1. Introduction
Gastric cancer (GC), as the third leading cause of cancer death in men and the fifth in women, remains one of the major public health problems worldwide [
1]. About two-thirds of the cases occur in developing countries and 42% in China alone, while the estimated high-risk areas in developed countries include Eastern Europe, and parts of Central and South America [
2]. In recent decades, its incidence rate has declined, and remarkable progress has been achieved in comprehensive treatment strategies of combined therapy. However, the prognosis for patients still remains poor, with 5-year overall survival rates of 30% [
3]. The development of gastric cancer is a multi-step, sequential process, which initiates from chronic gastritis, atrophy, intestinal metaplasia, dysplasia, and finally malignant transformation to invasive gastric cancer [
4]. Moreover, multiple genetic and epigenetic alterations are implicated in the multi-step process of human stomach carcinogenesis and development [
5]. Therefore, discovery of these biomarkers could be helpful in the improvement of early diagnosis, screening of high-risk individuals, as well as patient care [
6,
7]. In recent years, increased studies have focused on the detection of genetic variants that could play roles in the development, progression and prognoses of gastric cancer [
8].
The orphan nuclear receptor (NR5A2), also known as liver receptor homologue-1 (LRH-1) and fetoprotein transcription factor (FTF), belongs to the NR5A or FTZ-F1 subfamily of nuclear receptors. NR5A2 is expressed in developing and adult tissues of endodermal origin, including liver, pancreas, intestine and the ovary [
9,
10]. Functionally, NR5A2 has been implicated in the regulation of bile acid and cholesterol homeostasis [
9,
10] and the regulation of inflammatory responses in the liver and gut [
11]. Aromatase is an
NR5A2 target gene, which catalyses the conversion of androgens (testosterone and primarily androstenedione) to oestrogens.
NR5A2 may aid breast cancer progression in postmenopausal women by promoting local oestrogen biosynthesis [
12,
13,
14]. The expression of
NR5A2 is also elevated in pancreatic cancer and promotes pancreatic cancer cell growth through stimulation of
cyclin D1,
cyclin E1 and c-Myc [
15]. A recent genome-wide association study (GWAS) identified rs3790844 and rs3790843, located in the first intron of
NR5A2 in 1q32.11, as associated with pancreatic cancer susceptibility [
16]. In the gastrointestinal tract,
NR5A2 has been shown to participate in intestinal cell renewal [
17] and is expressed in the stomach epithelium [
18]. Therefore, we conducted this study to examine whether
NR5A2 rs3790844 and rs3790843 polymorphisms are associated with clinical outcomes of gastric cancer. The two SNPs may serve as potential molecular prognostic markers for gastric cancer, which will promote further defined sub-populations at higher risk of the disease. Consequently, these sub-populations may require more rigorous treatment and postoperative follow up.
3. Discussion
In the present study, we investigated the effects of two SNPs (rs3790843 and rs3790844) of the NR5A2 gene on the progression and survival of GC in Chinese populations. We found that NR5A2 rs3790843 polymorphism was significantly associated with the risk of GC when compared with regional lymph node metastasis and distant metastasis. Our results also indicated that rs3790844 polymorphism was associated with the increased OS of GC patients in the dominant model, suggesting a potential protective role of the variant A allele. No significant associations were observed between the rs3790843 genotypes and OS of GC patients in any genetic models. Additionally, in the stratified analysis, NR5A2 rs3790844 polymorphism was associated with significantly lower risk of death in the groups of female, tumor size >5 cm, lymph node metastasis, no distant metastasis, diffuse type and no chemotherapy history in the dominant model. Similar results were found among the female patients group and tumor size >5 cm group for the NR5A2 rs3790843 polymorphism in a dominant model.
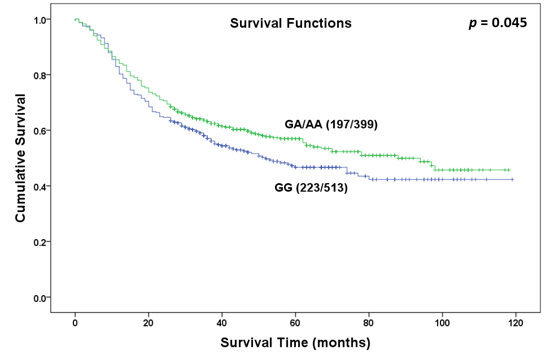
Figure 1.
Overall survival curve in relation to NR5A2 rs3790844 polymorphism in patients with gastric cancer in dominant model.
Figure 1.
Overall survival curve in relation to NR5A2 rs3790844 polymorphism in patients with gastric cancer in dominant model.
Nuclear receptor 5 subtype A2 (NR5A2), also known as Liver receptor homolog-1 (LRH-1), is a member of the orphan family of nuclear receptors that participates in a wide range of developmental processes [
9,
10]. Functionally, NR5A2 has been implicated in the regulation of bile acid and cholesterol homeostasis, and is expressed in developing and adult tissues of endodermal origin, including liver, pancreas, intestine and the ovary [
9,
10].
NR5A2 was first isolated as a transcriptional activator of the alpha-fetoprotein (
AFP) gene, which encodes a factor that plays a crucial rolein hepatic specification [
19]. Further,
NR5A2 expression is elevated in pancreatic cancer andpromotes pancreatic cancer cell growth through stimulation of
cyclin D1,
cyclin E1 and c-Myc [
15], while genome-wide association studies implicate mutations in the
NR5A2 gene in pancreatic ductal adenocarcinoma [
16]. In the gastrointestinal tract,
NR5A2 has been shown to participate in intestinal cell renewal [
17] and is expressed in the stomach epithelium [
18]. However, it seemed no study focused on the association between
NR5A2 and the progression and prognoses of gastric cancer. Thus, this study was conducted, and it indicates that the
NR5A2 polymorphism is significantly associated with the development and overall survival of gastric cancer.
In the gastrointestinal tract,
NR5A2 regulates proliferation through at least two different mechanisms: direct activation of the
cyclin E1 promoter and, in cooperation with
β-catenin, induction of
cyclin D1 and
c-Myc transcription [
20]. A previous study showed that
NR5A2, which interacts with
β-catenin, induces cell proliferation through the concomitant induction of
cyclin D1 and
E1, which are downstream
NR5A2 effectors whose induction may account for the diminished threshold for G/S transition [
20]. Whereas
β-catenin coactivates
NR5A2 on the
cyclin E1 promoter,
NR5A2 acts as a potent tissue-restricted coactivator of
β-catenin on the
cyclin D1 promoter.
Table 5.
Stratified analysis of rs3790843 and rs3790844 polymorphism among GC patients.
Table 5.
Stratified analysis of rs3790843 and rs3790844 polymorphism among GC patients.
| | rs3790843 | rs3790844 |
|---|
| | TT | TC/CC | HR (95% CI) e | p | GG | GA/AA | HR (95% CI) e | p |
|---|
| Age (years) | | | | | | | | |
| ≤60 | 88/192 | 108/237 | 0.977 (0.737–1.294) | 0.869 | 90/182 | 107/249 | 0.806 (0.609–1.067) | 0.132 |
| >60 | 108/222 | 115/256 | 0.873 (0.672–1.136) | 0.313 | 107/217 | 116/264 | 0.837 (0.644–1.089) | 0.185 |
| Sex | | | | | | | | |
| Male | 144/321 | 175/375 | 1.044 (0.838–1.302) | 0.700 | 149/311 | 172/391 | 0.877 (0.705–1.093) | 0.243 |
| Female | 52/93 | 48/118 | 0.619 (0.417–0.916) | 0.017 | 48/88 | 51/122 | 0.660 (0.445–0.981) | 0.040 |
| Location | | | | | | | | |
| Non–CardiaCancer | 133/279 | 148/320 | 0.948 (0.750–1.198) | 0.652 | 133/264 | 148/338 | 0.810 (0.641–1.024) | 0.077 |
| CardiaCancer | 63/135 | 75/173 | 0.861 (0.615–1.204) | 0.381 | 64/135 | 75/175 | 0.840 (0.601–1.174) | 0.308 |
| Tumor Size | | | | | | | | |
| ≤5 cm | 108/261 | 127/302 | 1.044 (0.807–1.349) | 0.745 | 106/243 | 132/324 | 0.927 (0.718–1.197) | 0.559 |
| >5 cm | 88/153 | 96/191 | 0.747 (0.558–0.999) | 0.049 | 91/156 | 91/189 | 0.693 (0.517–0.928) | 0.014 |
| Lymph Node Metastasis a | | | | | | | | |
| N0 | 64/180 | 60/182 | 0.947 (0.666–1.347) | 0.763 | 63/168 | 63/197 | 0.865 (0.610–1.226) | 0.414 |
| N1/N2/N3 | 132/234 | 163/311 | 0.829 (0.659–1.043) | 0.109 | 134/231 | 160/316 | 0.744 (0.591–0.937) | 0.012 |
| Distant Metastasis | | | | | | | | |
| M0 | 188/398 | 200/456 | 0.886 (0.726–1.081) | 0.232 | 188/382 | 202/478 | 0.793 (0.650–0.968) | 0.022 |
| M1 | 8/16 | 23/37 | 1.340 (0.599–2.998) | 0.476 | 7/9 | 21/35 | 1.189 (0.544–2.599) | 0.664 |
| Histological Types b | | | | | | | | |
| Intestinal | 71/168 | 80/187 | 1.041 (0.756–1.434) | 0.805 | 69/156 | 84/205 | 0.952 (0.692–1.309) | 0.762 |
| Diffuse | 108/207 | 127/270 | 0.826 (0.639–1.068) | 0.145 | 111/206 | 123/270 | 0.735 (0.568–0.950) | 0.019 |
| Differentiation b | | | | | | | | |
| Well to Moderate | 57/137 | 67/155 | 1.099 (0.772–1.565) | 0.601 | 56/127 | 69/170 | 0.962 (0.676–1.368) | 0.828 |
| Poorly | 104/208 | 126/267 | 0.882 (0.680–1.144) | 0.344 | 105/205 | 125/270 | 0.822 (0.634–1.067) | 0.140 |
| Others c | 18/30 | 14/35 | 0.522 (0.259–1.056) | 0.07 | 19/30 | 13/35 | 0.397 (0.195–0.808) | 0.011 |
| Lauren b | | | | | | | | |
| 1 | 69/182 | 78/199 | 1.049 (0.759–1.451) | 0.771 | 69/169 | 80/218 | 0.899 (0.652–1.241) | 0.655 |
| 2 | 125/229 | 144/293 | 0.824 (0.648–1.047) | 0.113 | 126/227 | 142/294 | 0.763 (0.600–0.970) | 0.027 |
| Chemotherapy | | | | | | | | |
| No | 132/275 | 151/333 | 0.908 (0.719–1.147) | 0.416 | 136/266 | 149/348 | 0.767 (0.608–0.968) | 0.025 |
| Yes | 64/139 | 72/160 | 0.957 (0.683–1.340) | 0.799 | 61/133 | 74/165 | 0.953 (0.679–1.338) | 0.783 |
| Depth of Invasion d | | | | | | | | |
| T1 | 25/86 | 34/93 | 1.297 (0.774–2.173) | 0.324 | 24/76 | 33/101 | 1.051 (0.621–1.779) | 0.852 |
| T2 | 31/60 | 24/69 | 0.669 (0.393–1.141) | 0.140 | 31/59 | 25/71 | 0.648 (0.382–1.099) | 0.107 |
| T3 | 1/3 | 2/3 | 1.405 (0.125–15.838) | 0.782 | 1/2 | 2/4 | 0.809 (0.071–9.157) | 0.864 |
| T4 | 134/257 | 159/320 | 0.880 (0.699–1.108) | 0.276 | 135/254 | 157/326 | 0.809 (0.642–1.019) | 0.072 |
| TNM Stage | | | | | | | | |
| I | 40/120 | 39/118 | 0.992 (0.638–1.542) | 0.972 | 40/111 | 40/128 | 0.862 (0.556–1.336) | 0.506 |
| II | 38/83 | 38/111 | 0.743 (0.474–1.166) | 0.196 | 37/78 | 41/119 | 0.711 (0.456–1.109) | 0.132 |
| III | 111/198 | 135/248 | 0.861 (0.670–1.108) | 0.245 | 113/198 | 132/250 | 0.800 (0.622–1.029) | 0.082 |
| IV | 4/10 | 8/13 | 1.855 (0.557–6.171) | 0.314 | 4/9 | 7/13 | 1.257 (0.368–4.302) | 0.715 |
Table 6.
Stepwise Cox regression analysis on the survival of GC.
Table 6.
Stepwise Cox regression analysis on the survival of GC.
| Variables | β | SE | HR b | 95% CI | p Value |
|---|
| Age a | 0.067 | 0.100 | 1.070 | (0.878–1.302) | 0.503 |
| Sex | 0.04 | 0.118 | 1.041 | (0.825–1.313) | 0.743 |
| Lymph Node Metastasis | 0.567 | 0.112 | 1.763 | (1.414–2.198) | <0.001 |
| rs3790844 (GG vs. GA/AA) | −0.243 | 0.099 | 0.784 | (0.646–0.951) | 0.014 |
| rs3790843 (TT vs. CT/CC) | −0.152 | 0.101 | 0.859 | (0.705–1.046) | 0.131 |
Tumor growth is the result of uncontrolled cell proliferation or a defective cell death program. For the proliferation of cells, the phosphorylation of pRb by
cyclin–CDK complexes to release the transcription factor E2F is an essential step. The
cyclin D1,
cyclin E and
c-Myc are important parts of expression peaks at the G1–S transition, and then decreases as cells proceed through the S phase [
21,
22,
23]. The G1–S transition is the major regulation point of the cell cycle, and during the G1–S transition, alterations in cell cycle regulators lead to the deregulation of the cell cycle, which can cause unbridled cell division, contributing to cancer development. Previously, overexpression of
cyclin E was demonstrated in many tumors and correlated with prognosis [
24,
25,
26,
27] including gastric cancer [
28]. Wang
et al. [
29] found that the mRNA expression of NR5A2 was significantly upregulated in gastric cancer, as compared with self-paired normal control. In addition, overexpression of NR5A2 was shown to promote the proliferation of gastric adenocarcinoma SGC-7901 cells via induction of
cyclin E1, which may lead to the tumorigenesis of gastric cancer [
29]. Botrugno
et al. [
20] found that
cyclin E was a direct
NR5A2 target gene, which could partly explain the mechanism of
NR5A2 polymorphism in the progression and prognoses of gastric cancer. On the other hand,
β-catenin has been proposed to act as a docking protein that assembles both general and specific factors required for the activation of target genes. Botrugno
et al. also found that
NR5A2 could act as a coactivator for
β-Catenin/Tcf4 to drive the expression of
cyclin D1 and other
β-catenin/Tcf target genes, such as
c-Myc [
30,
31,
32]. As reported,
cyclin D1 is a proto-oncogene that belongs to the family of G1 cyclins, and plays an important role in cell cycle G1 to S transition by binding its partners cyclin dependent kinase 4 and 6 to phosphorylate and inactivate the Rb protein [
33]. In addition,
cyclin D1 over-expression is usually an early event in carcinogenesis and a prognostic indicator associated with poor survival in cancers [
34]. Given above-mentioned evidences,
NR5A2 polymorphism could also affect the carcinogenesis and prognoses of cancer through acting as a coactivator for
β-catenin on the
cyclin D1 promoter.
Interestingly, in the stratified analyses by sex, our results indicated that both the
NR5A2 rs3790843 and rs3790844 polymorphisms were associated with significantly better prognosis in the female patient group.
NR5A2 is a direct estrogen receptor α (ERα) target gene [
35,
36], its expression correlates with ERα in breast tumours [
37] and it promotes breast cancer proliferation and invasion [
38]. Previous study showed that
NR5A2 is an important regulator of ERα target genes and it shares many binding sites with ERα. Importantly, at shared sites,
NR5A2 promotes ERα recruitment and
vice versa, ERα stimulates
NR5A2 recruitment [
39]. As a result, in the female patients with more ERα, more
NR5A2 polymorphisms, which act as a protective factor, contribute to a better prognosis.
Taken together, the presented findings of the potential involvement of the NR5A2 gene in anti-tumorigenesis prompts us to further characterize its structure, biological function and interaction with other partners by in vitro or in vivo studies. However, some limitations of the present study should be addressed. First, Helicobacter pylorus, a known crucial factor in gastric carcinogenesis, was not considered due to the lack of related follow-up information. Second, only two SNPs in NR5A2 are evaluated, and it is possible that some other important SNPs are neglected or the observed associations may be due to other polymorphisms in linkage disequilibrium with the rs3790843 and rs3790844 polymorphisms. Finally, for validation of the genotype–phenotype relationship, further investigation is underway to clarify the association between rs3790843 and rs3790844 polymorphisms and expression levels of NR5A2 protein in gastric cancer tissues, and will be reported separately.