New Hydrazones Bearing Thiazole Scaffold: Synthesis, Characterization, Antimicrobial, and Antioxidant Investigation
Abstract
:1. Introduction
2. Results and Discussion
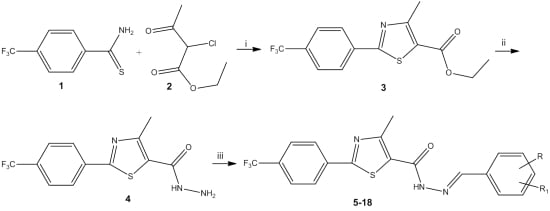
2.1. Chemistry

| Cp | R | R1 | Cp | R | R1 |
|---|---|---|---|---|---|
| 5 | H | 2-OCH3 | 12 | H | 4-Br |
| 6 | H | 3-OCH3 | 13 | H | 4-F |
| 7 | H | 4-OCH3 | 14 | H | 2-OH |
| 8 | H | 3-Cl | 15 | H | 3-OH |
| 9 | H | 4-Cl | 16 | H | 4-OH |
| 10 | H | 2-Br | 17 | 2-Cl | 4-Cl |
| 11 | H | 3-Br | 18 | 2-Cl | 6-Cl |
2.2. Antimicrobial Screening
| Cp (1 mg/mL) | Microbial Strain/Diameter of the Growth Inhibition Zone (mm) | ||||
|---|---|---|---|---|---|
| Gram-Negative Bacteria | Gram-Positive Bacteria | Fungus | |||
| S. enteritidis ATCC 13,076 | E. coli ATCC 25,922 | L. monocytogenes ATCC 13,932 | S. aureus ATCC 6538P | C. albicans ATCC 10,231 | |
| 3 | 20 | 18 | 16 | 12 | 18 |
| 4 | 20 | 18 |  |  | 20 |
| 5 | 10 | 16 | - | 12 | 25 |
| 6 | 10 | 18 | - | 12 | 20 |
| 7 | 18 | 18 | 14 | 16 | 14 |
| 8 | 10 | 18 | - | 14 | 20 |
| 9 | 18 | 18 | 14 | 14 | 18 |
| 10 | 18 | 20 | - | 14 | 22 |
| 11 | 18 | 20 | - | 14 | 20 |
| 12 |  | 20 | 14 | 14 |  |
| 13 | 20 | 18 | 14 | 16 | 14 |
| 14 | 12 | 16 | 16 | 16 | 18 |
| 15 | 18 | 18 | - | 12 | 18 |
| 16 | 18 | 20 | - | 12 | 20 |
| 17 | 12 | 20 | - | 12 | 18 |
| 18 | 18 | 20 | - | 12 | 20 |
| Gentamicin | 18 | 22 | 18 | 19 | NT |
| Fluconazole | NT | NT | NT | NT | 25 |
2.3. DPPH Based Free Radical Scavenging Activity

3. Experimental Section
3.1. Reagents and Solvents
3.2. Analytical Methods
3.3. Chemical Synthesis
3.3.1. General Procedure for the Synthesis of Ethyl-4-methyl-2-(4-(trifluoromethyl)phenyl)thiazole-5-carboxylate (3)
3.3.2. General Procedure for the Synthesis of 4-Methyl-2-(4-(trifluoromethyl)phenyl)thiazole-5-carbohydrazide (4)
3.3.3. General Procedure for the Synthesis of Acyl-hydrazones 5–18
3.4. Antimicrobial Screening
3.5. DPPH Based Free Radical Scavenging Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jomha, M.Y.; Yusef, H.; Holail, H. Antimicrobial and biocide resistance of bacteria in a Lebanese tertiary care hospital. J. Glob. Antimicrob. Resist. 2014, 2, 299–305. [Google Scholar] [CrossRef]
- Tang, S.S.; Apisarnthanarak, A.; Hsu, L.Y. Mechanisms of β-lactam antimicrobial resistance and epidemiology of major community and healthcare-associated multidrug-resistant bacteria. Adv. Drug Deliv. Rev. 2014, 78, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Ogbolu, D.O.; Webber, M.A. High-level and novel mechanisms of carbapenem resistance in Gramnegative bacteria from tertiary hospitals in Nigeria. Int. J. Antimicrob. Agents 2014, 43, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Nayak, N.; Lenka, R.K.; Padhy, R.N. Surveillance of multidrug resistant suppurative infection causing bacteria in hospitalized patients in an Indian tertiary care hospital. J. Acute Dis. 2014, 148–156. [Google Scholar] [CrossRef]
- WHO Library Cataloguing-in-Publication Data. Antimicrobial Resistance: Global Report on Surveillance; WHO Press, World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Popoola, O.K.; Marnewick, J.L.; Rautenbach, F.; Ameer, F.; Iwuoha, E.I.; Hussein, A.A. Inhibition of oxidative stress and skin aging-related enzymes by prenylated chalcones and other flavonoids from Helichrysum teretifolium. Molecules 2015, 20, 7143–7155. [Google Scholar] [CrossRef] [PubMed]
- Ni, N.; Liu, Q.; Ren, H.; Wu, D.; Luo, C.; Li, P.; Wan, J.B.; Su, H. Ginsenoside Rb1 protects rat neural progenitor cells against oxidative injury. Molecules 2014, 19, 3012–3024. [Google Scholar] [CrossRef] [PubMed]
- Shakir, R.M.; Ariffin, A.; Abdulla, M.A. Synthesis of new 2,5-di-substituted 1,3,4-oxadiazoles bearing 2,6-di-tert-butylphenol moieties and evaluation of their antioxidant activity. Molecules 2014, 19, 3436–3449. [Google Scholar] [CrossRef] [PubMed]
- Gwaram, N.S.; Ali, H.M.; Abdulla, M.A.; Buckle, M.J.C.; Sukumaran, S.D.; Chung, L.Y.; Othman, R.; Alhadi, A.A.; Yehye, W.A.; Hadi, A.H.A.; et al. Synthesis, characterization, X-ray crystallography, Acetyl cholinesterase inhibition and antioxidant activities of some novel ketone derivatives of gallic hydrazide-derived Schiff bases. Molecules 2012, 17, 2408–2427. [Google Scholar] [CrossRef] [PubMed]
- Kashinath, K.; Durg, V.; Baburao, K.; Angadi, S.D. Synthesis, characterization and biological activities of Schiff bases derived from hydrazide derivative and their Cu(II), Ni(II), Co(II), Zn(II), Cd(II) and Hg(II), complexes. Int. J. Res. Pharm. Chem. 2014, 4, 557–563. [Google Scholar]
- Husain, A.; Varshney, M.M.; Parcha, V.; Ahmad, A.; Khan, S.A. Synthesis and biological evaluation of new hydrazide-Schiff bases. Bangladesh J. Pharmacol. 2015, 10, 555–561. [Google Scholar] [CrossRef]
- Zayed, E.M.; Zayed, M.A. Synthesis of novel Schiff’s bases of highly potential biological activities and their structure investigation. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 143, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Hearn, M.J.; Cynamon, M.H.; Chen, M.F.; Coppins, R.; Davis, J.; Kang, H.J.-O.; Noble, A.; Tu-Sekine, B.; Terrot, M.S.; Trombino, D.; et al. Preparation and antitubercular activities in vitro and in vivo of novel Schiff bases of isoniazid. Eur. J. Med. Chem. 2009, 44, 4169–4178. [Google Scholar] [CrossRef] [PubMed]
- Hassan, F.A. Synthesis, characterization, anti-inflammatory, and antioxidant activities of some new thiazole derivatives. Int. J. Appl. Sci. Technol. 2012, 2, 180–187. [Google Scholar]
- Siddiqui, N.; Arshad, M.F.; Ahsan, W.; Alam, M.S. Thiazoles: A valuable insight into the recent advances and biological activities. Int. J. Pharm. Sci. Drug Res. 2009, 1, 136–143. [Google Scholar]
- Moldovan, C.M.; Oniga, O.; Pârvu, A.; Tiperciuc, B.; Verite, P.; Pîrnău, A.; Crişan, O.; Bojiţă, M.; Pop, R. Synthesis and anti-inflammatory evaluation of some new acyl-hydrazones bearing 2-aryl-thiazole. Eur. J. Med. Chem. 2011, 46, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Andreania, A.; Leoni, A.; Locatelli, A.; Morigi, R.; Rambaldi, M.; Cervellati, R.; Greco, E.; Kondratyuk, T.P.; Park, E.-J.; Huang, K.; et al. Chemopreventive and antioxidant activity of 6 substituted imidazo[2,1-b]thiazoles. Eur. J. Med. Chem. 2013, 68, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Jaishree, V.; Ramdas, N.; Sachin, J.; Ramesh, B. In vitro antioxidant properties of new thiazole derivatives. J. Saudi Chem. Soc. 2012, 16, 371–376. [Google Scholar] [CrossRef]
- Abdel-Wahab, B.F.; Awad, G.E.A.; Badria, F.A. Synthesis, antimicrobial, antioxidant, anti-hemolytic and cytotoxic evaluation of new imidazole-based heterocycles. Eur. J. Med. Chem. 2011, 46, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, H.; Reddy, P.V.N.; Monteleone, D.; Mayhoub, A.S.; Cushman, M.; Seleem, M.N. Synthesis and antibacterial evaluation of a novel series of synthetic phenylthiazole compounds against methicillin-resistant Staphylococcus aureus (MRSA). Eur. J. Med. Chem. 2015, 94, 306–316. [Google Scholar] [CrossRef] [PubMed]
- El-Wahab, H.A.; El-Fattah, M.A.; El-Khalik, N.A.; Nassar, H.S.; Abdelall, M.M. Synthesis and characterization of coumarin thiazole derivative 2-(2-amino-1,3-thiazol-4-yl)-3H-benzo[f]chromen-3-one with anti-microbial activity and its potential application in antimicrobial polyurethane coating. Prog. Org. Coat. 2014, 77, 1506–1511. [Google Scholar] [CrossRef]
- Yang, S.J.; Lee, W.J.; Kim, E.-A.; Namd, K.D.; Hahn, H.-G.; Choi, S.Y.; Cho, S.-W. Effects of N-adamantyl-4-methylthiazol-2-amine on hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced diabetic rats. Eur. J. Pharmacol. 2014, 736, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.L.; Song, L.X.; Li, Y.F.; Li, Y.L.; Guo, Y.Z.; Zhang, E.; Liu, H.M. Synthesis and biological evaluation of dehydroepiandrosterone-fused thiazole, imidazo[2,1-b]thiazole, pyridine steroidal analogues. Steroids 2014, 80, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Prashanth, T.; Thirusangu, P.; Avin, B.R.V.; Ranganatha, V.L.; Prabhakar, B.T.; Khanum, S.A. Synthesis and evaluation of novel benzophenone-thiazole derivatives as potent VEGF-A inhibitors. Eur. J. Med. Chem. 2014, 87, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Popsavin, M.; Koji, V.; Spai, S.; Svircev, M.; Bogdanovic, G.; Jakimov, D.; Aleksic, L.; Popsavin, V. 2-Substituted thiazole-4-carboxamide derivatives as tiazofurin mimics: Synthesis and in vitro antitumour activity. Tetrahedron 2014, 70, 2343–2350. [Google Scholar] [CrossRef]
- Tiperciuc, B.; Zaharia, V.; Colosi, I.; Moldovan, C.; Crisan, O.; Pirnau, A.; Vlase, L.; Duma, M.; Oniga, O. Synthesis and Evaluation of antimicrobial activity of some new hetaryl-azoles derivatives obtained from 2-aryl-4-methylthiazol-5-carbohydrazides and isonicotinic acid hydrazide. J. Heterocycl. Chem. 2012, 49, 1407–1414. [Google Scholar] [CrossRef]
- Bharti, S.K.; Nath, G.; Tilak, R.; Singh, S.K. Synthesis, anti-bacterial and anti-fungal activities of some novel Schiff bases containing 2,4-disubstituted thiazole ring. Eur. J. Med. Chem. 2010, 45, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, F.; Bizzarri, B.; Bolasco, A.; Secci, D.; Chimenti, P.; Granese, A.; Carradori, S.; D’Ascenzio, M.; Lilli, D.; Rivanera, D. Synthesis and biological evaluation of novel 2,4-disubstituted-1,3-thiazoles as anti-Candida spp. agents. Eur. J. Med. Chem. 2011, 46, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, N.D.; Patil, S.V.; Bobade, V.D. Hybrids of ravuconazole: Synthesis and biological evaluation. Eur. J. Med. Chem. 2012, 54, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, N.D.; Patil, S.V.; Bobade, V.D. Synthesis and biological evaluation of some novel thiazole substituted benzotriazole derivatives. Bioorg. Med. Chem. Lett. 2012, 22, 3449–3454. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.S.; Liu, L.; Lee, Y.E.; Lee, D.W. Synthesis, antibacterial activity and quantum-chemical studies of novel 2-arylidenehydrazinyl-4-arylthiazole analogues. Chem. Pharm. Bull. 2011, 59, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Carradori, S.; Secci, D.; Bolasco, A.; Rivanera, D.; Mari, E.; Zicari, A.; Lotti, L.V.; Bizzarri, B. Synthesis and cytotoxicity of novel (thiazol-2-yl)hydrazine derivatives as promising anti-Candida agents. Eur. J. Med. Chem. 2013, 65, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Secci, D.; Bizzarri, B.; Bolasco, A.; Carradori, S.; D’Ascenzio, M.; Rivanera, D.; Mari, E.; Polletta, L.; Zicari, A. Synthesis, anti-Candida activity, and cytotoxicity of new (4-(4-iodophenyl)thiazol-2-yl)hydrazine derivatives. Eur. J. Med. Chem. 2012, 53, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Benedec, D.; Vlase, L.; Oniga, I.; Mot, A.C.; Damian, G.; Hanganu, D.; Duma, M.; Silaghi-Dumitrescu, R. Polyphenolic Composition, antioxidant and antibacterial activities for two romanian subspecies of Achillea distans Waldst. et Kit. ex Willd. Molecules 2013, 18, 8725–8739. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.S.; Pinto, A.C.S.; Nogueira, K.L.; Rocha e Silva, L.F.; de Almeida, P.D.O.; de Vasconcellos, M.C.; Chaves, F.C.M.; Tadei, W.P.; Pohlit, A.M. Stability and antioxidant activity of semi-synthetic derivatives of 4-nerolidylcatechol. Molecules 2013, 18, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Simić, A.; Manojlović, D.; Šegan, D.; Todorović, M. Electrochemical behavior and antioxidant and prooxidant activity of natural phenolics. Molecules 2007, 12, 2327–2340. [Google Scholar] [CrossRef] [PubMed]
- Moalin, M.; Strijdonck, G.P.F.; Beckers, M.; Hagemen, G.J.; Borm, P.J.; Bast, A.; Haenen, G.R. A planar conformation and the hydroxyl groups in the B and C rings play a pivotal role in the antioxidant capacity of quercetin and quercetin derivatives. Molecules 2011, 16, 9636–9650. [Google Scholar] [CrossRef] [PubMed]
- National Committe for Clinical Laboratory Standards. NCCLS Approved Standard M27-PA; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 1997. [Google Scholar]
- Sample Availability: Samples of the compounds 3–18 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nastasă, C.; Tiperciuc, B.; Duma, M.; Benedec, D.; Oniga, O. New Hydrazones Bearing Thiazole Scaffold: Synthesis, Characterization, Antimicrobial, and Antioxidant Investigation. Molecules 2015, 20, 17325-17338. https://doi.org/10.3390/molecules200917325
Nastasă C, Tiperciuc B, Duma M, Benedec D, Oniga O. New Hydrazones Bearing Thiazole Scaffold: Synthesis, Characterization, Antimicrobial, and Antioxidant Investigation. Molecules. 2015; 20(9):17325-17338. https://doi.org/10.3390/molecules200917325
Chicago/Turabian StyleNastasă, Cristina, Brîndușa Tiperciuc, Mihaela Duma, Daniela Benedec, and Ovidiu Oniga. 2015. "New Hydrazones Bearing Thiazole Scaffold: Synthesis, Characterization, Antimicrobial, and Antioxidant Investigation" Molecules 20, no. 9: 17325-17338. https://doi.org/10.3390/molecules200917325