Effect of Pulsed Electric Field on Membrane Lipids and Oxidative Injury of Salmonella typhimurium
Abstract
:1. Introduction
2. Results
2.1. Salmonella typhimurium (S. typhimurium) Inactivation by Pulsed Electric Field (PEF) Treatment
2.2. Modification in Cytoplasmic Membrane Fluidity
2.3. Changes in Lipid Composition
2.4. Lipid Peroxidation and Cell Morphology
2.5. Alteration in Expression of Fatty Acid Biosynthesis-Associated Genes and Cytochrome Bo Oxidase Genes
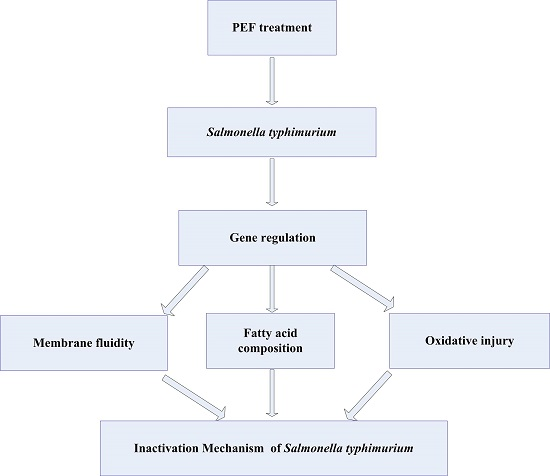
3. Discussion
3.1. The Effect of PEF Treatment Time on S. typhimurium Inactivation
3.2. The Effect of PEF on Cytoplasmic Membrane Fluidity of S. typhimurium
3.3. The Effect of PEF on on Lipid Composition and Oxidative Injury
3.4. The Effect of PEF on Relative Expression of Fatty Acid Biosynthesis-Associated Genes and Cytochrome Bo Oxidase Genes
4. Materials and Methods
4.1. Materials
4.2. Growth Condition of S. Typhimurium
4.3. PEF System and Treatment
4.4. Enumeration of Survivors
4.5. Measurement of Cytoplasmic Membrane Fluidity
4.6. Determination of Membrane Fatty Acid Composition
4.7. Detection of Membrane Lipid Peroxidation
4.8. RNA Extraction and Analysis of Gene Expression by Quantitative Real-Time RT-PCR (qRT-PCR)
4.9. Scanning Electron Microscope (SEM)
4.10. Statistics Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Aadil, R.M.; Zeng, X.-A.; Ali, A.; Zeng, F.; Farooq, M.A.; Han, Z.; Khalid, S.; Jabbar, S. Influence of different pulsed electric field strengths on the quality of the grapefruit juice. Int. J. Food Sci. Technol. 2015, 50, 2290–2296. [Google Scholar] [CrossRef]
- Aadil, R.M.; Zeng, X.-A.; Wang, M.-S.; Liu, Z.-W.; Han, Z.; Zhang, Z.-H.; Hong, J.; Jabbar, S. A potential of ultrasound on minerals, micro-organisms, phenolic compounds and colouring pigments of grapefruit juice. Int. J. Food Sci. Technol. 2015, 50, 1144–1150. [Google Scholar] [CrossRef]
- Aadil, R.M.; Zeng, X.-A.; Zhang, Z.-H.; Wang, M.-S.; Han, Z.; Jing, H.; Jabbar, S. Thermosonication: A potential technique that influences the quality of grapefruit juice. Int. J. Food Sci. Technol. 2015, 50, 1275–1282. [Google Scholar] [CrossRef]
- Zhang, Z.-H.; Yu, Q.; Zeng, X.-A.; Han, Z.; Sun, D.-W.; Muhammad-Aadil, R. Effects of pulsed electric field on selected properties of L-tryptophan. Int. J. Food Sci. Technol. 2015, 50, 1130–1136. [Google Scholar] [CrossRef]
- Saldana, G.; Monfort, S.; Condon, S.; Raso, J.; Alvarez, I. Effect of temperature, pH and presence of nisin on inactivation of Salmonella Typhimurium and Escherichia coli O157:H7 by pulsed electric fields. Food Res. Int. 2012, 45, 1080–1086. [Google Scholar] [CrossRef]
- Aadil, R.M.; Zeng, X.-A.; Sun, D.-W.; Wang, M.-S.; Liu, Z.-W.; Zhang, Z.-H. Combined effects of sonication and pulsed electric field on selected quality parameters of grapefruit juice. LWT Food Sci. Technol. 2015, 62, 890–893. [Google Scholar] [CrossRef]
- Faridnia, F.; Bekhit, A.E.-D.A.; Niven, B.; Oey, I. Impact of pulsed electric fields and post-mortem vacuum ageing on beef longissimus thoracis muscles. Int. J. Food Sci. Technol. 2014, 49, 2339–2347. [Google Scholar] [CrossRef]
- Shayanfar, S.; Chauhan, O.; Toepfl, S.; Heinz, V. Pulsed electric field treatment prior to freezing carrot discs significantly maintains their initial quality parameters after thawing. Int. J. Food Sci. Technol. 2014, 49, 1224–1230. [Google Scholar] [CrossRef]
- Stratakos, A.C.; Koidis, A. Suitability, efficiency and microbiological safety of novel physical technologies for the processing of ready-to-eat meals, meats and pumpable products. Int. J. Food Sci. Technol. 2015, 50, 1283–1302. [Google Scholar] [CrossRef]
- Walkling-Ribeiro, M.; Anany, H.; Griffiths, M.W. Effect of Heat-Assisted Pulsed Electric Fields and Bacteriophage on Enterohemorrhagic Escherichia coli O157:H7. Biotechnol. Prog. 2015, 31, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, C.; Somolinos, M.; Cebrian, G.; Condon, S.; Pagan, R. Pulsed electric fields cause sublethal injuries in the outer membrane of Enterobacter sakazakii facilitating the antimicrobial activity of citral. Lett. Appl. Microbiol. 2010, 51, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Boziaris, I.S.; Proestos, C.; Kapsokefalou, M.; Komaitis, M. Acid-induced injury renders Salmonella Enteritidis PT4 sensitive to the antimicrobial action of Filipendula ulmaria plant extract. Int. J. Food Sci. Technol. 2012, 47, 1784–1787. [Google Scholar] [CrossRef]
- Ferrario, M.; Alzamora, S.M.; Guerrero, S. Study of pulsed light inactivation and growth dynamics during storage of Escherichia coli ATCC 35218, Listeria innocua ATCC 33090, Salmonella Enteritidis MA44 and Saccharomyces cerevisiae KE162 and native flora in apple, orange and strawberry juices. Int. J. Food Sci. Technol. 2015, 50, 2498–2507. [Google Scholar] [CrossRef]
- Macias-Rodriguez, B.; Yang, W.; Schneider, K.; Rock, C. Pulsed UV light as a postprocessing intervention for decontamination of hard-cooked peeled eggs. Int. J. Food Sci. Technol. 2014, 49, 2472–2480. [Google Scholar] [CrossRef]
- Wang, M.S.; Zeng, X.A.; Sun, D.W.; Han, Z. Quantitative analysis of sublethally injured Saccharomyces cerevisiae cells induced by pulsed electric fields. LWT Food Sci. Technol. 2015, 60, 672–677. [Google Scholar] [CrossRef]
- Bonnafous, P.; Vernhes, M.-C.; Teissié, J.; Gabriel, B. The generation of reactive-oxygen species associated with long-lasting pulse-induced electropermeabilisation of mammalian cells is based on a non-destructive alteration of the plasma membrane. Biochim. Biophys. Acta 1999, 1461, 123–134. [Google Scholar] [CrossRef]
- Harutyunyan, H.A.; Sahakyan, G.V. Biological effects of the electrostatic field: Red blood cell-related alterations of oxidative processes in blood. Int. J. Biometeorol. 2016, 60, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Khoo, W.J.; Zheng, Q.; Chung, H.-J.; Yuk, H.-G. Growth temperature alters Salmonella Enteritidis heat/acid resistance, membrane lipid composition and stress/virulence related gene expression. Int. J. Food Microbiol. 2014, 172, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Chueca, B.; Pagan, R.; Garcia-Gonzalo, D. Transcriptomic analysis of Escherichia coli MG1655 cells exposed to pulsed electric fields. Innov. Food Sci. Emerg. 2015, 29, 78–86. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, X.A.; Wen, Q.B.; Li, L. Fluorescence polarization used to investigate the cell membrane fluidity of Saccharomyces cerevisiae treated by pulsed electric field. Spectrosc. Spectr. Anal. 2008, 28, 156–160. [Google Scholar]
- Zhao, W.; Yang, R.; Gu, Y.; Li, C. Effects of pulsed electric fields on cytomembrane lipids and intracellular nucleic acids of Saccharomyces cerevisiae. Food Control 2014, 39, 204–213. [Google Scholar] [CrossRef]
- Chen, W.; Golden, D.A.; Critzer, F.J. Salmonella survival and differential expression of fatty acid biosynthesis-associated genes in a low-water-activity food. Lett. Appl. Microbiol. 2014, 59, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Aronsson, K.; Rönner, U. Influence of pH, water activity and temperature on the inactivation of Escherichia coli and Saccharomyces cerevisiae by pulsed electric fields. Innov. Food Sci. Emerg. 2001, 2, 105–112. [Google Scholar] [CrossRef]
- Liu, Z.-W.; Zeng, X.-A.; Sun, D.-W.; Han, Z.; Aadil, R.M. Synergistic effect of thermal and pulsed electric field (PEF) treatment on the permeability of soya PC and DPPC vesicles. J. Food Eng. 2015, 153, 124–131. [Google Scholar] [CrossRef]
- Denich, T.J.; Beaudette, L.A.; Lee, H.; Trevors, J.T. Effect of selected environmental and physico-chemical factors on bacterial cytoplasmic membranes. J. Microbiol. Methods 2003, 52, 149–182. [Google Scholar] [CrossRef]
- Khakbaz, P.; Klauda, J.B. Probing the importance of lipid diversity in cell membranes via molecular simulation. Chem. Phys. Lipids 2015, 192, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Mihoub, M.; El May, A.; Aloui, A.; Chatti, A.; Landoulsi, A. Effects of static magnetic fields on growth and membrane lipid composition of Salmonella typhimurium wild-type and dam mutant strains. Int. J. Food Microbiol. 2012, 157, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Russell, N.J. Mechanisms of thermal adaptation in bacteria: Blueprints for survival. Trends Biochem. Sci. 1984, 9, 108–112. [Google Scholar] [CrossRef]
- Stepanyan, S.A.; Soloviev, V.R.; Starikovskaia, S.M. An electric field in nanosecond surface dielectric barrier discharge at different polarities of the high voltage pulse: Spectroscopy measurements and numerical modeling. J. Phys. D Appl. Phys. 2014, 47, 485201–485213. [Google Scholar] [CrossRef]
- Alvarez-Ordonez, A.; Fernandez, A.; Lopez, M.; Arenas, R.; Bernardo, A. Modifications in membrane fatty acid composition of Salmonella typhimurium in response to growth conditions and their effect on heat resistance. Int. J. Food Microbiol. 2008, 123, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Los, D.A.; Murata, N. Membrane fluidity and its roles in the perception of environmental signals. Biochim. Biophys. Acta 2004, 1666, 142–157. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, G.; Beljean-Leymarie, M.; Beyerle, F.; Bonnefont-Rousselot, D.; Cristol, J.P.; Therond, P.; Torreilles, J. Evaluation of lipid peroxidation by assaying the thiobarbituric acid-reactive substances. Ann. Biol. Clin. 1998, 56, 305–319. [Google Scholar]
- Garcia, D.; Manas, P.; Gomez, N.; Raso, J.; Pagan, R. Biosynthetic requirements for the repair of sublethal membrane damage in Escherichia coli cells after pulsed electric fields. J. Appl. Microbiol. 2006, 100, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, K.; Jackowski, S.; Rock, C.O.; Cronan, J.E., Jr. Regulation of fatty acid biosynthesis in Escherichia coli. Microbiol. Rev. 1993, 57, 522–542. [Google Scholar] [PubMed]
- Zhu, K.; Choi, K.H.; Schweizer, H.P.; Rock, C.O.; Zhang, Y.M. Two aerobic pathways for the formation of unsaturated fatty acids in Pseudomonas aeruginosa. Mol. Microbiol. 2006, 60, 260–273. [Google Scholar] [CrossRef] [PubMed]
- Chalova, V.I.; Hernandez-Hernandez, O.; Muthaiyan, A.; Sirsat, S.A.; Natesan, S.; Sanz, M.L.; Moreno, F.J.; O’Bryan, C.A.; Crandall, P.G.; Ricke, S.C. Growth and transcriptional response of Salmonella Typhimurium LT2 to glucose-lysine-based Maillard reaction products generated under low water activity conditions. Food Res. Int. 2012, 45, 1044–1053. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.H.; Kim, S.; Kim, H.G.; Lee, J.; Lee, I.S.; Park, Y.K. The formation of cyclopropane fatty acids in Salmonella enterica serovar Typhimurium. Microbiology 2005, 151, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Calderon, P.F.; Morales, E.H.; Acuna, L.G.; Fuentes, D.N.; Gil, F.; Porwollik, S.; McClelland, M.; Saavedra, C.P.; Calderon, I.L. The small RNA RyhB homologs from Salmonella typhimurium participate in the response to S-nitrosoglutathione-induced stress. Biochem. Biophys. Res. Commun. 2014, 450, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Wouters, P.C.; Bos, A.P.; Ueckert, J. Membrane permeabilization in relation to inactivation kinetics of Lactobacillus species due to pulsed electric fields. Appl. Environ. Microbiol. 2001, 67, 3092–3101. [Google Scholar] [CrossRef] [PubMed]
- García, D.; Gómez, N.; Mañas, P.; Raso, J.; Pagán, R. Pulsed electric fields cause bacterial envelopes permeabilization depending on the treatment intensity, the treatment medium pH and the microorganism investigated. Int. J. Food Microbiol. 2007, 113, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Somolinos, M.; Espina, L.; Pagán, R.; Garcia, D. sigB absence decreased Listeria monocytogenes EGD-e heat resistance but not its pulsed electric fields resistance. Int. J. Food Microbiol. 2010, 141, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Chen, R.; Zeng, X.-A.; Han, Z. Effect of pulsed electric fields assisted acetylation on morphological, structural and functional characteristics of potato starch. Food Chem. 2016, 192, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Zeng, X.-A.; Sun, D.-W.; Liu, D. Effects of pulsed electric field treatment on (+)-catechin–acetaldehyde condensation. Innov. Food Sci. Emerg. 2013, 20, 100–105. [Google Scholar] [CrossRef]
- Sasser, M.; Kunitsky, C.; Jackoway, G.; Ezzell, J.W.; Teska, J.D.; Harper, B.; Parker, S.; Barden, D.; Blair, H.; Breezee, J.; et al. Identification of Bacillus anthracis from culture using gas chromatographic analysis of fatty acid methyl esters. J. AOAC Int. 2005, 88, 178–181. [Google Scholar] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCt Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]





| Fatty Acid Composition | Content (%) | ||
|---|---|---|---|
| Control | PEF for 1.6 ms | PEF for 4.0 ms | |
| Saturated fatty acid (SFA) | |||
| C12:0 | 2.11 ± 0.32 a | 1.96 ± 0.15 a | 1.65 ± 0.12 b |
| C14:0 | 6.71 ± 0.48 a | 5.35 ± 0.12 ab | 5.81 ± 0.31 a |
| C16:0 | 46.04 ± 1.23 a | 50.90 ± 0.23 b | 55.68 ± 0.62 c |
| Unsaturated fatty acid (UFA) | |||
| C16:1 | 5.56 ± 0.41 a | 5.06 ± 0.98 a | 4.84 ± 0.37 a |
| C18:1 | 7.30 ± 0.12 a | 4.54 ± 0.21 b | 3.01 ± 0.16 c |
| Polyunsaturated fatty acid (PUFA) | |||
| C18:2 | 1.33 ± 0.11 a | 0.63 ± 0.09 b | 0.22 ± 0.08 c |
| Cyclic fatty acid (CFA) | |||
| C17:cyclo | 19.21 ± 0.37 a | 18.91 ± 0.93 a | 16.22 ± 0.21 b |
| C19:cyclo | 3.23 ± 0.62 a | 2.94 ± 0.21 a | 1.75 ± 0.14 b |
| C14:0 (3-OH) | 7.40 ± 0.12 a | 7.76 ± 0.15 a | 8.13 ± 0.16 ab |
| Total minor fatty acids | 1.11 ± 0.59 a | 1.95 ± 0.49 a | 2.69 ± 0.68 ab |
| Gene | Sequence (5′ to 3′) | Product Length (bp) |
|---|---|---|
| 16S rRNA | F: TCGTGTTGTGAAATGTTGGGTTA | 66 |
| R: ACCGCTGGCAACAAAGGAT | ||
| fabA | F: GGTTCTTCGGATGCCACTTTAT | 65 |
| R: CATAGCATCCAGACCCAGACAA | ||
| fabD | F: AGTGGACGAAGAGCGTGGAAT | 67 |
| R: CCTGGACCCACTTCATAAAGATG | ||
| cfa | F: CCCCCACCATGTTAAAGATACG | 74 |
| R: AGGCGCGTTTTTTACTTTGTAGA | ||
| cyoA | F: TGGTTTCGCCTGGAAGTATC | 64 |
| R: GTGTGACCAGTTCGGGCTAT | ||
| cyoB | F: GGCACCCATTTCTTTACCAA | 105 |
| R: GACCGGCAGAATCAGAATGT | ||
| cyoC | F: GGATGGCGGTGCTGATG | 67 |
| R: ATGATGCGGGTACGGTTAGTG |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, O.; Zeng, X.-A.; Brennan, C.S.; Han, Z. Effect of Pulsed Electric Field on Membrane Lipids and Oxidative Injury of Salmonella typhimurium. Int. J. Mol. Sci. 2016, 17, 1374. https://doi.org/10.3390/ijms17081374
Yun O, Zeng X-A, Brennan CS, Han Z. Effect of Pulsed Electric Field on Membrane Lipids and Oxidative Injury of Salmonella typhimurium. International Journal of Molecular Sciences. 2016; 17(8):1374. https://doi.org/10.3390/ijms17081374
Chicago/Turabian StyleYun, Ou, Xin-An Zeng, Charles S. Brennan, and Zhong Han. 2016. "Effect of Pulsed Electric Field on Membrane Lipids and Oxidative Injury of Salmonella typhimurium" International Journal of Molecular Sciences 17, no. 8: 1374. https://doi.org/10.3390/ijms17081374