Dissipation and Migration of Pyrethroids in Auricularia polytricha Mont. from Cultivation to Postharvest Processing and Dietary Risk
Abstract
:1. Introduction
2. Results and Discussion
2.1. Method Validation
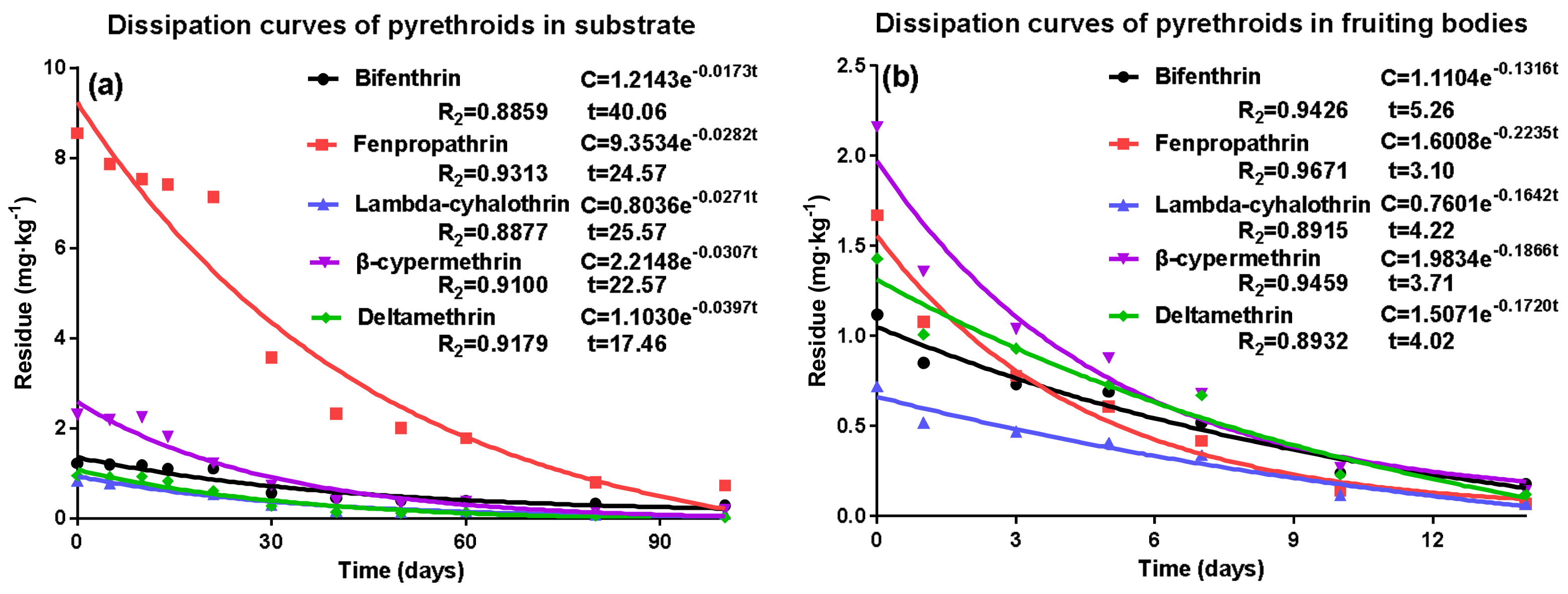
2.2. Dissipation of Pyrethroid Residues in Substrate and Fruiting Bodies
2.3. Terminal Residues of Pyrethroids in A. polytricha
2.4. Migration of Pyrethroids in the Substrate-Mushroom System
2.5. Effects of Processing
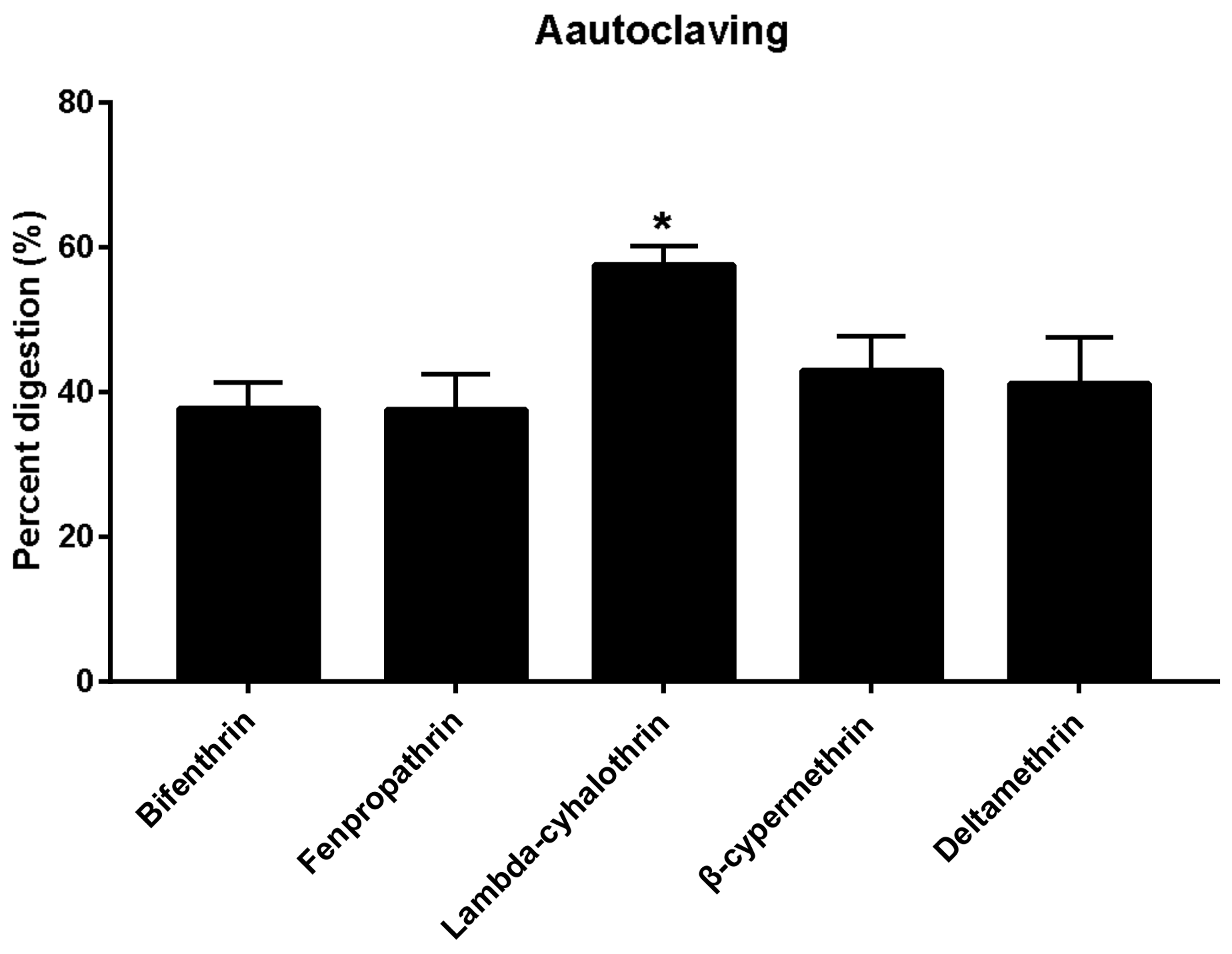
2.5.1. Effects of Autoclaving on the Residue Dissipation Behaviors
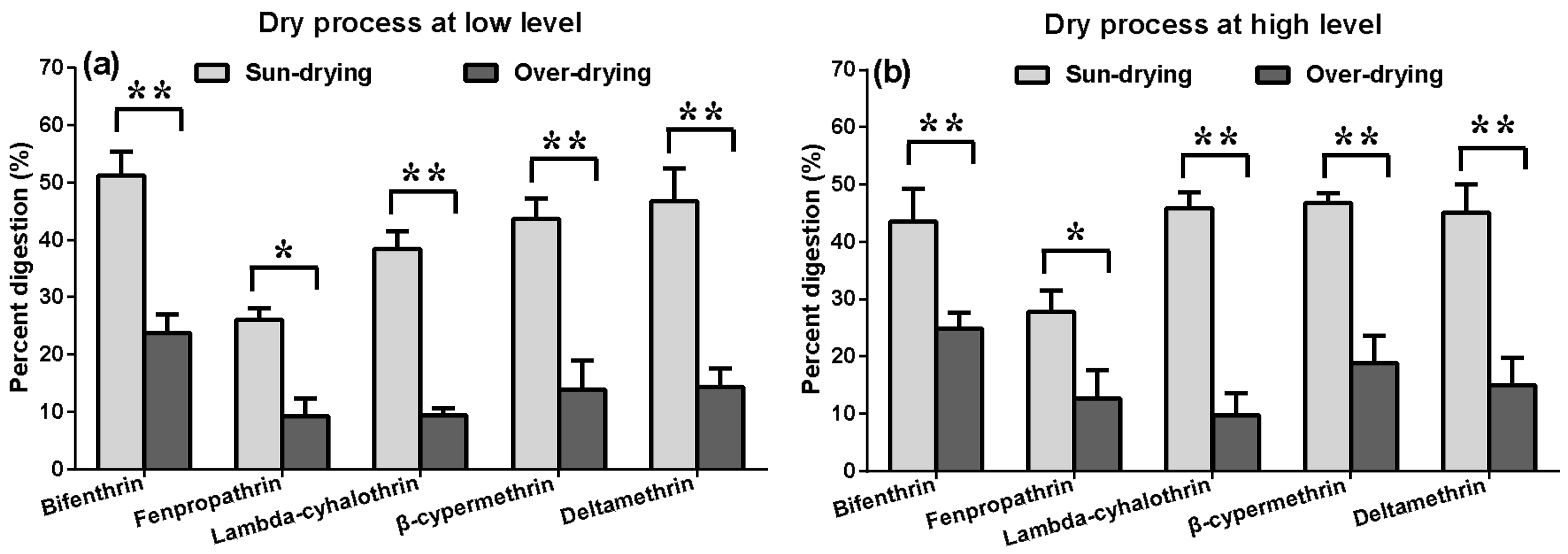
2.5.2. Effects of Two Drying Methods on the Residue Dissipation Behaviors
2.5.3. Effects of Storage on the Residue Dissipation Behaviors
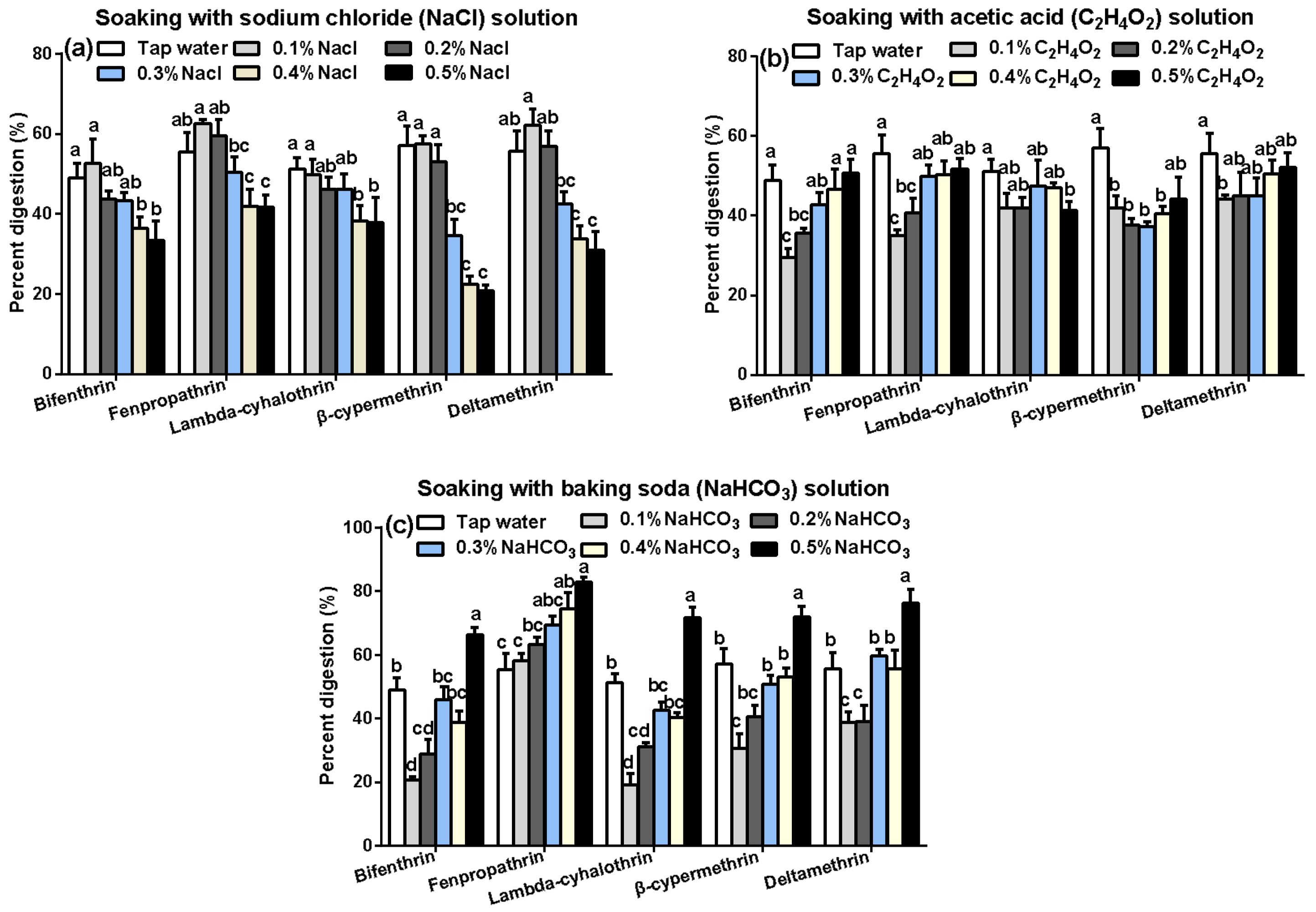
2.5.4. Effects of Soaking on the Residue Dissipation Behaviors
2.6. Processing Factors and Dietary Risk
3. Materials and Methods
3.1. Chemicals and Samples
3.2. Cultivation Conditions, Pest Surveys, and Field Trials
3.2.1. Pesticides for Trials of Premixing Treatment
3.2.2. Pesticides for Trials of Spraying Treatment
3.3. Postharvest Processing and Home-Soaking Experiments
3.4. Gas Chromatography (GC) Analysis
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liang, C.-H.; Wu, C.-Y.; Lu, P.-L.; Kuo, Y.-C.; Liang, Z.-C. Biological efficiency and nutritional value of the culinary-medicinal mushroom auricularia cultivated on a sawdust basal substrate supplement with different proportions of grass plants. Saudi J. Biol. Sci. 2016. [Google Scholar] [CrossRef]
- Kalac, P. Trace element contents in European species of wild growing edible mushrooms: A review for the period 2000–2009. Food Chem. 2010, 122, 2–15. [Google Scholar] [CrossRef]
- Cao, X.; Liu, S.; Yang, X.; Liu, Z.; Liu, L. A modified quechers sample preparation method for simultaneous determination of 62 pesticide residues in edible fungi using gas chromatography–triple quadrupole mass spectrometry. Food Anal. Methods 2016, 9, 263–274. [Google Scholar] [CrossRef]
- Wieland, M.; Hacker, K.; Bauer, N.; Schüle, E.; Wauschkuhn, C.; Roux, D.; Scherbaum, E.; Anastassiades, M. Pesticide Residues in Fresh and Dried Mushrooms on the German Market. Chemisches und Veterinaruntersuchungsamt, Stuttgart, Baden-Wü Rttemberg, Germany. Available online: http://cvusa.xn-untersuchungsmter-bw-nzb.de/pdf/MushroomPM036.pdf (accessed on 20 June 2010).
- Li, Y.J.; Ishikawa, Y.; Satake, T.; Kitazawa, H.; Qiu, X.L.; Rungchang, S. Effect of active modified atmosphere packaging with different initial gas compositions on nutritional compounds of shiitake mushrooms (Lentinus edodes). Postharvest Biol. Technol. 2014, 92, 107–113. [Google Scholar] [CrossRef]
- Ramezani, M.K.; Shahriari, D. Dissipation behaviour, processing factors and risk assessment for metalaxyl in greenhouse-grown cucumber. Pest Manag. Sci. 2015, 71, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Cruz, M.L.; Barreda, M.; Villarroya, M.; Peruga, A.; Llanos, S.; Garcia-Baudin, J.M. Captan and fenitrothion dissipation in field-treated cauliflowers and effect of household processing. Pest Manag. Sci. 2006, 62, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Caldas, E.D.; Tressou, J.; Boon, P.E. Dietary exposure of Brazilian consumers to dithiocarbamate pesticides—A probabilistic approach. Food Chem. Toxicol. 2006, 44, 1562–1571. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.W.; Liu, F.M.; Wu, L.M.; Xue, X.F.; Hou, F. Fate of triadimefon and its metabolite triadimenol in jujube samples during jujube wine and vinegar processing. Food Control 2017, 73, 468–473. [Google Scholar] [CrossRef]
- Fan, Z.-C.; Tang, J.; Cao, H.-Q.; Wu, X.-W.; Hua, R.-M.; Tang, F.; Yue, Y.-D. Degradation kinetics of 5 pyrethroid pesticide residues in pleurotus ostreatus and growing medium. Food Sci. 2013, 11, 009. [Google Scholar]
- Domingues, V.; Alves, A.; Cabral, M.; Delerue-Matos, C. Sorption behaviour of bifenthrin on cork. J. Chromatogr. A 2005, 1069, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Liao, M.; Shi, Y.H.; Cao, H.Q.; Hua, R.M.; Tang, F.; Wu, X.W.; Tang, J. Dissipation behavior of octachlorodipropyl ether residues during tea planting and brewing process. Environ. Monit. Assess. 2016, 188, 551. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.F.; Ross, D.G.; Starr, J.M.; Scollon, E.J.; Wolansky, M.J.; Crofton, K.M.; DeVito, M.J. Environmentally relevant pyrethroid mixtures: A study on the correlation of blood and brain concentrations of a mixture of pyrethroid insecticides to motor activity in the rat. Toxicology 2016, 359, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, J.; Kaneko, H.; Tsuji, R.; Okuno, Y. Pyrethroids, nerve poisons: How their risks to human health should be assessed. Toxicol. Lett. 1995, 82, 933–940. [Google Scholar] [CrossRef]
- Barik, S.R.; Ganguly, P.; Kunda, S.K.; Kole, R.K.; Bhattacharyya, A. Persistence behaviour of thiamethoxam and lambda cyhalothrin in transplanted paddy. Bull. Environ. Contam. Toxicol. 2010, 85, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Dordevic, T.M.; Siler-Marinkovic, S.S.; Durovic, R.D.; Dimitrijevic-Brankovic, S.I.; Umiljendic, J.S.G. Stability of the pyrethroid pesticide bifenthrin in milled wheat during thermal processing, yeast and lactic acid fermentation, and storage. J. Sci. Food Agric. 2013, 93, 3377–3383. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.P.; Sun, Y.; Gao, Y.; Xu, B.; Wu, Q.; Zhang, H.Q.; Song, D.Q. Determination of five pyrethroids in tea drinks by dispersive solid phase extraction with polyaniline-coated magnetic particles. Talanta 2014, 119, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Cabras, P.; Angioni, A.; Garau, L.; Melis, M.; Pirisi, F.M.; Cabitza, F.; Pala, M. Pesticide residues in raisin processing. J. Agric. Food Chem. 1998, 46, 2309–2311. [Google Scholar] [CrossRef]
- Ouzouni, P.K.; Veltsistas, P.G.; Paleologos, E.K.; Riganakos, K.A. Determination of metal content in wild edible mushroom species from regions of Greece. J. Food Compos. Anal. 2007, 20, 480–486. [Google Scholar] [CrossRef]
- Frangos, P.; Shen, W.H.; Wang, H.J.; Li, X.; Yu, G.; Deng, S.B.; Huang, J.; Wang, B.; Wang, Y.J. Improvement of the degradation of pesticide deethylatrazine by combining UV photolysis with electrochemical generation of hydrogen peroxide. Chem. Eng. J. 2016, 291, 215–224. [Google Scholar] [CrossRef]
- Clark, T.; Watkins, D.A.M.; Weerasinghe, D.K. Photolysis of fluotrimazole. Pestic. Sci. 1983, 14, 449–452. [Google Scholar] [CrossRef]
- Noh, H.H.; Kim, D.K.; Lee, E.Y.; Chang, M.I.; Im, M.H.; Lee, Y.D.; Kyung, K.S. Effects of oven drying on pesticide residues in field-grown chili peppers. J. Korean Soc. Appl. Biol. 2015, 58, 97–104. [Google Scholar] [CrossRef]
- Kujawski, M.W.; Namiesnik, J. Challenges in preparing honey samples for chromatographic determination of contaminants and trace residues. Trac-Trend Anal. Chem. 2008, 27, 785–793. [Google Scholar] [CrossRef]
- Chen, H.P.; Pan, M.L.; Pan, R.; Zhang, M.L.; Liu, X.; Lu, C.Y. Transfer rates of 19 typical pesticides and the relationship with their physicochemical property. J. Agric. Food Chem. 2015, 63, 723–730. [Google Scholar] [CrossRef] [PubMed]
- British Crop Protection Council. The Pesticide Manual: A World Compendium, 11th ed.; Tomlin, C.D.S., Ed.; British Crop Protection Council: Alton, UK, 1997. [Google Scholar]
- Kong, Z.Q.; Dong, F.S.; Xu, J.; Liu, X.G.; Zhang, C.P.; Li, J.; Li, Y.B.; Chen, X.; Shan, W.L.; Zheng, Y.Q. Determination of difenoconazole residue in tomato during home canning by UPLC-MS/MS. Food Control 2012, 23, 542–546. [Google Scholar] [CrossRef]
- Matsuo, N.; Miyamoto, J. Development of Synthetic Pyrethroids with Emphasis on Stereochemical Aspects; ACS Publications: Washington, DC, USA, 1997. [Google Scholar]
- Radwan, M.A.; Abu-Elamayem, M.M.; Shiboob, M.H.; Abdel-Aal, A. Residual behaviour of profenofos on some field-grown vegetables and its removal using various washing solutions and household processing. Food Chem. Toxicol. 2005, 43, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Hand, L.H.; Kuet, S.F.; Lane, M.C.G.; Maund, S.J.; Warinton, J.S.; Hill, I.R. Influences of aquatic plants on the fate of the pyrethroid insecticide lambida-cyhalothrin in aquatic environments. Environ. Toxicol. Chem. 2001, 20, 1740–1745. [Google Scholar] [PubMed]
- Wang, C.-F.; Chen, F.; Wei, H.; You, Y.; Xiu, D.-M. Removing methods for β-cypermethrin residue in contaminated vegetables. J. Agro-Environ. Sci. 2005, 24, 1018–1021. [Google Scholar]
- Peng, W.; Zhao, L.W.; Liu, F.M.; Xue, J.Y.; Li, H.C.; Shi, K.W. Effect of paste processing on residue levels of imidacloprid, pyraclostrobin, azoxystrobin and fipronil in winter jujube. Food Addit. Contam. A 2014, 31, 1562–1567. [Google Scholar] [CrossRef] [PubMed]
- Reiler, E.; Jors, E.; Baelum, J.; Huici, O.; Caero, M.M.A.; Cedergreen, N. The influence of tomato processing on residues of organochlorine and organophosphate insecticides and their associated dietary risk. Sci. Total Environ. 2015, 527, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Phan, C.W.; Sabaratnam, V. Potential uses of spent mushroom substrate and its associated lignocellulosic enzymes. Appl. Microbiol. Biotechnol. 2012, 96, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.-H.; Xiao, J.-J.; Feng, R.-P.; Liu, Y.-Y.; Liao, M.; Wu, X.-W.; Hua, R.-M.; Cao, H.-Q. In-vitro bioaccessibility of five pyrethroids after human ingestion and the corresponding gastrointestinal digestion parameters: A contribution for human exposure assessments. Chemosphere 2017, 182, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.N.; Sharma, D. Power of tukey’s test for non-additivity. J. R. Stat. Soc. Ser. B 1963, 25, 213–219. [Google Scholar]
- World Health Organization (WHO); Food and Agriculture Organization of the United Nations (FAO). Updating, the Principles and Methods of Risk Assessment Mrls for Pesticidas and Veterinary Drugs; FAO: Rome, Italy, 2006. [Google Scholar]
- Timme, G.; Walz-Tylla, B. Effects of food preparation and processing on pesticide residues in commodities of plant origin. In Pesticide Residues Food Drinking Water; John Wiley & Sons: Hoboken, NJ, USA, 2004; pp. 121–148. [Google Scholar]


| Pesticides | Fortified Level (mg·kg−1) | Dried Mushroom | Fresh Mushroom | Fortified Level (mg·kg−1) | Substrate | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Average Recovery (%) a | RSD (%) b | LQD/LOQ (μg·kg−1) | Average Recovery (%) | RSD (%) | LQD/LOQ (μg·kg−1) | Average Recovery (%) | RSD (%) | LQD/LOQ (μg·kg−1) | |||
| Bifenthrin | 0.005 | 82.51 ± 3.65 c | 3.99 | 0.91/2.93 | 95.02 ± 5.74 | 2.82 | 0.86/2.87 | 0.01 | 82.66 ± 4.01 | 2.58 | 0.85/2.44 |
| 0.05 | 76.89 ± 2.54 | 1.76 | 90.42 ± 4.52 | 3.64 | 0.1 | 93.93 ± 4.15 | 4.33 | ||||
| 0.5 | 83.56 ± 4.12 | 0.49 | 81.68 ± 3.25 | 2.30 | 1 | 88.52 ± 3.87 | 2.76 | ||||
| Fenpropathrin | 0.005 | 103.09 ± 5.31 | 2.16 | 0.75/2.61 | 94.71 ± 3.34 | 3.89 | 0.79/2.64 | 0.01 | 101.02 ± 3.65 | 1.67 | 0.66/2.55 |
| 0.05 | 80.44 ± 4.33 | 2.78 | 90.60 ± 4.26 | 4.35 | 0.1 | 93.22 ± 3.77 | 2.00 | ||||
| 0.5 | 82.66 ± 3.87 | 0.95 | 85.30 ± 3.39 | 3.45 | 1 | 87.93 ± 2.96 | 2.19 | ||||
| Lambda-cyhalothrin | 0.005 | 80.12 ± 3.26 | 1.72 | 0.50/1.62 | 85.96 ± 3.46 | 2.92 | 0.53/1.77 | 0.01 | 101.01 ± 3.82 | 2.49 | 0.50/1.61 |
| 0.05 | 76.04 ± 2.57 | 1.01 | 93.24 ± 4.55 | 3.73 | 0.1 | 96.74 ± 3.54 | 3.73 | ||||
| 0.5 | 90.13 ± 4.57 | 1.71 | 96.39 ± 4.12 | 1.87 | 1 | 87.07 ± 2.99 | 2.21 | ||||
| β-cypermethrin | 0.005 | 98.63 ± 5.44 | 3.71 | 1.60/5.34 | 84.73 ± 1.41 | 4.30 | 1.61/5.34 | 0.01 | 102.83 ± 5.54 | 2.87 | 1.63/5.37 |
| 0.05 | 98.13 ± 4.75 | 2.31 | 87.16 ± 3.25 | 3.26 | 0.1 | 94.22 ± 4.93 | 1.11 | ||||
| 0.5 | 82.67 ± 3.52 | 0.61 | 89.61 ± 5.75 | 2.35 | 1 | 87.07 ± 4.43 | 2.45 | ||||
| Deltamethrin | 0.005 | 93.57 ± 5.11 | 2.26 | 1.39/4.68 | 90.49 ± 3.76 | 3.62 | 1.42/4.73 | 0.01 | 101.47 ± 5.86 | 4.99 | 1.30/4.11 |
| 0.05 | 81.96 ± 3.57 | 2.70 | 86.52 ± 3.13 | 1.93 | 0.1 | 96.53 ± 4.37 | 3.63 | ||||
| 0.5 | 80.13 ± 2.54 | 0.87 | 92.08 ± 4.84 | 5.36 | 1 | 84.88 ± 2.58 | 2.55 | ||||
| Pesticides | Application Dosage (ai. a g/m2) | Spray Times | Days after Application (Mean ± SD b) | ||
|---|---|---|---|---|---|
| 3 | 5 | 7 | |||
| Bifenthrin | 1.25 | 1 | 0.49 ± 0.06 | 0.21 ± 0.01 | 0.11 ± 0.02 |
| 2 | 0.56 ± 0.02 | 0.29 ± 0.04 | 0.21 ± 0.01 | ||
| 0.83 c | 1 | 0.64 ± 0.10 | 0.22 ± 0.03 | 0.16 ± 0.02 | |
| 2 | 077 ± 0.05 | 0.34 ± 0.05 | 0.28 ± 0.03 | ||
| Fenpropathrin | 1.25 | 1 | 0.71 ± 0.04 | 0.33 ± 0.02 | 0.09 ± 0.04 |
| 2 | 0.79 ± 0.05 | 0.42 ± 0.08 | 0.21 ± 0.01 | ||
| 0.83 | 1 | 0.96 ± 0.12 | 0.28 ± 0.01 | 0.17 ± 0.02 | |
| 2 | 1.12 ± 0.09 | 0.51 ± 0.06 | 0.37 ± 0.01 | ||
| Lambda-cyhalothrin | 1.25 | 1 | 0.39 ± 0.02 | 0.14 ± 0.03 | 0.06 ± 0.02 |
| 2 | 0.44 ± 0.04 | 0.19 ± 0.06 | 0.11 ± 0.01 | ||
| 0.83 | 1 | 0.53 ± 0.05 | 0.14 ± 0.02 | 0.11 ± 0.05 | |
| 2 | 0.63 ± 0.06 | 0.24 ± 0.01 | 0.16 ± 0.03 | ||
| β-cypermethrin | 1.25 | 1 | 0.94 ± 0.02 | 0.37 ± 0.03 | 0.15 ± 0.01 |
| 2 | 1.06 ± 0.01 | 0.51 ± 0.05 | 0.27 ± 0.01 | ||
| 0.83 | 1 | 1.29 ± 0.17 | 0.35 ± 0.04 | 0.22 ± 0.02 | |
| 2 | 1.59 ± 0.06 | 0.67 ± 0.04 | 0.33 ± 0.04 | ||
| Deltamethrin | 1.25 | 1 | 0.84 ± 0.10 | 0.30 ± 0.05 | 0.13 ± 0.03 |
| 2 | 0.96 ± 0.02 | 0.43 ± 0.02 | 0.25 ± 0.01 | ||
| 0.83 | 1 | 1.14 ± 0.03 | 0.31 ± 0.04 | 0.23 ± 0.04 | |
| 2 | 1.34 ± 0.10 | 0.56 ± 0.01 | 0.31 ± 0.06 | ||
| Process | Bifenthrin | Fenpropathrin | Lambda-Cyhalothrin | β-Cypermethrin | Deltamethrin |
|---|---|---|---|---|---|
| Drying | 0.49 | 0.74 | 0.62 | 0.56 | 0.53 |
| Storage | 0.61 | 0.60 | 0.75 | 0.62 | 0.67 |
| Soaking | 0.34 | 0.17 | 0.28 | 0.28 | 0.24 |
| Overall process | 0.10 | 0.08 | 0.13 | 0.10 | 0.09 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, J.-J.; Duan, J.-S.; Wu, Y.-C.; Shi, Y.-H.; Fang, Q.-K.; Liao, M.; Hua, R.-M.; Cao, H.-Q. Dissipation and Migration of Pyrethroids in Auricularia polytricha Mont. from Cultivation to Postharvest Processing and Dietary Risk. Molecules 2018, 23, 791. https://doi.org/10.3390/molecules23040791
Xiao J-J, Duan J-S, Wu Y-C, Shi Y-H, Fang Q-K, Liao M, Hua R-M, Cao H-Q. Dissipation and Migration of Pyrethroids in Auricularia polytricha Mont. from Cultivation to Postharvest Processing and Dietary Risk. Molecules. 2018; 23(4):791. https://doi.org/10.3390/molecules23040791
Chicago/Turabian StyleXiao, Jin-Jing, Jin-Sheng Duan, Yan-Can Wu, Yan-Hong Shi, Qing-Kui Fang, Min Liao, Ri-Mao Hua, and Hai-Qun Cao. 2018. "Dissipation and Migration of Pyrethroids in Auricularia polytricha Mont. from Cultivation to Postharvest Processing and Dietary Risk" Molecules 23, no. 4: 791. https://doi.org/10.3390/molecules23040791
APA StyleXiao, J.-J., Duan, J.-S., Wu, Y.-C., Shi, Y.-H., Fang, Q.-K., Liao, M., Hua, R.-M., & Cao, H.-Q. (2018). Dissipation and Migration of Pyrethroids in Auricularia polytricha Mont. from Cultivation to Postharvest Processing and Dietary Risk. Molecules, 23(4), 791. https://doi.org/10.3390/molecules23040791






