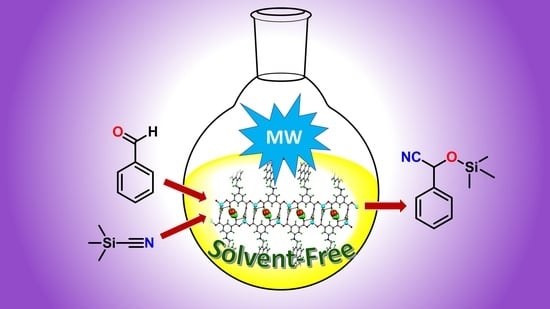
Pyrene Carboxylate Ligand Based Coordination Polymers for Microwave-Assisted Solvent-Free Cyanosilylation of Aldehydes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization
2.2. Thermogravimetric Analyses
2.3. Crystal Structure Analysis
2.4. Catalytic Activity Studies
3. Materials and Methods
3.1. Synthesis of 5-{(pyren-4-ylmethyl)amino}isophthalic Acid (H2L)
3.2. Synthesis of the Coordination Polymer [Zn(L)(H2O)2]n·n(H2O) (1)
3.3. Synthesis of the Coordination Polymer [Cd(L)(H2O)]n·n(H2O) (2)
3.4. Procedure for the Cyanosilylation Reaction of Aldehydes
3.5. Crystal Structure Determinations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Z.; Zaworotko, M.J. Template-directed synthesis of metal–organic materials. Chem. Soc. Rev. 2014, 43, 5444–5455. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, A.; Titi, H.M.; Goldberg, I. Coordination Polymers of 5-(2-Amino/Acetamido-4-carboxyphenoxy)-benzene-1,3-dioic Acids with Transition Metal Ions: Synthesis, Structure, and Catalytic Activity. Cryst. Growth Des. 2011, 11, 2621–2636. [Google Scholar] [CrossRef]
- Karmakar, A.; Martins, L.M.D.R.S.; Hazra, S.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. Metal–Organic Frameworks with Pyridyl-Based Isophthalic Acid and Their Catalytic Applications in Microwave Assisted Peroxidative Oxidation of Alcohols and Henry Reaction. Cryst. Growth Des. 2016, 16, 1837–1849. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Li, Z.; Garcia, H. Catalysis and photocatalysis by metal organic frameworks. Chem. Soc. Rev. 2018, 47, 8134–8172. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, A.; Pombeiro, A.J.L. Recent advances in amide functionalized metal organic frameworks for heterogeneous catalyt-ic applications. Coord. Chem. Rev. 2019, 395, 86–129. [Google Scholar] [CrossRef]
- Huang, Y.-B.; Liang, J.; Wang, X.-S.; Cao, R. Multifunctional metal–organic framework catalysts: Synergistic catalysis and tan-dem reactions. Chem. Soc. Rev. 2017, 46, 126–157. [Google Scholar] [CrossRef]
- Jin, F.-Z.; Zhao, C.-C.; Ma, H.-C.; Chen, G.-J.; Dong, Y.-B. Homochiral BINAPDA-Zr-MOF for Heterogeneous Asymmetric Cyanosilylation of Aldehydes. Inorg. Chem. 2019, 58, 9253–9259. [Google Scholar] [CrossRef]
- Karmakar, A.; Paul, A.; Mahmudov, K.T.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. pH dependent synthesis of Zn(ii) and Cd(ii) coordination polymers with dicarboxyl-functionalized arylhydrazone of barbituric acid: Photoluminescence properties and catalysts for Knoevenagel condensation. New J. Chem. 2016, 40, 1535–1546. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Metal–organic frameworks as heterogeneous catalysts for oxidation reactions. Catal. Sci. Technol. 2011, 1, 856–867. [Google Scholar] [CrossRef] [Green Version]
- Karmakar, A.; Oliver, C.L.; Roy, S.; Öhrström, L. The synthesis, structure, topology and catalytic application of a novel cub-ane-based copper(ii) metal–organic framework derived from a flexible amido tripodal acid. Dalton Trans. 2015, 44, 10156–10165. [Google Scholar] [CrossRef] [Green Version]
- Fan, Y.; Ren, Y.; Li, J.; Yu, C.; Jiang, H. Enhanced Activity and Enantioselectivity of Henry Reaction by the Postsynthetic Re-duction Modification for a Chiral Cu(salen)-Based Metal–Organic Framework. Inorg. Chem. 2018, 57, 11986–11994. [Google Scholar] [CrossRef]
- Jiang, N.; Mallat, T.; Krumeich, F.; Baiker, A. Copper-based metal-organic framework for the facile ring-opening of epoxides. J. Catal. 2008, 257, 390–395. [Google Scholar] [CrossRef]
- Karmakar, A.; Rúbio, G.M.D.M.; Guedes da Silva, M.F.C.; Ribeiro, A.P.C.; Pombeiro, A.J.L. ZnII and CdII MOFs based on an amidoisophthalic acid ligand: Synthesis, structure and catalytic application in transesterification. RSC Adv. 2016, 6, 89007–89018. [Google Scholar] [CrossRef]
- Karmakar, A.; Soliman, M.M.A.; Rúbio, G.M.D.M.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Synthesis and catalytic ac-tivities of a Zn(ii) based metallomacrocycle and a metal–organic framework towards one-pot deacetalization-Knoevenagel tandem reactions under different strategies: A comparative study. Dalton Trans. 2020, 49, 8075–8085. [Google Scholar] [CrossRef] [PubMed]
- Brunel, J.-M.; Holmes, I.P. Chemically Catalyzed Asymmetric Cyanohydrin Syntheses. Angew. Chem. Int. Ed. 2004, 43, 2752–2778. [Google Scholar] [CrossRef]
- Lacour, M.-A.; Rahier, N.J.; Taillefer, M. Mild and Efficient Trimethylsilylcyanation of Ketones Catalysed by PNP Chloride. Chem. A Eur. J. 2011, 17, 12276–12279. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, K.; Hao, M.; Liu, L.; Han, Z.-B.; Yuan, D. Two metal–organic frameworks based on pyridyl–tricarboxylate lig-ands as size-selective catalysts for solvent-free cyanosilylation reaction. CrystEngComm 2018, 20, 6070–6076. [Google Scholar] [CrossRef]
- Wu, X.; Lin, Z.; He, C.; Duan, C. Catalytic performance of lanthanide molecular solids containing well-modified metal–organic octahedra. New J. Chem. 2011, 36, 161–167. [Google Scholar] [CrossRef]
- Aguirre-Díaz, L.M.; Iglesias, M.; Snejko, N.; Gutiérrez-Puebla, E.; Monge, M.Á. Indium metal–organic frameworks as catalysts in solvent-free cyanosilylation reaction. CrystEngComm 2013, 15, 9562–9571. [Google Scholar] [CrossRef] [Green Version]
- Hu, T.-P.; Zhao, Y.-Q.; Mei, K.; Lin, S.-J.; Wang, X.-P.; Sun, D. A novel silver(i)-Keggin-polyoxometalate inorganic–organic hy-brid: A Lewis acid catalyst for cyanosilylation reaction. CrystEngComm 2015, 17, 5947–5952. [Google Scholar] [CrossRef]
- Kantam, M.L.; Mahendar, K.; Sreedhar, B.; Kumar, K.V.; Choudary, B.M. Cyanosilylation of Aldehydes and Ketones Cata-lyzed by Nanocrystalline Magnesium Oxide. Synth. Commun. 2008, 38, 3919–3936. [Google Scholar] [CrossRef]
- Karmakar, A.; Soliman, M.M.A.; Alegria, E.C.B.A.; Rúbio, G.M.D.M.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. A copper-amidocarboxylate based metal organic macrocycle and framework: Synthesis, structure and catalytic activities towards microwave assisted alcohol oxidation and Knoevenagel reactions. New J. Chem. 2019, 43, 9843–9854. [Google Scholar] [CrossRef]
- Karmakar, A.; Paul, A.; Rúbio, G.M.D.M.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. Zinc(II) and Copper(II) Metal-Organic Frameworks Constructed from a Terphenyl-4,4″-dicarboxylic Acid Derivative: Synthesis, Structure, and Catalytic Application in the Cyanosilylation of Aldehydes. Eur. J. Inorg. Chem. 2016, 2016, 5557–5567. [Google Scholar] [CrossRef]
- Yang, L.; Powell, D.R.; Houser, R.P. Structural variation in copper(i) complexes with pyridylmethylamide ligands: Structural analysis with a new four-coordinate geometry index, τ4. Dalton Trans. 2007, 955–964. [Google Scholar] [CrossRef]
- D’Vries, R.F.; Iglesias, M.; Snejko, N.; Gutiérrez-Puebla, E.; Monge, M.A. Lanthanide Metal–Organic Frameworks: Searching for Efficient Solvent-Free Catalysts. Inorg. Chem. 2012, 51, 11349–11355. [Google Scholar] [CrossRef] [PubMed]
- de Luis, R.F.; Karmele Urtiaga, M.; Mesa, J.L.; Larrea, E.S.; Iglesias, M.; Rojo, T.; Arriortua, M.I. Thermal Response, Cata-lytic Activity, and Color Change of the First Hybrid Vanadate Containing Bpe Guest Molecules. Inorg. Chem. 2013, 52, 2615–2626. [Google Scholar] [CrossRef]
- Larrea, E.S.; de Luis, R.F.; Orive, J.; Iglesias, M.; Arriortua, M.I. [NaCu(2,4-HPdc)(2,4-Pdc)] Mixed Metal–Organic Frame-work as a Heterogeneous Catalyst. Eur. J. Inorg. Chem. 2015, 28, 4699–4707. [Google Scholar] [CrossRef]
- Karmakar, A.; Hazra, S.; Rúbio, G.M.D.M.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. Packing polymorphism in 3-amino-2-pyrazinecarboxylate based tin(ii) complexes and their catalytic activity towards cyanosilylation of aldehydes. New J. Chem. 2018, 42, 17513–17523. [Google Scholar] [CrossRef]
- Karmakar, A.; Hazra, S.; Da Silva, M.F.C.G.; Pombeiro, A.J.L. Synthesis, structure and catalytic application of lead(ii) complexes in cyanosilylation reactions. Dalton Trans. 2014, 44, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Schlichte, K.; Kratzke, T.; Kaskel, S. Improved synthesis, thermal stability and catalytic properties of the metal-organic framework compound Cu3(BTC)2. Microporous Mesoporous Mater. 2004, 73, 81–88. [Google Scholar] [CrossRef]
- Choi, I.-H.; Kim, Y.; Lee, D.N.; Huh, S. Three-dimensional cobalt(II) and cadmium(II) MOFs containing 1,4-naphthalenedicarboxylate: Catalytic activity of Cd-MOF. Polyhedron 2016, 105, 96–103. [Google Scholar] [CrossRef]
- Phuengphai, P.; Youngme, S.; Chaichit, N.; Reedijk, J. New 3D supramolecular networks built from 1D and 2D frameworks via π–π and H-bonding interactions: Topology and catalytic properties. Inorg. Chim. Acta 2013, 403, 35–42. [Google Scholar] [CrossRef]
- Phuengphai, P.; Youngme, S.; Gamez, P.; Reedijk, J. Catalytic properties of a series of coordination networks: Cyanosilylation of aldehydes catalyzed by Zn(ii)-4,4′-bpy-carboxylato complexes. Dalton Trans. 2010, 39, 7936–7942. [Google Scholar] [CrossRef]
- Bruker APEX2; Bruker AXS Inc.: Madison, WI, USA, 2012.
- Sheldrick, G.M. SADABS. Program for Empirical Absorption Correction; University of Gottingen: Gottingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]










| Entry | Catalyst | Temperature (°C) | Catalyst Amount (mol%) | Solvent | Yield b (%) |
|---|---|---|---|---|---|
| 1 | 1 | 50 | 2.0 | Solvent free | 97 |
| 2 | 2 | 50 | 2.0 | Solvent free | 85 |
| 3 | 1 | 50 | 2.0 | MeOH | 78 |
| 4 | 1 | 50 | 2.0 | CHCl3 | 67 |
| 5 | 1 | 50 | 2.0 | CH3CN | 57 |
| 6 | 1 | 50 | 2.0 | THF | 81 |
| 7 | 2 | 50 | 2.0 | MeOH | 55 |
| 8 | 2 | 50 | 2.0 | CHCl3 | 43 |
| 9 | 2 | 50 | 2.0 | CH3CN | 41 |
| 10 | 2 | 50 | 2.0 | THF | 76 |
| 11 | 1 | 50 | 1.0 | Solvent free | 52 |
| 12 | 1 | 50 | 5.0 | Solvent free | 98 |
| 13 | 2 | 50 | 1.0 | Solvent free | 30 |
| 14 | 2 | 50 | 5.0 | Solvent free | 85 |
| 15 | 1 | 25 | 2.0 | Solvent free | 32 |
| 16 | 1 | 40 | 2.0 | Solvent free | 81 |
| 17 | 1 | 60 | 2.0 | Solvent free | 96 |
| 18 | 2 | 25 | 2.0 | Solvent free | 25 |
| 19 | 2 | 40 | 2.0 | Solvent free | 80 |
| 20 | 2 | 60 | 2.0 | Solvent free | 84 |
| 21 | Blank | 50 | - | Solvent free | 10 |
| 22 | H2L | 50 | 2.0 | Solvent free | 18 |
| 23 | Zn(NO3)3·6H2O | 50 | 2.0 | Solvent free | 28 |
| 24 | Cd(NO3)3·4H2O | 50 | 2.0 | Solvent free | 23 |
| Entry | Reactant | Yield b (%) with 1 | Yield b (%) with 2 |
|---|---|---|---|
| 1 | 4-Nitrobenzaldehyde | >99 | 96 |
| 2 | 3-Nitrobenzaldehyde | 98 | 95 |
| 3 | 4-Bromobenzaldehyde | >99 | 94 |
| 4 | 4-Chlorobenzaldehyde | 95 | 89 |
| 5 | 4-Hydroxybenzaldehyde | 91 | 75 |
| 6 | 3-Hydroxybenzaldehyde | 88 | 67 |
| 7 | 4-Methoxybenzaldehyde | 75 | 64 |
| 8 | 4-Methylbenzaldehyde | 71 | 58 |
| Entry | Catalyst | Solvent/Temp/Time | Aldehyde | Yield (%) | Reference |
|---|---|---|---|---|---|
| 1 | 1 | Solvent free/50 °C/1.5 h | Benzaldehyde | 97 | This work |
| 2 | [Pr(3,5-DSB)(Phen)]n | Solvent free/50 °C/3 h | Benzaldehyde | 78 | [23] |
| 3 | [{Co0.6Ni1.4(H2O)2(Bpe)2}(V4O12)]·4H2O·Bpe | Solvent free/50 °C/16 h | Benzaldehyde | 77 | [24] |
| 4 | [NaCu(2,4-HPdc)(2,4-Pdc)] | Solvent free/50 °C/24 h | Benzaldehyde | 85 | [25] |
| 5 | [Sn(L1)2] | Solvent free/50 °C/4 h | Benzaldehyde | 75 | [26] |
| 6 | 1 | THF/50 °C/1.5 h | Benzaldehyde | 81 | This work |
| 7 | [Pb(L1)2]2 | DCM/50 °C/6 h | Benzaldehyde | 97 | [27] |
| 8 | [Cu3(benzenetricarboxylate)2]n | pentane/40 °C/72 h | Benzaldehyde | 57 | [28] |
| 9 | [Cd2(1,4-NDC)2(DMF)2]n | Tolene/50 °C/72 h | 4-Nitrobenzaldehyde | 49 | [29] |
| 10 | {[Zn(dpe)(μ-OOCCH3)2](H2O)}n | DCM/RT/24 h | Benzaldehyde | 14 | [30] |
| 11 | {[Zn3(4,4′-bpy)4(μ-O2CCH2CH3)4](ClO4)2(4,4′-bpy)2(H2O)4}n | DCM/RT/24 h | Benzaldehyde | 22 | [31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karmakar, A.; Paul, A.; Sabatini, E.P.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Pyrene Carboxylate Ligand Based Coordination Polymers for Microwave-Assisted Solvent-Free Cyanosilylation of Aldehydes. Molecules 2021, 26, 1101. https://doi.org/10.3390/molecules26041101
Karmakar A, Paul A, Sabatini EP, Guedes da Silva MFC, Pombeiro AJL. Pyrene Carboxylate Ligand Based Coordination Polymers for Microwave-Assisted Solvent-Free Cyanosilylation of Aldehydes. Molecules. 2021; 26(4):1101. https://doi.org/10.3390/molecules26041101
Chicago/Turabian StyleKarmakar, Anirban, Anup Paul, Elia Pantanetti Sabatini, M. Fátima C. Guedes da Silva, and Armando J. L. Pombeiro. 2021. "Pyrene Carboxylate Ligand Based Coordination Polymers for Microwave-Assisted Solvent-Free Cyanosilylation of Aldehydes" Molecules 26, no. 4: 1101. https://doi.org/10.3390/molecules26041101