The ADAMTS5 Metzincin Regulates Zebrafish Somite Differentiation
Abstract
:1. Introduction
2. Results
2.1. The Secreted Metalloproteinase ADAMTS5 Is Expressed in Zebrafish Embryos
2.2. Silencing of ADAMTS5 Expression
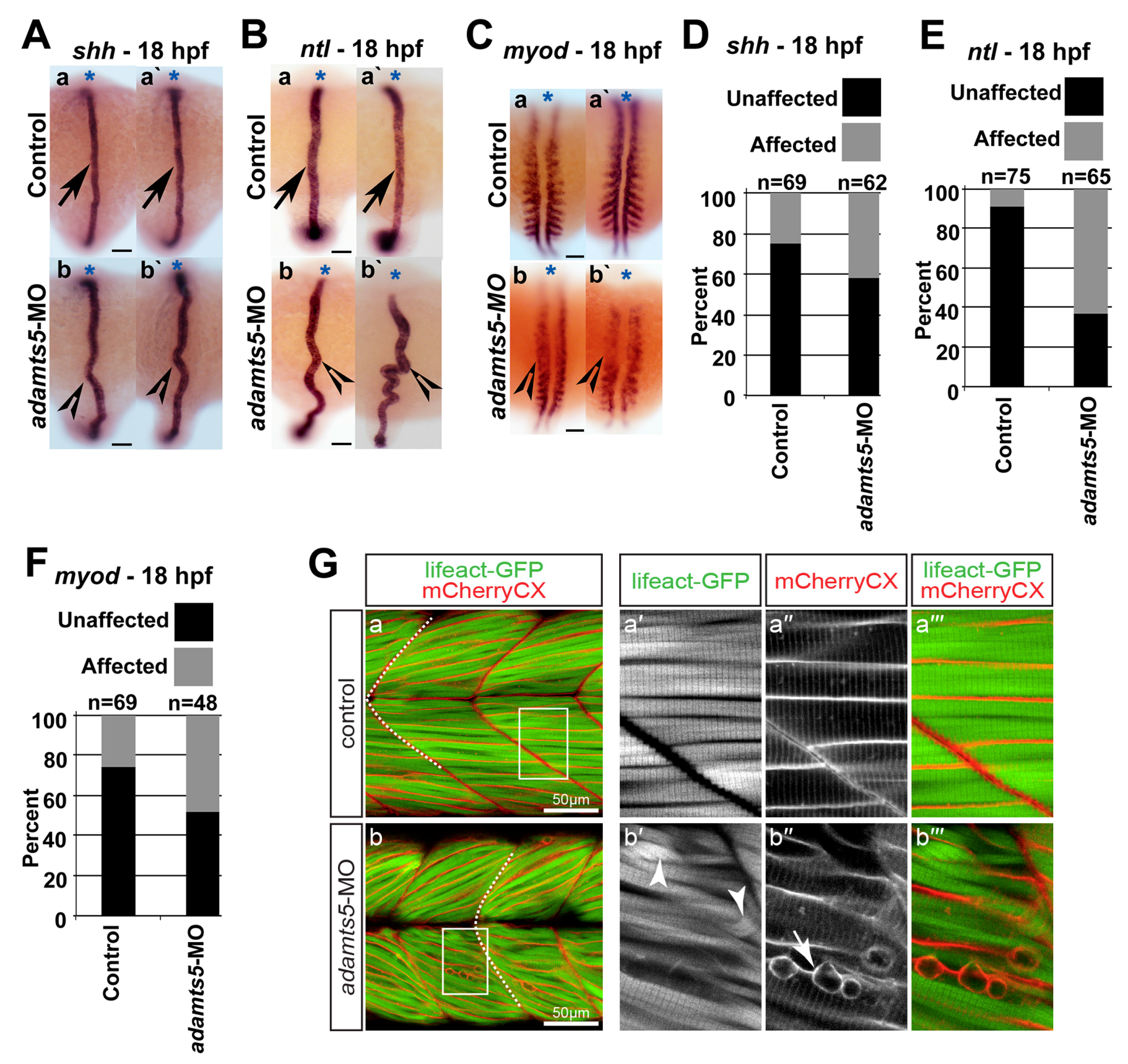
2.3. Notochord Morphology Is Perturbed in adamts5 Morphant Embryos
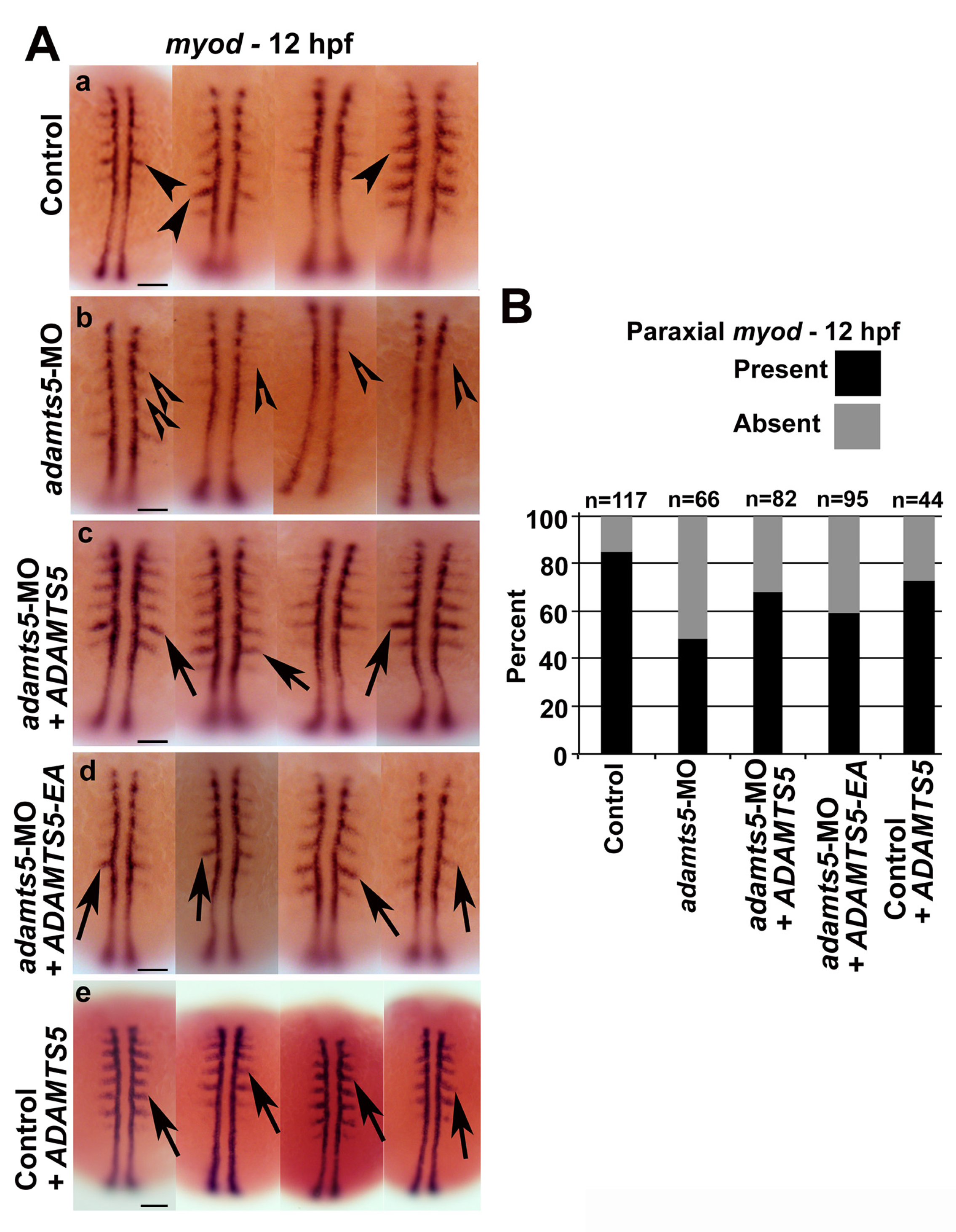
2.4. Skeletal Muscle Formation Is Disrupted in adamts5 Morphant Embryos
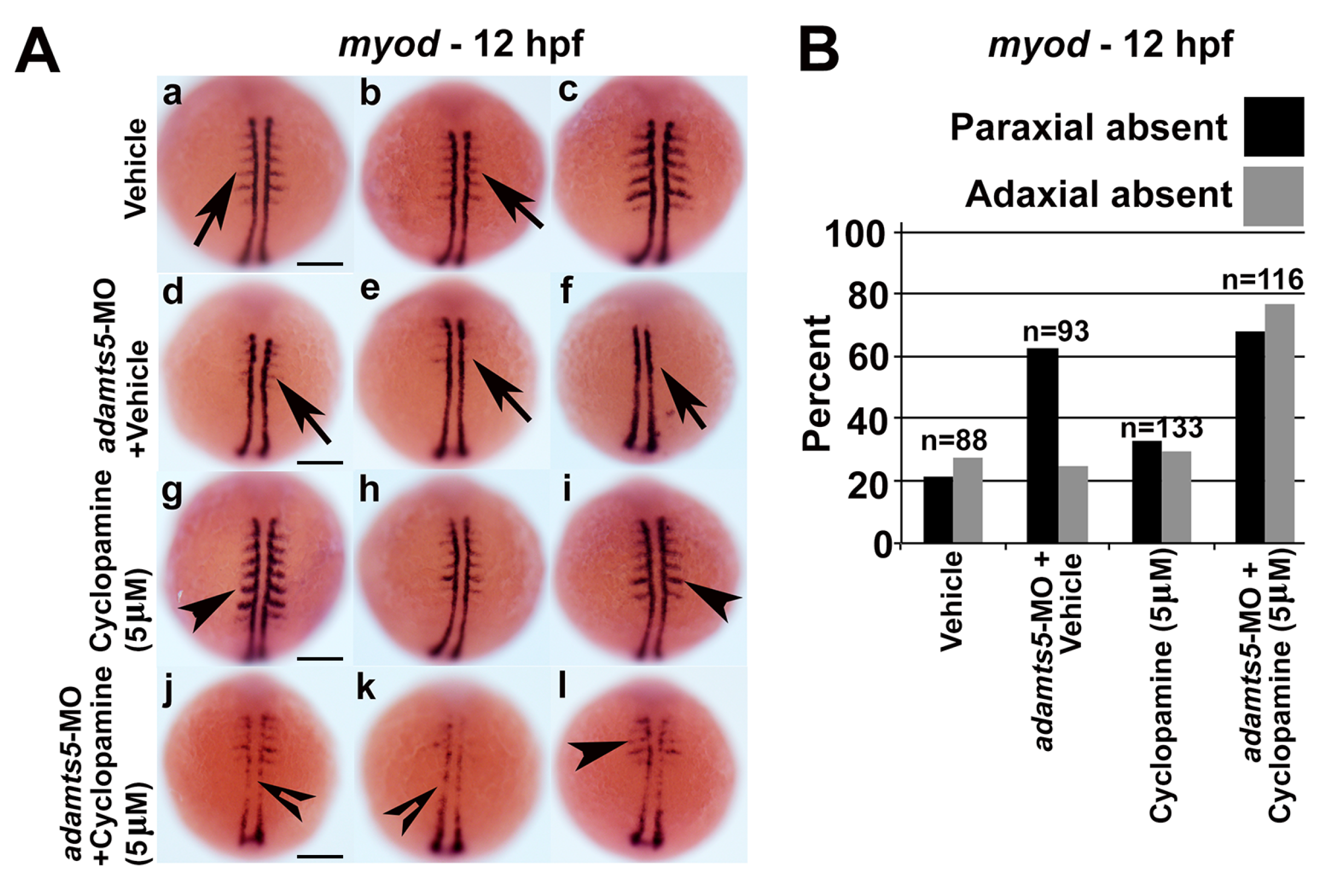
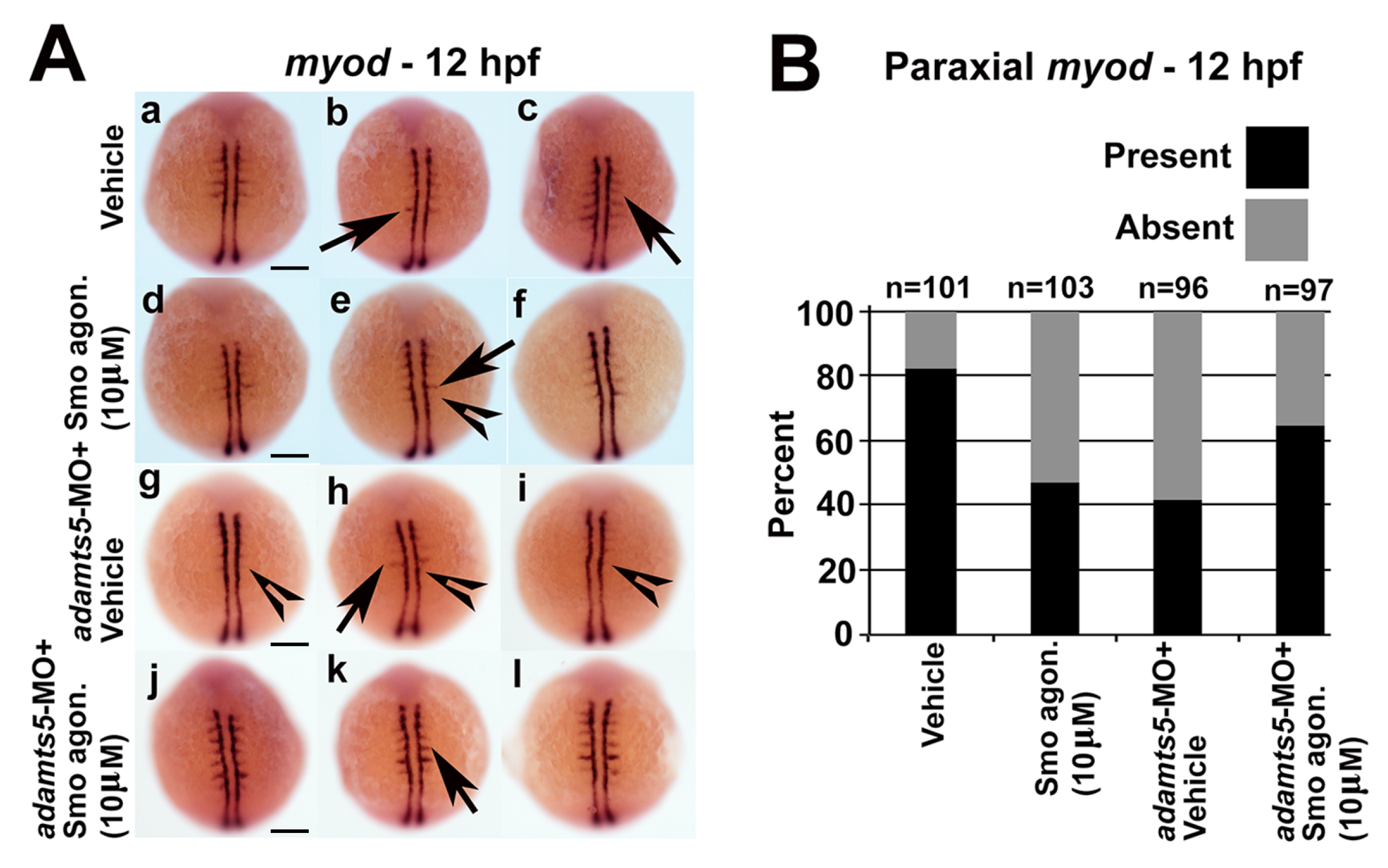
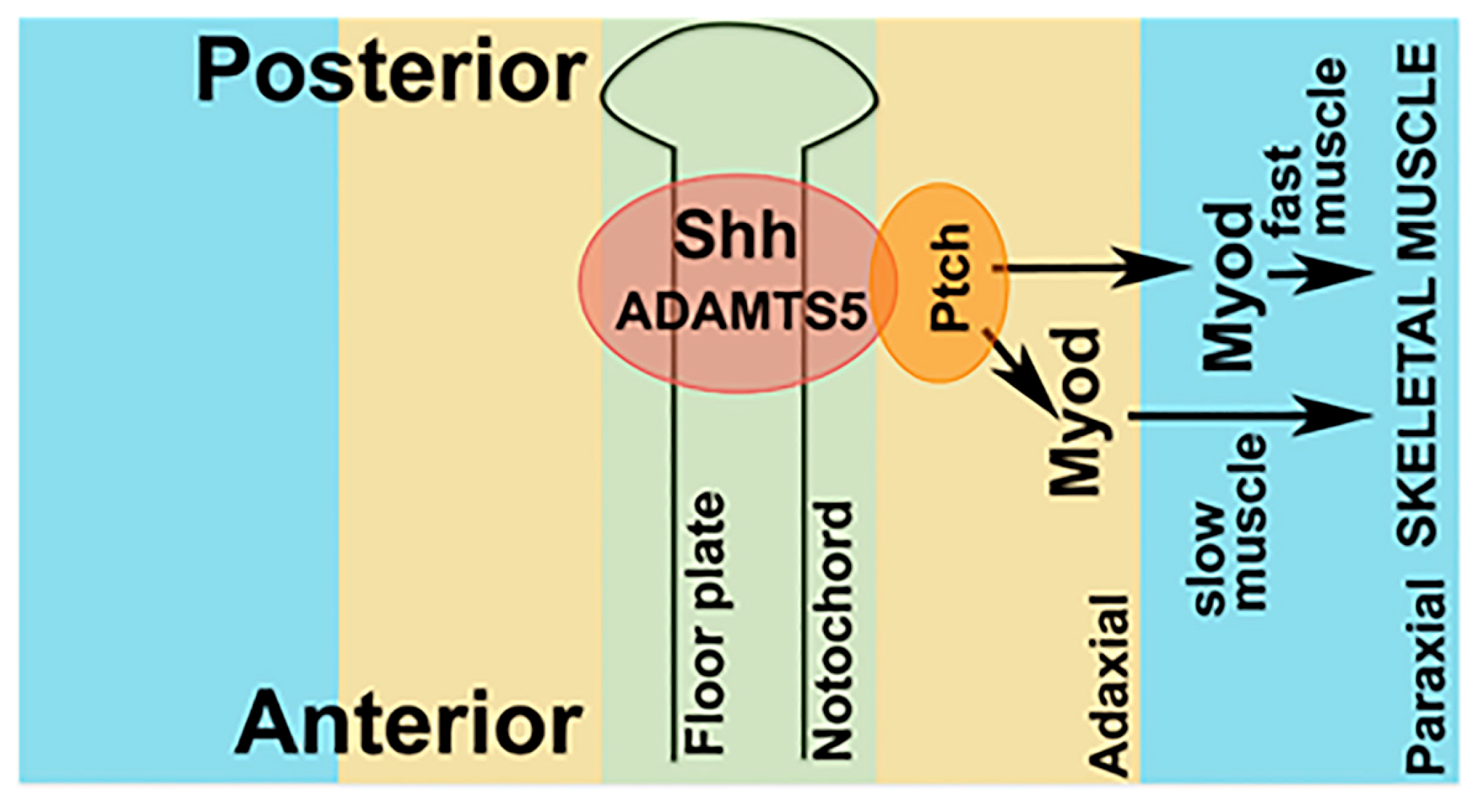
2.5. Receptor-Mediated Sonic Hedgehog Signaling Is Affected in adamts5 Morphants
3. Discussion
4. Materials and Methods
4.1. Zebrafish Lines and Maintenance
4.2. Embryo Microinjection and Other Treatments
4.3. Whole-Mount In Situ Hybridization and Immunofluorescence
4.4. Western Blotting
4.5. Statistics
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Silver, D.L.; Hou, L.; Somerville, R.; Young, M.E.; Apte, S.S.; Pavan, W.J. The secreted metalloprotease ADAMTS20 is required for melanoblast survival. PLoS Genet. 2008, 4, e1000003. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, H.; Nelson, C.M.; Somerville, R.P.; Mielke, K.; Dixon, L.J.; Powell, K.; Apte, S.S. Cooperation of two ADAMTS metalloproteases in closure of the mouse palate identifies a requirement for versican proteolysis in regulating palatal mesenchyme proliferation. Development 2010, 137, 4029–4038. [Google Scholar] [CrossRef] [PubMed]
- Jonsson-Rylander, A.C.; Nilsson, T.; Fritsche-Danielson, R.; Hammarstrom, A.; Behrendt, M.; Andersson, J.O.; Lindgren, K.; Andersson, A.K.; Wallbrandt, P.; Rosengren, B.; et al. Role of ADAMTS-1 in atherosclerosis: Remodeling of carotid artery, immunohistochemistry, and proteolysis of versican. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 180–185. [Google Scholar] [PubMed]
- Porter, S.; Span, P.N.; Sweep, F.C.; Tjan-Heijnen, V.C.; Pennington, C.J.; Pedersen, T.X.; Johnsen, M.; Lund, L.R.; Romer, J.; Edwards, D.R. ADAMTS8 and ADAMTS15 expression predicts survival in human breast carcinoma. Int. J. Cancer 2006, 118, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Iruela-Arispe, M.L.; Carpizo, D.; Luque, A. ADAMTS1: A matrix metalloprotease with angioinhibitory properties. Ann. N. Y. Acad. Sci. 2003, 995, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Kelwick, R.; Wagstaff, L.; Decock, J.; Roghi, C.; Cooley, L.S.; Robinson, S.D.; Arnold, H.; Gavrilovic, J.; Jaworski, D.M.; Yamamoto, K.; et al. Metalloproteinase-dependent and -independent processes contribute to inhibition of breast cancer cell migration, angiogenesis and liver metastasis by a disintegrin and metalloproteinase with thrombospondin motifs-15. Int. J. Cancer 2014, 136, E14–E26. [Google Scholar] [CrossRef] [PubMed]
- Hamel, M.G.; Ajmo, J.M.; Leonardo, C.C.; Zuo, F.; Sandy, J.D.; Gottschall, P.E. Multimodal signaling by the ADAMTSs (a disintegrin and metalloproteinase with thrombospondin motifs) promotes neurite extension. Exp. Neurol. 2008, 210, 428–440. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, D.R.; Nelson, C.M.; Dixon, L.J.; Silver, D.L.; Wylie, J.D.; Lindner, V.; Sasaki, T.; Cooley, M.A.; Argraves, W.S.; Apte, S.S. ADAMTS metalloproteases generate active versican fragments that regulate interdigital web regression. Dev. Cell 2009, 17, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Dubail, J.; Aramaki-Hattori, N.; Bader, H.L.; Nelson, C.M.; Katebi, N.; Matuska, B.; Olsen, B.R.; Apte, S.S. A new Adamts9 conditional mouse allele identifies its non-redundant role in interdigital web regression. Genesis 2014, 52, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, L.E.; McCulloch, D.R.; McGarity, J.D.; Bahan, A.; Wessels, A.; Weber, D.; Diminich, A.M.; Nelson, C.M.; Apte, S.S.; Kern, C.B. Altered versican cleavage in ADAMTS5 deficient mice; a novel etiology of myxomatous valve disease. Dev. Biol. 2011, 357, 152–164. [Google Scholar] [CrossRef] [PubMed]
- Stanton, H.; Rogerson, F.M.; East, C.J.; Golub, S.B.; Lawlor, K.E.; Meeker, C.T.; Little, C.B.; Last, K.; Farmer, P.J.; Campbell, I.K.; et al. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 2005, 434, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Askew, R.; Sheppard, B.; Carito, B.; Blanchet, T.; Ma, H.L.; Flannery, C.R.; Peluso, D.; Kanki, K.; Yang, Z.; et al. Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 2005, 434, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Didangelos, A.; Mayr, U.; Monaco, C.; Mayr, M. Novel role of ADAMTS-5 protein in proteoglycan turnover and lipoprotein retention in atherosclerosis. J. Biol. Chem. 2012, 287, 19341–19345. [Google Scholar] [CrossRef] [PubMed]
- Yayon, A.; Klagsbrun, M.; Esko, J.D.; Leder, P.; Ornitz, D.M. Cell surface, heparin-like molecules are required for binding of basic fibroblast growth factor to its high affinity receptor. Cell 1991, 64, 841–848. [Google Scholar] [CrossRef]
- Rapraeger, A.C.; Krufka, A.; Olwin, B.B. Requirement of heparan sulfate for bFGF-mediated fibroblast growth and myoblast differentiation. Science 1991, 252, 1705–1708. [Google Scholar] [CrossRef] [PubMed]
- Witt, R.M.; Hecht, M.L.; Pazyra-Murphy, M.F.; Cohen, S.M.; Noti, C.; van Kuppevelt, T.H.; Fuller, M.; Chan, J.A.; Hopwood, J.J.; Seeberger, P.H.; et al. Heparan sulfate proteoglycans containing a glypican 5 core and 2-O-sulfo-iduronic acid function as sonic hedgehog co-receptors to promote proliferation. J. Biol. Chem. 2013, 288, 26275–26288. [Google Scholar] [CrossRef] [PubMed]
- Nandadasa, S.; Nelson, C.M.; Apte, S.S. ADAMTS9-mediated extracellular matrix dynamics regulates umbilical cord vascular smooth muscle differentiation and rotation. Cell Rep. 2015, 11, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.C.; Seeto, B.L.; Bartoszko, J.M.; Khoury, M.A.; Whetstone, H.; Ho, L.; Hsu, C.; Ali, S.A.; Alman, B.A. Modulating hedgehog signaling can attenuate the severity of osteoarthritis. Nat. Med. 2009, 15, 1421–1425. [Google Scholar] [CrossRef] [PubMed]
- Stupka, N.; Kintakas, C.; White, J.D.; Fraser, F.W.; Hanciu, M.; Aramaki-Hattori, N.; Martin, S.; Coles, C.; Collier, F.; Ward, A.C.; et al. Versican processing by a disintegrin-like and metalloproteinase domain with thrombospondin-1 repeats proteinases-5 and -15 facilitates myoblast fusion. J. Biol. Chem. 2013, 288, 1907–1917. [Google Scholar] [CrossRef] [PubMed]
- Brunet, F.G.; Fraser, F.W.; Binder, M.J.; Smith, A.D.; Kintakas, C.; Dancevic, C.M.; Ward, A.C.; McCulloch, D.R. The evolutionary conservation of the A Disintegrin-like and Metalloproteinase domain with Thrombospondin-1 motif metzincins across vertebrate species and their expression in teleost zebrafish. BMC Evol. Biol. 2015, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Longpre, J.M.; McCulloch, D.R.; Koo, B.H.; Alexander, J.P.; Apte, S.S.; Leduc, R. Characterization of proADAMTS5 processing by proprotein convertases. Int. J. Biochem. Cell Biol. 2009, 41, 1116–1126. [Google Scholar] [CrossRef] [PubMed]
- Brunet, F.; Kintakas, C.; Smith, A.D.; McCulloch, D.R. The function of the hyalectan class of proteoglycans and their binding partners during vertebrate development. In Advances in Medicine and Biology; Nova Science Publishers Inc.: Hauppauge, NY, USA, 2012; Volume 52, pp. 49–96. [Google Scholar]
- Dancevic, C.M. Deakin University, Waurn Ponds, Victoria, Australia. Unpublished data, 2015. [Google Scholar]
- Blagden, C.S.; Currie, P.D.; Ingham, P.W.; Hughes, S.M. Notochord induction of zebrafish slow muscle mediated by Sonic hedgehog. Genes Dev. 1997, 11, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Halpern, M.E.; Ho, R.K.; Walker, C.; Kimmel, C.B. Induction of muscle pioneers and floor plate is distinguished by the zebrafish no tail mutation. Cell 1993, 75, 99–111. [Google Scholar] [CrossRef]
- Berger, J.; Tarakci, H.; Berger, S.; Li, M.; Hall, T.E.; Arner, A.; Currie, P.D. Loss of Tropomodulin4 in the zebrafish mutant trage causes cytoplasmic rod formation and muscle weakness reminiscent of nemaline myopathy. Dis. Model. Mech. 2014, 7, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Schauerte, H.E.; van Eeden, F.J.; Fricke, C.; Odenthal, J.; Strahle, U.; Haffter, P. Sonic hedgehog is not required for the induction of medial floor plate cells in the zebrafish. Development 1998, 125, 2983–2993. [Google Scholar] [PubMed]
- Lewis, C.; Krieg, P.A. Reagents for developmental regulation of Hedgehog signaling. Methods 2014, 66, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Stickney, H.L.; Barresi, M.J.; Devoto, S.H. Somite development in zebrafish. Dev. Dyn. 2000, 219, 287–303. [Google Scholar] [CrossRef]
- Rida, P.C.; Le Minh, N.; Jiang, Y.J. A Notch feeling of somite segmentation and beyond. Dev. Biol. 2004, 265, 2–22. [Google Scholar] [CrossRef] [PubMed]
- Von Maltzahn, J.; Chang, N.C.; Bentzinger, C.F.; Rudnicki, M.A. Wnt signaling in myogenesis. Trends Cell Biol. 2012, 22, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Devoto, S.H.; Melancon, E.; Eisen, J.S.; Westerfield, M. Identification of separate slow and fast muscle precursor cells in vivo, prior to somite formation. Development 1996, 122, 3371–3380. [Google Scholar] [PubMed]
- Long, J.H.; Adcock, B.; Root, R.G. Force transmission via axial tendons in undulating fish: A dynamic analysis. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2002, 133, 911–929. [Google Scholar] [CrossRef]
- Goody, M.F.; Sher, R.B.; Henry, C.A. Hanging on for the ride: Adhesion to the extracellular matrix mediates cellular responses in skeletal muscle morphogenesis and disease. Dev. Biol. 2015, 401, 75–91. [Google Scholar] [CrossRef] [PubMed]
- Dancevic, C.M.; McCulloch, D.R.; Ward, A.C. The ADAMTS hyalectanase family: Biological insights from diverse species. Biochem. J. 2016, 473, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Le Goff, C.; Morice-Picard, F.; Dagoneau, N.; Wang, L.W.; Perrot, C.; Crow, Y.J.; Bauer, F.; Flori, E.; Prost-Squarcioni, C.; Krakow, D.; et al. ADAMTSL2 mutations in geleophysic dysplasia demonstrate a role for ADAMTS-like proteins in TGF-β bioavailability regulation. Nat. Genet. 2008, 40, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Kroeldrup, L.; Kjaergaard, S.; Kirchhoff, M.; Kock, K.; Brasch-Andersen, C.; Kibaek, M.; Ousager, L.B. Duplication of 7q36.3 encompassing the Sonic Hedgehog (SHH) gene is associated with congenital muscular hypertrophy. Eur. J. Med. Genet. 2012, 55, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Balczerski, B.; Zakaria, S.; Tucker, A.S.; Borycki, A.G.; Koyama, E.; Pacifici, M.; Francis-West, P. Distinct spatiotemporal roles of hedgehog signalling during chick and mouse cranial base and axial skeleton development. Dev. Biol. 2012, 371, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Riddle, R.D.; Johnson, R.L.; Laufer, E.; Tabin, C. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell 1993, 75, 1401–1416. [Google Scholar] [CrossRef]
- Chiang, C.; Litingtung, Y.; Harris, M.P.; Simandl, B.K.; Li, Y.; Beachy, P.A.; Fallon, J.F. Manifestation of the limb prepattern: Limb development in the absence of sonic hedgehog function. Dev. Biol. 2001, 236, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.M.; Tessier-Lavigne, M. Patterning of mammalian somites by surface ectoderm and notochord: Evidence for sclerotome induction by a hedgehog homolog. Cell 1994, 79, 1175–1186. [Google Scholar] [CrossRef]
- Johnson, R.L.; Laufer, E.; Riddle, R.D.; Tabin, C. Ectopic expression of Sonic hedgehog alters dorsal-ventral patterning of somites. Cell 1994, 79, 1165–1173. [Google Scholar] [CrossRef]
- Duprez, D.; Fournier-Thibault, C.; Le Douarin, N. Sonic Hedgehog induces proliferation of committed skeletal muscle cells in the chick limb. Development 1998, 125, 495–505. [Google Scholar] [PubMed]
- Henry, C.A.; Amacher, S.L. Zebrafish slow muscle cell migration induces a wave of fast muscle morphogenesis. Dev. Cell 2004, 7, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Tamamura, Y.; Otani, T.; Kanatani, N.; Koyama, E.; Kitagaki, J.; Komori, T.; Yamada, Y.; Costantini, F.; Wakisaka, S.; Pacifici, M.; et al. Developmental regulation of Wnt/β-catenin signals is required for growth plate assembly, cartilage integrity, and endochondral ossification. J. Biol. Chem. 2005, 280, 19185–19195. [Google Scholar] [CrossRef] [PubMed]
- Julich, D.; Geisler, R.; Holley, S.A. Tübingen 2000 Screen Consortium. Integrinalpha5 and delta/notch signaling have complementary spatiotemporal requirements during zebrafish somitogenesis. Dev. Cell 2005, 8, 575–586. [Google Scholar] [PubMed]
- Gansner, J.M.; Madsen, E.C.; Mecham, R.P.; Gitlin, J.D. Essential role for fibrillin-2 in zebrafish notochord and vascular morphogenesis. Dev. Dyn. 2008, 237, 2844–2861. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.S.; Wilm, T.P.; Smith, J.; Bagnat, M.; Dale, R.M.; Topczewski, J.; Johnson, S.L.; Solnica-Krezel, L. Loss of col8a1a function during zebrafish embryogenesis results in congenital vertebral malformations. Dev. Biol. 2014, 386, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Parsons, M.J.; Pollard, S.M.; Saude, L.; Feldman, B.; Coutinho, P.; Hirst, E.M.; Stemple, D.L. Zebrafish mutants identify an essential role for laminins in notochord formation. Development 2002, 129, 3137–3146. [Google Scholar] [PubMed]
- Pollard, S.M.; Parsons, M.J.; Kamei, M.; Kettleborough, R.N.; Thomas, K.A.; Pham, V.N.; Bae, M.K.; Scott, A.; Weinstein, B.M.; Stemple, D.L. Essential and overlapping roles for laminin alpha chains in notochord and blood vessel formation. Dev. Biol. 2006, 289, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Snow, C.J.; Peterson, M.T.; Khalil, A.; Henry, C.A. Muscle development is disrupted in zebrafish embryos deficient for fibronectin. Dev. Dyn. 2008, 237, 2542–2553. [Google Scholar] [CrossRef] [PubMed]
- Peterson, M.T.; Henry, C.A. Hedgehog signaling and laminin play unique and synergistic roles in muscle development. Dev. Dyn. 2010, 239, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, M.H.; Alrowaished, S.S.; Goody, M.F.; Crawford, B.D.; Henry, C.A. Laminin and Matrix metalloproteinase 11 regulate Fibronectin levels in the zebrafish myotendinous junction. Skelet. Muscle 2016, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Nasevicius, A.; Ekker, S.C. Effective targeted gene ‘knockdown’ in zebrafish. Nat Genet 2000, 26, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Thisse, C.; Thisse, B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat. Protoc. 2008, 3, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Gibert, Y.; Gajewski, A.; Meyer, A.; Begemann, G. Induction and prepatterning of the zebrafish pectoral fin bud requires axial retinoic acid signaling. Development 2006, 133, 2649–2659. [Google Scholar] [CrossRef] [PubMed]
- Begemann, G.; Schilling, T.F.; Rauch, G.J.; Geisler, R.; Ingham, P.W. The zebrafish neckless mutation reveals a requirement for raldh2 in mesodermal signals that pattern the hindbrain. Development 2001, 128, 3081–3094. [Google Scholar] [PubMed]
- Lange, M.; Norton, W.; Coolen, M.; Chaminade, M.; Merker, S.; Proft, F.; Schmitt, A.; Vernier, P.; Lesch, K.P.; Bally-Cuif, L. The ADHD-susceptibility gene lphn3.1 modulates dopaminergic neuron formation and locomotor activity during zebrafish development. Mol. Psychiatry 2012, 17, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Dancevic, C.M.; Fraser, F.W.; Smith, A.D.; Stupka, N.; Ward, A.C.; McCulloch, D.R. Biosynthesis and expression of a Disintegrin-like and Metalloproteinase domain with Thrombospondin-1 repeats-15: A novel versican-cleaving proteoglycanase. J. Biol. Chem. 2013, 288, 37267–37276. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dancevic, C.M.; Gibert, Y.; Berger, J.; Smith, A.D.; Liongue, C.; Stupka, N.; Ward, A.C.; McCulloch, D.R. The ADAMTS5 Metzincin Regulates Zebrafish Somite Differentiation. Int. J. Mol. Sci. 2018, 19, 766. https://doi.org/10.3390/ijms19030766
Dancevic CM, Gibert Y, Berger J, Smith AD, Liongue C, Stupka N, Ward AC, McCulloch DR. The ADAMTS5 Metzincin Regulates Zebrafish Somite Differentiation. International Journal of Molecular Sciences. 2018; 19(3):766. https://doi.org/10.3390/ijms19030766
Chicago/Turabian StyleDancevic, Carolyn M., Yann Gibert, Joachim Berger, Adam D. Smith, Clifford Liongue, Nicole Stupka, Alister C. Ward, and Daniel R. McCulloch. 2018. "The ADAMTS5 Metzincin Regulates Zebrafish Somite Differentiation" International Journal of Molecular Sciences 19, no. 3: 766. https://doi.org/10.3390/ijms19030766





