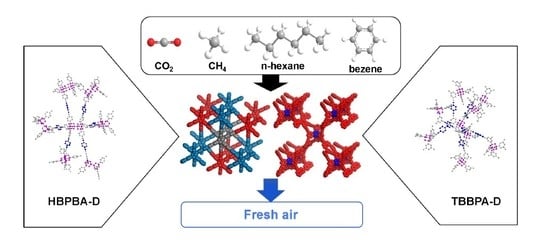
Adamantane-Based Micro- and Ultra-Microporous Frameworks for Efficient Small Gas and Toxic Organic Vapor Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Measurements
2.3. Synthesis of HBPBA-D and TBBPA-D
3. Results and Discussion
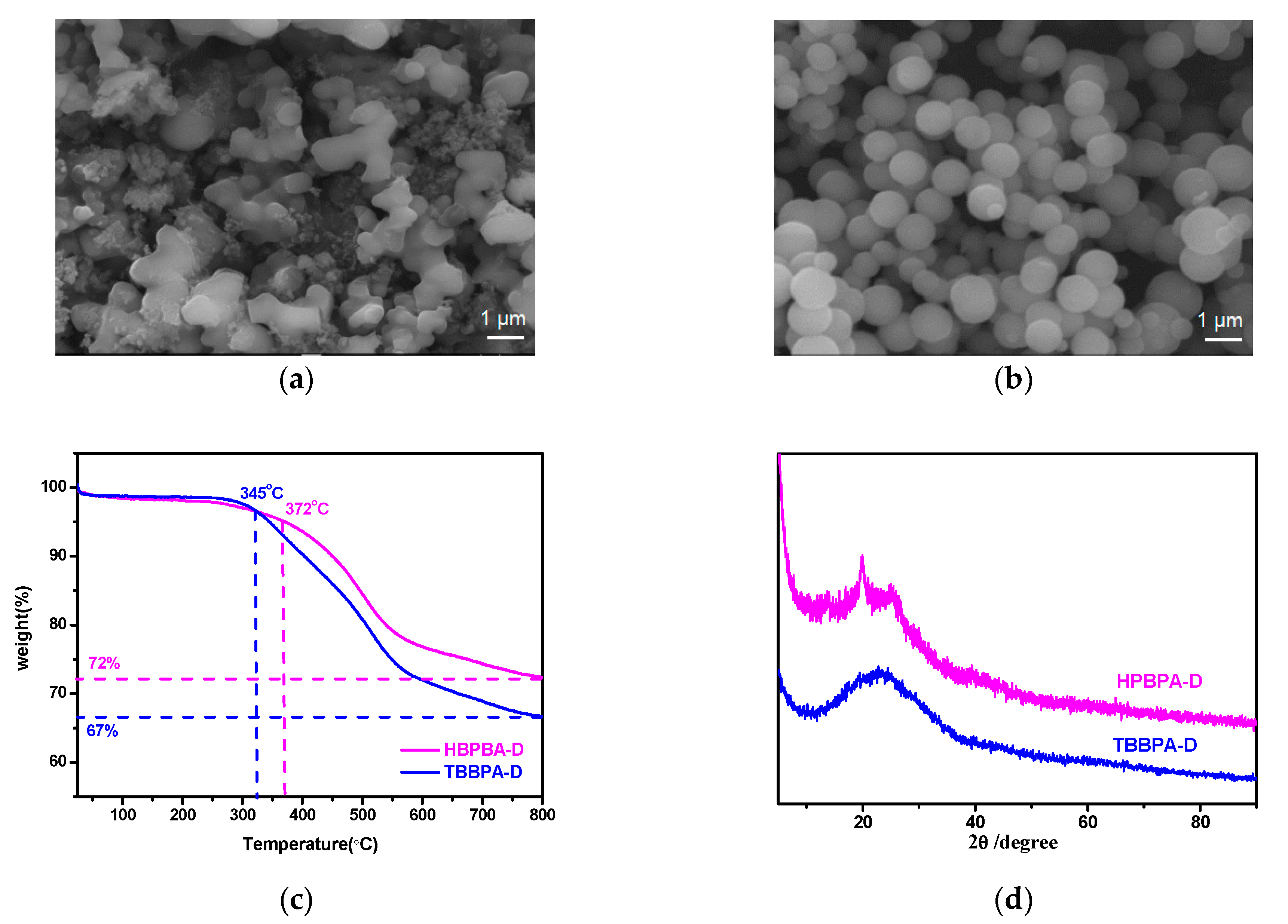
3.1. Synthesis and Characterization of HBPBA-D and TBBPA-D
3.2. Porosity of HBPBA-D and TBBPA-D
3.3. Small Gas and Toxic Organic Vapor Adsorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jiang, J.X.; Cooper, A.I. Microporous organic polymers: Design, synthesis, and function. Top. Curr. Chem. 2009, 293, 1–33. [Google Scholar] [CrossRef]
- Huang, N.; Day, G.; Yang, X.; Drake, H.; Zhou, H.C. Engineering porous organic polymers for carbon dioxide capture. Sci. China Chem. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- Zhang, G.; Lan, Z.A.; Wang, X. Conjugated polymers: Catalysts for photocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 2016, 55, 15712–15727. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Huang, S.; Chen, L.; Li, Y.; Yang, X.; Yuan, Z. Applications of hierarchically structured porous materials from energy storage and conversion, catalysis, photocatalysis, adsorption, separation, and sensing to biomedicine. Cheminform 2016, 45, 3479–3563. [Google Scholar] [CrossRef] [PubMed]
- Vyas, V.S.; Vishwakarma, M.; Moudrakovski, I.; Haase, F.; Savasci, G.; Ochsenfeld, C. Exploiting noncovalent interactions in an imine-based covalent organic framework for quercetin delivery. Adv. Mater. 2016, 28, 8749–8754. [Google Scholar] [CrossRef] [PubMed]
- Diercks, C.S.; Yaghi, O.M. The atom, the molecule, and the covalent organic framework. Science 2017, 355, eaal1585. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.X.; Tan, B. Hypercrosslinked porous polymer materials: Design, synthesis, and applications. Chem. Soc. Rev. 2017, 46, 3322–3356. [Google Scholar] [CrossRef] [PubMed]
- Mckeown, N.B.; Budd, P.M. Exploitation of intrinsic microporosity in polymer-based materials. Macromolecules 2010, 43, 5163–5176. [Google Scholar] [CrossRef]
- Xu, Y.; Jin, S.; Xu, H.; Nagai, A.; Jiang, D. Conjugated microporous polymers: Design, synthesis and application. Chem. Soc. Rev. 2013, 42, 8012–8031. [Google Scholar] [CrossRef]
- Dey, S.; Bhunia, A.; Esquivelb, D.; Janiak, C.J. Covalent triazine-based frameworks (CTFs) from triptycene and fluorene motif for CO2 adsorption. Mater. Chem. A 2016, 4, 6259–6263. [Google Scholar] [CrossRef]
- Furukawa, H.; Yaghi, O.M. Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications. J. Am. Chem. Soc. 2009, 131, 8875–8883. [Google Scholar] [CrossRef]
- Zhang, B.; Li, G.Y.; Yan, J.; Wang, Z.G. Tetraphenyladamantane-based microporous Polybenzimidazoles for adsorption of carbon dioxide, hydrogen, and organic vapors. J. Phys. Chem. C 2015, 119, 13080–13087. [Google Scholar] [CrossRef]
- Dawson, R.; Stöckel, E.; Holst, J.R.; Adams, D.J.; Cooper, A.I. Microporous organic polymers for carbon dioxide capture. Energy Environ. Sci. 2011, 4, 4239–4245. [Google Scholar] [CrossRef]
- Pandey, P.; Farha, O.K.; Spokoyny, A.M.; Mirkin, C.A.; Kanatzidis, M.G.; Hupp, J.T.; Nguyen, S.T. A “click-based” porous organic polymer from tetrahedral building blocks. J. Mater. Chem. 2011, 21, 1700–1703. [Google Scholar] [CrossRef]
- Totten, R.K.; Olenick, L.L.; Kim, Y.S.; Chakraborty, S.; Weston, M.H.; Farha, O.K.; Hupp, J.T.; Nguyen, S.T. A dual approach to tuning the porosity of porous organic polymers: Controlling the porogen size and supercritical CO2 processing. Chem. Sci. 2014, 5, 782–787. [Google Scholar] [CrossRef]
- Wang, D.; Yang, W.; Li, L.; Zhao, X.; Fend, S.; Liu, H. Hybrid networks constructed from tetrahedral silicon-centered precursors and cubic POSS-based building blocks via Heck reaction: Porosity, gas sorption, and luminescence. J. Mater. Chem. 2013, 1, 13549–13558. [Google Scholar] [CrossRef]
- Lim, H.; Chang, J.Y. Preparation of clickable microporous hydrocarbon particles based on adamantane. Macromolecules 2010, 43, 6943–6945. [Google Scholar] [CrossRef]
- Lim, H.; Cha, M.C.; Chang, J.Y. Synthesis of microporous polymers by Friedel-Crafts reaction of 1-bromoadamantane with aromatic compounds and their surface modification. Polym. Chem. 2012, 3, 868–870. [Google Scholar] [CrossRef]
- Yuan, D.; Lu, W.; Zhao, D.; Zhou, H.C.; Zhou, H.C. Highly stable porous polymer networks with exceptionally high gas-uptake capacities. Adv. Mater. 2011, 23, 3723–3725. [Google Scholar] [CrossRef]
- Li, X.; Guo, J.W.; Yue, H.B.; Wang, J.W.; Topham, P.D. Synthesis of thermochemically stable tetraphenyladamantane-based microporous polymers as gas storage materials. RSC Adv. 2017, 7, 16174–16180. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.W.; Lai, X.F.; Fu, S.Q.; Yue, H.B.; Wang, J.W.; Topham, P.D. Microporous organic polymers based on hexaphenylbiadamantane: Synthesis, ultra-high stability and gas capture. Mater. Lett. 2017, 187, 76–79. [Google Scholar] [CrossRef] [Green Version]
- Yue, H.B.; Guo, J.W.; Fu, S.Q.; Li, X.; Wen, W.Q.; Jiang, W.Z.; Tong, R. Structural design, preparation and characterization of light, isotropic and robust statically determined organic frameworks as reusable adsorbents. Chem. Eng. J. 2017, 335, 887–895. [Google Scholar] [CrossRef]
- Mahmood, J.; Kim, S.J.; Noh, H.J.; Jung, S.M.; Ahmad, I.; Le, F.; Seo, J.M.; Baek, J.B. A robust 3D cage-like ultramicroporous network structure with high gas-uptake capacity. Angew. Chem. Int. Ed. 2018, 57, 3415–3420. [Google Scholar] [CrossRef]
- Jun, Y.; Zhang, B.; Wang, Z.G. Monodispersed ultramicroporous semi-cycloaliphatic polyimide for CO2, H2, and organic vapors. Polym. Chem. 2017, 7, 7295–7303. [Google Scholar] [CrossRef]
- Marissen, R. Strong materials—A structural view of material strength down to a molecular scale: Concept of a novel strong and extremely light weight molecular space frame-polymer. Mater. Werkst. 2006, 37, 704–709. [Google Scholar] [CrossRef]
- Shi, Q.; Sun, H.; Yang, R.; Zhu, Z.Q.; Liang, W.D.; Tan, D.Z.; Yang, B.P.; Li, A.; Deng, W.Q. Synthesis of conjugated microporous polymers for gas storage and selective adsorption. J. Mater. Sci. 2015, 50, 6388–6394. [Google Scholar] [CrossRef]
- Chen, D.Y.; Fu, Y.; Yu, W.G.; Yu, G.P.; Pan, C.Y. Versatile adamantane-based porous polymers with enhanced microporosity for efficient CO2 capture and iodine removal. Chem. Eng. J. 2017, 334, 900–906. [Google Scholar] [CrossRef]
- Martin, R.L.; Shahrak, M.N.; Swisher, J.A.; Simon, C.M.; Sculley, J.P.; Zhou, H.C. Modeling methane adsorption in interpenetrating porous polymer networks. J. Phys. Chem. C 2013, 117, 20037–20042. [Google Scholar] [CrossRef]
- Teng, B.; Li, Y.; Zhu, L.; Zhang, D.; Cao, D.; Xiang, Z. Selective adsorption of carbon dioxide by carbonized porous aromatic framework (PAF). Energ. Environ. Sci. 2012, 5, 8370–8376. [Google Scholar] [CrossRef]
- Zhang, C.; Zhu, P.; Tan, L.; Luo, L.; Liu, Y.; Liu, J. Synthesis and properties of organic microporous polymers from the monomer of hexaphenylbenzene based triptycene. Polymer 2016, 82, 100–104. [Google Scholar] [CrossRef]
- Modak, A.; Maegawa, Y.; Goto, Y.; Inagaki, S. Synthesis of 9,9′-spirobifluorene-based conjugated microporous polymers by FeCl3-mediated polymerization. Polym. Chem. 2016, 7, 1290–1296. [Google Scholar] [CrossRef]
- Ma, Q.Y.; Yang, B.X.; Li, J.Q. Porous organic polymers derived from tetrahedral silicon-centered monomers and a stereocontorted spirobifluorene-based precursor: Synthesis, porosity and carbon dioxide sorption. RSC Adv. 2015, 5, 64163–64169. [Google Scholar] [CrossRef]
- Zhu, Y.; Long, H.; Zhang, W. Imine-Linked porous polymer frameworks with high small gas (H2, CO2, CH4, C2H2) uptake and CO2/N2 selectivity. Chem. Mater. 2013, 25, 1630–1635. [Google Scholar] [CrossRef]
- Shen, C.J.; Yan, J.; Deng, G.Y.; Zhang, B.; Wang, Z.G. Synthetic modulation of micro- and mesopores in polycyanurate networks for adsorptions of gases and organic hydrocarbons. Polym. Chem. 2017, 8, 1074–1083. [Google Scholar] [CrossRef]
- Yu, M.; Wang, X.Y.; Yang, X.; Zhao, Y.; Jiang, J.X. Conjugated microporous copolymer networks with enhanced gas adsorption. Polym. Chem. 2015, 6, 3217–3223. [Google Scholar] [CrossRef]
- Zhang, H.J.; Zhang, C.; Wang, X.C. Microporous organic polymers based on tetraethynyl building blocks with N-functionalized pore surfaces: Synthesis, porosity and carbon dioxide sorption. RSC Adv. 2016, 6, 113826–113833. [Google Scholar] [CrossRef]
- Yang, X.; Yao, S.W.; Yu, M.; Jiang, J.X. Synthesis and gas adsorption properties of tetra-armed microporous organic polymer networks based on triphenylamine. Macromol. Rapid. Commun. 2014, 35, 834–839. [Google Scholar] [CrossRef]
- Yang, S.J.; Ding, X.; Han, B.H. Conjugated microporous polymers with extended π-structures for organic vapor adsorption. Macromolecules 2018, 51, 947–953. [Google Scholar] [CrossRef]
- Li, G.Y.; Zhang, B.; Yan, J.; Wang, Z.G. Tetraphenyladamantane-based polyaminals for highly efficient captures of CO2 and organic vapors. Macromolecules 2014, 47, 6664–6670. [Google Scholar] [CrossRef]
- Li, G.Y.; Zhang, B.; Yan, J.; Wang, Z.G. Micro- and mesoporous poly(schiff-base)s constructed from different building blocks and their adsorption behaviors towards organic vapors and CO2 gas. J. Mater. Chem. A 2014, 2, 18881–18888. [Google Scholar] [CrossRef]



| HBPBA-D | TBBPA-D | |
|---|---|---|
| SBET 1 (m2 g−1) | 488 | 395 |
| Smicro 2 (m2 g−1) | 243.2 | 298 |
| Vtotal 3 (cm3 g−1) | 0.29 | 0.29 |
| VMicro 4 (cm3 g−1) | 0.11 | 0.13 |
| VMicro/Vtotal | 0.38 | 0.45 |
| D 5 (nm) | 0.92–1.1 | 0.42 |
| D’ 6 (nm) | --- | 0.59 |
| Gas/Organic Vapor | Molecular Size | Uptake Capacities | |
|---|---|---|---|
| HBPBA-D Pore Size: 0.92–1.1 nm | TBBPA-D Pore Size: 0.42–0.59 nm | ||
| CO2 1 | 0.33 nm | 9 wt % | 8.92 wt % |
| CH4 1 | 0.38 nm | 1.63 wt % | 1.43 wt % |
| n-hexane 2 | 0.72 nm | 172.5 mg g−1 | 104.8 mg g−1 |
| Benzene 2 | 0.65–0.68 nm | 272.3 mg g−1 | 144.3 mg g−1 |
| Selectivity 3 (CO2/CH4) | --- | 4.1 | 4.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, W.; Yue, H.; Shuttleworth, P.S.; Xie, P.; Li, S.; Guo, J. Adamantane-Based Micro- and Ultra-Microporous Frameworks for Efficient Small Gas and Toxic Organic Vapor Adsorption. Polymers 2019, 11, 486. https://doi.org/10.3390/polym11030486
Jiang W, Yue H, Shuttleworth PS, Xie P, Li S, Guo J. Adamantane-Based Micro- and Ultra-Microporous Frameworks for Efficient Small Gas and Toxic Organic Vapor Adsorption. Polymers. 2019; 11(3):486. https://doi.org/10.3390/polym11030486
Chicago/Turabian StyleJiang, Wenzhao, Hangbo Yue, Peter S. Shuttleworth, Pengbo Xie, Shanji Li, and Jianwei Guo. 2019. "Adamantane-Based Micro- and Ultra-Microporous Frameworks for Efficient Small Gas and Toxic Organic Vapor Adsorption" Polymers 11, no. 3: 486. https://doi.org/10.3390/polym11030486