Evaluation of the Virulence of Low Pathogenic H9N2 Avian Influenza Virus Strains in Broiler Chickens
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Clinical Signs and Mortality
2.3. Sample Collection
2.4. Histopathological Staining
2.5. Immunohistochemical Staining
2.6. Real-Time PCR
2.7. Statistical Analysis
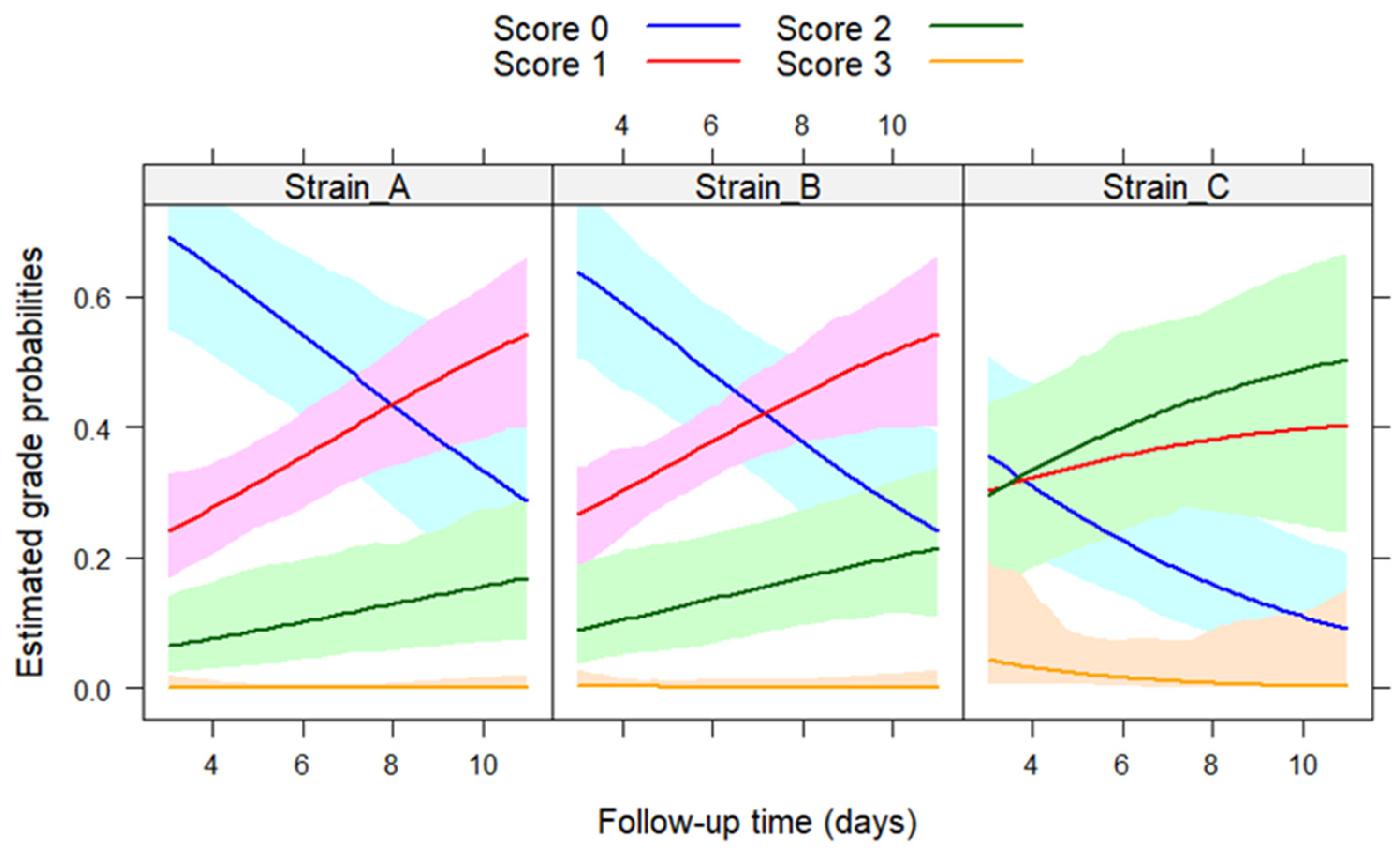
3. Results
3.1. Clinical Signs and Mortality
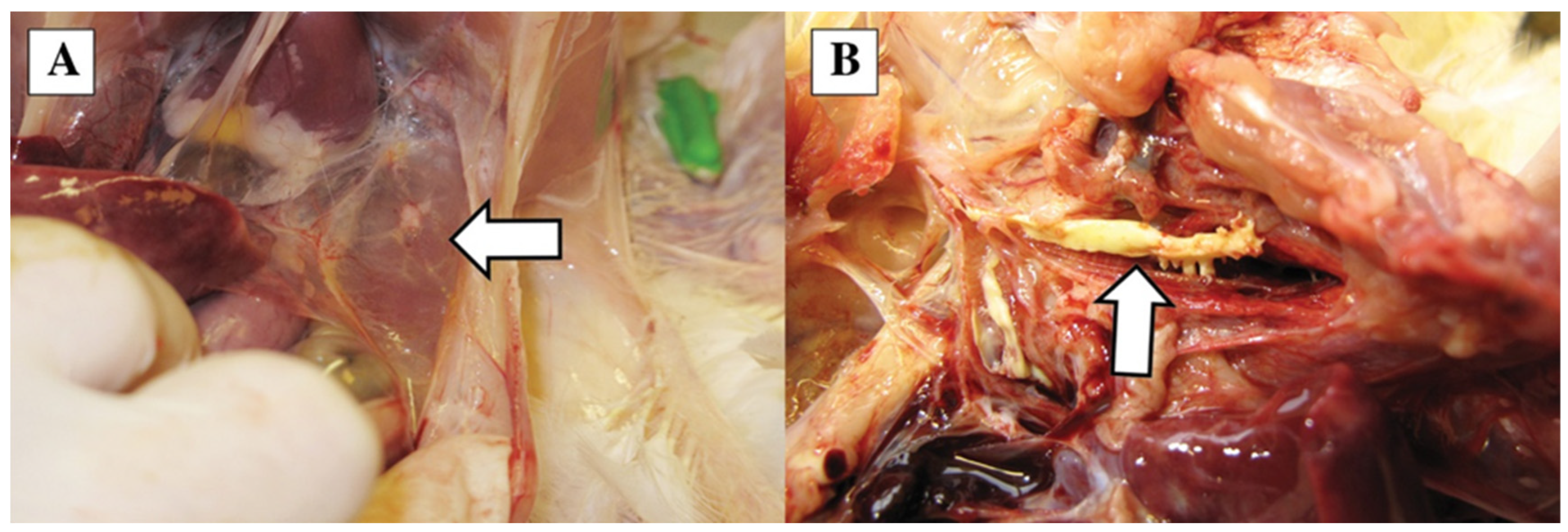
3.2. Gross Pathology
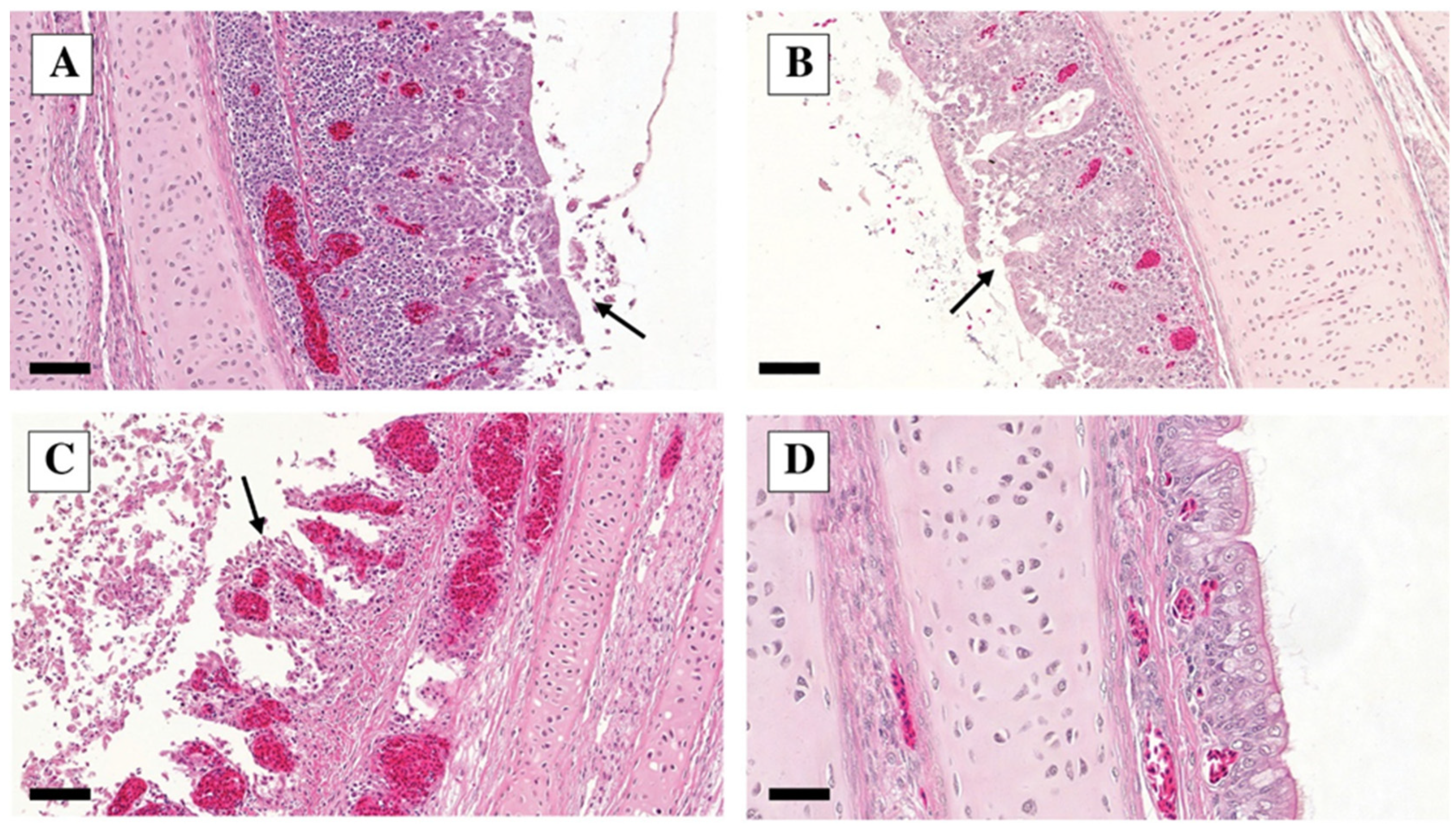
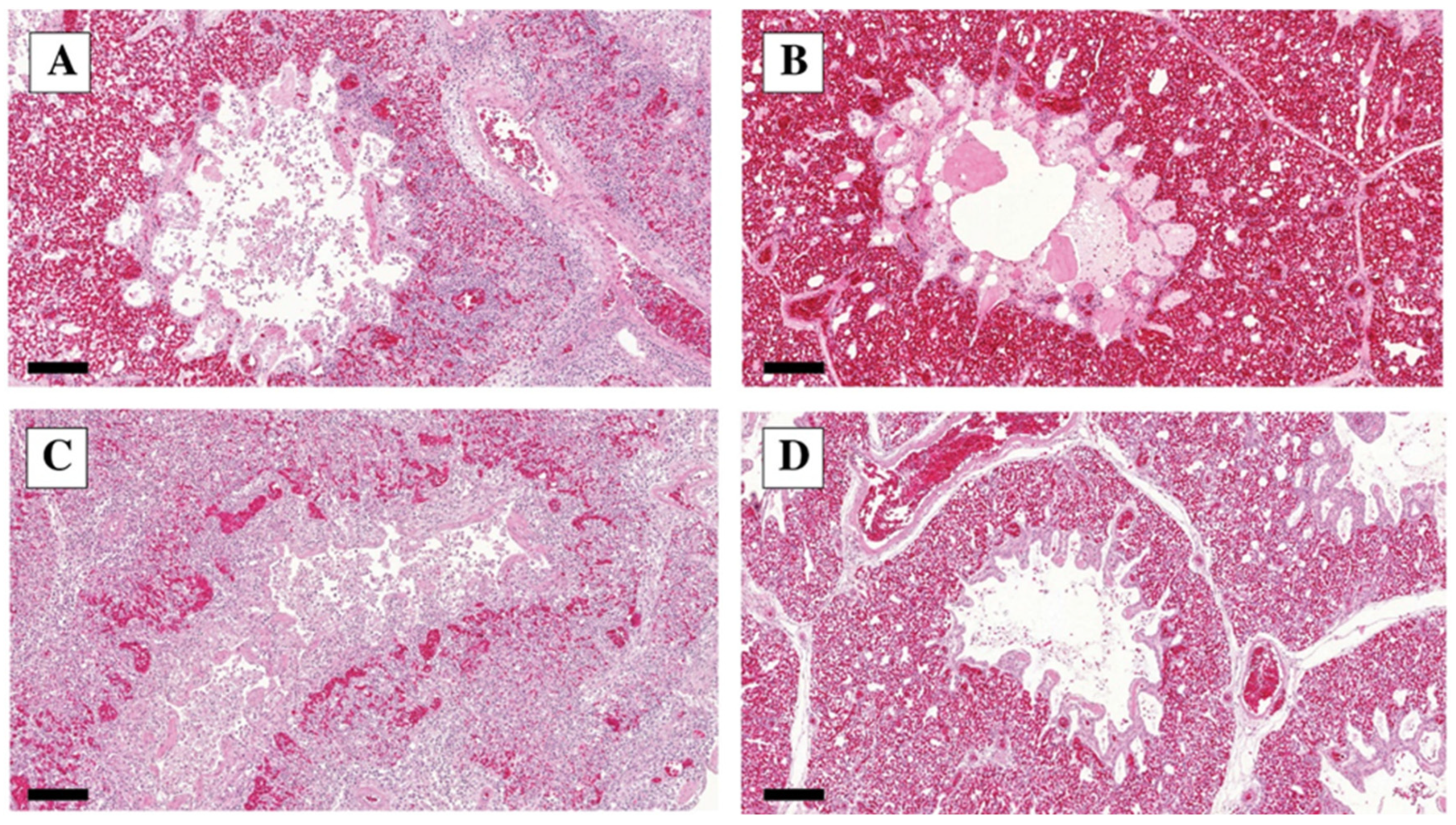
3.3. Histopathology
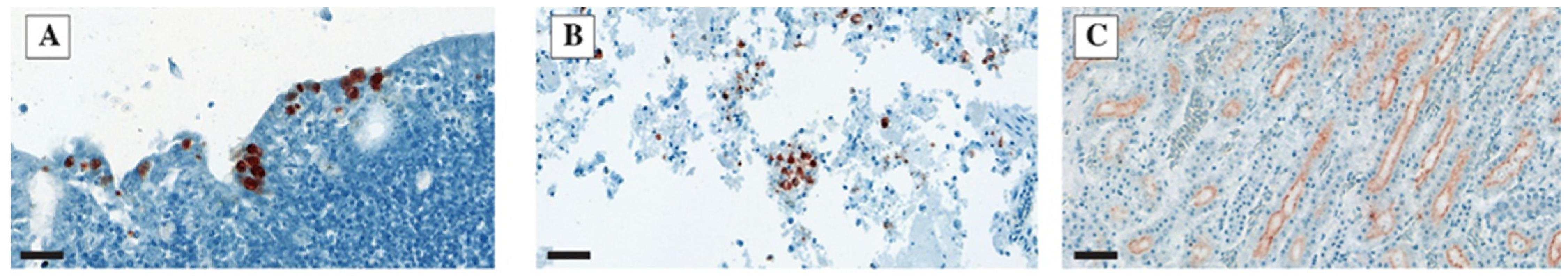
3.4. IHC
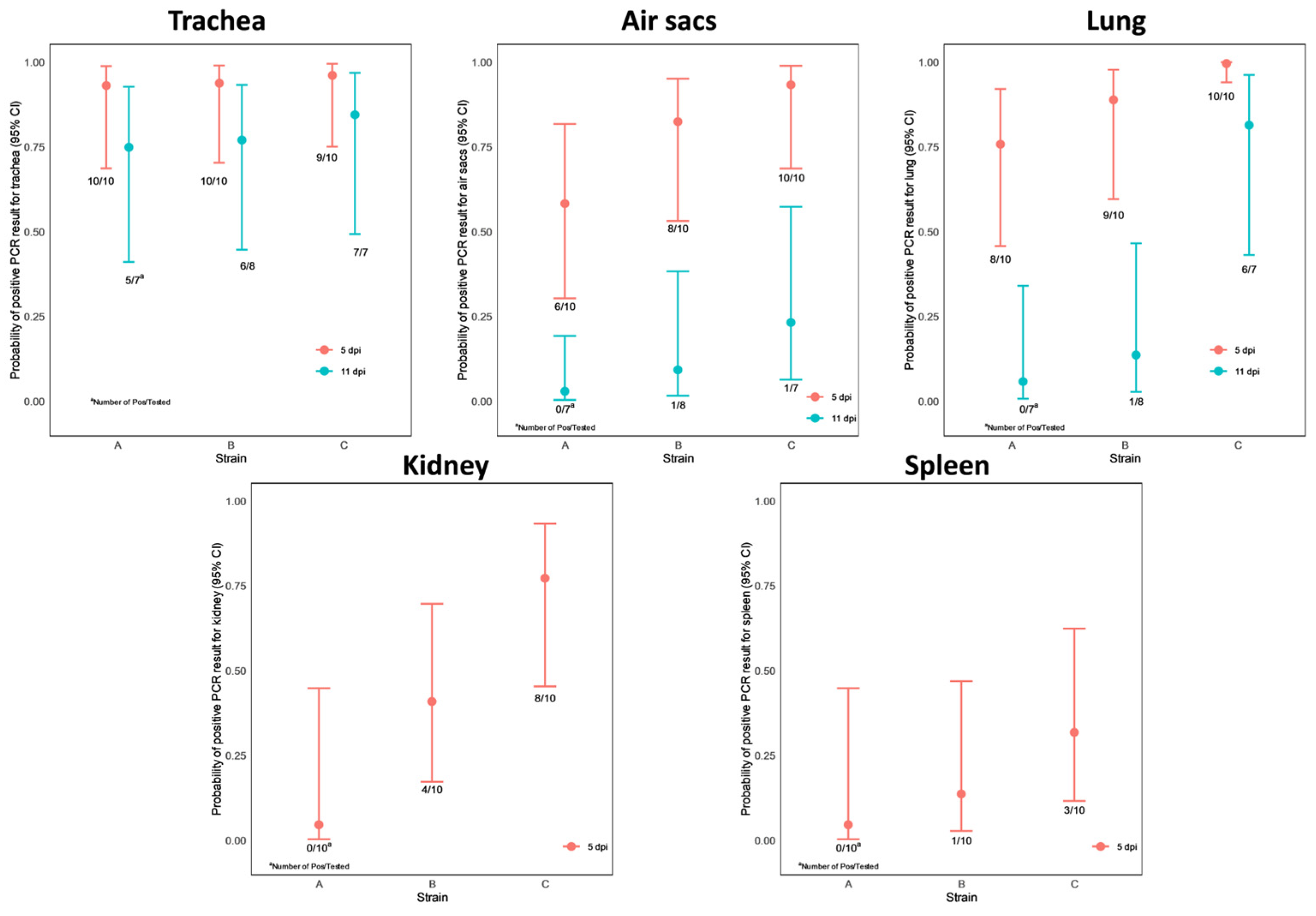
3.5. PCR
- In the trachea: all birds but one on dpi 5 and in the majority of them (18 out of 22, 82%) on dpi 11. This observation is supported by the model that neither the dpi nor the strain has a significant effect on the positive PCR result;
- In the air sacs: 80% (24 out of 30) of the chickens were found positive on dpi 5 and 9% (2 out of 22) on dpi 11. Accordingly, the dpi variable appeared to be significant (p < 0.0001), but no significant difference was obtained between the three strains;
- In the lungs: 90% (27 out of 30) of the chickens were found positive on dpi 5 and 32% (7 out of 22) on dpi 11. In the case of the lung PCR results, the dpi variable appeared to be significant (p < 0.0001), and a significant difference was obtained between strains A and C (p = 0.0093) and strains B and C (p = 0.0257).
3.6. Correlation between the Diagnostic Methods
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bóna, M.; Tatár-Kis, T.; Mándoki, M.; Farsang, A.; Kiss, I. Increasing impact of H9N2 avian influenza virus in the world. Hung. Vet. J. 2023, 145, 19–36. [Google Scholar] [CrossRef]
- Zhang, S.; Yu, J.L.; He, L.; Gong, L.; Hou, S.; Shu, M.; Wu, J.B.; Su, B.; Liu, J.; Wu, G.; et al. Molecular Characteristics of the H9N2 Avian Influenza Viruses in Live Poultry Markets in Anhui Province, China, 2013 to 2018. Health Sci. Rep. 2021, 4, e230. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Jin, Y.; Zhou, J.; Huang, Z.; Li, B.; Zhou, W.; Ren, H.; Yue, J.; Liang, L. Genetic Characteristic and Global Transmission of Influenza A H9N2 Virus. Front. Microbiol. 2017, 8, 2611. [Google Scholar] [CrossRef] [PubMed]
- Peacock, T.H.P.; James, J.; Sealy, J.E.; Iqbal, M. A Global Perspective on H9N2 Avian Influenza Virus. Viruses 2019, 11, 620. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Mettenleiter, T.C.; Abdelwhab, E.M. A Brief Summary of the Epidemiology and Genetic Relatedness of Avian Influenza H9N2 Virus in Birds and Mammals in the Middle East and North Africa. Epidemiol. Infect. 2017, 145, 3320–3333. [Google Scholar] [CrossRef] [PubMed]
- Adel, A.; Abdelmagid, M.A.; Mohamed, A.A.; Wasberg, A.; Mosaad, Z.; Selim, K.; Shaaban, A.; Tarek, M.; Hagag, N.M.; Lundkvist, A.; et al. Genetic Variations among Different Variants of G1-like Avian Influenza H9N2 Viruses and Their Pathogenicity in Chickens. Viruses 2022, 14, 1030. [Google Scholar] [CrossRef] [PubMed]
- El Houadfi, M.; Fellahi, S.; Nassik, S.; Guérin, J.-L.; Ducatez, M.F. First Outbreaks and Phylogenetic Analyses of Avian Influenza H9N2 Viruses Isolated from Poultry Flocks in Morocco. Virol. J. 2016, 13, 140. [Google Scholar] [CrossRef] [PubMed]
- El Khantour, A.; Soulaymani, A.; Salek, M.; Maltouf, A.F.; El Mellouli, F.; Ducatez, M.F.; Fellahi, S. Molecular Characterization of the Hemagglutinin Gene of H9N2 Avian Influenza Viruses Isolated from Broiler Flocks in Morocco from 2016 to 2018. Vet. Arh. 2020, 90, 477–484. [Google Scholar] [CrossRef]
- El Mellouli, F.; Mouahid, M.; Fusaro, A.; Zecchin, B.; Zekhnini, H.; El Khantour, A.; Giussani, E.; Palumbo, E.; Rguibi Idrissi, H.; Monne, I.; et al. Spatiotemporal Dynamics, Evolutionary History and Zoonotic Potential of Moroccan H9N2 Avian Influenza Viruses from 2016 to 2021. Viruses 2022, 14, 509. [Google Scholar] [CrossRef]
- Sikht, F.-Z.; Ducatez, M.; Touzani, C.D.; Rubrum, A.; Webby, R.; El Houadfi, M.; Tligui, N.-S.; Camus, C.; Fellahi, S. Avian Influenza a H9N2 Viruses in Morocco, 2018–2019. Viruses 2022, 14, 529. [Google Scholar] [CrossRef]
- Barberis, A.; Boudaoud, A.; Gorrill, A.; Loupias, J.; Ghram, A.; Lachheb, J.; Alloui, N.; Ducatez, M.F. Full-Length Genome Sequences of the First H9N2 Avian Influenza Viruses Isolated in the Northeast of Algeria. Virol. J. 2020, 17, 108. [Google Scholar] [CrossRef] [PubMed]
- Jeevan, T.; Darnell, D.; Gradi, E.A.; Benali, Y.; Kara, R.; Guetarni, D.; Rubrum, A.; Seiler, P.J.; Crumpton, J.C.; Webby, R.J.; et al. A(H9N2) Influenza Viruses Associated with Chicken Mortality in Outbreaks in Algeria 2017. Influenza Other Respir. Viruses 2019, 13, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Fusade-Boyer, M.; Djegui, F.; Batawui, K.; Byuragaba, D.K.; Jones, J.C.; Wabwire-Mangeni, F.; Erima, B.; Atim, G.; Ukuli, Q.A.; Tugume, T.; et al. Antigenic and Molecular Characterization of Low Pathogenic Avian Influenza A(H9N2) Viruses in Sub-Saharan Africa from 2017 through 2019. Emerg. Microbes Infect. 2021, 10, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Kariithi, H.M.; Welch, C.N.; Ferreira, H.L.; Pusch, E.A.; Ateya, L.O.; Binepal, Y.S.; Apopo, A.A.; Dulu, T.D.; Afonso, C.L.; Suarez, D.L. Genetic Characterization and Pathogenesis of the First H9N2 Low Pathogenic Avian Influenza Viruses Isolated from Chickens in Kenyan Live Bird Markets. Infect. Genet. Evol. 2020, 78, 104074. [Google Scholar] [CrossRef]
- Awadin, W.; Said, H.; Abdin, S.; El-Sawak, A.A. Pathological and Molecular Studies on Avian Influenza Virus (H9N2) in Broilers. Asian J. Anim. Vet. Adv. 2018, 13, 232–244. [Google Scholar] [CrossRef]
- Zaker, S.R.; Nili, H.; Asasi, K. Apoptosis in Field and Experimental Cases of Avian Influenza H9N2 Infection in Broiler Chickens in Iran. Vet. Res. Forum Int. Q. J. 2022, 3, 439–441. [Google Scholar] [CrossRef]
- Chrzastek, K.; Lee, D.; Gharaibeh, S.; Zsak, A.; Kapczynski, D.R. Characterization of H9N2 Avian Influenza Viruses from the Middle East Demonstrates Heterogeneity at Amino Acid Position 226 in the Hemagglutinin and Potential for Transmission to Mammals. Virology 2018, 518, 195–201. [Google Scholar] [CrossRef]
- Kaboudi, K. Low Pathogenic Avian Influenza Virus Subtype H9N2 in Poultry in North Africa: Current Status. Vet. Sci. Res. Rev. 2019, 5, 73–79. [Google Scholar] [CrossRef]
- Gu, M.; Xu, L.; Wang, X.; Liu, X. Current Situation of H9N2 Subtype Avian Influenza in China. Vet. Res. 2017, 48, 49. [Google Scholar] [CrossRef]
- Luna, L.G. (Ed.) Manual of Histologic Staining Methods, 3rd ed.; McGraw-Hill: New York, NY, USA, 1968. [Google Scholar]
- Bóna, M.; Kiss, I.; Dénes, L.; Szilasi, A.; Mándoki, M. Tissue Tropism of H9N2 Low-Pathogenic Avian Influenza Virus in Broiler Chickens by Immunohistochemistry. Animals 2023, 13, 1052. [Google Scholar] [CrossRef]
- Spackman, E.; Senne, D.A.; Myers, T.J.; Bulaga, L.L.; Garber, L.P.; Perdue, M.L.; Lohman, K.; Daum, L.T.; Suarez, D.L. Development of a Real-Time Reverse Transcriptase PCR Assay for Type A Influenza Virus and the Avian H5 and H7 Hemagglutinin Subtypes. J. Clin. Microbiol. 2002, 40, 3256–3260. [Google Scholar] [CrossRef] [PubMed]
- Landmann, M.; Scheibner, D.; Graaf, A.; Gischke, M.; Koethe, S.; Fatola, O.I.; Raddatz, B.; Mettenleiter, T.C.; Beer, M.; Grund, C.; et al. A Semiquantitative Scoring System for Histopathological and Immunohistochemical Assessment of Lesions and Tissue Tropism in Avian Influenza. Viruses 2021, 13, 868. [Google Scholar] [CrossRef]
- Rizopoulos, D. 2022. Available online: https://drizopoulos.github.io/GLMMadaptive/articles/Ordinal_Mixed_Models.html (accessed on 16 June 2023).
- Liu, X. Ordinal Regression Analysis: Fitting the Continuation Ratio Model to Educational Data Using Stata. NERA Conf. Proc. 2010, 35. [Google Scholar]
- Firth, D. Bias Reduction of Maximum Likelihood Estimates. Biometrika 1993, 80, 27–38. [Google Scholar] [CrossRef]
- Ananth, C.V.; Kleinbaum, D.G. Regression Models for Ordinal Responses: A Review of Methods and Applications. Int. J. Epidemiol. 1997, 26, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Kosmidis, I. Improved estimation in cumulative link models. J. R. Stat. Soc. B 2014, 76 Pt 1, 169–196. [Google Scholar] [CrossRef][Green Version]
- Shahid, M.F.; Yaqub, T.; Tipu, M.Y.; Aslam, A.; Yaqub, S.; Rahman, A.; Ali, M. Comparative Pathogenic Potential of Avian Influenza A/H9N2 Viruses Isolated from Commercial, Backyard and Fancy Birds. Punjab Univ. J. Zool. 2019, 34, 143–148. [Google Scholar] [CrossRef]
- Subtain, S.; Chaudhry, Z.; Anjum, A.; Maqbool, A.; Sadique, U. Study on Pathogenesis of Low Pathogenic Avian Influenza Virus H9 in Broiler Chickens. Pak. J. Zool. 2011, 43, 999–1008. [Google Scholar]
- Pazani, J.; Vasfi Marandi, M.; Ashrafihelan, J.; Marjanmehr, S.; Farid, G. Pathological Studies of A/Chicken/Tehran/ZMT-173/99 (H9N2) Influenza Virus in Commercial Broiler Chickens of Iran. Int. J. Poult. Sci. 2008, 7, 502–510. [Google Scholar] [CrossRef]
- Begum, J.A.; Hossain, I.; Nooruzzaman, M.; King, J.; Chowdhury, E.H.; Harder, T.C.; Parvin, R. Experimental Pathogenicity of H9N2 Avian Influenza Viruses Harboring a Tri-Basic Hemagglutinin Cleavage Site in Sonali and Broiler Chickens. Viruses 2023, 15, 461. [Google Scholar] [CrossRef]


| Group * | Challenge Strain | No. of Chickens Examined | |
|---|---|---|---|
| dpi 5 ** | dpi 11 | ||
| A | A/chicken/Saudi Arabia/8616/2016 H9N2 | 11(1) *** | 7 |
| B | A/chicken/Egypt/4531/2016 H9N2 | 10 | 8 |
| C | A/chicken/Morocco/2021/2016 H9N2 | 10(4) *** | 7 |
| Control | PBS buffer | 3 | 6 |
| H9N2 Strain | ‘A’ | ‘B’ | ‘C’ | ‘A’ | ‘B’ | ‘C’ |
|---|---|---|---|---|---|---|
| dpi 5 | dpi 11 | |||||
| Trachea | ||||||
| No * | 2 ** | 6 | 1 | 4 | 1 | 7 |
| Mild | 1 | 1 | 1 | 1 | 0 | 0 |
| Moderate | 7 | 1 | 3 | 2 | 7 | 0 |
| Severe | 1 | 2 | 6 | 0 | 0 | 0 |
| Air sacs | ||||||
| No | 0 | 1 | 0 | 5 | 1 | 0 |
| Mild | 11 | 5 | 3 | 2 | 3 | 4 |
| Moderate | 0 | 4 | 6 | 0 | 4 | 1 |
| Severe | 0 | 0 | 2 | 0 | 0 | 2 |
| H9N2 Strain | ‘A’ | ‘B’ | ‘C’ | ‘A’ | ‘B’ | ‘C’ |
|---|---|---|---|---|---|---|
| dpi 5 | dpi 11 | |||||
| Lymphocytic inflammation | ||||||
| No * | 0 ** | 0 | 0 | 7 | 1 | 3 |
| Mild | 0 | 1 | 0 | 0 | 3 | 3 |
| Moderate | 7 | 5 | 1 | 0 | 4 | 1 |
| Severe | 4 | 4 | 9 | 0 | 0 | 0 |
| Epithelial degeneration | ||||||
| No | 0 | 0 | 0 | 7 | 8 | 7 |
| Yes | 11 | 10 | 10 | 0 | 0 | 0 |
| H9N2 Strain | ‘A’ | ‘B’ | ‘C’ | ‘A’ | ‘B’ | ‘C’ |
|---|---|---|---|---|---|---|
| dpi 5 | dpi 11 | |||||
| Interstitial pneumonia | ||||||
| No * | 1 ** | 2 | 1 | 7 | 8 | 7 |
| Mild | 3 | 2 | 4 | 0 | 0 | 0 |
| Moderate | 5 | 3 | 2 | 0 | 0 | 0 |
| Severe | 2 | 3 | 3 | 0 | 0 | 0 |
| Bronchitis | ||||||
| No | 1 | 0 | 0 | 4 | 6 | 3 |
| Mild | 0 | 0 | 1 | 3 | 1 | 3 |
| Moderate | 5 | 7 | 5 | 0 | 1 | 1 |
| Severe | 5 | 3 | 4 | 0 | 0 | 0 |
| Catarrhal infiltration | ||||||
| No | 1 | 2 | 1 | 6 | 7 | 6 |
| Mild | 5 | 4 | 1 | 0 | 0 | 0 |
| Moderate | 5 | 2 | 4 | 0 | 1 | 1 |
| Severe | 0 | 2 | 4 | 1 | 0 | 0 |
| Edema | ||||||
| No | 0 | 1 | 0 | 7 | 8 | 7 |
| Mild | 1 | 4 | 0 | 0 | 0 | 0 |
| Moderate | 8 | 5 | 5 | 0 | 0 | 0 |
| Severe | 2 | 0 | 5 | 0 | 0 | 0 |
| H9N2 Strain | ‘A’ | ‘B’ | ‘C’ | ‘A’ | ‘B’ | ‘C’ |
|---|---|---|---|---|---|---|
| dpi 5 | dpi 11 | |||||
| Lymphocytic inflammation | ||||||
| No * | 9 ** | 8 | 8 | 7 | 8 | 7 |
| Mild | 2 | 1 | 2 | 0 | 0 | 0 |
| Moderate | 0 | 1 | 0 | 0 | 0 | 0 |
| Severe | 0 | 0 | 0 | 0 | 0 | 0 |
| Tubulonephrosis | ||||||
| No | 1 | 3 | 0 | 7 | 8 | 7 |
| Mild | 3 | 5 | 0 | 0 | 0 | 0 |
| Moderate | 7 | 2 | 3 | 0 | 0 | 0 |
| Severe | 0 | 0 | 7 | 0 | 0 | 0 |
| Glomerulonephritis | ||||||
| No | 3 | 5 | 0 | 7 | 8 | 7 |
| Yes | 7 | 5 | 10 | 0 | 0 | 0 |
| H9N2 Strain | ‘A’ | ‘B’ | ‘C’ | ‘A’ | ‘B’ | ‘C’ |
|---|---|---|---|---|---|---|
| dpi 5 | dpi 11 | |||||
| Trachea | ||||||
| No * | 5 ** | 7 | 4 | 7 | 8 | 7 |
| Low | 6 | 2 | 2 | 0 | 0 | 0 |
| High | 0 | 1 | 4 | 0 | 0 | 0 |
| Lung | ||||||
| No | 7 | 10 | 3 | 7 | 8 | 7 |
| Low | 3 | 0 | 5 | 0 | 0 | 0 |
| High | 1 | 0 | 2 | 0 | 0 | 0 |
| Kidney | ||||||
| No | 9 | 6 | 1 | 7 | 8 | 7 |
| Low | 2 | 4 | 4 | 0 | 0 | 0 |
| High | 0 | 0 | 5 | 0 | 0 | 0 |
| Spleen | ||||||
| No | 10 | 10 | 9 | 7 | 8 | 7 |
| Low | 1 | 0 | 1 | 0 | 0 | 0 |
| High | 0 | 0 | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bóna, M.; Földi, J.; Dénes, L.; Harnos, A.; Paszerbovics, B.; Mándoki, M. Evaluation of the Virulence of Low Pathogenic H9N2 Avian Influenza Virus Strains in Broiler Chickens. Vet. Sci. 2023, 10, 671. https://doi.org/10.3390/vetsci10120671
Bóna M, Földi J, Dénes L, Harnos A, Paszerbovics B, Mándoki M. Evaluation of the Virulence of Low Pathogenic H9N2 Avian Influenza Virus Strains in Broiler Chickens. Veterinary Sciences. 2023; 10(12):671. https://doi.org/10.3390/vetsci10120671
Chicago/Turabian StyleBóna, Márta, József Földi, Lilla Dénes, Andrea Harnos, Bettina Paszerbovics, and Míra Mándoki. 2023. "Evaluation of the Virulence of Low Pathogenic H9N2 Avian Influenza Virus Strains in Broiler Chickens" Veterinary Sciences 10, no. 12: 671. https://doi.org/10.3390/vetsci10120671
APA StyleBóna, M., Földi, J., Dénes, L., Harnos, A., Paszerbovics, B., & Mándoki, M. (2023). Evaluation of the Virulence of Low Pathogenic H9N2 Avian Influenza Virus Strains in Broiler Chickens. Veterinary Sciences, 10(12), 671. https://doi.org/10.3390/vetsci10120671







