The Hematopoietic Effect of Ninjinyoeito (TJ-108), a Traditional Japanese Herbal Medicine, in Pregnant Women Preparing for Autologous Blood Storage
Abstract
:1. Introduction
2. Materials and Methods
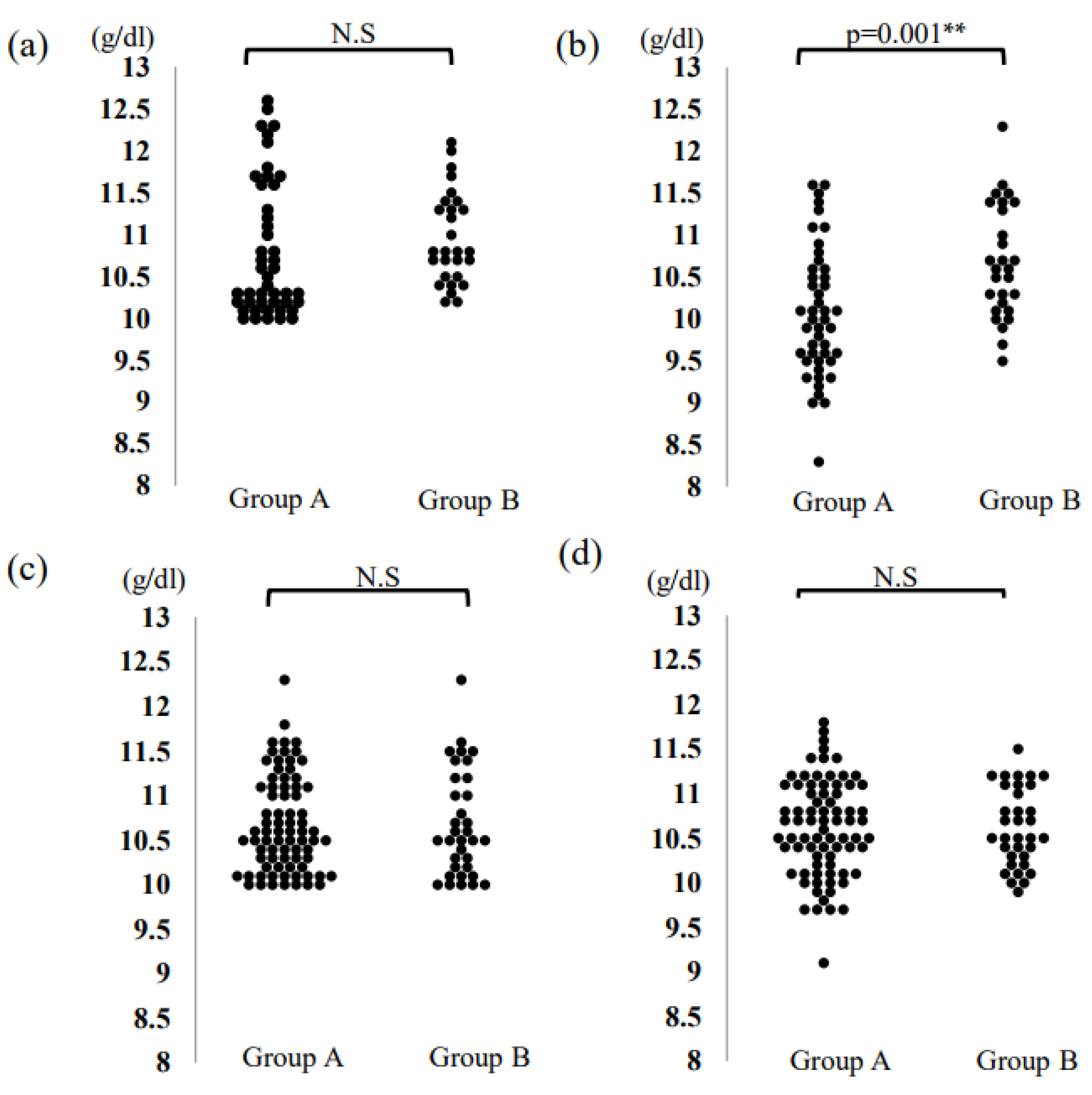
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Say, L.; Chou, D.; Gemmill, A.; Tuncalp, O.; Moller, A.B.; Daniels, J.; Gulmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A WHO systematic analysis. Lancet Glob. Health 2014, 2, e323–e333. [Google Scholar] [CrossRef]
- Chua, S.C.; Joung, S.J.; Aziz, R. Incidence and risk factors predicting blood transfusion in caesarean section. Aust. New Zealand J. Obstet. Gynaecol. 2009, 49, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Vetter, T.R.; Adhami, L.F.; Porterfield, J.R., Jr.; Marques, M.B. Perceptions about blood transfusion: A survey of surgical patients and their anesthesiologists and surgeons. Anesth. Analg. 2014, 118, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Silver, R.M. Abnormal Placentation: Placenta Previa, Vasa Previa, and Placenta Accreta. Obstet. Gynecol. 2015, 126, 654–668. [Google Scholar] [CrossRef] [PubMed]
- Oyelese, Y.; Smulian, J.C. Placenta previa, placenta accreta, and vasa previa. Obstet. Gynecol. 2006, 107, 927–941. [Google Scholar] [CrossRef] [PubMed]
- Billote, D.B.; Glisson, S.N.; Green, D.; Wixson, R.L. Efficacy of preoperative autologous blood donation: Analysis of blood loss and transfusion practice in total hip replacement. J. Clin. Anesth. 2000, 12, 537–542. [Google Scholar] [CrossRef]
- Dinsmoor, M.J.; Hogg, B.B. Autologous blood donation with placenta previa: Is it feasible? Am. J. Perinatol. 1995, 12, 382–384. [Google Scholar] [CrossRef]
- Fruchart, M.F.; Rolland, E.; Courtois, F.; Meier, F.; Besse-Moreau, M.; Foucher, E.; Engelmann, P. Programmed autologous transfusion in obstetrics. Blood samples in 100 patients in the last month of pregnancy. J. Gynecol. Obstet. Biol. Reprod. 1995, 24, 204–208. [Google Scholar]
- Yamada, T.; Mori, H.; Ueki, M. Autologous blood transfusion in patients with placenta previa. Acta Obstet. Gynecol. Scand 2005, 84, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Spahn, D.R.; Casutt, M. Eliminating blood transfusions: New aspects and perspectives. Anesthesiology 2000, 93, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Miyano, K.; Nonaka, M.; Uzu, M.; Ohshima, K.; Uezono, Y. Multifunctional Actions of Ninjinyoeito, a Japanese Kampo Medicine: Accumulated Scientific Evidence Based on Experiments With Cells and Animal Models, and Clinical Studies. Front. Nutr. 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Takano, F.; Ohta, Y.; Tanaka, T.; Sasaki, K.; Kobayashi, K.; Takahashi, T.; Yahagi, N.; Yoshizaki, F.; Fushiya, S.; Ohta, T. Oral Administration of Ren-Shen-Yang-Rong-Tang ‘Ninjin’yoeito’ Protects Against Hematotoxicity and Induces Immature Erythroid Progenitor Cells in 5-Fluorouracil-induced Anemia. Evid. Based Complement Alternat. Med. 2009, 6, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Motoo, Y.; Mouri, H.; Ohtsubo, K.; Yamaguchi, Y.; Watanabe, H.; Sawabu, N. Herbal medicine Ninjinyoeito ameliorates ribavirin-induced anemia in chronic hepatitis C: A randomized controlled trial. World J. Gastroenterol. 2005, 11, 4013–4017. [Google Scholar] [CrossRef] [PubMed]
- Waugh, R.E.; Mantalaris, A.; Bauserman, R.G.; Hwang, W.C.; Wu, J.H. Membrane instability in late-stage erythropoiesis. Blood 2001, 97, 1869–1875. [Google Scholar] [CrossRef] [PubMed]
- Muckenthaler, M.U.; Rivella, S.; Hentze, M.W.; Galy, B. A Red Carpet for Iron Metabolism. Cell 2017, 168, 344–361. [Google Scholar] [CrossRef]
- Hatano, R.; Takano, F.; Fushiya, S.; Michimata, M.; Tanaka, T.; Kazama, I.; Suzuki, M.; Matsubara, M. Water-soluble extracts from Angelica acutiloba Kitagawa enhance hematopoiesis by activating immature erythroid cells in mice with 5-fluorouracil-induced anemia. Exp. Hematol. 2004, 32, 918–924. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.A.; Lupattelli, A.; Koren, G.; Nordeng, H. Herbal medicine use in pregnancy: Results of a multinational study. BMC Complement Altern. Med. 2013, 13, 355. [Google Scholar] [CrossRef] [PubMed]
- John, L.J.; Shantakumari, N. Herbal medicines use during pregnancy: A review from the Middle East. Oman. Med. J. 2015, 30, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Hwang, J.H.; Choi, S.; Han, D. Safety classification of herbal medicines used among pregnant women in Asian countries: A systematic review. BMC Complement Altern. Med. 2017, 17, 489. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Obara, T.; Ishikawa, T.; Noda, A.; Matsuzaki, F.; Arita, R.; Ohsawa, M.; Mano, N.; Kikuchi, A.; Takayama, S.; et al. Prescription of Kampo Formulations for Pre-natal and Post-partum Women in Japan: Data From an Administrative Health Database. Front. Nutr. 2021, 12, 762895. [Google Scholar] [CrossRef] [PubMed]
| Variables | Group A | Group B | p Value |
|---|---|---|---|
| n = 38 | n = 27 | ||
| Number or Median (Range) | Number or Median (Range) | ||
| Age (year) | 35 (28–44) | 34 (24–44) | 0.310 |
| Height (cm) | 160 (140–165) | 160 (151–169) | 0.329 |
| Body weight before pregnancy (kg) | 52 (41–65) | 51 (44–87) | 0.669 |
| BMI at before pregnancy (kg/m2) | 20.7 (16.4–27.8) | 20.4 (17.5–32.7) | 0.926 |
| Body weight at birth (kg) | 61 (47–72) | 59 (50–89) | 0.680 |
| BMI at birth (kg/m2) | 24 (21.0–28.5) | 24.2 (19.3–33.5) | 0.963 |
| Gestational age (week) | 37.0 (34.4–37.3) | 37 (32.4–37.7) | 0.942 |
| Primigravida (%) | 15 (39.5) | 15 (55.6) | 0.200 |
| ART (%) | 6 (15.8) | 7 (25.9) | 0.314 |
| Emergency cesarean delivery (%) | 7 (18.4) | 6 (22.2) | 0.706 |
| Apgar score 1 min | 8 (1–9) | 8 (3–9) | 0.660 |
| Apgar score 5 min | 9 (5–9) | 9 (6–9) | 0.225 |
| Birth weight (g) | 2643 (1966–3440) | 2500 (1917–3035) | 0.250 |
| Male (%) | 24 (63.2) | 11 (40.7) | 0.074 |
| Female (%) | 14 (36.8) | 16 (59.3) | 0.074 |
| Variables | Group A | Group B | p Value | |
|---|---|---|---|---|
| n = 38 | n = 27 | |||
| Number or Median (Range) | Number or Median (Range) | |||
| Total of autologous blood storage (mL) | 600 (300–1200) | 600 (300–1200) | 0.589 | |
| Blood transfusion during cesarean section | Autologous transfusion (mL) | 300 (0–1200) | 300 (0–1200) | 0.336 |
| Allogeneic transfusion (%) | 8 (21.1) | 8 (29.7) | 0.121 | |
| RBC (unit) | 0 (0–10) | 0 (0–8) | 0.729 | |
| FFP (unit) | 0 (0–10) | 0 (0–6) | 0.779 | |
| PC (unit) | 0 (0–10) | 0 (0–0) | 0.919 | |
| Infusion (mL) | 1950 (850–4300) | 1450 (700–3400) | 0.186 | |
| Blood loss (mL) | 1835 (340–7500) | 1760 (895–5500) | 0.863 | |
| Urine output (mL) | 110 (0–550) | 80 (0–500) | 0.105 | |
| Operation time (min) | 65 (39–148) | 68 (41–104) | 0.739 | |
| Group A | Group B | p Value | |
|---|---|---|---|
| 38 Storage (n = 38) | 27 Storage (n = 27) | ||
| Median (Range) | Median (Range) | ||
| Cancellation rate of next storage (%) | 17 (44.7) | 3 (11.1) | 0.004 * |
| Hb level before storage (g/dL) | 10.5 (10.0–12.6) | 10.8 (10.2–12.1) | 0.070 |
| Hb level after 7 days of storage (g/dL) | 10.0 (8.3–11.6) | 10.6 (9.5–12.3) | 0.001 ** |
| Amount of change in Hb (g/dL) | −0.6 (−2.1–0.4) | −0.2 (−1.5–0.6) | 0.012 * |
| Ht level before storage (%) | 31.9 (29.6–39.5) | 32.3 (29.8–36.3) | 0.394 |
| Ht level after 7 days of storage (%) | 30.5 (27.7–36.5) | 32.1 (28.4–36.4) | 0.015 * |
| Amount of change in Ht (%) | −1.8 (−5.8–1.9) | −0.7 (−2.5–1.2) | 0.015 * |
| RBC level before storage (×104/μL) | 356 (296–433) | 354 (318–430) | 0.730 |
| RBC level after 7 days of storage (×104/μL) | 333 (270–396) | 349 (299–398) | 0.125 |
| Amount of change in RBC (×104/μL) | −24 (−69–12) | −11.5 (−45–12) | 0.003 ** |
| Group A | Group B | p Value | |
|---|---|---|---|
| 55 Storage (n = 38) | 38 Storage (n = 27) | ||
| Median (Range) | Median (Range) | ||
| Cancel rate of next storage (%) | 6 (10.9) | 1 (2.7) | 0.137 |
| Hb level before storage (g/dL) | 10.6 (10.0–11.8) | 10.5 (10.0–12.3) | 0.769 |
| Hb level after 7 days of storage (g/dL) | 10.7 (9.1–11.8) | 10.5 (9.9–11.5) | 1.000 |
| Amount of change in Hb (g/dL) | −0.1 (−1.0–1.1) | 0.0 (−1.4–0.6) | 0.692 |
| Ht level before storage (%) | 32.2 (29.0–37.2) | 31.8 (28.9–36.4) | 0.367 |
| Ht level after 7 days of storage (%) | 32.2 (28.8–37.2) | 31.7 (28.6–34.1) | 0.259 |
| Amount of change in Ht (%) | 0.2 (−3.4–3.2) | −0.1 (−3.1–2.7) | 0.629 |
| RBC level before storage (×104/μL) | 343 (305–404) | 337 (298–398) | 0.205 |
| RBC level after 7 days of storage (×104/μL) | 344 (292–392) | 332 (297–384) | 0.082 |
| Amount of change in RBC (×104/μL) | −4 (−36–24) | −8 (−34–18) | 0.841 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukuda, E.; Misugi, T.; Kitada, K.; Fudaba, M.; Kurihara, Y.; Tahara, M.; Hamuro, A.; Nakano, A.; Koyama, M.; Tachibana, D. The Hematopoietic Effect of Ninjinyoeito (TJ-108), a Traditional Japanese Herbal Medicine, in Pregnant Women Preparing for Autologous Blood Storage. Medicina 2022, 58, 1083. https://doi.org/10.3390/medicina58081083
Fukuda E, Misugi T, Kitada K, Fudaba M, Kurihara Y, Tahara M, Hamuro A, Nakano A, Koyama M, Tachibana D. The Hematopoietic Effect of Ninjinyoeito (TJ-108), a Traditional Japanese Herbal Medicine, in Pregnant Women Preparing for Autologous Blood Storage. Medicina. 2022; 58(8):1083. https://doi.org/10.3390/medicina58081083
Chicago/Turabian StyleFukuda, Eriko, Takuya Misugi, Kohei Kitada, Megumi Fudaba, Yasushi Kurihara, Mie Tahara, Akihiro Hamuro, Akemi Nakano, Masayasu Koyama, and Daisuke Tachibana. 2022. "The Hematopoietic Effect of Ninjinyoeito (TJ-108), a Traditional Japanese Herbal Medicine, in Pregnant Women Preparing for Autologous Blood Storage" Medicina 58, no. 8: 1083. https://doi.org/10.3390/medicina58081083







