A 3D QSAR Study of Betulinic Acid Derivatives as Anti-Tumor Agents Using Topomer CoMFA: Model Building Studies and Experimental Verification
Abstract
:1. Introduction
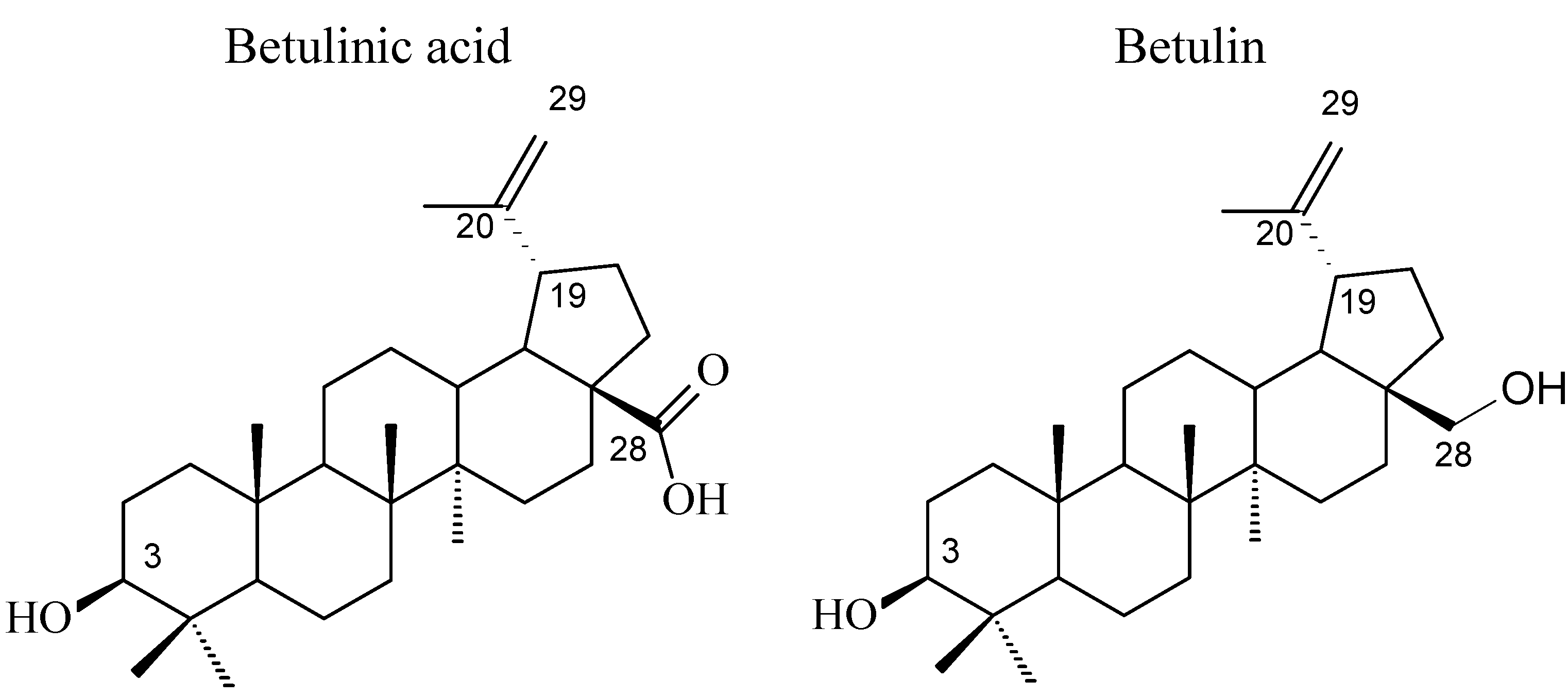
2. Results and Discussion
2.1. 3D-QSAR Model using a Topomer CoMFA Method
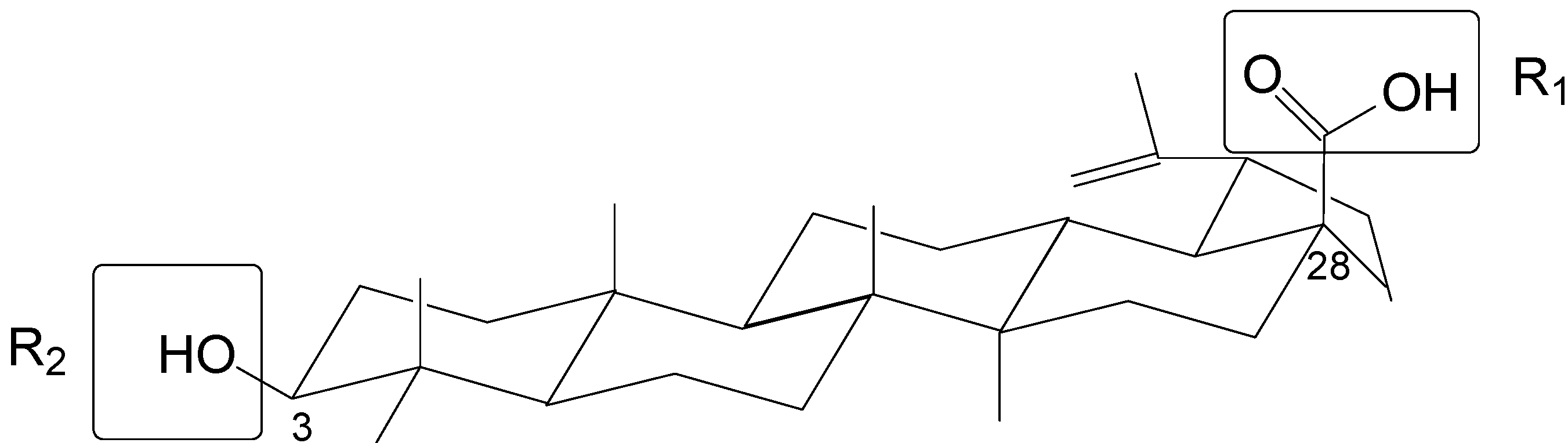
| Comp. | Substituent | pIC50 | Res. | ||
|---|---|---|---|---|---|
| R1 | R2 | Experimental | Pred. | ||
| Test set | |||||
| 1 | ―COOH | ―OH | 4.856 | 4.627 | 0.229 |
| 2 |  | ―OCOCH3 | 4.999 | 4.855 | 0.144 |
| 3 |  | ―OCOCH3 | 5.276 | 5.244 | 0.032 |
| 4 |  | ―OH | 5.310 | 5.150 | 0.160 |
| 5 |  | ―OCOCH3 | 4.936 | 5.053 | −0.118 |
| 6 |  | ―OCOCH3 | 4.921 | 4.953 | −0.032 |
| 7 |  | ―OH | 5.229 | 4.990 | 0.239 |
| 8 |  | ―OCOCH3 | 4.733 | 4.722 | 0.011 |
| 9 | ―COOH | ―OCONHC2H5 | 4.776 | 4.806 | −0.030 |
| 10 | ―COOH | ―OCOC5H11 | 4.000 | 4.486 | −0.486 |
| 11 | ―CH2OCOCH3 | ―OCOCH3 | 4.590 | 4.782 | −0.192 |
| Training set | |||||
| 12 | ―COOH | ―OCOCH3 | 4.792 | 4.818 | −0.026 |
| 13 |  | ―OCOCH3 | 5.611 | 5.384 | 0.227 |
| 14 |  | ―OH | 5.051 | 5.192 | −0.141 |
| 15 | ―CH2OCOCH3 |  | 5.009 | 4.970 | 0.039 |
| 16 | ―CH2OCOCH3 |  | 4.997 | 4.970 | 0.026 |
| 17 |  | ―OH | 5.056 | 5.052 | 0.004 |
| 18 |  | ―OCOCH3 | 5.301 | 5.341 | −0.040 |
| 19 |  | ―OCH3 | 5.328 | 5.334 | −0.006 |
| 20 |  | ―OCOCH3 | 4.770 | 4.728 | 0.042 |
| 21 |  | ―OCOCH3 | 4.740 | 4.775 | −0.035 |
| 22 |  | ―OCOCH3 | 5.244 | 5.296 | −0.052 |
| 23 |  | ―OCOCH3 | 5.292 | 5.316 | −0.024 |
| 24 |  | ―OCOCH3 | 5.409 | 5.421 | −0.012 |
| 25 |  | ―OCOCH3 | 4.799 | 4.825 | −0.026 |
| 26 |  | ―OH | 4.813 | 4.711 | 0.102 |
| 27 |  | ―OH | 4.740 | 4.711 | 0.029 |
| 28 |  | ―OCH3 | 4.775 | 4.715 | 0.060 |
| 29 |  | ―OCOCH3 | 4.524 | 4.527 | −0.003 |
| 30 | ―COCH2COOC2H5 | ―OCOCH3 | 4.921 | 4.914 | 0.007 |
| 31 |  | ―OCOCH3 | 4.830 | 4.776 | 0.054 |
| 32 | ―OCOOCH3 | ―OCONHC6H5 | 4.161 | 4.125 | 0.036 |
| 33 | ―CH2OCONHC2H5 | ―OCONHC2H5 | 5.208 | 5.180 | 0.028 |
| 34 | ―CH2OCONHC2H5 | ―OCONHC6H5 | 4.160 | 4.176 | −0.016 |
| 35 | ―CH2OCOCH3 | ―OCONHC6H5 | 4.117 | 4.116 | 0.001 |
| 36 | ―COOH | ―OCOC9H19 | 4.279 | 4.287 | −0.008 |
| 37 | ―CH2OCOCH2CI | ―OH | 4.559 | 4.586 | −0.027 |

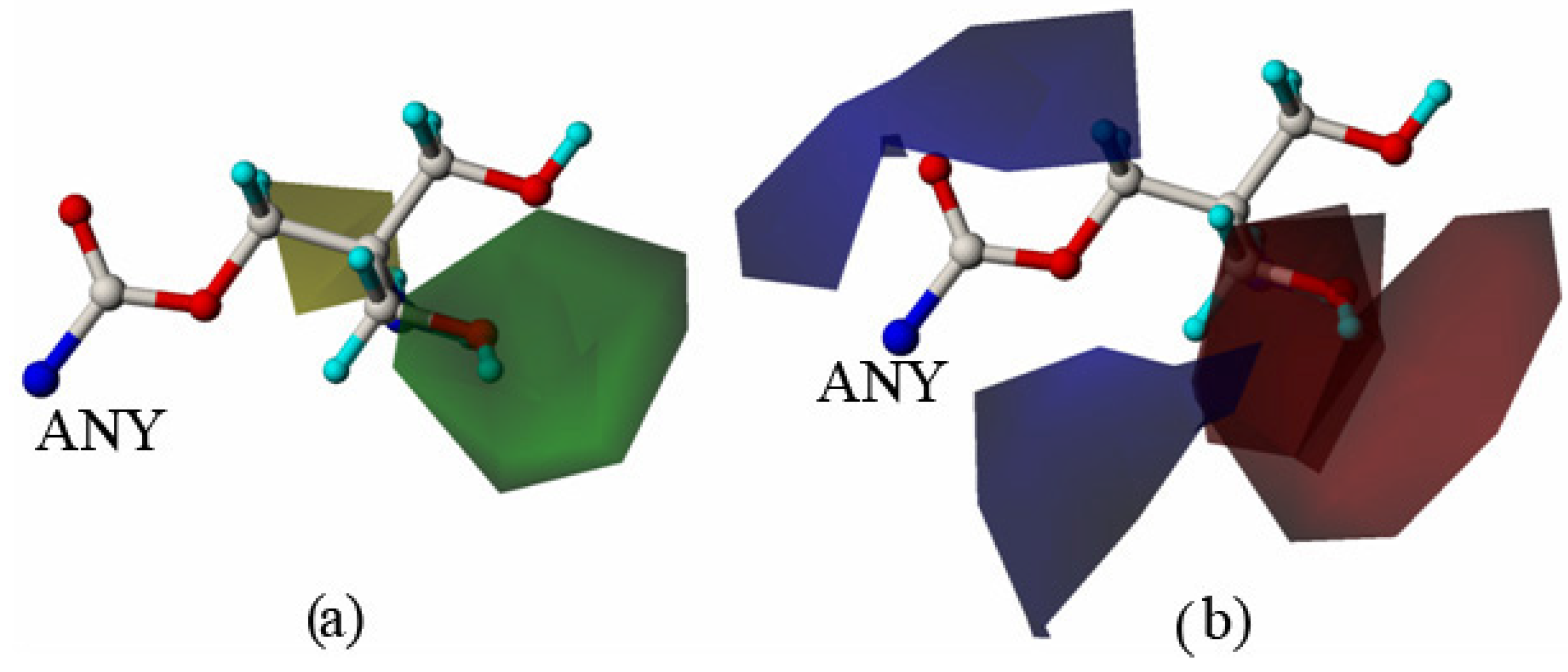
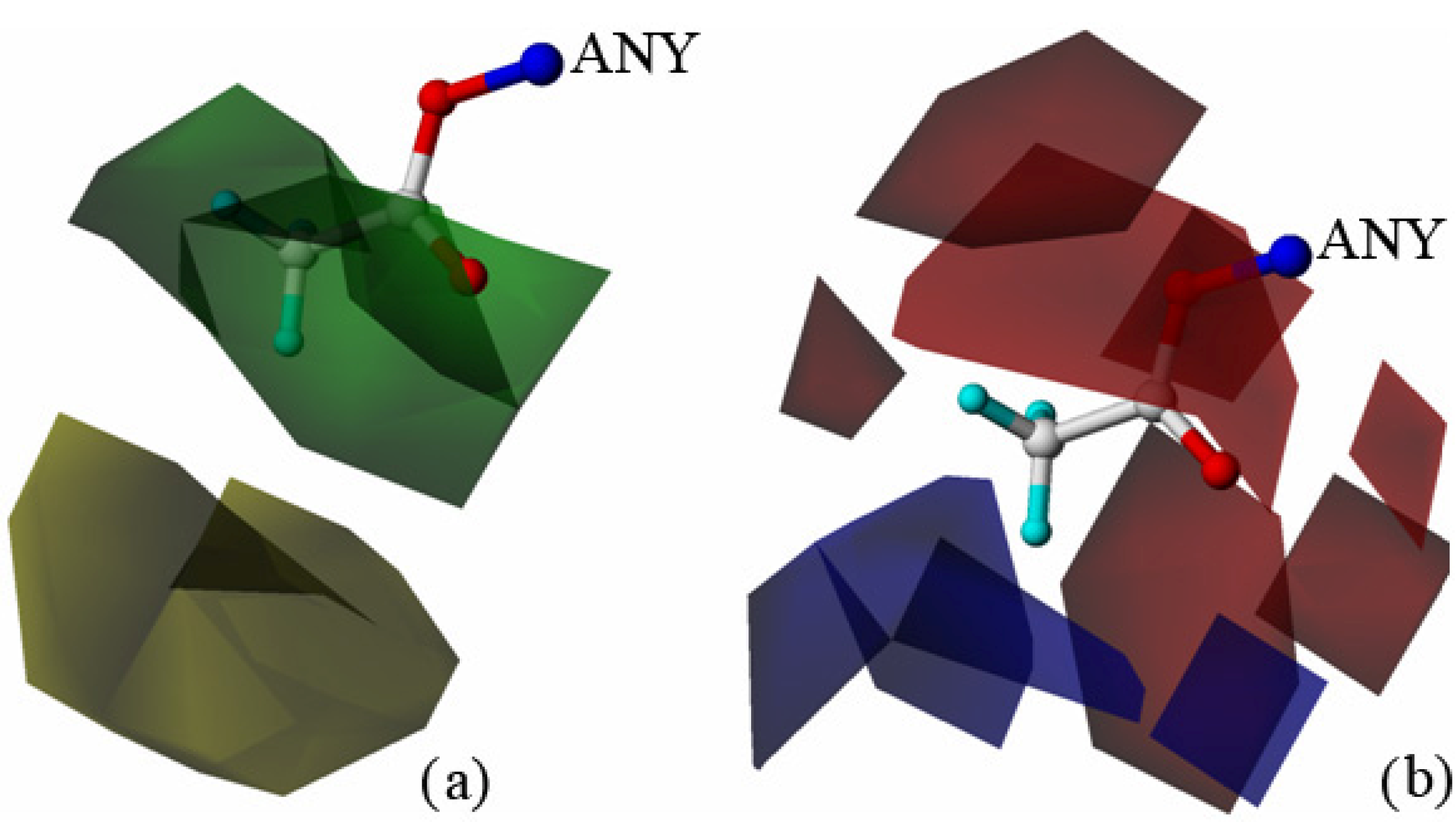
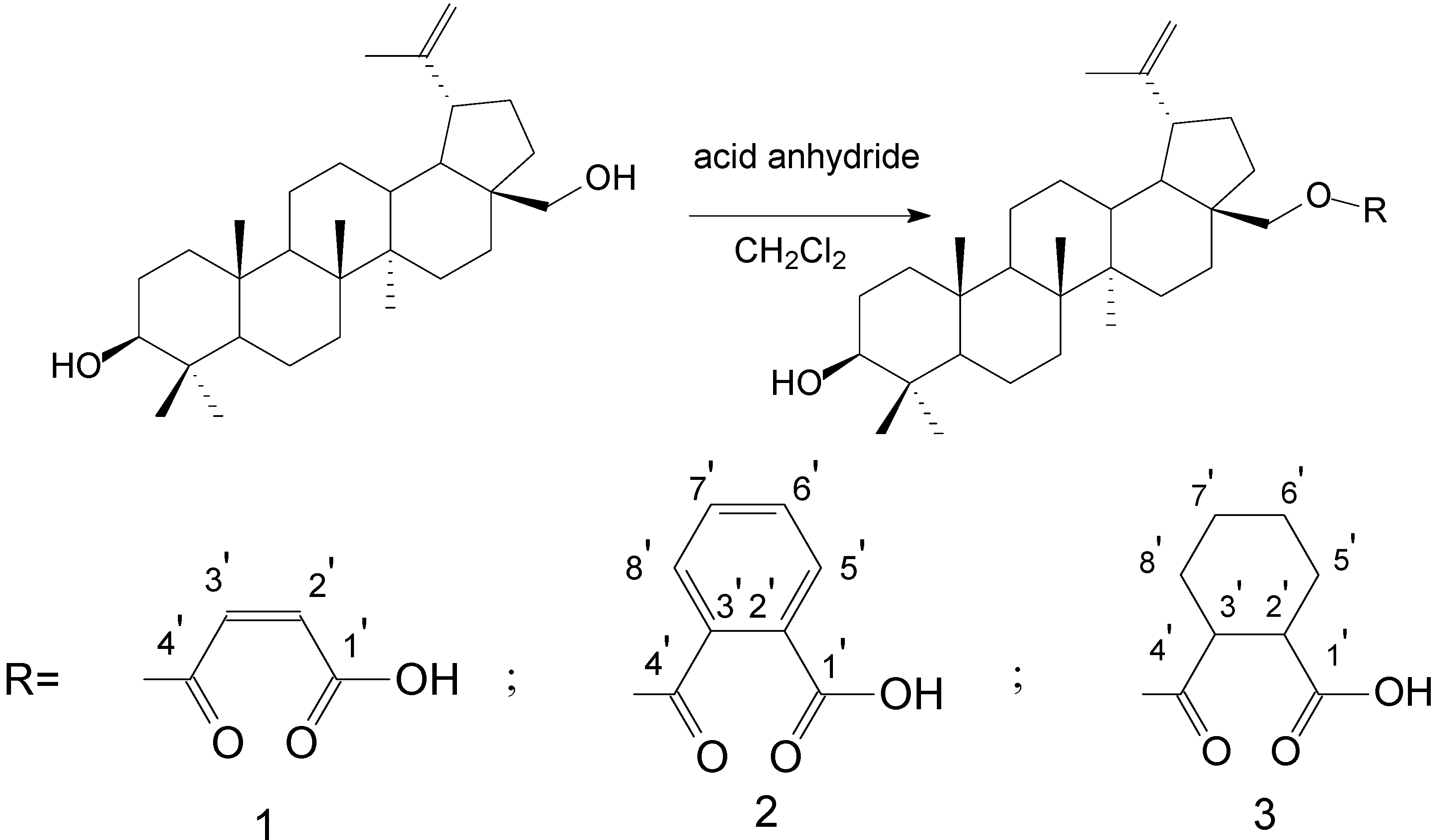
2.2. Molecular Design and Experimental Verification
| Compd. | IC50 (μM) | pIC50− (Exp.) | pIC50 (Pred.) |
|---|---|---|---|
| 1 | 83.09 ± 7.53 | 4.080 | 4.925 |
| 2 | 35.33 ± 1.04 | 4.452 | 5.071 |
| 3 | 30.05 ± 1.54 | 4.522 | 5.127 |
| BA | 32.66 ± 0.62 | 4.486 | 4.627 |
| 13.93 ± 0.46 * | 4.856 * |
| Compd. | IC50 (μM) for cancer cell lines | ||||
|---|---|---|---|---|---|
| MPC2 (pancreatic cancer) | DU145 (prostate cancer) | NCI-H520 (lung carcinoma) | HeLa (cervical cancer) | 2774 (ovarian cancer) | |
| BA | 38.58 ± 2.91 | 23.27 ± 2.20 | 19.60 ± 2.44 | 25.93 ± 1.87 | 39.54 ± 2.19 |
| 1 | 75.07 ± 4.96 | 92.54 ± 1.45 | 84.83 ± 2.15 | 99.62 ± 4.64 | 67.93 ± 2.00 |
| 2 | 31.28 ± 3.43 | 36.19 ± 2.77 | 31.24 ± 0.46 | 40.05 ± 3.37 | 27.02 ± 2.92 |
| 3 | 22.79 ± 0.35 | 37.99 ± 2.50 | 18.55 ± 0.38 | 17.47 ± 1.48 | 25.85 ± 1.34 |
3. Experimental
3.1. Data Sets
3.2. Topomer CoMFA Setup


3.3. Materials and Instruments
3.4. General Procedure for Synthesizing BE Esters
3.4.1. BE 28-O-Maleate 1
3.4.2. BE 28-O-Phthalate 2
3.4.3. BE 28-O-Hexahydrophthalate 3
3.5. Cell Culture and in Vitro Cytotoxicity Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Fulda, S. Betulinic acid for cancer treatment and prevention. Int. J. Mol. Sci. 2008, 9, 1096–1107. [Google Scholar] [CrossRef]
- Udeani, G.O.; Zhao, G.M.; Geun Shin, Y.; Cooke, B.P.; Graham, J.; Beecher, C.W.; Kinghorn, A.D.; Pezzuto, J.M. Pharmacokinetics and tissue distribution of betulinic acid in CD-1 mice1. Biopharm. Drug Dispos. 1999, 20, 379–383. [Google Scholar] [CrossRef]
- Zuco, V.; Supino, R.; Righetti, S.C.; Cleris, L.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar] [CrossRef]
- Pisha, E.; Chai, H.; Lee, I.S.; Chagwedera, T.E.; Farnsworth, N.R.; Cordell, G.A.; Beecher, C.W.; Fong, H.H.; Kinghorn, A.D.; Brown, D.M.; et al. Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nat. Med. 1995, 1, 1046–1051. [Google Scholar] [CrossRef]
- Patočka, J. Biologically active pentacyclic triterpenes and their current medicine signification. J. Appl. Biomed. 2003, 1, 7–12. [Google Scholar]
- Alakurtti, S.; Makela, T.; Koskimies, S.; Yli-Kauhaluoma, J. Pharmacological properties of the ubiquitous natural product betulin. Eur. J. Pharm. Sci. 2006, 29, 1–13. [Google Scholar]
- Csuk, R.; Barthel, A.; Kluge, R.; Strohl, D. Synthesis, cytotoxicity and liposome preparation of 28-acetylenic betulin derivatives. Bioorg. Med. Chem. 2010, 18, 7252–7259. [Google Scholar] [CrossRef]
- Csuk, R.; Barthel, A.; Kluge, R.; Strohl, D.; Kommera, H.; Paschke, R. Synthesis and biological evaluation of antitumour-active betulin derivatives. Bioorg. Med. Chem. 2010, 18, 1344–1355. [Google Scholar] [CrossRef]
- Csuk, R.; Barthel, A.; Schwarz, S.; Kommera, H.; Paschke, R. Synthesis and biological evaluation of antitumor-active gamma-butyrolactone substituted betulin derivatives. Bioorg. Med. Chem. 2010, 18, 2549–2558. [Google Scholar] [CrossRef]
- Kommera, H.; Kaluderovic, G.N.; Bette, M.; Kalbitz, J.; Fuchs, P.; Fulda, S.; Mier, W.; Paschke, R. In vitro anticancer studies of alpha- and beta-D-glucopyranose betulin anomers. Chem. Biol. Interact. 2010, 185, 128–136. [Google Scholar] [CrossRef]
- Kommera, H.; Kaluderovic, G.N.; Dittrich, S.; Kalbitz, J.; Drager, B.; Mueller, T.; Paschke, R. Carbamate derivatives of betulinic acid and betulin with selective cytotoxic activity. Bioorg. Med. Chem. Lett. 2010, 20, 3409–3412. [Google Scholar] [CrossRef]
- Kommera, H.; Kaluderovic, G.N.; Kalbitz, J.; Drager, B.; Paschke, R. Small structural changes of pentacyclic lupane type triterpenoid derivatives lead to significant differences in their anticancer properties. Eur. J. Med. Chem. 2010, 45, 3346–3353. [Google Scholar] [CrossRef]
- Kommera, H.; Kaluderovic, G.N.; Kalbitz, J.; Paschke, R. Lupane triterpenoids—Betulin and betulinic acid derivatives induce apoptosis in tumor cells. Invest. New Drugs 2011, 29, 266–272. [Google Scholar] [CrossRef]
- Kvasnica, M.; Sarek, J.; Klinotova, E.; Dzubak, P.; Hajduch, M. Synthesis of phthalates of betulinic acid and betulin with cytotoxic activity. Bioorg. Med. Chem. 2005, 13, 3447–3454. [Google Scholar] [CrossRef]
- Rajendran, P.; Jaggi, M.; Singh, M.K.; Mukherjee, R.; Burman, A.C. Pharmacological evaluation of C-3 modified Betulinic acid derivatives with potent anticancer activity. Invest. New Drugs 2008, 26, 25–34. [Google Scholar] [CrossRef]
- Salin, O.; Alakurtti, S.; Pohjala, L.; Siiskonen, A.; Maass, V.; Maass, M.; Yli-Kauhaluoma, J.; Vuorela, P. Inhibitory effect of the natural product betulin and its derivatives against the intracellular bacterium Chlamydia pneumoniae. Biochem. Pharmacol. 2010, 80, 1141–1151. [Google Scholar] [CrossRef]
- Santos, R.C.; Salvador, J.A.; Cortes, R.; Pachon, G.; Marin, S.; Cascante, M. New betulinic acid derivatives induce potent and selective antiproliferative activity through cell cycle arrest at the S phase and caspase dependent apoptosis in human cancer cells. Biochimie 2011, 93, 1065–1075. [Google Scholar] [CrossRef]
- Santos, R.C.; Salvador, J.A.; Marin, S.; Cascante, M.; Moreira, J.N.; Dinis, T.C. Synthesis and structure-activity relationship study of novel cytotoxic carbamate and N-acylheterocyclic bearing derivatives of betulin and betulinic acid. Bioorg. Med. Chem. 2010, 18, 4385–4396. [Google Scholar] [CrossRef]
- Kubinyi, H. 3D Qsar in Drug Design: Theory Methods and Applications; Springer: Ludwigshafen, Germany, 1993; Volume 1. [Google Scholar]
- Kubinyi, H. QSAR and 3D QSAR in drug design Part 1: Methodology. Drug Discov. Today 1997, 2, 457–467. [Google Scholar] [CrossRef]
- Cramer, R.D. Topomer CoMFA: A design methodology for rapid lead optimization. J. Med. Chem. 2003, 46, 374–388. [Google Scholar] [CrossRef]
- Cramer, R.D.; Clark, R.D.; Patterson, D.E.; Ferguson, A.M. Bioisosterism as a molecular diversity descriptor: Steric fields of single “topomeric” conformers. J. Med. Chem. 1996, 39, 3060–3069. [Google Scholar] [CrossRef]
- Cramer, R.D. R-group template CoMFA combines benefits of “ad hoc” and topomer alignments using 3D-QSAR for lead optimization. J. Comput. Aided Mol. Des. 2012, 26, 805–819. [Google Scholar] [CrossRef]
- Aptula, A.O.; Jeliazkova, N.G.; Schultz, T.W.; Cronin, M.T. The better predictive model: High q2 for the training set or low root mean square error of prediction for the test set? QSAR Comb. Sci. 2005, 24, 385–396. [Google Scholar] [CrossRef]
- Roy, P.P.; Roy, K. On some aspects of variable selection for partial least squares regression models. QSAR Comb. Sci. 2008, 27, 302–313. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, R.; Shao, C.; Hu, Z.; Wang, D; Yu, T.; Yan, X.; Wang, Y. Development and validation of a RP-HPLC method with fluorescence detection for simultaneous determination of 10-methoxycamptothecin and its metabolite 10-hydroxycamptothecin in rat plasma. J. Chromatogr. B 2012, 903, 81–87. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ding, W.; Sun, M.; Luo, S.; Xu, T.; Cao, Y.; Yan, X.; Wang, Y. A 3D QSAR Study of Betulinic Acid Derivatives as Anti-Tumor Agents Using Topomer CoMFA: Model Building Studies and Experimental Verification. Molecules 2013, 18, 10228-10241. https://doi.org/10.3390/molecules180910228
Ding W, Sun M, Luo S, Xu T, Cao Y, Yan X, Wang Y. A 3D QSAR Study of Betulinic Acid Derivatives as Anti-Tumor Agents Using Topomer CoMFA: Model Building Studies and Experimental Verification. Molecules. 2013; 18(9):10228-10241. https://doi.org/10.3390/molecules180910228
Chicago/Turabian StyleDing, Weimin, Miao Sun, Shaman Luo, Tao Xu, Yibo Cao, Xiufeng Yan, and Yang Wang. 2013. "A 3D QSAR Study of Betulinic Acid Derivatives as Anti-Tumor Agents Using Topomer CoMFA: Model Building Studies and Experimental Verification" Molecules 18, no. 9: 10228-10241. https://doi.org/10.3390/molecules180910228




