Synthesis, Evaluation of Antioxidant Activity and Crystal Structure of 2,4-Dimethylbenzoylhydrazones
Abstract
:1. Introduction
2. Results and Discussion
Chemistry
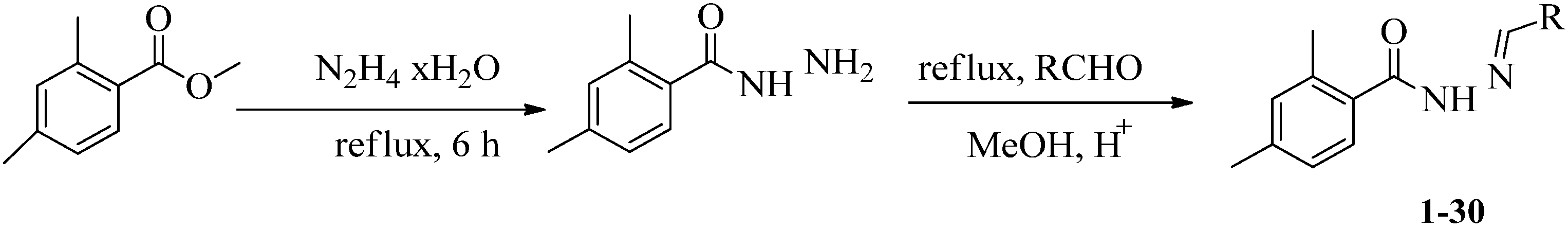
3. Antioxidant Activity
3.1. DPPH Scavenging Activity
| R | Comp. No. | R | |||||
|---|---|---|---|---|---|---|---|
| No. | (%) | (µM ± SEM a) | (%) | (µM ± SEM a) | |||
| 1 |  | 84 | 25.6 ± 0.5 | 10 |  | 88 | NA b |
| 2 |  | 82 | 29.3 ± 1.0 | 11 |  | 90 | 60.1 ± 2.2 |
| 3 |  | 78 | 29.8 ± 1.1 | 12 |  | 87 | 34.2 ± 2.1 |
| 4 |  | 84 | 28.14 ± 0.8 | 13 |  | 90 | NA b |
| 5 |  | 85 | 34.1 ± 1.0 | 14 |  | 87 | 52.2 ± 2.8 |
| 6 |  | 86 | 30.1 ± 1.3 | 15 |  | 82 | NA b |
| 7 |  | 81 | 30.0 ± 1.2 | 16 |  | 82 | NA b |
| 8 |  | 83 | 34.3 ± 1.5 | 17 |  | 84 | NA b |
| 9 |  | 92 | 40.0 ± 1.8 | 18 |  | 83 | NA b |
| 19 |  | 85 | NA b | 25 |  | 82 | NA b |
| 20 |  | 87 | 130.0 ± 4.8 | 26 |  | 88 | NA b |
| 21 |  | 88 | NA b | 27 |  | 90 | 190.0 ± 5.1 |
| 22 |  | 90 | NA b | 28 |  | 92 | NA b |
| 23 |  | 92 | NAb | 29 |  | 91 | 180.0 ± 4.6 |
| 24 |  | 90 | NAb | 30 |  | 84 | NAb |
| Standard drug n-propyl gallate c | 30.30 ± 0.2 | ||||||
3.2. Superoxide Scavenging Activity
| Comp. No. | IC50 (μM ± SEM a) | Comp. No. | IC50 (μM ± SEM a) |
|---|---|---|---|
| 1 | 98.3 ± 1.2 | 16 | NA b |
| 2 | 102.6 ± 1.5 | 17 | NA b |
| 3 | 105.6 ± 1.7 | 18 | NA b |
| 4 | 145 ± 2.1 | 19 | NA b |
| 5 | 170.2 ± 3.2 | 20 | 315.1 ± 8.4 |
| 6 | 175.0 ± 3.5 | 21 | NA b |
| 7 | 180.1 ± 3.8 | 22 | NA b |
| 8 | 190.1 ± 3.9 | 23 | NA b |
| 9 | 208.9 ± 5.4 | 24 | NA b |
| 10 | NA b | 25 | NA b |
| 11 | NA b | 26 | NA b |
| 12 | 210.1 ± 4.4 | 27 | NA b |
| 13 | NA b | 28 | NA b |
| 14 | 260.3 ± 6.4 | 29 | NA b |
| 15 | NA b | 30 | NA b |
| n-propyl gallate c | 106.34 ± 1.6 | ||
4. Experimental
4.1. General
4.2. X-ray Crystallography Studies
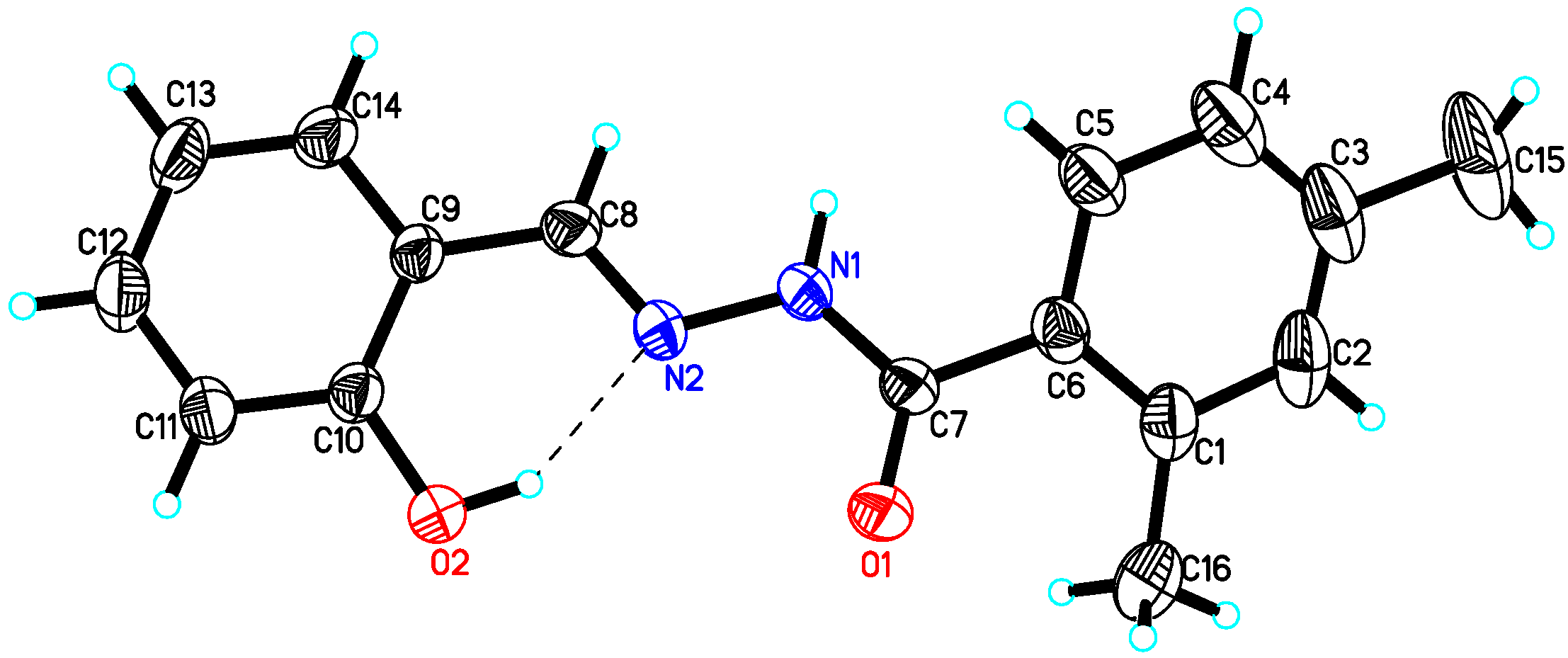
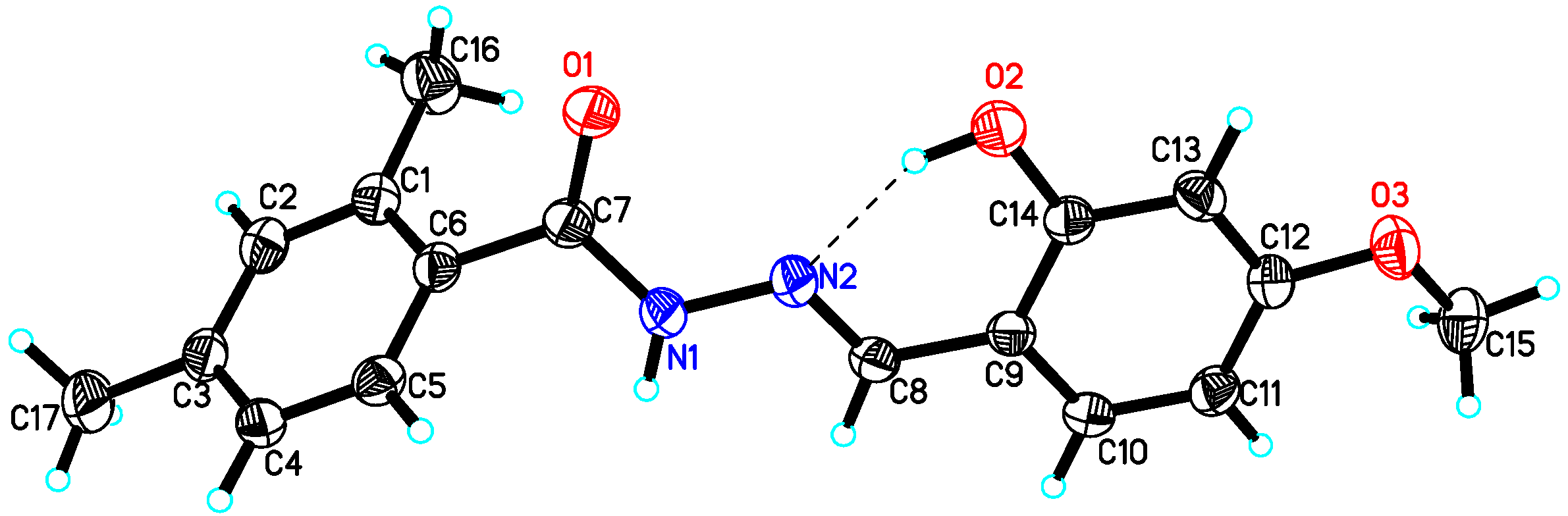
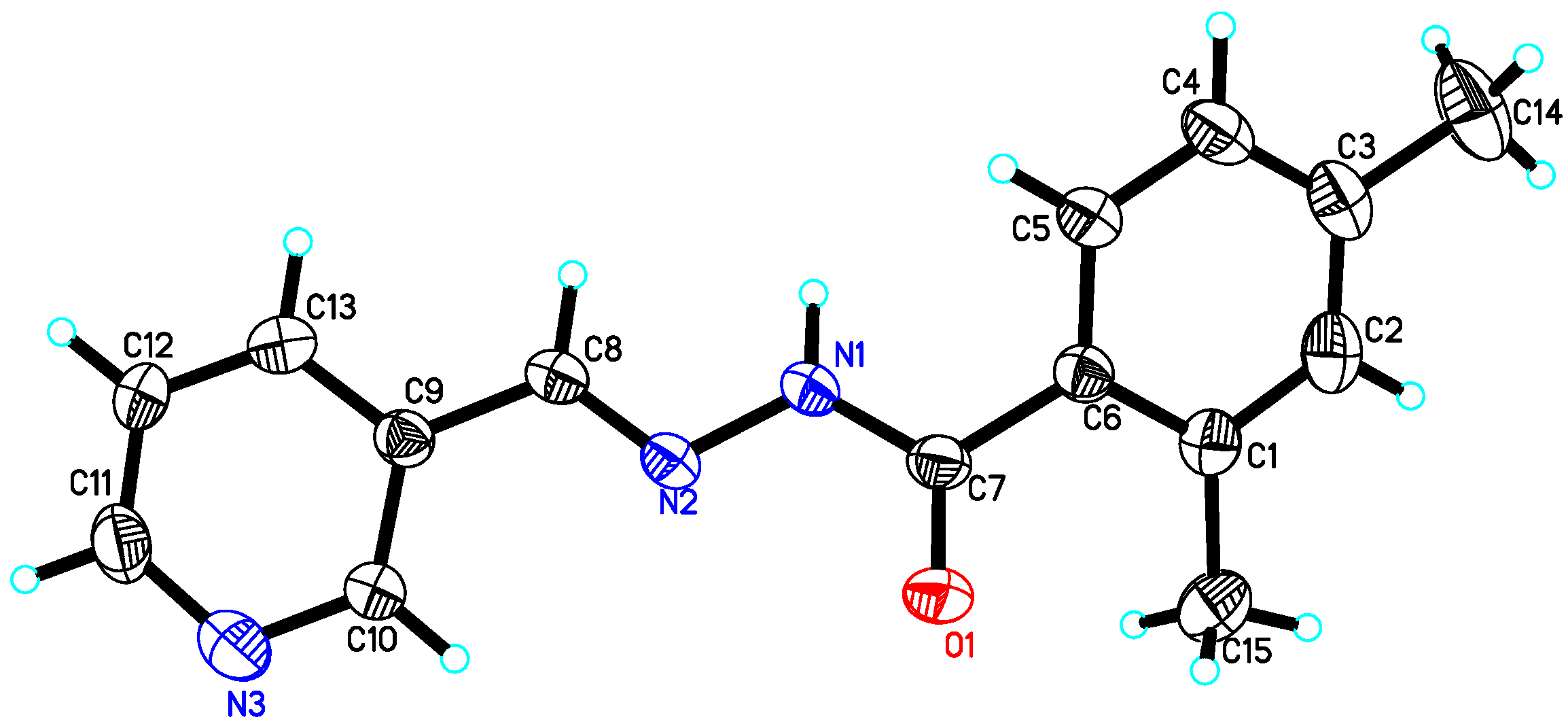
| Compound 10 | Compound 15 | Compound 22 | |
|---|---|---|---|
| Empirical formula | C16H16N2O2 | C17H18N2O3 | C15H15N1O3 |
| Formula weight | 268.31 | 298.33 | 253.30 |
| Temperature | 273(2) | 273(2) | 273(2) |
| Wavelength | 0.71073 Å | 0.71073 Å | 0.71073 Å |
| Crystal system | Monoclinic | Monoclinic | Monoclinic |
| Space group | P21/c | P21/n | P21/c |
| A | 12.7293(17) Å | 11.1984(9) Å | 14.0960(14) Å |
| B | 13.0700(17) Å | 10.0703(9) Å | 11.8440(12) A |
| C | 8.8762(12) Å | 13.8665(12) Å | 8.2813(8) Å |
| β | 94.845(3)° | 102.535(2)° | 100.889(2)° |
| Volume | 1,463.2(3) A3 | 1,526.5(2) A3 | 1,357.7(2) A3 |
| Z | 4 | 4 | 4 |
| Calculated density | 1.218 mg/m3 | 1.298 mg/m3 | 1.239 mg/m3 |
| Absorption coefficient | 0.082 mm−1 | 0.090 mm−1 | 0.080 mm−1 |
| F(000) | 568 | 632 | 536 |
| Crystal size | 0.52 × 0.12 × 0.11 mm | 0.56 × 0.43 × 0.12 mm | 0.56 × 0.18 × 0.13 mm |
| θ range | 2.24 to 25.49° | 2.13 to 25.49° | 1.47 to 25.40° |
| Reflections Collected | 8546 | 8849 | 7827 |
| Reflections Unique | 2713 | 2758 | 2522 |
| (Rint) | 0.0275 | 0.0212 | 0.0313 |
| R1 with I > 2σ(I) | 0.0463 | 0.0438 | 0.0536 |
| R2 with I > 2σ(I) | 0.1124 | 0.1248 | 0.1508 |
| R1 for all data | 0.0774 | 0.0520 | 0.0702 |
| R2 for all data | 0.1323 | 0.1337 | 0.1607 |
| Goodness of fit | 1.030 | 1.057 | 1.014 |
| max/min ρ eA˚−3 | 0.140 and −0.110 | 0.232 and −0.018 | 0.265 and −0.258 |
4.3. DPPH (1,1-Diphenyl-2-picryl hydrazyl) Free Radical Scavenging Activity
4.4. In Vitro Assay for Superoxide Anion Radical Scavenging Activity
4.5. General Procedure for the Synthesis of 2,4-Dimethylbenzohydrazide
4.6. General Procedure for the Synthesis of 2,4-Dimethylbenzohydrazones
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Tsai, M.D.; Herschel, J.; Weintraub, R.; Byrn, S.R.; Chang, C.; Floss, H.G. Conformation-reactivity relationship for pyridoxal Schiff’s bases. Rates of racemization and alpha-hydrogen exchange of the pyridoxal Schiff’s bases of amino acids. Biochemistry 1978, 17, 3183–3188. [Google Scholar] [CrossRef]
- Schirch, L.; Slotter, R.A. Spectral properties of Schiff bases of amino acid esters with pyridoxal and pyridoxal N-methochloride in ethanol. Biochemistry 1966, 5, 3175–3181. [Google Scholar] [CrossRef]
- Popp, F.D.; Kirsch, W.J. Synthesis of potential anticancer agents. V. Schiff bases and related compounds1–2. J. Org. Chem. 1961, 26, 3858–3860. [Google Scholar] [CrossRef]
- Kumar, S.; Niranjan, M.S.; Chaluvaraju, K.C.; Jamakhandi, C.M.; Kadadevar, D. Synthesis and antimicrobial study of some Schiff bases of sulfonamides. J. Curr. Pharm. Res. 2010, 1, 39–42. [Google Scholar]
- Mallikarjun, S.Y.; Sangamesh, A.P. Synthesis, characterization and biological studies of cobalt(II) and nickel(II) complexes with new Schiff bases. Trans. Met. Chem. 1997, 22, 220–224. [Google Scholar] [CrossRef]
- Akelah, A.; Kenawy, E.R.; Sherrington, D.C. Agricultural polymers with herbicide/fertilizer function-III. Polyureas and poly(Schiff base)s based systems. Eur. Polym. J. 1993, 29, 1041–1045. [Google Scholar] [CrossRef]
- Jain, J.S.; Srivastava, R.S.; Aggarwal, N.; Sinha, R. Synthesis and evaluation of Schiff bases for anticonvulsant and behavioral depressant properties. Cent. Nerv. Syst. Agents Med. Chem. 2007, 7, 200–204. [Google Scholar] [CrossRef]
- Fareed, G.; Versiani, M.A.; Afza, N.; Fareed, N.; Iqbal, L.; Lateef, M. Structure activity relationship: Antioxidant potential of some novel Schiff bases containing benzophenone moiety. Int. J. Curr. Pharm. Res. 2013, 5, 61–64. [Google Scholar]
- Mishra, P.; Gupta, P.N.; Shakya, A.K.; Shukla, R.; Srimal, R.C. Anti-inflammatory and diuretic activity of a new class of compounds—Schiff bases of 3-amino-2-methylquinazolin 4(3H)-ones. Indian J. Physiol. Pharmacol. 1995, 39, 169–172. [Google Scholar]
- Supuran, C.T.; Barboiu, M.; Luca, C.; Pop, E.; Brewster, M.E.; Dinculescu, A. Carbonic anhydrase activators. Part 14. Syntheses of mono and bis pyridinium salt derivatives of 2-amino-5-(2-aminoethyl)- and 2-amino-5-(3-aminopropyl)-1,3,4-thiadiazole and their interaction with isozyme II. Eur. J. Med. Chem. 1996, 31, 597–606. [Google Scholar] [CrossRef]
- Tarafder, M.T.; Kasbollah, A.; Saravanan, N.; Crouse, K.A.; Ali, A.M.; OoK, T. S-Methyldithiocarbazate and its Schiff bases: Evaluation of bondings and biological properties. Biochem. Mol. Bio. Biophy. 2002, 6, 85–91. [Google Scholar] [CrossRef]
- Walcourt, A.; Loyevsky, M.; Lovejoy, D.B.; Gordeuk, V.R.; Richardson, D.R. Novel aroylhydrazone and thiosemicarbazone iron chelators with anti-malarial activity against chloroquine-resistant and -sensitive parasites. Int. J. Biochem. Cell Biol. 2004, 36, 401–407. [Google Scholar] [CrossRef]
- Vicini, P.; Geronikaki, A.; Incerti, M.; Busonera, B.; Poni, G.; Cabras, C.A.; Colla, P.L. Synthesis and biological evaluation of benzo[d]isothiazole, benzothiazole and thiazole Schiff bases. Bioorg. Med. Chem. 2003, 11, 4785–4789. [Google Scholar] [CrossRef]
- Andreani, A.; Rambaldi, M.; Bonazzi, D.; Greci, L.; Andreani, F. Study on compounds with potential antitumor activity. III. Hydrazone derivatives of 5-substituted 2-chloro-3-formyl-6-methylindole. Farmaco Sci. 1979, 34, 132–138. [Google Scholar]
- Gaur, S. Physico-chemical and Biological properties of Mn(II), Co(II), Ni(II) and Cu(II) chelates of Schiff Bases. Asian J. Chem. 2003, 15, 250–254. [Google Scholar]
- Gemi, M.J.; Biles, C.; Keiser, B.J.; Poppe, S.M.; Swaney, S.M.; Tarapley, W.G.; Romeso, D.L.; Yage, Y. Novel 1,5-diphenylpyrazole nonnucleoside HIV-1 reverse transcriptase inhibitors with enhanced activity versus the delavirdine-resistant P236L mutant: Lead identification and SAR of 3- and 4-substituted derivatives. J. Med. Chem. 2000, 43, 1034–1040. [Google Scholar] [CrossRef]
- Al-Amiery, A.; Al-Majedy, Y.K.; Ibrahim, H.H.; Al-Tamimi, A. Antioxidant, antimicrobial, and theoretical studies of the thiosemicarbazone derivative Schiff base 2-(2-imino-1-methylimidazolidin-4-ylidene)hydrazinecarbothioamide (IMHC). Org. Med. Chem. Lett. 2012, 2, 4. [Google Scholar] [CrossRef]
- Corona, A.B.; Viveros, J.P.; Flores, A.P.; Peraza, A.C.; Martínez, J.M.; Sumaya, M.M.; Organillo, R. Antioxidant activity of butyl- and phenylstannoxanes derivedfrom 2-, 3- and 4-pyridinecarboxylic acids. Molecules 2010, 15, 5445–5459. [Google Scholar] [CrossRef]
- Dharmaraj, N.; Viswanathamurthi, P.; Natarajan, K. Ruthenium(II) complexes containing bidentate Schiff bases and their antifungal activity. Trans. Met. Chem. 2001, 26, 105–109. [Google Scholar] [CrossRef]
- Ozdemir, A.; Turan-Zitouni, G.; Kaplancikli, Z.A.; Demirci, F.; Iscan, G. Studies on hydrazone derivatives as antifungal agents. J. Enzyme Inhib. Med. Chem. 2008, 23, 470–475. [Google Scholar] [CrossRef]
- Ergenç, N.; Günay, N.S.; Demirdamar, R. Synthesis and antidepressant evaluation of new 3-phenyl-5-sulfonamideindole derivatives. Eur. J. Med. Chem. 1998, 33, 143–148. [Google Scholar] [CrossRef]
- Silva, A.G.; Zapata-Suto, G.; Kummerle, A.E.; Fraga, C.A.M.; Barreiro, E.J.; Sudo, R.T. Synthesis and vasodilatory activity of new N-acylhydrazone derivatives, designed as LASSBio-294 analogues. Bioorg. Med. Chem. 2005, 13, 3431–3437. [Google Scholar] [CrossRef]
- Cavier, R.; Rips, R. Dihydrazides, a new class of anthelmintics. J. Med. Chem. 1965, 8, 706–708. [Google Scholar] [CrossRef]
- Rollas, S.; Kuçukguzel, S.G. Biological activities of hydrazone derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar] [CrossRef]
- Lima, P.C.; Lima, L.M.; Silva, K.C.; Leda, P.H.; Miranda, A.L.P.; Fraga, C.A.M.; Barreiro, E.J. Synthesis and analgesic activity of novel N-acylarylhydrazones and isosters, derived from natural safrole. Eur. J. Med. Chem. 2000, 35, 187–203. [Google Scholar] [CrossRef]
- Fraga, A.G.M.; Rodrigues, C.R.; Miranda, A.L.P.; Barreiro, E.J.; Fraga, C.A.M. Synthesis and pharmacological evaluation of novel heterotricyclic acylhydrazone derivatives, designed as PAF antagonists. Eur. J. Pharm. Sci. 2000, 11, 285–290. [Google Scholar] [CrossRef]
- Satyanarayana, V.S.V.; Sivakumar, A.; Ghosh, A.R. Synthesis, characterization of some new five membered heterocycles based on imidazole moiety and their applications on therapeutics. Lett. Drug. Des. Discov. 2011, 8, 276–283. [Google Scholar] [CrossRef]
- Tumer, M.; Toroglu, E.S.A.; Kayraldiz, A.; Donbak, L. Synthesis and characterization of Schiff base metal complexes: Their antimicrobial, genotoxicity and electrochemical properties. J. Coord. Chem. 2008, 61, 2935–2949. [Google Scholar] [CrossRef]
- Barton, D.H.R.; Yadav-Bhatnagar, N.; Finet, J.P.; Khamsi, J. Phenylation of aromatic and aliphatic amines by phenyllead triacetate using copper catalysis. Tetrahedron Lett. 1987, 28, 3111–3114. [Google Scholar] [CrossRef]
- Aranha, P.E.; Dos Santos, M.P.; Romera, S.; Dockal, E.R. Synthesis, characterization, and spectroscopic studies of tetradentate Schiff base chromium(III) complexes. Polyhedron 2007, 26, 1373–1382. [Google Scholar] [CrossRef]
- Prakash, A.; Adhikari, D. Application of Schiff bases and their metal complexes—A review. Int. J. Chem. Tech. Res. 2011, 3, 1891–1896. [Google Scholar]
- Capasso, R.; Evidente, A.; Tremblay, E.; Sala, A.; Santoro, C.; Cristinzio, G. Direct and mediated effects on Bactrocera oleae (Gmelin) (Diptera; Tephritidae) of natural polyphenols and some of related synthetic compounds: Structure-activity relationships. J. Chem. Ecol. 1994, 20, 1189–1199. [Google Scholar] [CrossRef]
- Sun-Waterhouse, D.; Chen, J.; Chuah, C.; Wibisono, R.; Melton, L.D.; Laing, W.; Ferguson, L.R.; Skinner, M.A. Kiwifruit-based polyphenols and related antioxidants for functional foods: Mkiwifruit extract-enhabnced gluten-free bread. Int. J. Food Sci. Nutr. 2009, 60, 251–264. [Google Scholar] [CrossRef]
- Taha, M.; Baharudin, M.S.; Ismail, N.H.; Khan, K.M.; Jaafar, F.M.; Samreen; Siddiqui, S.; Choudhary, M.I. Synthesis of 2-methoxybenzoylhydrazone and evaluation of their antileishmanial activity. Bioorg. Med. Chem. Lett. 2013, 23, 3463–3466. [Google Scholar] [CrossRef]
- Khan, K.M.; Taha, M.; Rahim, F.; Fakhri, M.I.; Jamil, W.; Khan, M.; Rasheed, S.; Karim, A.; Perveen, S.; choudhary. Acylhydrazide Schiff Bases: Synthesis and Antiglycation Activity. J. Pak. Chem Soc. 2013, 35, 929–937. [Google Scholar]
- Khan, K.M.; Rahim, F.; Ambreen, N.; Taha, M.; Khan, M.; Jahan, H.; Najeebullah; Shaikh, A.; Iqbal, S.; Perveen, S.; Choudhary, M.I. Synthesis of Benzophenonehydrazone Schiff Bases and their In Vitro Antiglycating Activities. Med. Chem. 2013, 9, 588–595. [Google Scholar] [CrossRef]
- Taha, M.; Ismail, N.H.; Jaafar, F.M.; Aziz, A.N.; Yousuf, S. (E)-N'-(3,4-Dihydroxybenzylidene)-2,4-dimethylbenzohydrazide monohydrate. Acta Cryst. 2013, E69, o490. [Google Scholar]
- Baharudin, M.S.; Taha, M.; Ismail, N.H.; Shah, S.A.A.; Yousuf, S. (E)-N'-(4-Chlorobenzylidene)-2-methoxybenzohydrazide. Acta Cryst. 2013, E69, o276. [Google Scholar]
- Taha, M.; Baharudin, M.S.; Ismail, N.H.; Shah, S.A.A.; Yousuf, S. (E)-2-Methoxy-N'-(2,4,6-trihydroxybenzylidene)Benzohydrazide. Acta Cryst. 2013, E69, o277. [Google Scholar]
- Taha, M.; Ismail, N.H.; Jaafar, F.M.; Khan, K.M.; Yousuf, S. (E)-2,4-Dimethyl-N'-(2-methylbenzylidene) benzohydrazide. Acta Cryst. 2013, E69, o400. [Google Scholar]
- Baharudin, M.S.; Taha, M.; Ismail, N.H.; Shah, S.A.A.; Yousuf, S. N-[(E)-2-Hydroxy-5-methoxybenzylidene]-2-methoxybenzohydra-zide. Acta Cryst. 2012, E68, o3255. [Google Scholar]
- Taha, M.; Baharudin, M.S.; Ismail, N.H.; Shah, S.A.A.; Yousuf, S. N'-[(E)-2,3-Dihydroxybenzylidene]-2-methoxybenzohydrazide. Acta Cryst. 2012, E68, o3256. [Google Scholar]
- Khan, K.M.; Shah, Z.; Ahmad, V.U.; Khan, M.; Taha, M.; Ali, S.; Perveen, S.; Choudhary, M.I.; Voelter, W. 2,4,6-Trichlorophenylhydrazine Schiff bases as DPPH radical and super oxide anion scavengers. Med. Chem. 2012, 8, 452–461. [Google Scholar] [CrossRef]
- Khan, K.M.; Taha, M.; Naz, F.; Ali, S.; Perveen, S.; Choudhary, M.I. Acylhydrazide Schiff bases: DPPH radical and superoxide anion scavengers. Med. Chem. 2012, 8, 705–710. [Google Scholar] [CrossRef]
- Khan, K.M.; Khan, M.; Ali, M.; Taha, M.; Hameed, A.; Ali, S.; Perveen, S.; Choudhary, M.I. Synthesis and DPPH radical scavenging activity of 5-Arylidene-N,NDimethylbarbiturates. Med. Chem. 2011, 7, 231–236. [Google Scholar] [CrossRef]
- Siemens SMART and SAINT; Siemens Analytical X-ray Instruments Inc.: Madison, WI, USA, 1996.
- Sheldrick, G.M. A Program for Refinement of Crystal Structures; SHELXL 97: Göttingen, Germany, 1997. [Google Scholar]
- Johnson, C.K. ORTEP II. In Report ORNL-5138; Oak Ridge National Laboratory: Tennessee, USA, 1976. [Google Scholar]
- Sample Availability: Contact authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Taha, M.; Ismail, N.H.; Jamil, W.; Yousuf, S.; Jaafar, F.M.; Ali, M.I.; Kashif, S.M.; Hussain, E. Synthesis, Evaluation of Antioxidant Activity and Crystal Structure of 2,4-Dimethylbenzoylhydrazones. Molecules 2013, 18, 10912-10929. https://doi.org/10.3390/molecules180910912
Taha M, Ismail NH, Jamil W, Yousuf S, Jaafar FM, Ali MI, Kashif SM, Hussain E. Synthesis, Evaluation of Antioxidant Activity and Crystal Structure of 2,4-Dimethylbenzoylhydrazones. Molecules. 2013; 18(9):10912-10929. https://doi.org/10.3390/molecules180910912
Chicago/Turabian StyleTaha, Muhammad, Nor Hadiani Ismail, Waqas Jamil, Sammer Yousuf, Faridahanim Mohd Jaafar, Muhammad Imran Ali, Syed Muhammad Kashif, and Ejaz Hussain. 2013. "Synthesis, Evaluation of Antioxidant Activity and Crystal Structure of 2,4-Dimethylbenzoylhydrazones" Molecules 18, no. 9: 10912-10929. https://doi.org/10.3390/molecules180910912





