Different Effects of Empagliflozin on Markers of Mineral-Bone Metabolism in Diabetic and Non-Diabetic Patients with Stage 3 Chronic Kidney Disease
Abstract
:1. Introduction
2. Materials and Methods
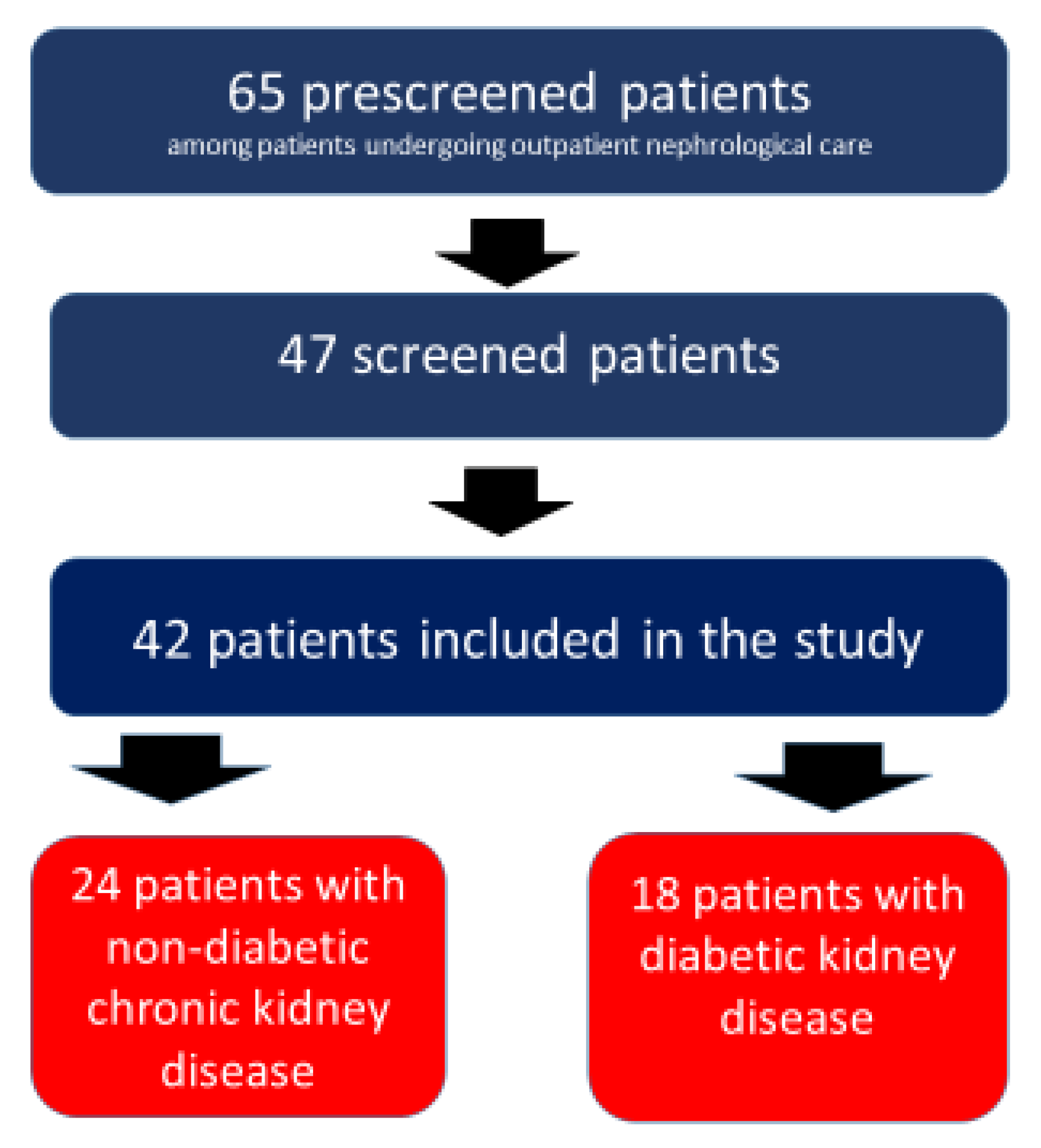
2.1. Patients
2.2. Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Juppner, H. Phosphate and FGF-23. Kidney Int. Suppl. 2011, 79, S24–S27. [Google Scholar] [CrossRef] [Green Version]
- Tentori, F.; Blayney, M.J.; Albert, J.M.; Gillespie, B.W.; Kerr, P.G.; Bommer, J.; Young, E.W.; Akizawa, T.; Akiba, T.; Pisoni, R.L.; et al. Mortality risk for dialysis patients with different levels of serum calcium, phosphate, and PTH: The Dialysis Outcomes and Practice Patterns Study (DOPPS). Am. J. Kidney Dis. 2008, 52, 519–530. [Google Scholar] [CrossRef]
- Tonelli, M.; Sacks, F.; Pfeffer, M.; Gao, Z.; Curhan, G. Relation between serum phosphate level and cardiovascular event rate in people with coronary disease. Circulation 2005, 25, 2627–2633. [Google Scholar] [CrossRef] [Green Version]
- Dhingra, R.; Sullivan, L.M.; Fox, C.S.; Wang, T.J.; D’Agostino, R.B.; Gaziano, J.M.; Ramachandran, S.V. Relations of serum phosphate and calcium levels to the incidence of cardiovascular disease in the community. Arch. Intern. Med. 2007, 14, 879–885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kandula, P.; Dobre, M.; Schold, J.D.; Schreiber, M.J.; Mehrotra, R.; Navaneethan, S.D. Vitamin D supplementation in chronic kidney disease: A systematic review and meta-analysis of observational studies and randomized controlled trials. Clin. J. Am. Soc. Nephrol. 2011, 6, 50–62. [Google Scholar] [CrossRef] [Green Version]
- Pittas, A.G.; Dawson-Hughes, B. Vitamin D and Diabetes. J. Steroid. Biochem. Mol. Biol. 2010, 121, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Bakris, G.L.; Fonseca, V.; Sharma, K.; Wright, E. Renal sodium-glucose transport: Role in diabetes mellitus and potential clinical implications. Kidney Int. 2009, 75, 1272–1277. [Google Scholar] [CrossRef] [Green Version]
- Shubrook, J.H.; Bokaie, B.B.; Adkins, S.E. Empagliflozin in the treatment of type 2 diabetes: Evidence to date. Drug Des. Dev. Ther. 2015, 9, 5793–5803. [Google Scholar] [CrossRef] [Green Version]
- Triplitt, C.; Cornell, S. Canagliflozin Treatment in Patients with Type 2 Diabetes Mellitus. Clin. Med. Insights Endocrinol. Diabetes 2015, 8, 73–81. [Google Scholar] [CrossRef]
- Scheen, A.J. Pharmacodynamics, efficacy and safety of sodium-glucose co-transporter type 2 (SGLT2) inhibitors for the treatment of type 2 diabetes mellitus. Drugs 2015, 75, 33–59. [Google Scholar] [CrossRef] [PubMed]
- Fioretto, P.; Giaccari, A.; Sesti, G. Efficacy and safety of dapagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor in diabetes mellitus. Cardiovasc. Diabetol. 2015, 14, 142. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.W.; Chou, C.L.; Knepper, M.A. Deep Sequencing in Microdissected Renal Tubules Identifies Nephron Segment-Specific Transcriptomes. J. Am. Soc. Nephrol. 2015, 26, 2669–2677. [Google Scholar] [CrossRef]
- Dennis, V.W.; Brazy, P.C. Sodium, phosphate, glucose, bicarbonate, and alanine interactions in the isolated proximal convoluted tubule of the rabbit kidney. J. Clin. Investig. 1978, 62, 387–397. [Google Scholar] [CrossRef] [Green Version]
- Skeith, M.D.; Healey, L.A.; Cutler, R.E. Effect of phloridzin on uric acid excretion in man. Am. J. Physiol. 1970, 219, 1080–1082. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.R.; Kline, I.; Xie, J.; Edwards, R.; Usiskin, K. Effect of canagliflozin on serum electrolytes in patients with type 2 diabetes in relation to estimated glomerular filtration rate (eGFR). Curr. Med. Res. Opin. 2014, 30, 1759–1768. [Google Scholar] [CrossRef]
- Kwon, H. Canagliflozin: Clinical Efficacy and Safety. In Proceedings of the Endocrinologic and Metabolic Drugs Advisory Committee, Silver Spring, MD, USA, 11 December 2013; Available online: www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/UCM336234.pdf (accessed on 13 April 2018).
- Burnett, S.A.; Gunawardene, S.C.; Bringhurst, F.R.; Juppner, H.; Lee, H.; Finkelstein, J.S. Regulation of C-terminal and intact FGF-23 by dietary phosphate in men and women. J. Bone Min. Res 2006, 21, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Quarles, L.D. Skeletal secretion of FGF-23 regulates phosphate and vitamin D metabolism. Nat. Rev. Endocrinol. 2012, 8, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Mirams, M.; Robinson, B.G.; Mason, R.M.; Nelson, A.E. Bone as a source of FGF23: Regulation by phosphate? Bone 2004, 35, 1192–1199. [Google Scholar] [CrossRef]
- Urena, P.; Hruby, M.; Ferreira, A.; Ang, K.S.; de Vernejoul, M.C. Plasma total versus bone alkaline phosphatase as markers of bone turnover in hemodialysis patients. J. Am. Soc. Nephrol. 1996, 7, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Jean, G.; Souberbielle, J.C.; Zaoui, E.; Lorriaux, C.; Mayor, B.; Hurot, J.M.; Deleaval, P.; Chazot, C. Total and bone-specific alkaline phosphatases in haemodialysis patients with chronic liver disease. Clin. Biochem. 2012, 45, 436–439. [Google Scholar] [CrossRef]
- Sardiwal, S.; Gardham, C.; Coleman, A.E.; Stevens, P.E.; Delaney, M.P.; Lamb, E.J. Bone-specific alkaline phosphatase concentrations are less variable than those of parathyroid hormone in stable hemodialysis patients. Kidney Int. 2012, 82, 100–105. [Google Scholar] [CrossRef] [Green Version]
- Perkovic, V.; Jardine, M.J.; Neal, B.; Bompoint, S.; Heerspink, H.J.L.; Charytan, D.M.; Robert Edwards, M.P.H.; Rajiv Agarwal, M.D.; George Bakris, M.D.; Scott Bull, P.D.; et al. For the CREDENCE Trial Investigators. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306. [Google Scholar] [CrossRef] [Green Version]
- Perkovic, V.; de Zeeuw, D.; Mahaffey, K.W.; Fulcher, G.; Erondu, N.; Shaw, W.; Barrett, T.D.; Weidner-Wells, M.; Deng, H.; Matthews, D.R.; et al. Canagliflozin and renal outcomes in type 2 diabetes: Result from the CANVAS Program randomised clinical trials. Lancet Diabetes Endocrinol. 2018, 6, 691–704. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Michael, G.; Silverman, M.D.; Thomas, A.; Zelniker, M.D.; et al. For the DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Haring, H.U.; Merker, L.; Seewaldt-Becker, E.; Weimer, M.; Meinicke, T.; Broedl, U.C.; Woerle, H. EMPA-REG Trial Investigators. Empagliflozin as add-on to metformin in patients with type 2 diabetes: A 24-week randomized, double-blind, placebo-controlled trial. Diabetes Care 2014, 37, 1650–1659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wahl, P.; Xie, H.; Scialla, J.; Anderson, C.; Bellovich, K.; Brecklin, C.; Chen, J.; Feldman, H.; Gutierrez, O.M.; Lash, J.; et al. Earlier Onset and Greater Severity of Disordered Mineral Metabolism in Diabetic Patients With Chronic Kidney Disease. Diabetes Care 2012, 35, 994–1001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- KDiGO. KDIGO 2020 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease. 2020. Available online: https://kdigo.org/wp-content/uploads/2020/10/KDIGO-2020-Diabetes-in-CKD-GL.pdf (accessed on 10 December 2021).
- Food and Nutrition Board. Phosphorus. In Dietary Reference Intakes: Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride; Institute of Medicine, Ed.; National Academy Press: Washington, DC, USA, 1997; pp. 146–189. [Google Scholar]
- Levey, A.S.; Stevens, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; Coresh, J. CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation in estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Kenny, A.P.; Glen, A.C. Tests of phosphate reabsorption. Lancet 1973, 2, 158. [Google Scholar] [CrossRef]
- Chong, W.H.; Molinolo, A.A.; Chen, C.C.; Collins, M.T. Tumor-induced osteomalacia. Endocr. Relat. Cancer 2011, 18, R53–R77. [Google Scholar] [CrossRef] [Green Version]
- Blau, J.E.; Bauman, V.; Conway, E.M. Canagliflozin triggers the FGF23/1,25-dihydoxywitamin D/PTH axis in healthy volunteers in a randomized crossover study. JCI Insight. 2018, 3, e99123. [Google Scholar] [CrossRef]
- Taylor, S.I.; Blau, J.; Rother, K.I. Possible adverse effects of SGLT2 inhibitors on bone. Lancet Diabetes Endocrinol. 2015, 3, 8–10. [Google Scholar] [CrossRef] [Green Version]
- Gkentzi, D.; Efthymiadou, A.; Kritikou, D.; Chrysis, D. Fibroblast growth factor 23 and Klotho serum levels in healthy children. Bone 2014, 66, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Payne, R.B. Renal tubular reabsorption of phosphate (TmP/GFR): Indication and interpretation. Ann. Clin. Biochem. 1998, 35, 201–206. [Google Scholar] [CrossRef]
- Komo, T. Relation between serum FGF-23 and phosphate or bone metabolism in adults. J. Osaka City Med. Assoc. 2007, 56, 35–41. (In Japanese) [Google Scholar]
- Ozeki, M.; Fujita, S.; Kizawa, S.; Morita, H.; Sohmiya, K.; Hoshiga, M.; Ishizaka, N. Association of serum level of FGF23 and α-Klotho with glomerular filtration rate and proteinuria among cardiac patients. BMC Nephrol. 2014, 15, 147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vervloet, M.G.; van Ittersum, F.J.; Buttler, R.M.; Heijboer, A.C.; Blankenstein, M.A.; ter Wee, P.M. Effects of dietary phosphate and calcium intake on fibroblast growth factor-23. Clin. J. Am. Soc. Nephrol. 2011, 6, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Perwad, F.; Azam, N.; Zhang, M.Y.H.; Yamashita, T.; Tenenhouse, H.S.; Portale, A.A. Dietary and serum phosphate regulate fibroblast growth factor 23 expression and 1,25-dihydroxyvitamin D metabolism in mice. Endocrinology 2005, 146, 5358–5364. [Google Scholar] [CrossRef] [Green Version]
- Shimada, T.; Hasegawa, H.; Yamazaki, Y.; Muto, T.; Hino, R.; Takeuchi, Y.; Nakahara, K.; Fukumoto, S.; Yamashita, T. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J. Bone. Miner. Res. 2004, 19, 429–435. [Google Scholar] [CrossRef] [Green Version]
- Bikle, D.D. Vitamin D metabolism, mechanism of action, and clinical applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef] [Green Version]
- List, J.F.; Woo, V.; Morales, E.; Tang, W.; Fiedorek, F.T. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care 2009, 32, 650–657. [Google Scholar] [CrossRef] [Green Version]
- Kohan, D.E.; Fioretto, P.; Tang, W.; List, J.F. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 2014, 85, 962–971. [Google Scholar] [CrossRef] [Green Version]
- Alba, M.; Xie, J.; Fung, A.; Desai, M. The effects of canagliflozin, a sodium glucose co-transporter 2 inhibitor, on mineral metabolism and bone in patients with type 2 diabetes mellitus. Curr. Med. Res. Opin. 2016, 32, 1375–1385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blevins, T.C.; Farooki, A. Bone effects of canagliflozin, a sodium glucose cotransporter 2 inhibitor, in patients with type 2 diabetes mellitus. Posrtgr. Med. 2017, 129, 159–168. [Google Scholar] [CrossRef]
- Ferrannini, E.; Berk, A.; Hantel, S.; Pinnetti, S.; Hach, T.; Woerle, H.J.; Broedl, U.C. Long-term safety and efficacy of empagliflozin, sitagliptin, and metformin: An active-controlled, parallel-group, randomized, 78-week open-label extension study in patients with type 2 diabetes. Diabetes Care 2013, 36, 4015–4021. [Google Scholar] [CrossRef] [Green Version]
- Rosenstock, J.; Jelaska, A.; Zeller, C.; Kim, G.; Broedl, U.C.; Woerle, H.J. EMPA-REG BASALTM trial investigators. Impact of empagliflozin added on to basal insulin in type 2 diabetes, inadequately controlled on basal insulin: A 78-week randomized, double-blind, placebo-controlled trial. Diabetes Obes. Metab. 2015, 17, 936–948. [Google Scholar] [CrossRef] [Green Version]
- de Jong, M.A.; Petrykiv, S.I.; Laverman, G.D.; van Herwaarden, A.E.; de Zeeuw, D.; Bakker, S.J.L.; Heerspink, H.J.L.; de Borst, M.H. Effects of dapagliflozin on circulating markers of phosphate homeostasis. Clin. J. Am. Soc. Nephrol. 2019, 14, 66–73. [Google Scholar] [CrossRef]
- Watanabe, C.; Akuta, N.; Suzuki, Y.; Kobayashi, M.; Mori, Y.; Kumada, H. Changes of the Bone Metabolism Markers and Body Composition in Nonalcoholic Fatty Liver Disease by the SGLT2 inhibitor. Diabetes 2018, 65, 782. [Google Scholar] [CrossRef]
- Yale, J.F.; Bakris, G.; Cariou, B.; Yue, D.; David-Neto, E.; Xi, L.; Figueroa, K.; Wajs, E.; Usiskin, K.; Meininger, G. Efficacy and safety of canagliflozin in subjects with type 2 diabetes and chronic kidney disease. Diabetes Obes. Metab. 2013, 15, 463–473. [Google Scholar] [CrossRef]
- Heerspink, H.J.L.; Johnsson, E.; Gause-Nilsson, I.; Cain, V.A.; Sjöström, C.D. Dapagliflozin reduces albuminuria in patients with diabetes and hypertension receiving renin-angiotensin blockers. Diabetes Obes. Metab. 2016, 18, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H. Canagliflozin: Clinical Efficacy and Safety; United States Food and Drug Administration: Silver Spring, MD, USA, 2013. [Google Scholar]
- Ljunggrren, O.; Bolinder, J.; Johansson, L.; Wilding, J.; Langkilde, A.M.; Sjöström, C.D.; Sugg, J.; Parikh, S. Dapagliflozin has no effect on markers of bone formation and resorption or bone mineral density in patients with inadequately controlled type 2 diabetes mellitus on metformin. Diabetes Obes. Metab. 2012, 14, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Meier, C.; Schwartz, A.V.; Egger, A.; Lecka-Czernik, B. Effects of diabetes drugs on the skeleton. Bone 2016, 82, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Zinman, B.; Lachin, J.M.; Haffner, S.M.; Herman, W.H.; Holman, R.R.; Kravitz, B.G.; Yu, D.; Heise, M.A.; Aftring, R.P.; et al. For the A Diabetes Outcome Progression Trial (ADOPT) Study Group. Rosiglitazone-associated fractures in type 2 diabetes: An Analysis from A Diabetes Outcome Progression Trial (ADOPT). Diabetes Care 2008, 31, 845–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandran, M. Diabetes drug effects on the skeleton. Calcif. Tissue Int. 2017, 100, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Egger, A.; Kraenzlin, M.E.; Meier, C. Effect on incretin-based therapies and SGLT2 inhibitors on skeletal health. Curr. Osteoporos. Rep. 2016, 14, 345–350. [Google Scholar] [CrossRef]
- Kestenbaum, B.; Sampson, J.N.; Rudser, K.D.; Patterson, D.J.; Seliger, S.L. Young B, Sherrard DJ, Andress DL: Serum phosphate levels and mortality risk among people with chronic kidney disease. J. Am. Soc. Nephrol. 2005, 16, 520–528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eddington, H.; Hoefield, R.; Sinha, S.; Chrysochou, C.; Lane, B.; Foley, R.N.; Hegarty, J.; New, J.; O’Donoghue, D.J.; Middleton, R.J.; et al. Serum phosphate and mortality in patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 2251–2257. [Google Scholar] [CrossRef] [Green Version]
- Wanner, C.; Inzucchi, S.E.; Lachin, J.M.; Fitchett, D.; von Eynatten, M.; Mattheus, M.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Zinman, B.; et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N. Engl. J. Med. 2016, 375, 323–334. [Google Scholar] [CrossRef]
- Cherney, D.Z.I.; Dekkers, C.C.J.; Barbour, S.J.; Cattran, D.; Abdul Gafor, A.H.; Greasley, P.J.; Laverman, G.D.; Lim, S.K.; Di Tanna, G.C.; Reich, H.N.; et al. DIAMOND investigators. Effects of the SGLT2 inhibitor dapaglifozin on proteinuria in non-diabetic patients with chronic kidney disease (DIAMOND): A randomized, double-blind, crossover trial. Lancet. Diabetes Endocrinol. 2020, 8, 582–593. [Google Scholar] [CrossRef]
- Fioretto, P.; Del Prato, S.; Buse, J.B.; Goldenberg, R.; Giorgino, F.; Reyner, D.; Langkilde, A.M.; Sjöström, C.D.; Sartipy, P. Derive Study Investigators. Efficacy and safety of dapaglifozin in patients with type 2 diabetes and moderate renal impairment (chronic kidney disease stage 3A): The DERIVE Study. Diabetes Obes. Metab. 2018, 20, 2532–2540. [Google Scholar] [CrossRef] [Green Version]
- Kohan, D.E.; Fioretto, P.; Johnsson, K.; Parikh, S.; Ptaszynska, A.; Ying, L. The effect of dapaglifozin on renal function in patients with type 2 diabetes. J. Nephrol. 2016, 29, 391–400. [Google Scholar] [CrossRef]
- Ferrannini, E.; Seman, L.; Seewaldt-Becker, E.; Hantel, S.; Pinnetti, S.; Woerle, H.A. Phase IIb, randomized, placebo-controlled study of the SGLT2 inhibitor empaglifozin in patients with type 2 diabetes. Diabetes Obes. Metab. 2013, 15, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Kashiwagi, A.; Takahashi, H.; Ishikawa, H.; Yoshida, S.; Kazuta, K.; Utsuno, A.; Ueyama, E. A randomized, double-blind, placebo-controlled study on long-term efficacy and safety of ipraglifozin treatment in patients with type 2 diabetes mellitus and renal impairment: Results of the long-term ASP1941 safety evaluation in patients with type 2 diabetes with renal impairment (LANTERN) study. Diabetes Obes. Metab. 2015, 17, 152–160. [Google Scholar] [PubMed]
- Rajasekeran, H.; Reich, H.N.; Hladunewich, M.A.; Cattran, D.; Lovshin, J.A.; Lytvyn, Y.; Bjornstad, P.; Lai, V.; Tse, J.; Cham, L.; et al. Dapagliflozin in focal segmental glomerulosclerosis: A combined human-rodent pilot study. Am. J. Physiol. Renal. Physiol. 2018, 314, F412–F422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bays, H.E.; Weinstein, R.; Law, G.; Canovatchel, W. Canagliflozin: Effects in overweight and obese subjects without diabetes mellitus. Obesity 2014, 22, 1042–1049. [Google Scholar] [CrossRef] [Green Version]

| All Patients (n = 42) | Non-Diabetic (n = 24) | Diabetic (n = 18) | p Value (Non-Diabetic vs. Diabetic) | |
|---|---|---|---|---|
| Men/Women | 25/17 | 13/11 | 12/6 | NS |
| Age, years | 55.9 ± 9 | 53.7 ± 9 | 58.8 ± 8.4 | NS |
| eGFR CKD-EPI, ml/min per 1.73 m2 | 38.6 ± 6.3 | 38.5 ± 6.3 | 38.8 ± 7.7 | NS |
| HbA1c, % | 6.6 ± 0.2 | |||
| Serum calcium (Ca), mmol/L | 2.1 ± 0.02 | 2.1 ± 0.08 | 2.1 ± 0.30 | NS |
| Serum phosphate (PO4), mmol/L | 1.3 ± 0.08 | 1.3 ± 0.1 | 1.2 ± 0.20 | NS |
| Serum PTH, pg/mL | 43.4 ± 20.5 | 34.6 ± 2.6 | 54.8 ± 27.1 | 0.02 |
| Plasma 1.25(OH)2D, pg/ml | 27.7 ± 8.3 | 26.4 ± 11.7 | 29.4 ± 22.4 | NS |
| Serum FGF-23, pg/mL | 107.2 ± 34.9 | 106.2 ± 6.4 | 108.4 ± 87.1 | NS |
| Serum BAP, µg/L | 19.5 ± 3.3 | 17.3 ± 11.3 | 22.2 ± 1.7 | NS |
| Serum albumin, g/L | 33.6 ± 3.04 | 34.5 ± 6.6 | 32.3 ± 0.2 | NS |
| Urine-Ca/creatinine, mmol/g | 0.6 [0.2, 0.9] | 0.6 [0.3, 0.9] | 0.5 [0.3, 1.1] | NS |
| Urine-PO4/creatinine, mmol/g | 15.7 ± 2.3 | 15 ± 14.7 | 16.6 ± 1.2 | NS |
| TRP, % | 67.6 ± 2.9 | 68.9 ±11.6 | 65.8 ± 17.3 | NS |
| TmP/GFR, mmol/L | 0.77 ± 0.14 | 0.80 ± 0.12 | 0.74 ± 0.06 | NS |
| Fractional excretion of phosphate, % | 32.4 ± 2.9 | 31.1 ± 11.6 | 34.1 ± 1.7 | NS |
| Fractional excretion of calcium, % | 0.5 [0.2, 1.0] | 0.6 [0.2, 1.2] | 0.4 [0.2, 0.8] | NS |
| Urine-albumin/creatinine, mg/g | 209.0 ± 31.4 | 239.6 ± 45.6 | 169.9 ± 42.7 | NS |
| Parameter | Subjects | Before Empagliflozin | After Empagliflozin | p-Value after vs. before Empagliflozin Administration |
|---|---|---|---|---|
| Serum total calcium (mmol/L) | All (n = 42) | 2.1 [2.0, 2.3] | 2.13 [2.1, 2.34] | NS |
| ND-CKD (n = 24) | 2.1 ± 0.08 | 2.2 ± 0.1 | NS | |
| DKD (n = 18) | 2.1 ± 0.3 | 2.1 [2.0, 2.2] | NS | |
| Serum phosphate (mmol/L) | All (n = 42) | 1.2 [1.1, 1.5] | 1.2 [1.1, 1.34] | 0.02 |
| ND-CKD (n = 24) | 1.24 [1.1, 1.5] | 1.25 [1.0, 1.39] | NS | |
| DKD (n = 18) | 1.1 ± 0.1 | 1.2 ± 0.2 | 0.02 | |
| PTH (pg/mL) | All (n = 42) | 34.7 [18.7, 57.8] | 34.7 [18.7, 57.8] | NS |
| ND-CKD (n = 24) | 21.3 [14.5, 57.8] * | 21.3 [14.55, 52.9] | NS | |
| DKD (n = 18) | 54.8 ± 27.1 | 62.9 ± 43.2 | 0.0016 | |
| 1.25(OH)2D (pg/mL) | All (n = 42) | 27.2 [18.7, 36.7] | 24.8 [20.1, 39.4] | NS |
| ND-CKD (n = 24) | 26.4 ± 11.7 | 26.8 ± 13.3 | NS | |
| DKD (n = 18) | 29.4 ± 22.4 | 13.8 ± 17.7 | NS | |
| FGF-23 (pg/mL) | All (n = 42) | 98.4 [75.6, 130.3] | 98 [75.6, 130.3] | NS |
| ND-CKD (n = 24) | 106.2 ± 6.4 | 98.7 [76.7, 128.7] | NS | |
| DKD (n = 18) | 98.3 [76.4, 128.3] | 98.3 [76.4, 128.3] | 0.03 | |
| BAP (µg/L) | All (n = 4 2) | 19.5 [12.9, 24.7] | 20.3 [14.3, 25.8] | NS |
| ND-CKD (n = 24) | 13.6 [10.4, 23.8] | 15.8 [11.8, 22.0] | NS | |
| DKD (n = 18) | 22.2 ± 1.7 | 22.5 ± 1.4 | NS | |
| Urine-Ca/Cr, (mmol/g) | All (n = 42) | 0.6 [0.2, 0.9] | 0.42 [0.26, 0.93] | NS |
| ND-CKD (n = 24) | 0.6 [0.3, 0.9] | 0.5 [0.33, 1.4] | NS | |
| DKD (n = 18) | 0.5 [0.3, 1.1] | 0.28 [0.22, 0.8] | NS | |
| Urine-PO4/Cr, (mmol/g) | All (n = 42) | 1.94 [1.5, 2.3] | 1.8 [1.4, 2.2] | NS |
| ND-CKD (n = 24) | 1.9 ± 0.09 | 1.9 ± 1.3 | NS | |
| DKD (n = 18) | 2.0 ± 0.02 | 1.7 [1.5, 2.1] | NS | |
| TRP, % | All (n = 42) | 67.6 ± 2.9 | 64.5 ± 4.9 | NS |
| ND-CKD (n = 24) | 68.9 ±11.6 | 66.5 ± 42.1 | NS | |
| DKD (n = 18) | 65.8 ± 17.3 | 61.8 ± 2.0 | NS | |
| TmP/GFR, mmol/L | All (n = 42) | 0.75 [0.6, 0.9] | 0.86 [0.68, 1.0] | NS |
| ND-CKD (n = 24) | 0.80 ± 0.12 | 0.92 [0.66, 1.0] | NS | |
| DKD (n = 18) | 0.74 ± 0.06 | 0.83 ± 0.28 | NS | |
| Fractional excretion of phosphate, % | All (n = 42) | 31.5 [21.2, 41.9] | 29.2 [19.9, 42.8] | NS |
| ND-CKD (n = 24) | 31.1 ± 11.6 | 33.4 ± 42.2 | NS | |
| DKD (n = 18) | 34.1 ± 1.7 | 26.6 [20.4, 41.7] | NS | |
| Fractional excretion of calcium, % | All (n = 42) | 0.5 [0.2, 1.0] | 0.4 [0.2, 1.0] | NS |
| ND-CKD (n = 24) | 0.6 [0.2, 1.2] | 0.7 [0.2, 1.3] | NS | |
| DKD (n = 18) | 0.4 [0.2, 0.8] | 0.4 [0.2, 0.7] | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masajtis-Zagajewska, A.; Hołub, T.; Pęczek, K.; Makówka, A.; Nowicki, M. Different Effects of Empagliflozin on Markers of Mineral-Bone Metabolism in Diabetic and Non-Diabetic Patients with Stage 3 Chronic Kidney Disease. Medicina 2021, 57, 1352. https://doi.org/10.3390/medicina57121352
Masajtis-Zagajewska A, Hołub T, Pęczek K, Makówka A, Nowicki M. Different Effects of Empagliflozin on Markers of Mineral-Bone Metabolism in Diabetic and Non-Diabetic Patients with Stage 3 Chronic Kidney Disease. Medicina. 2021; 57(12):1352. https://doi.org/10.3390/medicina57121352
Chicago/Turabian StyleMasajtis-Zagajewska, Anna, Tomasz Hołub, Katarzyna Pęczek, Agnieszka Makówka, and Michał Nowicki. 2021. "Different Effects of Empagliflozin on Markers of Mineral-Bone Metabolism in Diabetic and Non-Diabetic Patients with Stage 3 Chronic Kidney Disease" Medicina 57, no. 12: 1352. https://doi.org/10.3390/medicina57121352







