Biol. Chem. 2001, 276, 4527–4530. [Google Scholar] [CrossRef]
- Carpenter, M.C.; Palmer, A.E. Native and engineered sensors for Ca2+ and Zn2+: Lessons from calmodulin and MTF1. Essays Biochem. 2017, 61, 237–243. [Google Scholar] [CrossRef]
- Forbes, I.J.; Zalewski, P.D.; Giannakis, C. Role for zinc in a cellular response mediated by protein kinase C in human B lymphocytes. Exp. Cell Res. 1991, 195, 224–229. [Google Scholar] [CrossRef]
- Ansari, N.; Hadi-Alijanvand, H.; Sabbaghian, M.; Kiaei, M.; Khodagholi, F. Interaction of 2-APB, dantrolene, and TDMT with IP3R and RyR modulates ER stress-induced programmed cell death I and II in neuron-like PC12 cells: An experimental and computational investigation. J. Biomol. Struct. Dyn. 2014, 32, 1211–1230. [Google Scholar] [CrossRef]
- Bertolo, R.F.P.; Bettger, W.J.; Atkinson, S.A. Calcium competes with zinc for a channel mechanism on the brush border membrane of piglet intestine. J. Nutr. Biochem. 2001, 12, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Burkhart, J.M.; Vaudel, M.; Gambaryan, S.; Radau, S.; Walter, U.; Martens, L.; Geiger, J.; Sickmann, A.; Zahedi, R.P. The first comprehensive and quantitative analysis of human platelet protein composition allows the comparative analysis of structural and functional pathways. Blood 2012, 120, e73–e82. [Google Scholar] [CrossRef] [PubMed]
- Hogstrand, C.; Kille, P.; Nicholson, R.I.; Taylor, K.M. Zinc transporters and cancer: A potential role for ZIP7 as a hub for tyrosine kinase activation. Trends Mol. Med. 2009, 15, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.M.; Vichova, P.; Jordan, N.; Hiscox, S.; Hendley, R.; Nicholson, R.I. ZIP7-mediated intracellular zinc transport contributes to aberrant growth factor signaling in antihormone-resistant breast cancer Cells. Endocrinology 2008, 149, 4912–4920. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, S.; Sakata-Sogawa, K.; Hasegawa, A.; Suzuki, T.; Kabu, K.; Sato, E.; Kurosaki, T.; Yamashita, S.; Tokunaga, M.; Nishida, K.; et al. Zinc is a novel intracellular second messenger. J. Cell Biol. 2007, 177, 637–645. [Google Scholar] [CrossRef]
- Stork, C.J.; Li, Y.V. Zinc release from thapsigargin/IP3-sensitive stores in cultured cortical neurons. J. Mol. Signal. 2010, 5, 5. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).