Gender-Dependent Effects of Enriched Environment and Social Isolation in Ischemic Retinal Lesion in Adult Rats
Abstract
:1. Introduction
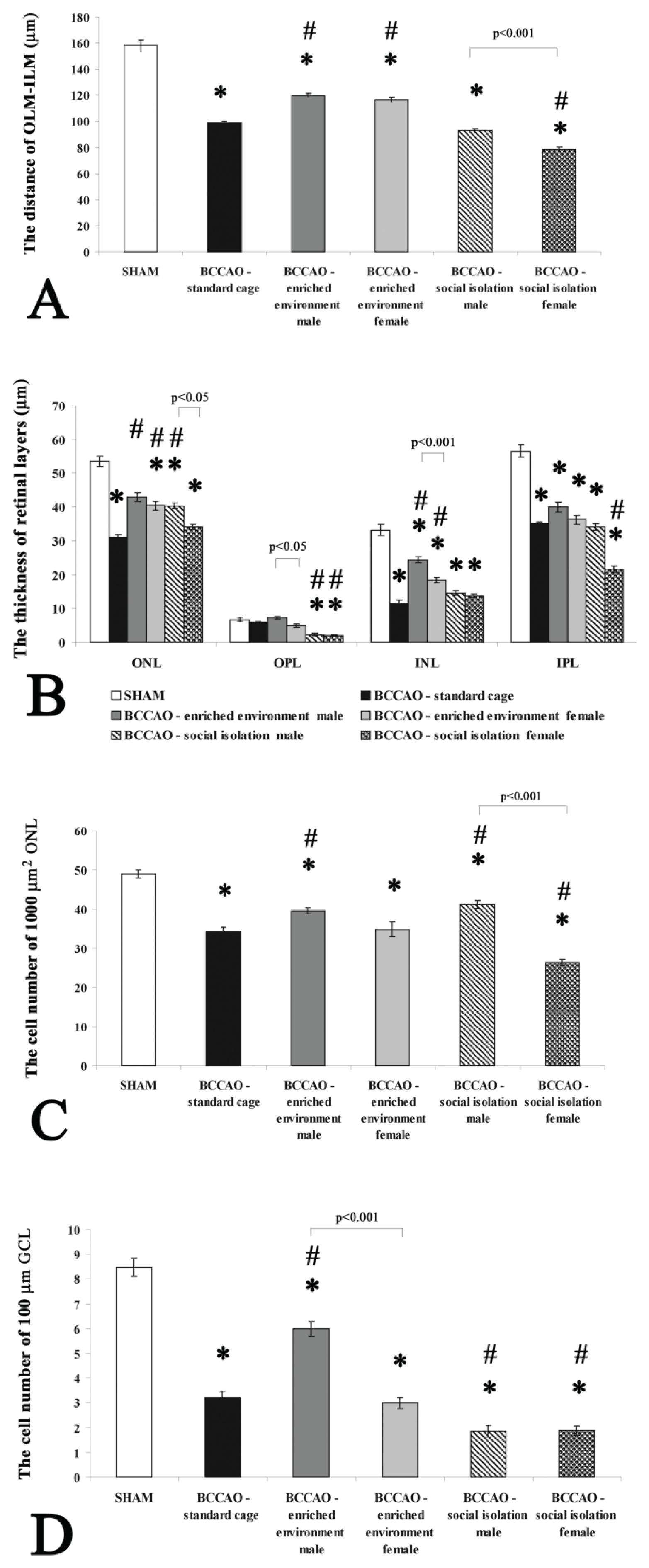
2. Results and Discussion
3. Experimental Section
3.1. Animals
3.2. Permanent Bilateral Common Carotid Artery Occlusion
3.3. Environmental Enrichment and Social Isolation Paradigm
3.4. Histology
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Bishnoi, M.; Chopra, K.; Rongzhu, L.; Kulkarni, S.K. Protective effect of curcumin and its combination with piperine (bioavailability enhancer) against haloperidol-associated neurotoxicity: cellular and neurochemical evidence. Neurotox. Res 2011, 20, 215–225. [Google Scholar]
- Del Barco, D.G.; Pérez-Saad, H.; Rodríguez, V.; Marín, J.; Falcón, V.; Martín, J.; Cibrian, D.; Berlanga, J. Therapeutic effect of the combined use of growth hormone releasing peptide-6 and epidermal growth factor in an axonopathy model. Neurotox. Res 2011, 19, 195–209. [Google Scholar]
- Kalonia, H.; Mishra, J.; Kumar, A. Targeting neuro-inflammatory cytokines and oxidative stress by minocycline attenuates quinolinic-acid-induced Huntington’s disease-like symptoms in rats. Neurotox. Res 2012, 22, 310–320. [Google Scholar]
- van Praag, H.; Kempermann, G.; Gage, F.H. Neural consequences of environmental enrichment. Nat. Rev. Neurosci 2000, 1, 191–198. [Google Scholar]
- Kazl, C.; Foote, L.T.; Kim, M.J.; Koh, S. Early-life experience alters response of developing brain to seizures. Brain Res 2009, 1285, 174–181. [Google Scholar]
- Schneider, T.; Lee, M.H.; Anderson, D.W.; Zuck, L.; Lidsky, T.I. Enriched environment during development is protective against lead-induced neurotoxicity. Brain Res 2001, 896, 48–55. [Google Scholar]
- Soeda, F.; Tanaka, A.; Shirasaki, T.; Takahama, K. An enriched environment mitigates the brain-disruptive effects of prenatal diethylstilbestrol exposure in mice. Neuroscience 2010, 169, 223–228. [Google Scholar]
- Johansson, B.B. Functional outcome in rats transferred to an enriched environment 15 days after focal brain ischemia. Stroke 1996, 27, 324–326. [Google Scholar]
- Ronnback, A.; Dahlqvist, P.; Svensson, P.A.; Jernas, M.; Carlsson, B.; Carlsson, L.M.; Olsson, T. Gene expression profiling of the rat hippocampus one month after focal cerebral ischemia followed by enriched environment. Neurosci. Lett 2005, 385, 173–178. [Google Scholar]
- Zhu, H.; Zhang, J.; Sun, H.; Zhang, L.; Liu, H.; Zeng, X.; Yang, Y.; Yao, Z. An enriched environment reverses the synaptic plasticity deficit induced by chronic cerebral hypoperfusion. Neurosci. Lett 2011, 502, 71–75. [Google Scholar]
- Kovesdi, E.; Gyorgy, A.B.; Kwon, S.K.; Wingo, D.L.; Kamnaksh, A.; Long, J.B.; Kasper, C.E.; Agoston, D.V. The effect of enriched environment on the outcome of traumatic brain injury; a behavioral, proteomics, and histological study. Front. Neurosci 2011, 5, 42. [Google Scholar]
- Johansson, B.B.; Belichenko, P.V. Neuronal plasticity and dendritic spines: Effect of environmental enrichment on intact and postischemic rat brain. J. Cereb. Blood Flow Metabolism 2002, 22, 89–96. [Google Scholar]
- Nithianantharajah, J.; Hannan, A.J. Enriched environments, experience-dependent plasticity and disorders of the nervous system. Nat. Rev. Neurosci 2006, 7, 697–709. [Google Scholar]
- Qiu, X.; Huang, C.X.; Lu, W.; Yang, S.; Li, C.; Shi, X.Y.; Chen, L.; Xiu, Y.; Yang, J.Q.; Tang, Y. Effects of a 4 month enriched environment on the hippocampus and the myelinated fibers in the hippocampus of middle-aged rats. Brain Res 2012, 1465, 26–33. [Google Scholar]
- Landi, S.; Ciucci, F.; Maffei, L.; Berardi, N.; Cenni, M.C. Setting the pace for retinal development: Environmental enrichment acts through insulin-like growth factor 1 and brain-derived neurotrophic factor. J. Neurosci 2009, 29, 10809–10819. [Google Scholar]
- Ortuzar, N.; Argandoña, E.G.; Bengoetxea, H.; Lafuente, J.V. Combination of intracortically administered VEGF and environmental enrichment enhances brain protection in developing rats. J. Neural Transm 2011, 118, 135–144. [Google Scholar]
- Landi, S.; Sale, A.; Berardi, N.; Viegi, A.; Maffei, L.; Cenni, M.C. Retinal functional development is sensitive to environmental enrichment: a role for BDNF. FASEB J 2007, 21, 130–139. [Google Scholar]
- Sale, A.; Cenni, M.C.; Ciucci, F.; Putignano, E.; Chierzi, S.; Maffei, L. Maternal enrichment during pregnancy accelerates retinal development of the fetus. PLoS One 2007, 2, e1160. [Google Scholar]
- Kiss, P.; Atlasz, T.; Szabadfi, K.; Horvath, G.; Griecs, M.; Farkas, J.; Matkovits, A.; Toth, G.; Lubics, A.; Tamas, A.; et al. Comparison between PACAP- and enriched environment-induced retinal protection in MSG-treated newborn rats. Neurosci. Lett 2011, 487, 400–405. [Google Scholar]
- Szabadfi, K.; Atlasz, T.; Horvath, G.; Kiss, P.; Hamza, L.; Farkas, J.; Tamas, A.; Lubics, A.; Gabriel, R.; Reglodi, D. Early postnatal enriched environment decreases retinal degeneration induced by monosodium glutamate treatment. Brain Res 2009, 1259, 107–112. [Google Scholar]
- Atlasz, T.; Babai, N.; Reglodi, D.; Kiss, P.; Tamas, A.; Bari, F.; Domoki, F.; Gabriel, R. Diazoxide is protective in the rat retina against ischemic injury induced by bilateral carotid occlusion and glutamate-induced degeneration. Neurotox. Res 2007, 12, 105–111. [Google Scholar]
- Szabadfi, K.; Mester, L.; Reglodi, D.; Kiss, P.; Babai, N.; Racz, B.; Kovacs, K.; Szabo, A.; Tamas, A.; Gabriel, R.; Atlasz, T. Novel neuroprotective strategies in ischemic retinal lesions. Int. J. Mol. Sci 2010, 11, 544–561. [Google Scholar]
- Osborne, N.N.; Casson, R.J.; Wood, J.P.M.; Chidlow, G.; Graham, M.; Melena, J. Retinal ischemia: Mechanisms of damage and potential therapeutic strategies. Prog. Retin. Eye Res 2004, 223, 91–147. [Google Scholar]
- Barone, H.; Novelli, E.; Piano, I.; Gargini, C.; Strettoi, E. Environmental enrichment extends photoreceptor survival and visual function in a mouse model of retinitis pigmentosa. PLoS One 2012, 11, e50726. [Google Scholar]
- Dorfman, D.; Fernandez, D.C.; Chianelli, M.; Miranda, M.; Aranda, M.L.; Rosenstein, R.E. Post-ischemic environmental enrichment protects the retina from ischemic damage in adult rats. Exp. Neurol 2013, 240C, 146–156. [Google Scholar]
- Tamas, A.; Lubics, A.; Lengvari, I.; Reglodi, D. Effects of age, gender, and gonadectomy on neurochemistry and behavior in animal models of Parkinson’s disease. Endocrine 2006, 29, 275–287. [Google Scholar]
- Leasure, J.L.; Decker, L. Social isolation prevents exercise-induced proliferation of hippocampal progenitor cells in female rats. Hippocampus 2009, 19, 907–912. [Google Scholar]
- Takemoto, T.I.; Suzuki, T.; Miyama, T. Effects of isolation on mice in relation to age and sex. Tohoku J. Exp. Med 1975, 117, 153–165. [Google Scholar]
- Chourbaji, S.; Hörtnagl, H.; Molteni, R.; Riva, M.A.; Gass, P.; Hellweg, R. The impact of environmental enrichment on sex-specific neurochemical circuitries—Effects on brain-derived neurotrophic factor and the serotonergic system. Neuroscience 2012, 220, 267–276. [Google Scholar]
- Reglodi, D.; Lubics, A.; Kiss, P.; Lengvari, I.; Gaszner, B.; Toth, G.; Hegyi, O.; Tamas, A. Effect of PACAP in 6-OHDA-induced injury of the substantia nigra in intact young and ovariectomized female rats. Neuropeptides 2006, 40, 265–274. [Google Scholar]
- Brown, K.J.; Grunberg, N.E. Effects of housing on male and female rats: Crowding stresses male but calm females. Physiol. Behav 1995, 58, 1085–1089. [Google Scholar]
- Markham, J.A.; Herting, M.M.; Luszpak, A.E.; Juraska, J.M.; Greenough, W.T. Myelination of the corpus callosum in male and female rats following complex environment housing during adulthood. Brain Res 2009, 1288, 9–17. [Google Scholar]
- Peña, Y.; Prunell, M.; Dimitsantos, V.; Nadal, R.; Escorihuela, R.M. Environmental enrichment effects in social investigation in rats are gender dependent. Behav. Brain Res 2006, 174, 181–187. [Google Scholar]
- Westenbroek, C.; den Boer, J.A.; Veenhuis, M.; ter Horst, G.J. Chronic stress and social housing differentially affect neurogenesis in male and female rats. Brain Res. Bull 2004, 64, 303–308. [Google Scholar]
- Westenbroek, C.; Snijders, T.A.; den Boer, J.A.; Gerrits, M.; Fokkema, D.S.; ter Horst, G.J. Pair-housing of male and female rats during chronic stress exposure results in gender-specific behavioral responses. Horm. Behav 2005, 47, 620–628. [Google Scholar]
- Arranz, L.; de Castro, N.M.; Baeza, I.; Giménez-Llort, L.; de la Fuente, M. Effect of environmental enrichment on the immunoendocrine aging of male and female triple-transgenic 3×Tg-AD mice for Alzheimer’s disease. J. Alzheimers Dis 2011, 25, 727–737. [Google Scholar]
- Mainardi, M.; Landi, S.; Gianfranceschi, L.; Baldini, S.; de Pasquale, R.; Berardi, N.; Maffei, L.; Caleo, M. Environmental enrichment potentiates thalamocortical transmission and plasticity in the adult rat visual cortex. J. Neurosci. Res 2010, 88, 3048–3059. [Google Scholar]
- Yang, S.; Lu, W.; Zhou, D.S.; Tang, Y. Enriched environment and white matter in aging brain. Anat. Rec. (Hoboken) 2012, 295, 1406–1414. [Google Scholar]
- Young, D.; Lawlor, P.A.; Leone, P.; Dragunow, M.; During, M.J. Environmental enrichment inhibits spontaneous apoptosis, prevents seizures and is neuroprotective. Behav. Brain Res 2012, 230, 92–99. [Google Scholar]
- Martinez, M.; Brezun, J.M.; Xerri, C. Sensorimotor experience influences recovery of forelimb abilities but not tissue loss after focal cortical compression in adult rats. PLoS One 2011, 6, e16726. [Google Scholar]
- Farrell, R.; Evans, S.; Corbett, D. Environmental enrichment enhances recovery of function but exacerbates ischemic cell death. Neuroscience 2001, 107, 585–592. [Google Scholar]
- Varga, B.; Szabadfi, K.; Kiss, P.; Fabian, E.; Tamas, A.; Griecs, M.; Gabriel, R.; Reglodi, D.; Kemeny-Beke, A.; Pamer, Z.; et al. PACAP improves functional outcome in excitotoxic retinal lesion: an electroretinographic study. J. Mol. Neurosci 2011, 43, 44–50. [Google Scholar]
- Sale, A.; Maya Vetencourt, J.F.; Medini, P.; Cenni, M.C.; Baroncelli, L.; de Pasquale, R.; Maffei, L. Environmental enrichment in adulthood promotes amblyopia recovery through a reduction of intracortical inhibition. Nat. Neurosci 2007, 10, 679–681. [Google Scholar]
- Jain, V.; Baitharu, I.; Prasad, D.; Ilavazhagan, G. Enriched Environment prevents hypobaric hypoxia induced memory impairment and neurodegeneration: Role of BDNF/PI3K/GSK3β pathway coupled with CREB activation. PLoS One 2013, 8, e62235. [Google Scholar]
- Lee, M.Y.; Yu, J.H.; Kim, J.Y.; Seo, J.H.; Park, E.S.; Kim, C.H.; Kim, H.; Cho, S.R. Alteration of synaptic activity-regulating genes underlying functional improvement by long-term exposure to an enriched environment in the adult brain. Neurorehabil. Neural. Repair 2013, 27, 561–574. [Google Scholar]
- Fabricius, K.; Steiniger-Brach, B.; Helboe, L.; Fink-Jensen, A.; Wörtwein, G. Socially isolated rats exhibit changes in dopamine homeostasis pertinent to schizophrenia. Int. J. Dev. Neurosci 2011, 29, 347–350. [Google Scholar]
- Zakharova, E.; Starosciak, A.; Wade, D.; Izenwasser, S. Sex differences in the effects of social and physical environment on novelty-induced exploratory behavior and cocaine-stimulated locomotor activity in adolescent rats. Behav. Brain Res 2012, 230, 92–99. [Google Scholar]
- Prilloff, S.; Henrich-Noack, P.; Kropf, S.; Sabel, B.A. Experience-dependent plasticity and vision restoration in rats after optic nerve crush. J. Neurotrauma 2010, 27, 2295–2307. [Google Scholar]
- Alkayed, N.J.; Harukuni, I.; Kimes, A.S.; London, E.D.; Traystman, R.J.; Hurn, P.D. Gender-linked brain injury in experimental stroke. Stroke 1998, 29, 159–165. [Google Scholar]
- Chen, X.; Li, Y.; Kline, A.E.; Dixon, C.E.; Zafonte, R.D.; Wagner, A.K. Gender and environmental effects on regional brain-derived neurotrophic factor expression after experimental traumatic brain injury. Neuroscience 2005, 135, 11–17. [Google Scholar]
- Stein, D.G. Brain damage, sex hormones and recovery: A new role for progesterone and estrogens? Trends Neurosci 2001, 24, 386–391. [Google Scholar]
- Suzuki, T.; Bramlett, H.M.; Dietrich, D. The importance of gender on the beneficial effects of posttraumatic hypothermia. Exp. Neurol 2003, 184, 1017–1026. [Google Scholar]
- Lang, J.T.; McCullough, L.D. Pathways to ischemic neuronal cell death: Are sex differences relevant? J. Transl. Med 2008, 6, 33. [Google Scholar]
- Zeynalov, E.; Nemoto, M.; Hurn, P.D.; Koehler, R.C.; Bhardwaj, A. Neuroprotective effect of kappa opioid receptor agonist is gender specific and linked to reduced neuronal nitric oxide. J. Cereb. Blood Flow Metabolism 2006, 26, 414–420. [Google Scholar]
- Saucier, D.M.; Yager, J.Y.; Armstrong, E.A. Housing environment and sex affect behavioral recovery from ischemic brain damage. Behav. Brain Res 2010, 214, 48–54. [Google Scholar]
- Saucier, D.M.; Yager, J.Y.; Armstrong, E.A.; Keller, A.; Shultz, S. Enriched environment and the effect of age on ischemic brain damage. Brain Res 2007, 1170, 31–38. [Google Scholar]
- Wagner, A.K.; Kline, A.E.; Sokoloski, J.; Zafonte, R.D.; Capulong, E.; Dixon, C.E. Intervention with environmental enrichment after experimental brain trauma enhances cognitive recovery in male but not female rats. Neurosci. Lett 2002, 334, 165–168. [Google Scholar]
- Hong, S.; Flashner, B.; Chiu, M.; ver Hoeve, E.; Luz, S.; Bhatnagar, S. Social isolation in adolescence alters behaviors in the forced swim and sucrose preference tests in female but not in male rats. Physiol. Behav 2012, 105, 269–275. [Google Scholar]
- Mester, L.; Szabo, A.; Atlasz, T.; Szabadfi, K.; Reglodi, D.; Kiss, P.; Racz, B.; Tamas, A.; Gallyas, F., Jr; Sumegi, B.; et al. Protection against chronic hypoperfusion-induced retinal neurodegeneration by PARP inhibition via activation of PI-3-kinase Akt pathway and suppression of JNK and p38 MAP kinases. Neurotox. Res. 2009, 16, 68–76. [Google Scholar]
- Mracskó, E.; Hugyecz, M.; Institóris, A.; Farkas, E.; Bari, F. Changes in pro-oxidant and antioxidant enzyme levels during cerebral hypoperfusion in rats. Brain Res 2010, 1321, 13–19. [Google Scholar]
- Yamamoto, H.; Schmidt-Kasner, R.; Hamasaki, D.I.; Yamamoto, H.; Parel, J.M. Complex neurodegeneration in retina following moderate ischemia induced by bilateral common carotid artery occlusion in Wistar rats. Exp. Eye Res 2006, 82, 767–779. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kiss, P.; Szabadfi, K.; Horvath, G.; Tamas, A.; Farkas, J.; Gabriel, R.; Reglodi, D. Gender-Dependent Effects of Enriched Environment and Social Isolation in Ischemic Retinal Lesion in Adult Rats. Int. J. Mol. Sci. 2013, 14, 16111-16123. https://doi.org/10.3390/ijms140816111
Kiss P, Szabadfi K, Horvath G, Tamas A, Farkas J, Gabriel R, Reglodi D. Gender-Dependent Effects of Enriched Environment and Social Isolation in Ischemic Retinal Lesion in Adult Rats. International Journal of Molecular Sciences. 2013; 14(8):16111-16123. https://doi.org/10.3390/ijms140816111
Chicago/Turabian StyleKiss, Peter, Krisztina Szabadfi, Gabor Horvath, Andrea Tamas, Jozsef Farkas, Robert Gabriel, and Dora Reglodi. 2013. "Gender-Dependent Effects of Enriched Environment and Social Isolation in Ischemic Retinal Lesion in Adult Rats" International Journal of Molecular Sciences 14, no. 8: 16111-16123. https://doi.org/10.3390/ijms140816111




