Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats
Abstract
1. Introduction
2. Results
2.1. Extraction and Phytochemical Analysis of Phenols
2.2. In Vitro Antioxidant Assay
2.3. Effects of the JSP Extract on the Body Weight, Water, and Feed Intake of the Rats
2.4. Biochemical Analysis
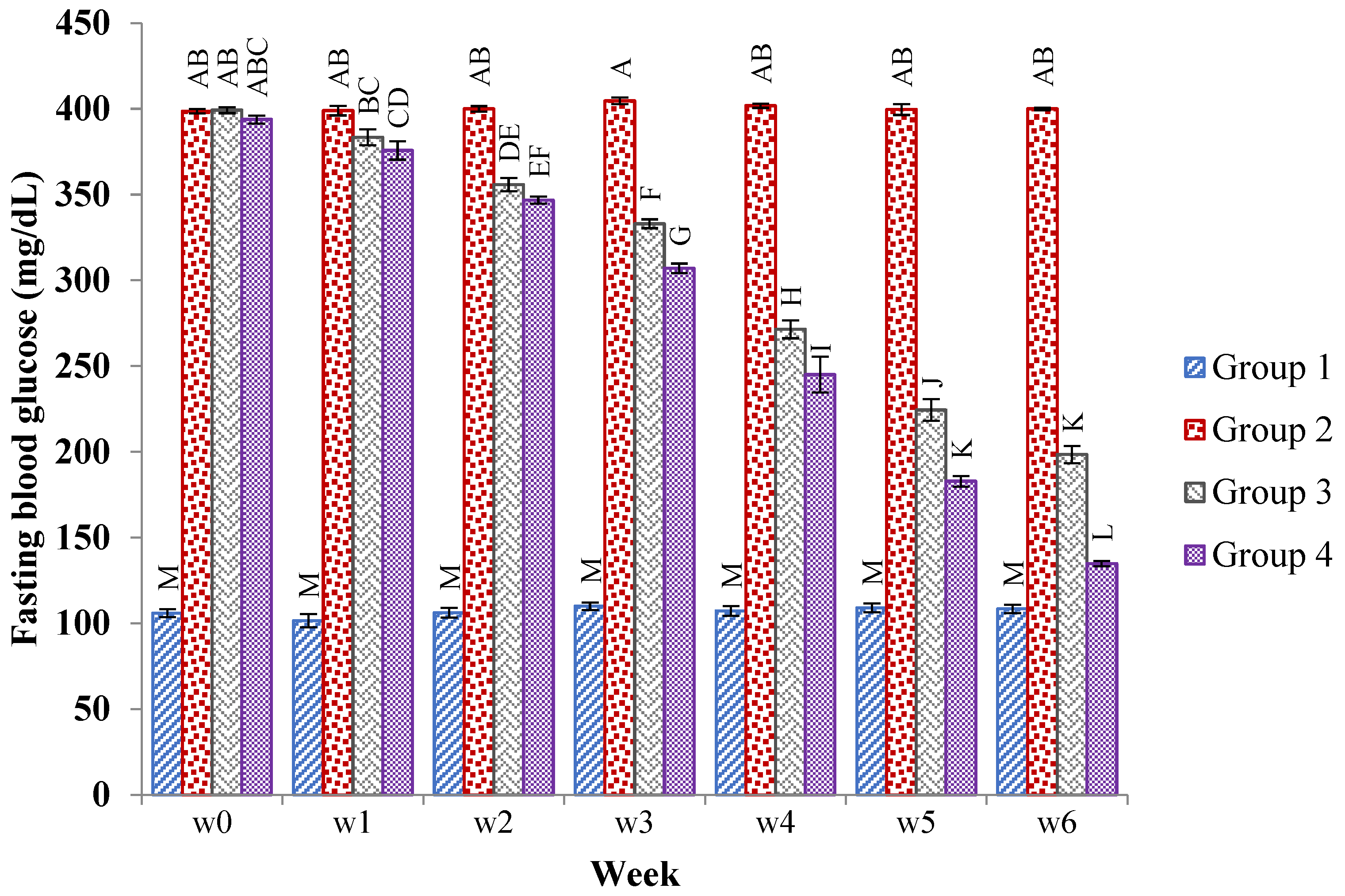
2.4.1. Effects of the JSP Extract on the Fasting Blood Glucose Level in the Rats
2.4.2. Effects of the JSP Extract on the Serum Glucose, Serum Insulin, and Glycosylated Hemoglobin Level of the Rats
2.4.3. Effects of the JSP Extract on Serum Lipid Profile and Liver Function Enzymes
2.4.4. Effects of JSP on Cardiac Oxidative Stress and Antioxidant Enzyme Level
2.4.5. Effects of the JSP Extract on Cardiac Function Biomarkers
2.4.6. Histopathological Examination
Pancreas
Heart
2.4.7. Gene Expression Analysis
3. Discussion
4. Methods
4.1. Extraction Procedure of Phenols
4.2. Qualitative Phytochemical Analysis of Phenols and Flavonoids
4.3. Quantitative Phytochemical Analysis of Phenols and Flavonoids
4.4. Quantification of the Phenols in the Jasminum sambac Extract
4.5. In Vitro Antioxidant Analysis of the JSP Extract
4.5.1. Total Antioxidant Capacity (TAC)
4.5.2. DPPH Assay
4.5.3. ABTS•+ Radical Scavenging Assay
4.6. Experiment Animals
4.7. Acute Oral Toxicity Test
4.8. Experimental Design
- -
- Group I: Untreated normal control group on a routine diet;
- -
- Group II: Untreated diabetic (positive control) given alloxan (150 mg/kg);
- -
- Group III: Diabetic group treated with the JSP extract (250 mg/kg) for six weeks;
- -
- Group IV: Diabetic group treated with the JSP extract (500 mg/kg) for six weeks.
4.9. Physical Parameters
4.9.1. Body Weight
4.9.2. Feed and Water Intake
4.10. Blood Sampling
4.11. Preparation of Tissue Homogenate
4.12. Biochemical Analysis
4.13. Oxidative Stress and Antioxidant Enzyme Measurements
4.14. Determination of Cardiac Biomarkers
4.15. Histopathological Examination
4.16. RNA Extraction and Real-Time Quantitative PCR
4.17. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Xiong, W.; Xiao, M.Y.; Zhang, M.; Chang, F. Efficacy and safety of canagliflozin in patients with type 2 diabetes: A meta-analysis of randomized controlled trials. Medicine 2016, 95, e5473. [Google Scholar] [CrossRef] [PubMed]
- Harding, J.L.; Pavkov, M.E.; Magliano, D.J.; Shaw, J.E.; Gregg, E.W. Global trends in diabetes complications: A review of current evidence. Diabetologia 2019, 62, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Murthy, V.L.; Naya, M.; Foster, C.R.; Gaber, M.; Hainer, J.; Klein, J.; Dorbala, S.; Blankstein, R.; Di Carli, M.F. Association between coronary vascular dysfunction and cardiac mortality in patients with and without diabetes mellitus. Circulation 2012, 126, 1858–1868. [Google Scholar] [CrossRef]
- Chen, J.; Mangelinckx, S.; Adams, A.; Wang, Z.-T.; Li, W.-L.; De Kimpe, N. Natural Flavonoids as Potential Herbal Medication for the Treatment of Diabetes Mellitus and its Complications. Nat. Prod. Commun. 2015, 10, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Chen, S.; Wang, Y.; Feng, J.; Wang, H.; Deng, Y. Complete chloroplast genome of Jasminum sambac L. (Oleaceae). Braz. J. Bot. 2020, 43, 855–867. [Google Scholar] [CrossRef]
- Jia, G.; Whaley-Connell, A.; Sowers, J.R. Diabetic cardiomyopathy: A hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 2017, 61, 21–28. [Google Scholar] [CrossRef]
- Khan, I.A.; Hussain, M.; Munawar, S.H.; Iqbal, M.O.; Arshad, S.; Manzoor, A.; Shah, M.A.; Abbas, K.; Shakeel, W.; Syed, S.K. Jasminum sambac: A Potential Candidate for Drug Development to Cure Cardiovascular Ailments. Molecules 2021, 26, 5664. [Google Scholar] [CrossRef]
- Matowa, P.R.; Gundidza, M.; Gwanzura, L.; Nhachi, C.F.B. A survey of ethnomedicinal plants used to treat cancer by traditional medicine practitioners in Zimbabwe. BMC Complement. Med. Ther. 2020, 20, 278. [Google Scholar] [CrossRef]
- Kang, G.G.; Francis, N.; Hill, R.; Waters, D.; Blanchard, C.; Santhakumar, A.B. Dietary Polyphenols and Gene Expression in Molecular Pathways Associated with Type 2 Diabetes Mellitus: A Review. Int. J. Mol. Sci. 2019, 21, 140. [Google Scholar] [CrossRef]
- Xu, X.; Shan, B.; Liao, C.-H.; Xie, J.-H.; Wen, P.-W.; Shi, J.-Y. Anti-diabetic properties of Momordica charantia L. polysaccharide in alloxan-induced diabetic mice. Int. J. Biol. Macromol. 2015, 81, 538–543. [Google Scholar] [CrossRef]
- Yang, C.S.; Zhang, J.; Zhang, L.; Huang, J.; Wang, Y. Mechanisms of body weight reduction and metabolic syndrome alleviation by tea. Mol. Nutr. Food Res. 2015, 60, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Rollins, K.E.; Varadhan, K.K.; Dhatariya, K.; Lobo, D.N. Systematic review of the impact of HbA1c on outcomes following surgery in patients with diabetes mellitus. Clin. Nutr. 2016, 35, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Jafarnejad, S.; Keshavarz, S.A.; Mahbubi, S.; Saremi, S.; Arab, A.; Abbasi, S.; Djafarian, K. Effect of ginger (Zingiber officinale) on blood glucose and lipid concentrations in diabetic and hyperlipidemic subjects: A meta-analysis of randomized controlled trials. J. Funct. Foods 2017, 29, 127–134. [Google Scholar] [CrossRef]
- Gad-Elkareem, M.A.; Abdelgadir, E.H.; Badawy, O.M.; Kadri, A. Potential antidiabetic effect of ethanolic and aqueous-ethanolic extracts of Ricinus communis leaves on streptozotocin-induced diabetes in rats. PeerJ 2019, 7, e6441. [Google Scholar] [CrossRef]
- Garg, P.; Morris, P.; Fazlanie, A.L.; Vijayan, S.; Dancso, B.; Dastidar, A.G.; Plein, S.; Mueller, C.; Haaf, P. Cardiac biomarkers of acute coronary syndrome: From history to high-sensitivity cardiac troponin. Intern. Emerg. Med. 2017, 12, 147–155. [Google Scholar] [CrossRef]
- Hijazi, Z.; Oldgren, J.; Wallentin, L.; Andersson, U.; Connolly, S.J.; Yusuf, S.; Ezekowitz, M.D.; Hohnloser, S.H.; Reilly, P.A.; Vinereanu, D.; et al. Response to letter regarding article, “Cardiac biomarkers are associated with an increased risk of stroke and death in patients with atrial fibrillation: A randomized evaluation of long-term anticoagulation therapy (RE-LY) substudy”. Circulation 2012, 125, 1605–1616. [Google Scholar] [CrossRef] [PubMed]
- Sandamali, J.A.N.; Hewawasam, R.P.; Jayatilaka, K.A.P.W.; Mudduwa, L.K.B. Cinnamomum zeylanicum Blume (Ceylon cinnamon) bark extract attenuates doxorubicin induced cardiotoxicity in Wistar rats. Saudi Pharm. J. 2021, 29, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhang, T.; Zhu, B.; Cao, Z. Diagnostic role of postmortem CK-MB in cardiac death: A systematic review and meta-analysis. Forensic Sci. Med. Pathol. 2020, 16, 287–294. [Google Scholar] [CrossRef]
- Moravej Aleali, A.; Amani, R.; Shahbazian, H.; Namjooyan, F.; Latifi, S.M.; Cheraghian, B. The effect of hydroalcoholic Saffron (Crocus sativus L.) extract on fasting plasma glucose, HbA1c, lipid profile, liver, and renal function tests in patients with type 2 diabetes mellitus: A randomized double-blind clinical trial. Phytother. Res. 2019, 33, 1648–1657. [Google Scholar] [CrossRef]
- Tan, Y.; Zhang, Z.; Zheng, C.; Wintergerst, K.A.; Keller, B.B.; Cai, L. Mechanisms of diabetic cardiomyopathy and potential therapeutic strategies: Preclinical and clinical evidence. Nat. Rev. Cardiol. 2020, 17, 585–607. [Google Scholar] [CrossRef]
- Ali, S.S.; Ahsan, H.; Zia, M.K.; Siddiqui, T.; Khan, F.H. Understanding oxidants and antioxidants: Classical team with new players. J. Food Biochem. 2020, 44, e13145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shi, E.; Yang, L.; Fu, W.; Hu, F.; Zhou, X. Gentiopicroside attenuates diabetic retinopathy by inhibiting inflammation, oxidative stress, and NF-κB activation in rat model. Eur. J. Inflamm. 2019, 17, 2058739219847837. [Google Scholar] [CrossRef]
- Hameister, R.; Kaur, C.; Dheen, S.T.; Lohmann, C.H.; Singh, G. Reactive oxygen/nitrogen species (ROS/RNS) and oxidative stress in arthroplasty. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 2073–2087. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Xu, L.; Zhang, J.; Mo, J.; Zhuang, P.; Zheng, L. Oxyresveratrol from mulberry branch extract protects HUVECs against oxidized Low-density Lipoprotein-induced oxidative injury via activation of the Nrf-2/HO-1 pathway. J. Funct. Foods 2023, 100, 105371. [Google Scholar] [CrossRef]
- Raish, M.; Ahmad, A.; Jardan, Y.A.B.; Shahid, M.; Alkharfy, K.M.; Ahad, A.; Ansari, M.A.; Abdelrahman, I.A.; Al-Jenoobi, F.I. Sinapic acid ameliorates cardiac dysfunction and cardiomyopathy by modulating NF-κB and Nrf2/HO-1 signaling pathways in streptozocin induced diabetic rats. Biomed. Pharmacother. 2022, 145, 112412. [Google Scholar] [CrossRef]
- Lind, M.; Hayes, A.; Caprnda, M.; Petrovic, D.; Rodrigo, L.; Kruzliak, P.; Zulli, A. Inducible nitric oxide synthase: Good or bad? Biomed. Pharmacother. 2017, 93, 370–375. [Google Scholar] [CrossRef]
- Kitakata, H.; Endo, J.; Hashimoto, S.; Mizuno, E.; Moriyama, H.; Shirakawa, K.; Goto, S.; Katsumata, Y.; Fukuda, K.; Sano, M. Imeglimin prevents heart failure with preserved ejection fraction by recovering the impaired unfolded protein response in mice subjected to cardiometabolic stress. Biochem. Biophys. Res. Commun. 2021, 572, 185–190. [Google Scholar] [CrossRef]
- Wang, X.; Tan, Y.; Xu, B.; Lu, L.; Zhao, M.; Ma, J.; Liang, H.; Liu, J.; Yu, S. GPR30 Attenuates Myocardial Fibrosis in Diabetic Ovariectomized Female Rats: Role of iNOS Signaling. DNA Cell Biol. 2018, 37, 821–830. [Google Scholar] [CrossRef]
- Moradipour, A.; Dariushnejad, H.; Ahmadizadeh, C.; Lashgarian, H.E. Dietary flavonoid carvacrol triggers the apoptosis of human breast cancer MCF-7 cells via the p53/Bax/Bcl-2 axis. Med. Oncol. 2022, 40, 46. [Google Scholar] [CrossRef]
- Zheng, Q.; Wang, H.; Hou, W.; Zhang, Y. Use of Anti-angiogenic Drugs Potentially Associated with an Increase on Serum AST, LDH, CK, and CK-MB Activities in Patients with Cancer: A Retrospective Study. Front. Cardiovasc. Med. 2021, 8, 755191. [Google Scholar] [CrossRef]
- Okagu, I.U.; Ndefo, J.C.; Aham, E.C.; Udenigwe, C.C. Zanthoxylum Species: A comprehensive review of traditional uses, phytochemistry, pharmacological and nutraceutical applications. Molecules 2021, 26, 4023. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, A.; Aslam, B.; Iftikhar, M.; Majeed, W.; Batool, M.; Zahoor, B.; Amna, N.; Gohar, H.; Latif, I. Effect of Caesalpinia bonduc Polyphenol Extract on Alloxan-Induced Diabetic Rats in Attenuating Hyperglycemia by Upregulating Insulin Secretion and Inhibiting JNK Signaling Pathway. Oxidative Med. Cell. Longev. 2020, 2020, 9020219. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Prakash, V.; Saxena, S.; Gupta, S.; Saxena, A.; Yadav, R.; Singh, S. Preliminary phytochemical screening and biological activities of Adina cardifolia. J. Microb. Biochem. Technol. 2015, 7, 33–38. [Google Scholar]
- Ganzera, M.; Greifeneder, V.; Schwaiger, S.; Stuppner, H. Chemical profiling of Edelweiss (Leontopodium alpinum Cass.) extracts by micellar electrokinetic capillary chromatography. Fitoterapia 2012, 83, 1680–1686. [Google Scholar] [CrossRef] [PubMed]
- Olegário, L.S.; Andrade, J.K.S.; Andrade, G.R.S.; Denadai, M.; Cavalcanti, R.L.; da Silva, M.A.A.P.; Narain, N. Chemical characterization of four Brazilian brown propolis: An insight in tracking of its geographical location of production and quality control. Food Res. Int. 2019, 123, 481–502. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric Quantitation of Antioxidant Capacity through the Formation of a Phosphomolybdenum Complex: Specific Application to the Determination of Vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; El-Hefnawy, H.M.; Osman, S.M.; El-Raey, M.A.; Mokhtar Ali, F.A. Phenolic profiling of different Jasminum species cultivated in Egypt and their antioxidant activity. Nat. Prod. Res. 2021, 35, 4663–4668. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- No, O.T. 423: Acute oral toxicity-acute toxic class method. In OECD Guidelines for the Testing of Chemicals, Section 4; OECD Publishing: Paris, France, 2002; Volume 4, p. 14. [Google Scholar] [CrossRef]
- Alelign, T.; Chalchisa, D.; Fekadu, N.; Solomon, D.; Sisay, T.; Debella, A.; Petros, B. Evaluation of acute and sub-acute toxicity of selected traditional antiurolithiatic medicinal plant extracts in Wistar albino rats. Toxicol. Rep. 2020, 7, 1356–1365. [Google Scholar] [CrossRef]
- Barry, A.J.; Groseclose, M.R.; Castellino, S. Quantification and assessment of detection capability in imaging mass spectrometry using a revised mimetic tissue model. Bioanalysis 2019, 11, 1099–1116. [Google Scholar] [CrossRef] [PubMed]
- De Leon, J.A.D.; Borges, C.R. Evaluation of oxidative stress in biological samples using the thiobarbituric acid reactive substances assay. JoVE 2020, 159, e61122. [Google Scholar]
- Adeyi, O.A.; Idowu, A.B.; Mafiana, C.F.; Oluwalana, A.S.; Ajayi, O.L.; Akinloye, A.O. Rat model of food-induced non-obese-type 2 diabetes mellitus: Comparative pathophysiology and histopathology. Int. J. Physiol. Pathophysiol. Pharmacol. 2012, 4, 51–58. [Google Scholar] [PubMed]
- Siddique, T.; Awan, F.R. Effects of Reg3 Delta Bioactive Peptide on Blood Glucose Levels and Pancreatic Gene Expression in an Alloxan-Induced Mouse Model of Diabetes. Can. J. Diabetes 2016, 40, 198–203. [Google Scholar] [CrossRef]
- Abdel-Wahab, N.; Tayar, J.H.; Fa’Ak, F.; Sharma, G.; Lopez-Olivo, M.A.; Yousif, A.; Shagroni, T.; Al-Hawamdeh, S.; Rojas-Hernandez, C.M.; Suarez-Almazor, M.E. Systematic review of observational studies reporting antiphospholipid antibodies in patients with solid tumors. Blood Adv. 2020, 4, 1746–1755. [Google Scholar] [CrossRef]
- Ritchie, R.H.; Love, J.E.; Huynh, K.; Bernardo, B.C.; Henstridge, D.C.; Kiriazis, H.; Tham, Y.K.; Sapra, G.; Qin, C.; Cemerlang, N.; et al. Enhanced phosphoinositide 3-kinase(p110α) activity prevents diabetes-induced cardiomyopathy and superoxide generation in a mouse model of diabetes. Diabetologia 2012, 55, 3369–3381. [Google Scholar] [CrossRef]






| Sample | Total Phenolic Content (mg GAE/g) | Total Flavonoid Content (mg CE/g) |
|---|---|---|
| JSP extract | 129.6 ± 3.0 | 63.026 ± 3.0 |
| No. Sample | Name of Phenolics | Quantity (μg/g) |
|---|---|---|
| 1 | Rosmarinic acid | 788.7 ± 1.42 |
| 2 | p-coumaric acid | 410.2 ± 5.04 |
| 3 | p-hydroxybenzoic acid | 188.1 ± 2.36 |
| 4 | Syringic acids | 72.3 ± 3.53 |
| 5 | Chlorogenic acid | 30.7 ± 2.14 |
| Parameters | Normal Control | Diabetic Control | 250 mg/kg of JSP Extract | 500 mg/kg of JSP Extract |
|---|---|---|---|---|
| Body weight (g) | 164.94 ± 1.37 A | 104.29 ± 3.65 B * | 125.51 ± 2.55 D # | 136.31 ± 3.57 C ## |
| Feed intake (g) | 27.17 ± 0.29 A | 20.17 ± 0.57 BC * | 22.63 ± 0.48 C # | 25.03 ± 0.67 B ## |
| Water intake (mL) | 45.43 ± 0.59 C | 66.17 ± 1.53 A * | 57.09 ± 1.29 B # | 49.89 ± 1.79 BC ## |
| Parameters | Normal Control | Diabetic Control | 250 mg/kg of JSP Extract | 500 mg/kg of JSP Extract |
|---|---|---|---|---|
| Serum glucose (mL) | 104.60 ± 1.18 D | 354.07 ± 9.83 A * | 321.53 ± 19.52 B # | 283.47 ± 29.47 C ## |
| Serum insulin (mg/dL) | 12.22 ± 0.28 A | 6.49 ± 0.22 D * | 8.25 ± 0.50 C # | 9.51 ± 0.58 B ## |
| Glycosylated hemoglobin (mmol/mol) | 5.75 ± 0.13 C | 12.82 ± 0.14 A * | 11.17 ± 0.48 AB # | 10.22 ± 0.70 B # |
| Parameters | Normal Control | Diabetic Control | 250 mg/kg of JSP Extract | 500 mg/kg of JSP Extract |
|---|---|---|---|---|
| Serum cholesterol (mg/dL) | 144.27 ± 2.15 D | 252.27 ± 7.92 A * | 223.60 ± 4.03 B # | 203.40 ± 8.30 C ## |
| Serum triglycerides (mg/dL) | 99.07 ± 1.70 D | 176.73 ± 3.19 A * | 149.47 ± 2.64 B # | 130.47 ± 6.21 C ## |
| Serum HDL (mg/dL) | 44.27 ± 1.07 A | 30.40 ± 0.68 D * | 34.67 ± 1.47 C # | 37.80 ± 1.95 B ## |
| Serum LDL (mg/dL) | 118.13 ± 2.27 D | 177.60 ± 2.45 A * | 155.87 ± 3.40 B # | 146.67 ± 5.05 C ## |
| Serum ALT (U/L) | 17.80 ± 0.86 C | 42.67 ± 1.37 A * | 35.67 ± 1.82 B # | 34.67 ± 3.14 B # |
| Serum AST (U/L) | 21.15 ± 0.63 C | 38.40 ± 3.45 A * | 36.53 ± 2.31 A NS | 32.60 ± 2.59 B # |
| Parameters | Normal Control | Diabetic Control | 250 mg/kg of JSP Extract | 500 mg/kg of JSP Extract |
|---|---|---|---|---|
| TBRAS (μM/mg protein) | 4.27 ± 0.15 C | 10.54 ± 2.1 A * | 6.21 ± 1.03 BC # | 7.40 ± 1.40 B # |
| GPx (units/mg protein) | 148 ± 4.35 A | 115.4 ± 3.6 D * | 123.5 ± 9.19 C # | 141.34 ± 3.11 B ## |
| SOD (units/gram tissue) | 9.68 ± 1.05 C | 15.40 ± 1.42 A * | 12.42 ± 1.47 B # | 14.64 ± 1.95 AB # |
| Catalase (μmol H2O2/min/mg protein) | 53 ± 2.03 A | 23.5 ± 1.48 C * | 34 ± 1.04 BC # | 43.5 ± 1.54 B ## |
| Names | Forward: | Reverse |
|---|---|---|
| Nrf2 | 5′TTTGTAGATGACCATGAGTCGC-3′ | 5′TGTCCTGCTGTATGCTGCTT-3′ |
| Bax | 5′CCTGAGCTGACCTTGGAGCA-3′ | 5′GGTGGTTGCCCTTTTCTACT-3′ |
| Bcl-2 | 5′TGATAACCGGGAGATCGTGA | 5′AAAGCACATCCAATAAAAAGC |
| caspase-3 | 5′CTCGGTCTGGTACAGATGTCGATG | 5′GGTTAACCCGGGTAAGAATGTGCA |
| Beta-actin | CGAGTACAACCTTCTTGCAGC | TATCGTCATCCATGGCGAACTG |
| HO-1 | 5′TCTGCAGGGGAGAATCTTGC | 5′TTGGTGACGGAAATGTGCCA |
| iNOS | 5′-CATTCAGATCCCGAAACGTAC-3′ | 5′-AGCCTCATGGTGAACACGTTCT-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umar, U.; Ahmed, S.; Iftikhar, A.; Iftikhar, M.; Majeed, W.; Liaqat, A.; Shahzad, S.; Abbas, M.; Mehmood, T.; Anwar, F. Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats. Molecules 2023, 28, 5453. https://doi.org/10.3390/molecules28145453
Umar U, Ahmed S, Iftikhar A, Iftikhar M, Majeed W, Liaqat A, Shahzad S, Abbas M, Mehmood T, Anwar F. Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats. Molecules. 2023; 28(14):5453. https://doi.org/10.3390/molecules28145453
Chicago/Turabian StyleUmar, Urooj, Sibtain Ahmed, Asra Iftikhar, Maryam Iftikhar, Wafa Majeed, Atika Liaqat, Sana Shahzad, Mateen Abbas, Tahir Mehmood, and Farooq Anwar. 2023. "Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats" Molecules 28, no. 14: 5453. https://doi.org/10.3390/molecules28145453
APA StyleUmar, U., Ahmed, S., Iftikhar, A., Iftikhar, M., Majeed, W., Liaqat, A., Shahzad, S., Abbas, M., Mehmood, T., & Anwar, F. (2023). Phenolics Extracted from Jasminum sambac Mitigates Diabetic Cardiomyopathy by Modulating Oxidative Stress, Apoptotic Mediators and the Nfr-2/HO-1 Pathway in Alloxan-Induced Diabetic Rats. Molecules, 28(14), 5453. https://doi.org/10.3390/molecules28145453







