Mitochondrial Proteins Unveil the Mechanism by Which Physical Exercise Ameliorates Memory, Learning and Motor Activity in Hypoxic Ischemic Encephalopathy Rat Model
Abstract
:1. Introduction
2. Results
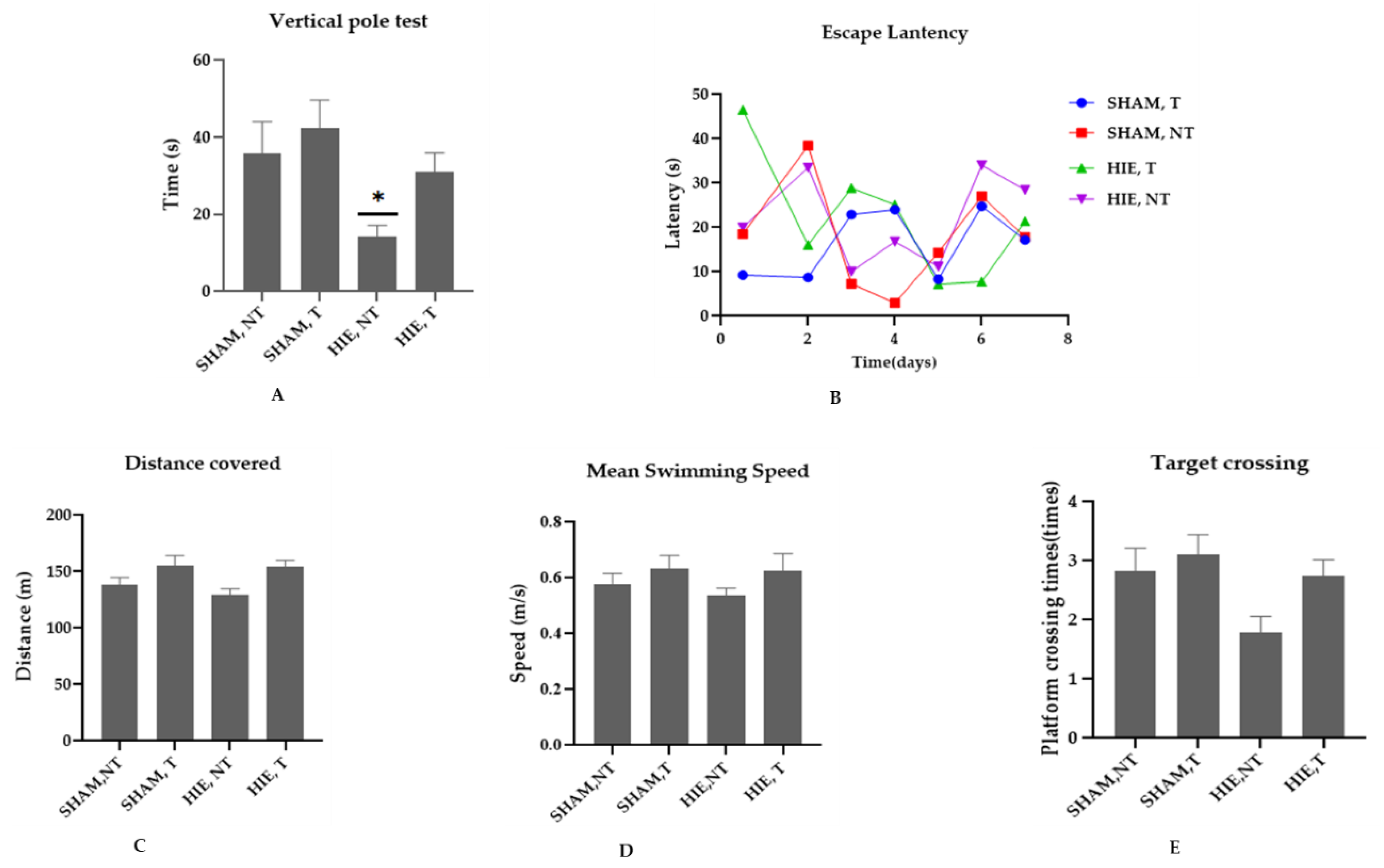
2.1. Swimming Exercise Promotes Motor and Cognitive Performance
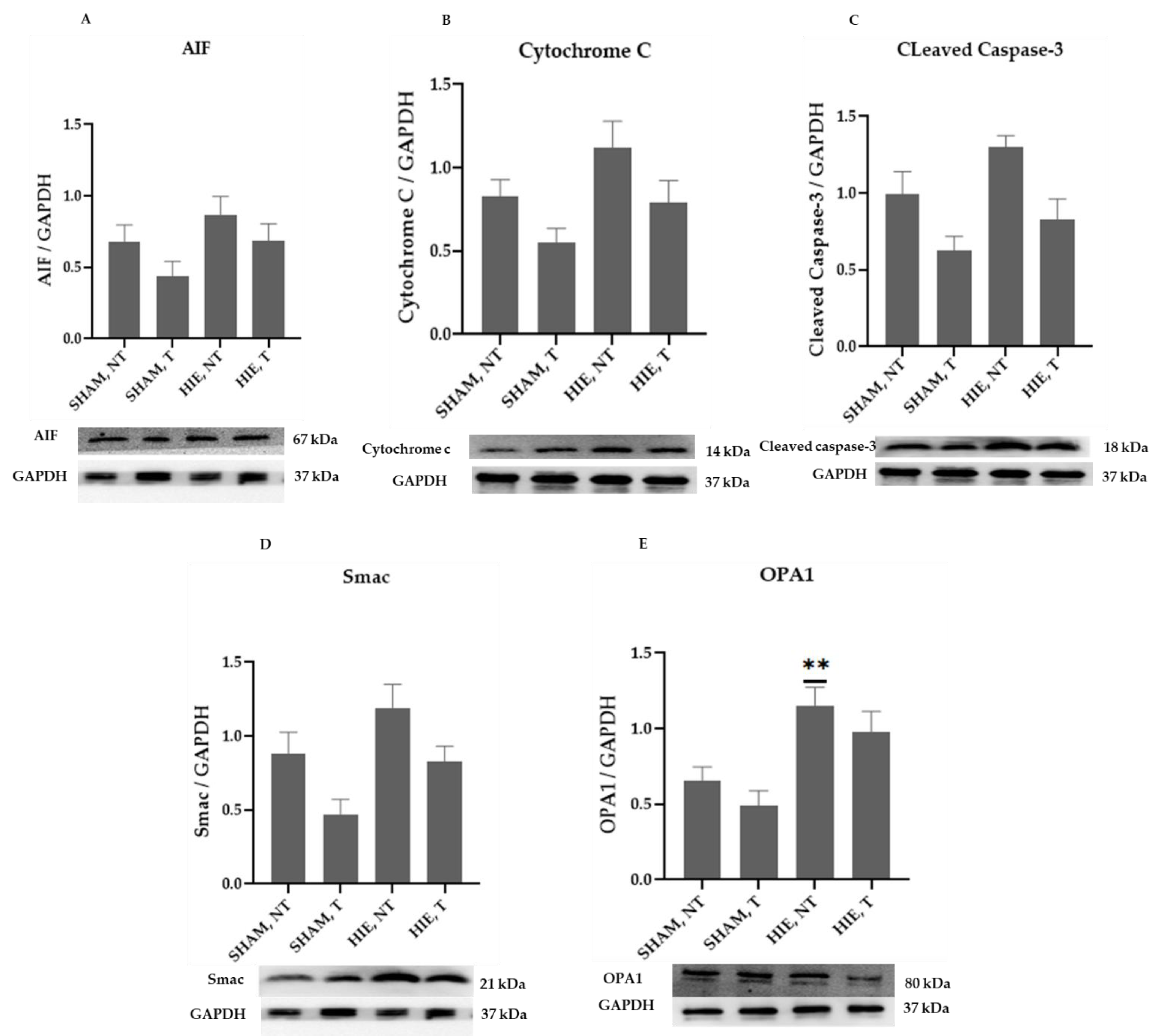
2.2. Mitochondrial Proteins in the Cytoplasm of the Hippocampus
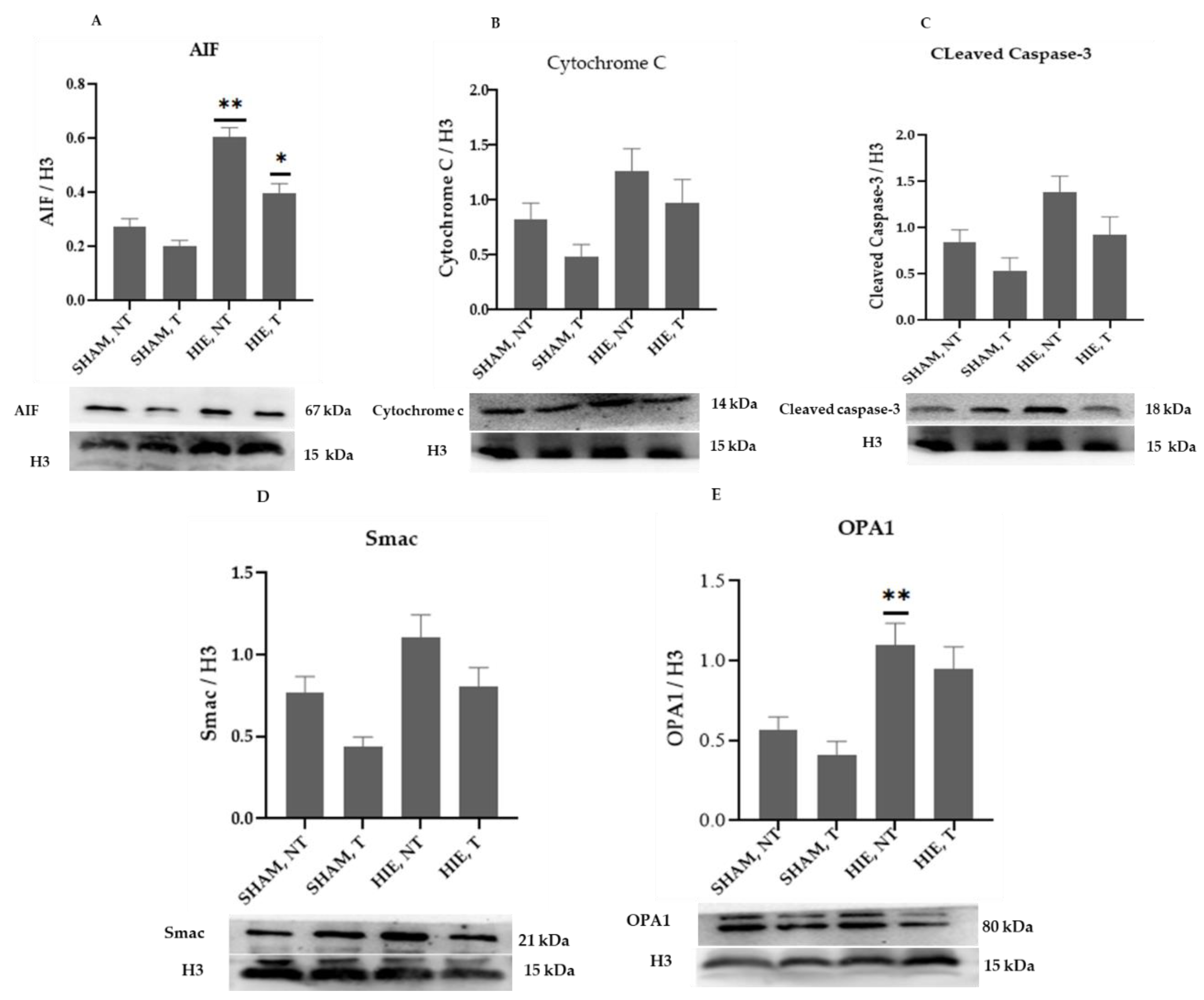
2.3. Mitochondrial Proteins in the Nuclei of the Hippocampus
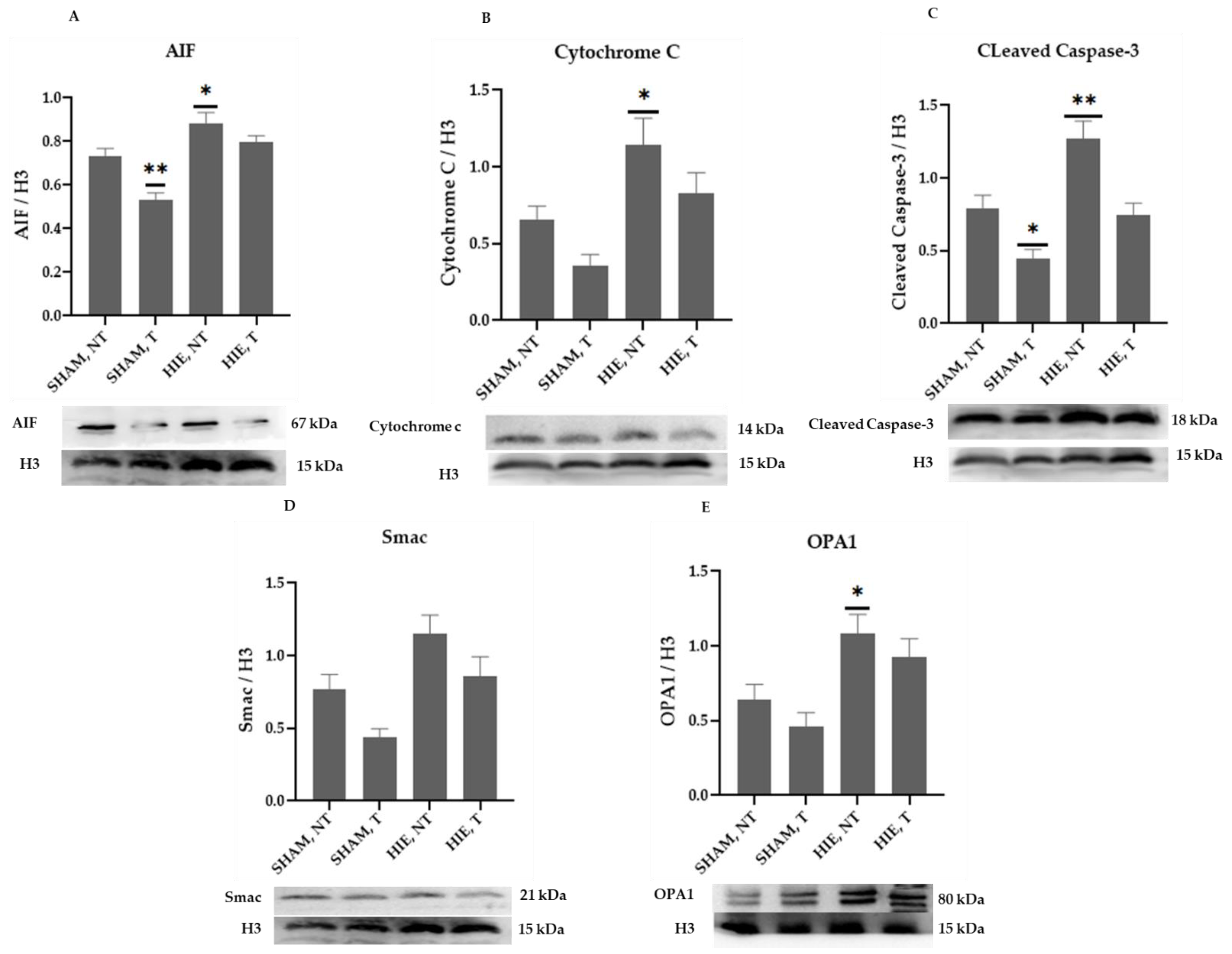
2.4. Mitochondrial Proteins in the Cytoplasm of the Cerebral Cortex
2.5. Mitochondrial Proteins in the Nuclei of the Cerebral Cortex
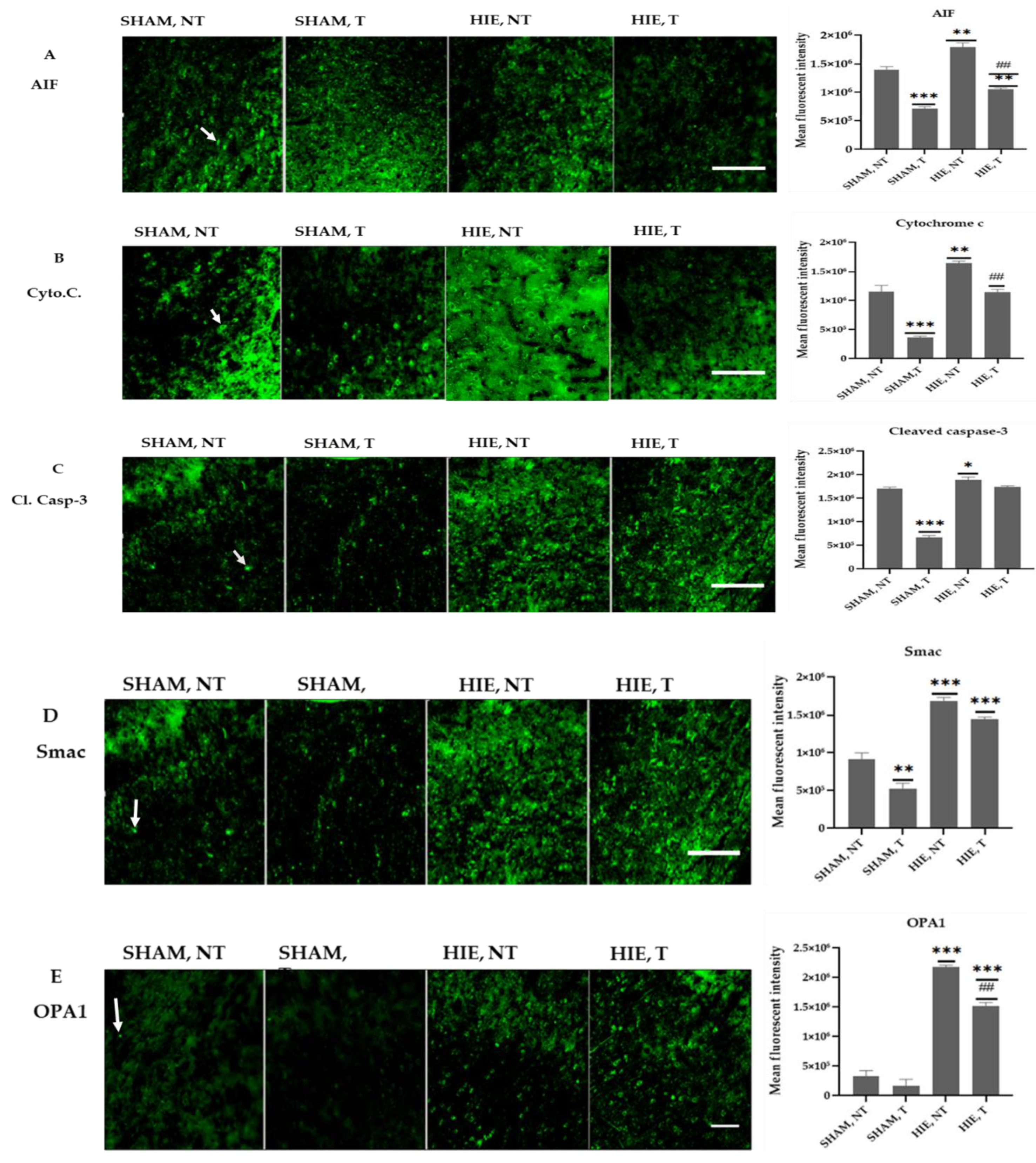
2.6. Immunofluorescence Analysis of Proteins in the Motor Cortex
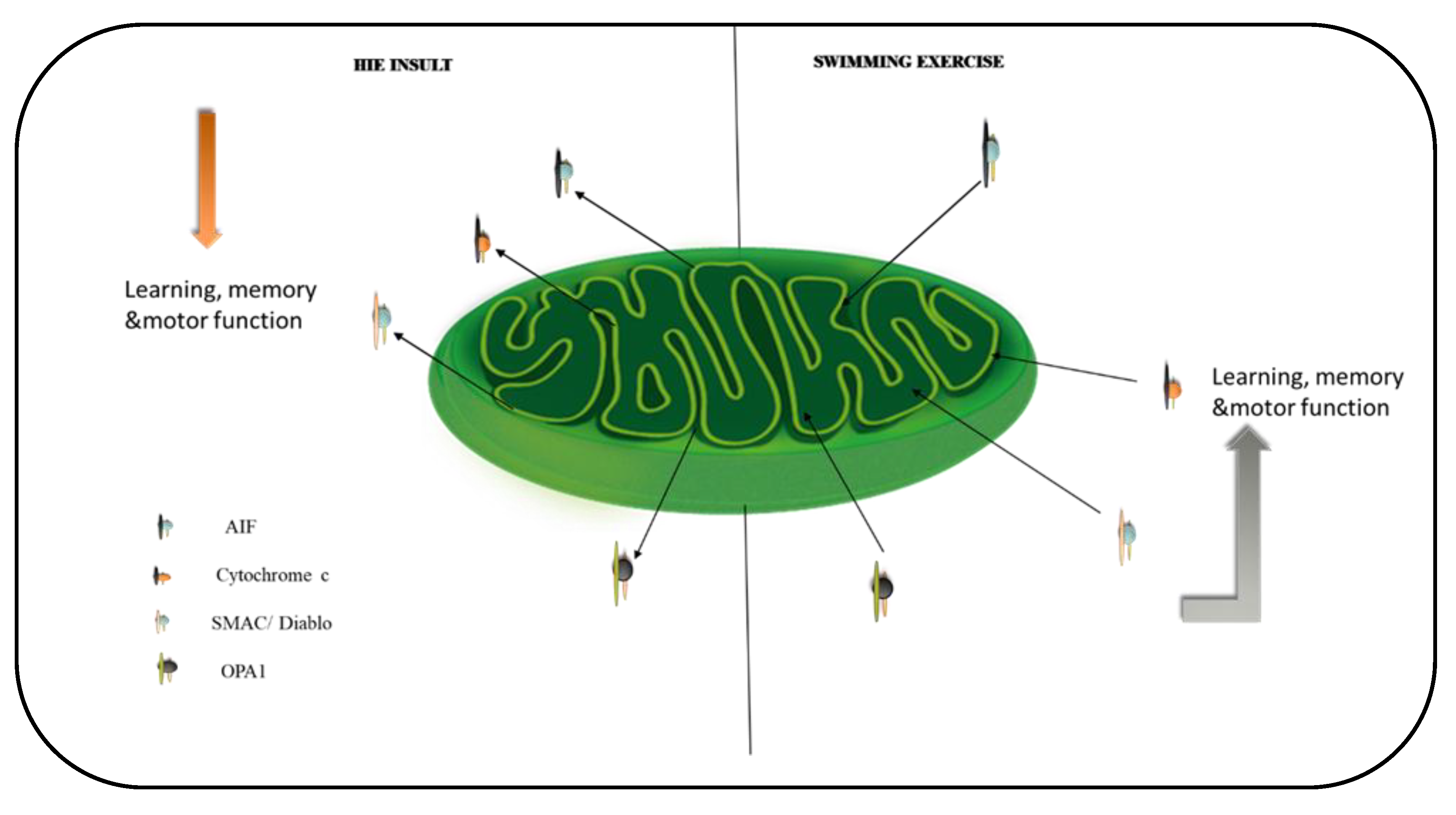
3. Discussion
4. Materials and Methods
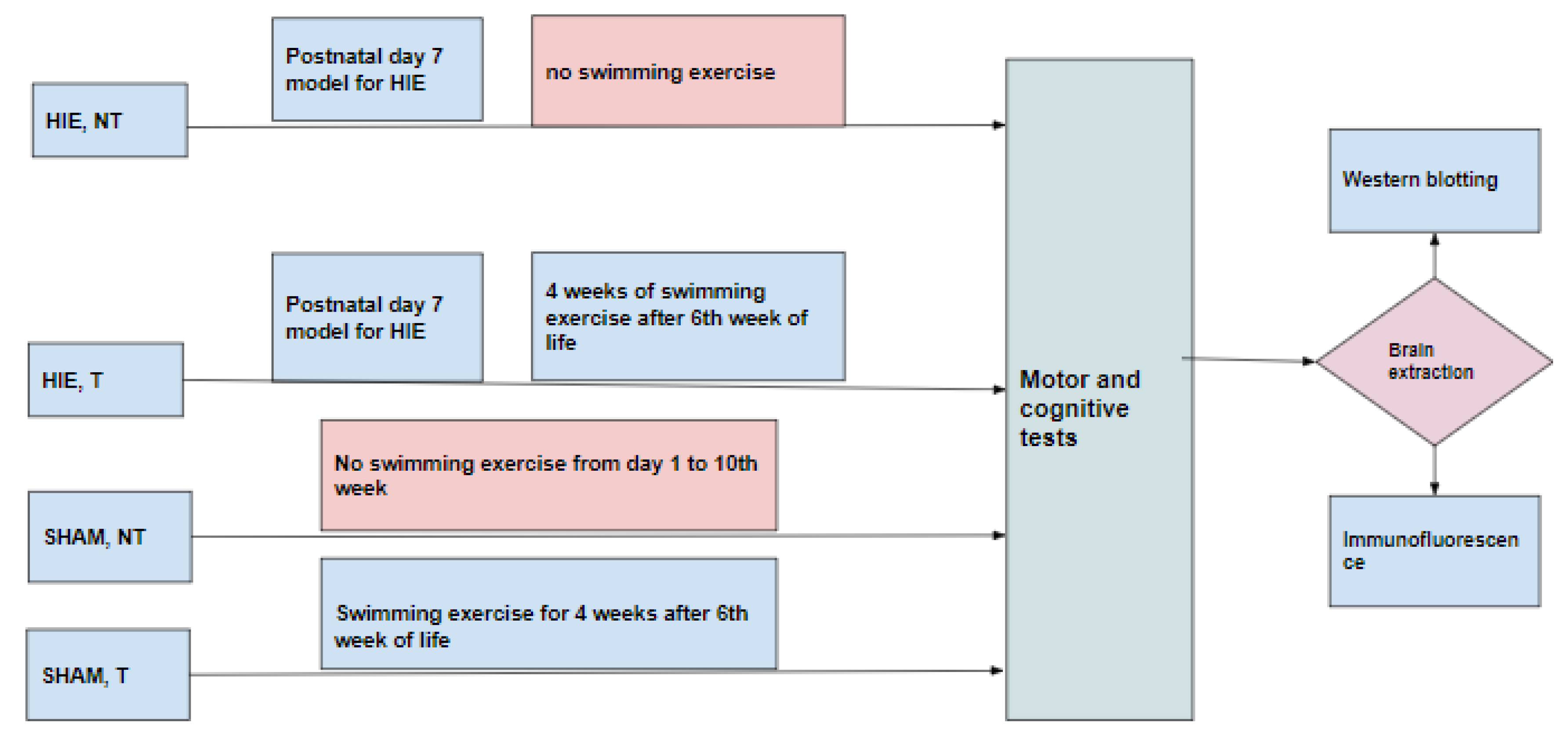
4.1. Animals and Experimental Groups
4.2. Exercise Paradigm
4.3. Vertical Pole Motor Test
4.4. Morris Water Maze Test (MWM)
4.5. Western Blotting
4.6. Immunofluorescence
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AIF | Apoptosis-inducing factor |
| Cyto. C | Cytochrome C |
| Endo G | Endonuclease G |
| HIE | Hypoxic ischemic encephalopathy |
| HIE, NT | Hypoxia ischemic encephalopathy with no exercise training |
| HIE, T | Hypoxia ischemic encephalopathy with exercise training |
| MFI | Mean fluorescence intensity |
| MWM | MWM Morris water maze |
| SHAM, NT | Sham group with no exercise training |
| SHAM, T | Sham group with exercise training |
| Smac | Second mitochondria-derived activator of caspase |
| OMM | Outer mitochondrial membrane |
| OPA1 | Optic atrophy-1 |
References
- Li, B.; Concepcion, K.; Meng, X.; Zhang, L. Brain-immune interactions in perinatal hypoxic-ischemic brain injury. Prog. Neurobiol. 2017, 159, 50–68. [Google Scholar] [CrossRef]
- Douglas-Escobar, M.; Weiss, M.D. Hypoxic-ischemic encephalopathy: A review for the clinician. JAMA Pediatr. 2015, 169, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Black, R.E.; Cousens, S.; Johnson, H.L.; Lawn, J.; Rudan, I.; Bassani, D.; Jha, P.; Campbell, H.; Walker, C.F.; Cibulskis, R.; et al. Global, regional, and national causes of child mortality in 2008: A systematic analysis. Lancet 2010, 375, 1969–1987. [Google Scholar] [CrossRef]
- Juul, S.E.; Ferriero, D.M. Pharmacologic Neuroprotective Strategies in Neonatal Brain Injury. Clin. Perinatol. 2014, 41, 119–131. [Google Scholar] [CrossRef] [Green Version]
- Zhu, C.; Rodriguez, J.I.; Li, T.; Xu, Y.; Sun, Y. Role of apoptosis-inducing factor in perinatal hypoxic-ischemic brain injury. Neural Regen. Res. 2021, 16, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Song, S.-H.; Jee, Y.-S.; Ko, I.-G.; Lee, S.-W.; Sim, Y.-J.; Kim, D.-Y.; Lee, S.-J.; Cho, Y.S. Treadmill exercise and wheel exercise improve motor function by suppressing apoptotic neuronal cell death in brain inflammation rats. J. Exerc. Rehabil. 2018, 14, 911–919. [Google Scholar] [CrossRef]
- Kelly, M.; Darrah, J. Aquatic exercise for children with cerebral palsy. Dev. Med. Child. Neurol. 2005, 47, 838–842. [Google Scholar] [CrossRef]
- Müller, P.; Duderstadt, Y.; Lessmann, V.; Müller, N.G. Lactate and BDNF: Key Mediators of Exercise Induced Neuroplasticity? J. Clin. Med. 2020, 9, 1136. [Google Scholar] [CrossRef]
- Intlekofer, K.A.; Cotman, C.W. Exercise counteracts declining hippocampal function in aging and Alzheimer’s disease. Neurobiol. Dis. 2013; in press. [Google Scholar] [CrossRef]
- Real, C.; Ferreira, A.; Chaves-Kirsten, G.; Torrão, A.; Pires, R.; Britto, L. BDNF receptor blockade hinders the beneficial effects of exercise in a rat model of Parkinson’s disease. Neuroscience 2013, 237, 118–129. [Google Scholar] [CrossRef]
- Chae, S.Y.; Jang, J.H.; HoIm, G.; Jeong, J.-H.; Jung, W.-B.; Ko, S.; Jie, H.; Kim, J.H.; Chang, Y.S.; Chung, S.; et al. Physical exercise enhances adult cortical plasticity in a neonatal rat model of hypoxic-ischemic injury: Evidence from BOLD-fMRI and electrophysiological recordings. NeuroImage 2019, 188, 335–346. [Google Scholar] [CrossRef]
- Fernandes, M.S.D.S.; Ordônio, T.F.; Santos, G.C.J.; Santos, L.E.R.; Calazans, C.T.; Gomes, D.A.; Santos, T.M. Effects of Physical Exercise on Neuroplasticity and Brain Function: A Systematic Review in Human and Animal Studies. Neural Plast. 2020, 2020, 8856621. [Google Scholar] [CrossRef] [PubMed]
- Kujach, S.; Olek, R.A.; Byun, K.; Suwabe, K.; Sitek, E.; Ziemann, E.; Laskowski, R.; Soya, H. Acute Sprint Interval Exercise Increases Both Cognitive Functions and Peripheral Neurotrophic Factors in Humans: The Possible Involvement of Lactate. Front. Neurosci. 2020, 13, 1455. [Google Scholar] [CrossRef] [PubMed]
- Tarassova, O.; Ekblom, M.M.; Moberg, M.; Lövdén, M.; Nilsson, J. Peripheral BDNF Response to Physical and Cognitive Exercise and Its Association With Cardiorespiratory Fitness in Healthy Older Adults. Front. Physiol. 2020, 11, 1080. [Google Scholar] [CrossRef] [PubMed]
- Roberts, F.L.; Markby, G.R. New Insights into Molecular Mechanisms Mediating Adaptation to Exercise; A Review Focusing on Mitochondrial Biogenesis, Mitochondrial Function, Mitophagy and Autophagy. Cells 2021, 10, 2639. [Google Scholar] [CrossRef] [PubMed]
- Baburamani, A.A.; Hurling, C.; Stolp, H.; Sobotka, K.; Gressens, P.; Hagberg, H.; Thornton, C. Mitochondrial Optic Atrophy (OPA) 1 Processing Is Altered in Response to Neonatal Hypoxic-Ischemic Brain Injury. Int. J. Mol. Sci. 2015, 16, 22509–22526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Y.; Tucker, D.; Dong, Y.; Zhao, N.; Zhuo, X.; Zhang, Q. Role of Mitochondria in Neonatal Hypoxic-Ischemic Brain Injury. J. Neurosci. Rehabil. 2015, 2, 1. [Google Scholar] [PubMed]
- Niizuma, K.; Yoshioka, H.; Chen, H.; Kim, G.S.; Jung, J.E.; Katsu, M.; Okami, N.; Chan, P.H. Mitochondrial and apoptotic neuronal death signaling pathways in cerebral ischemia. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2010, 1802, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Del Re, D.P.; Amgalan, D.; Linkermann, A.; Liu, Q.; Kitsis, R.N. Fundamental Mechanisms of Regulated Cell Death and Implications for Heart Disease. Physiol. Rev. 2019, 99, 1765–1817. [Google Scholar] [CrossRef]
- Steiner, J.L.; Murphy, E.A.; McClellan, J.L.; Carmichael, M.D.; Davis, J.M. Exercise training increases mitochondrial biogenesis in the brain. J. Appl. Physiol. 2011, 111, 1066–1071. [Google Scholar] [CrossRef] [Green Version]
- Yun, H.-S.; Park, M.-S.; Ji, E.-S.; Kim, T.-W.; Ko, I.-G.; Kim, H.-B.; Kim, H. Treadmill exercise ameliorates symptoms of attention deficit/hyperactivity disorder through reducing Purkinje cell loss and astrocytic reaction in spontaneous hypertensive rats. J. Exerc. Rehabil. 2014, 10, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Andreska, T.; Rauskolb, S.; Schukraft, N.; Lüningschrör, P.; Sasi, M.; Signoret-Genest, J.; Behringer, M.; Blum, R.; Sauer, M.; Tovote, P.; et al. Induction of BDNF Expression in Layer II/III and Layer V Neurons of the Motor Cortex Is Essential for Motor Learning. J. Neurosci. 2020, 40, 6289–6308. [Google Scholar] [CrossRef]
- Huang, M.L.-H.; Chiang, S.; Kalinowski, D.S.; Bae, D.-H.; Sahni, S.; Richardson, D.R. The Role of the Antioxidant Response in Mitochondrial Dysfunction in Degenerative Diseases: Cross-Talk between Antioxidant Defense, Autophagy, and Apoptosis. Oxidative Med. Cell. Longev. 2019, 2019, 6392763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Memme, J.M.; Hood, D.A. Molecular Basis for the Therapeutic Effects of Exercise on Mitochondrial Defects. Front. Physiol. 2021, 11, 615038. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.; Zhou, Z.; Cohn, W.; Norheim, F.; Lin, A.J.; Kalajian, N.; Strumwasser, A.R.; Cory, K.; Whitney, K.; Ho, T.; et al. The impact of exercise on mitochondrial dynamics and the role of Drp1 in exercise performance and training adaptations in skeletal muscle. Mol. Metab. 2019, 21, 51–67. [Google Scholar] [CrossRef]
- Misrani, A.; Tabassum, S.; Yang, L. Mitochondrial Dysfunction and Oxidative Stress in Alzheimer’s Disease. Front. Aging Neurosci. 2021, 13, 617588. [Google Scholar] [CrossRef]
- Li, T.; Li, K.; Zhang, S.; Wang, Y.; Xu, Y.; Cronin, S.J.F.; Sun, Y.; Zhang, Y.; Xie, C.; Rodriguez, J.I.; et al. Overexpression of apoptosis inducing factor aggravates hypoxic-ischemic brain injury in neonatal mice. Cell Death Dis. 2020, 11, 77. [Google Scholar] [CrossRef] [Green Version]
- Cheng, J.; Shen, W.; Jin, L.; Pan, J.; Zhou, Y.; Pan, G.; Xie, Q.; Hu, Q.; Wu, S.; Zhang, H.; et al. Treadmill exercise promotes neurogenesis and myelin repair via upregulating Wnt/β-catenin signaling pathways in the juvenile brain following focal cerebral ischemia/reperfusion. Int. J. Mol. Med. 2020, 45, 1447–1463. [Google Scholar] [CrossRef] [Green Version]
- El Hayek, L.; Khalifeh, M.; Zibara, V.; Abi Assaad, R.; Emmanuel, N.; Karnib, N.; El-Ghandour, R.; Nasrallah, P.; Bilen, M.; Ibrahim, P.; et al. Lactate mediates the effects of exercise on learning and memory through SIRT1-dependent activation of hippocampal brain-derived neurotrophic factor (BDNF). J. Neurosci. 2019, 39, 2369–2382. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.; Smith, S.B.; Yoon, Y. The short variant of the mitochondrial dynamin OPA1 maintains mitochondrial energetics and cristae structure. J. Biol. Chem. 2017, 292, 7115–7130. [Google Scholar] [CrossRef] [Green Version]
- Olichon, A.; Baricault, L.; Gas, N.; Guillou, E.; Valette, A.; Belenguer, P.; Lenaers, G. Loss of OPA1 Perturbates the Mitochondrial Inner Membrane Structure and Integrity, Leading to Cytochrome c Release and Apoptosis. J. Biol. Chem. 2003, 278, 7743–7746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Methods 1984, 11, 47–60. [Google Scholar] [CrossRef]
- Shihan, M.H.; Novo, S.G.; Le Marchand, S.J.; Wang, Y.; Duncan, M.K. A simple method for quantitating confocal fluorescent images. Biochem. Biophys. Rep. 2021, 25, 100916. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gendi, F.; Pei, F.; Wang, Y.; Li, H.; Fu, J.; Chang, C. Mitochondrial Proteins Unveil the Mechanism by Which Physical Exercise Ameliorates Memory, Learning and Motor Activity in Hypoxic Ischemic Encephalopathy Rat Model. Int. J. Mol. Sci. 2022, 23, 4235. https://doi.org/10.3390/ijms23084235
Gendi F, Pei F, Wang Y, Li H, Fu J, Chang C. Mitochondrial Proteins Unveil the Mechanism by Which Physical Exercise Ameliorates Memory, Learning and Motor Activity in Hypoxic Ischemic Encephalopathy Rat Model. International Journal of Molecular Sciences. 2022; 23(8):4235. https://doi.org/10.3390/ijms23084235
Chicago/Turabian StyleGendi, Fred, Feifei Pei, Yuan Wang, Haoye Li, Jia Fu, and Cheng Chang. 2022. "Mitochondrial Proteins Unveil the Mechanism by Which Physical Exercise Ameliorates Memory, Learning and Motor Activity in Hypoxic Ischemic Encephalopathy Rat Model" International Journal of Molecular Sciences 23, no. 8: 4235. https://doi.org/10.3390/ijms23084235






