Role of the Short Distance Order in Glass Reactivity
Abstract
:1. Introduction
2. Traditional Characterization of Bioactive Glasses
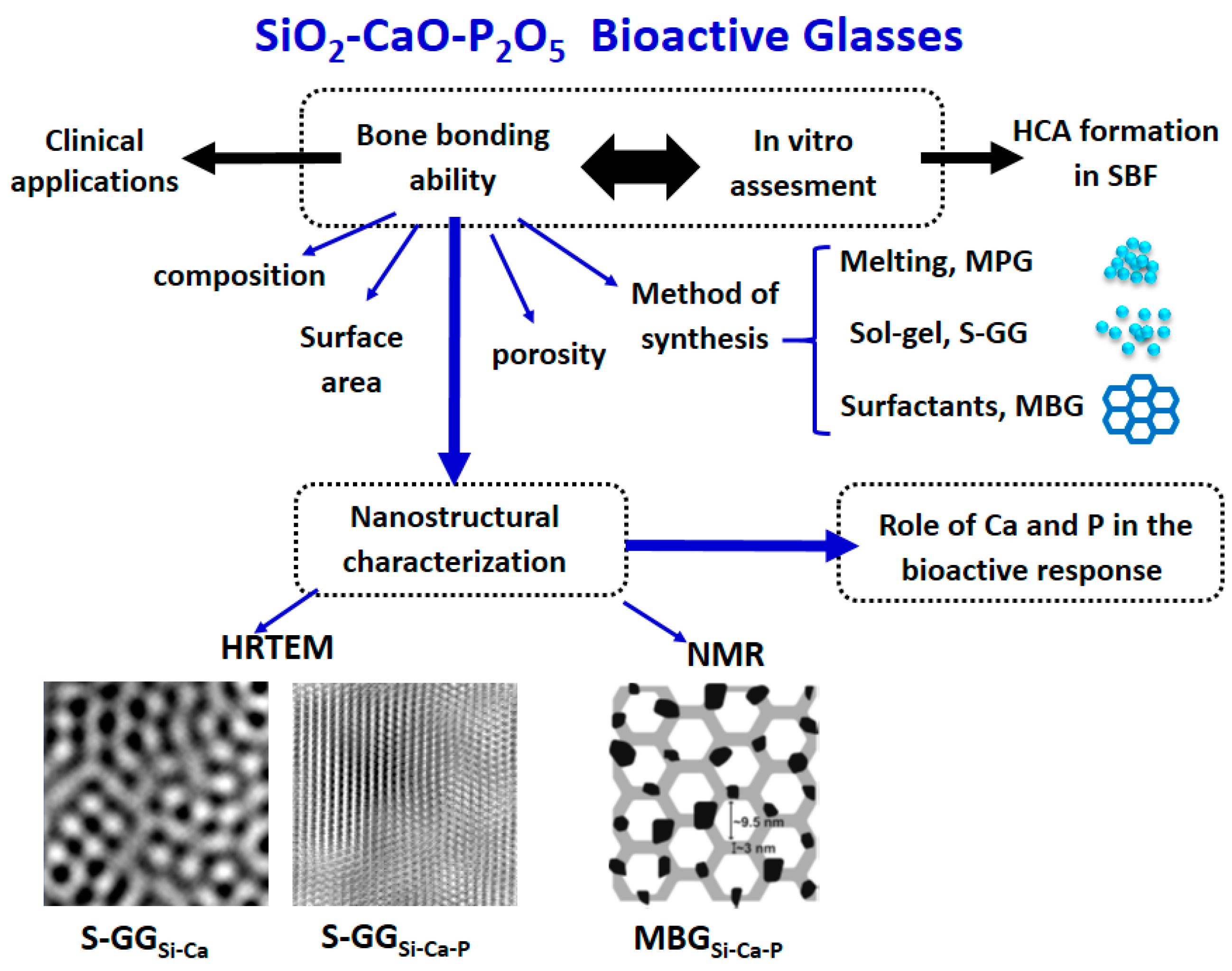
3. Nanostructure of Bioactive Glasses and Relationship with Their Bioactivity
3.1. Nanostructure of Bioactive Glasses from High-Resolution Transmission Electron Microscopy Analysis
3.2. Mechanism of Bioactivity of Bioactive Glasses Explained from Their Nanostructure
3.3. Recent Advances in the Characterization of the Nanostructure of Bioactive Glasses
3.4. Nanostructural Characterization of Bioactive Glasses by Nuclear Magnetic Resonance
4. Characterization of HCA Formed In Vitro on Bioactive Glasses Mainly Using NMR
5. Future Perspectives and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hench, L.L. Bioglass: 10 milestones from concept to commerce. J. Non-Cryst. Solids 2016, 432, 2–8. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Salinas, A.J. Mesoporous Bioactive Glasses in Tissue Engineering and Drug Delivery. In Bioactive Glasses: Fundamentals, Technology and Applications; Boccaccini, A.R., Brauer, D.S., Hupa, L., Eds.; The Royal Society of Chemistry: London, UK, 2017; pp. 393–419. [Google Scholar]
- Salinas, A.J.; Vallet-Regí, N. Glasses in bone regeneration: A multiscale issue. J. Non-Cryst. Solids 2016, 432, 9–14. [Google Scholar] [CrossRef]
- Jones, J.R. Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, N.C.; Koski, I.; Heikkilä, J.T.; Mattila, K.; Aho, A.J. A prospective randomized 14-year follow-up study of bioactive glass and autogenous bone as bone graft substitutes in benign bone tumors. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 4, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; Ragel, C.V.; Salinas, A.J. Glasses with medical applications. Eur. J. Inorg. Chem. 2003, 1029–1042. [Google Scholar] [CrossRef]
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 2, 117–141. [Google Scholar] [CrossRef]
- Li, R.; Clark, A.E.; Hench, L.L. An investigation of bioactive glass powders by sol-gel processing. J. Appl. Biomater. 1991, 2, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Yu, C.; Zhou, X.; Tang, J.; Zhao, D. Highly ordered mesoporous bioactive glasses with superior in vitro bone forming bioactivities. Angew. Chem. Int. Ed. 2004, 43, 5980–5984. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Barba, I.; Salinas, A.J.; Vallet-Regí, M. Effect of the continuous solution exchange on the in vitro reactivity of a CaO-SiO2 sol-gel glass. J. Biomed. Mater. Res. A 2000, 51, 191–199. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Maçon, A.L.B.; Kim, T.B.; Valliant, E.M.; Goetschius, K.; Brow, R.K.; Day, D.E.; Hoppe, A.; Boccaccini, A.R.; Kim, I.Y.; Ohtsuki, C.; et al. A unified in vitro evaluation for apatite-forming ability of bioactive glasses and their variants. J. Mater. Sci. Mater. Med. 2015, 26, 115. [Google Scholar] [CrossRef] [PubMed]
- Zadpoor, A.A. Relationship between in vitro apatite-forming ability measured using simulated body fluid and in vivo bioactivity of biomaterials. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 35, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; Salinas, A.J.; Ramírez-Castellanos, J.; González-Calbet, J.M. Nanostructure of bioactive sol–gel glasses and organic–inorganic hybrids. Chem. Mater. 2005, 17, 1874–1879. [Google Scholar] [CrossRef]
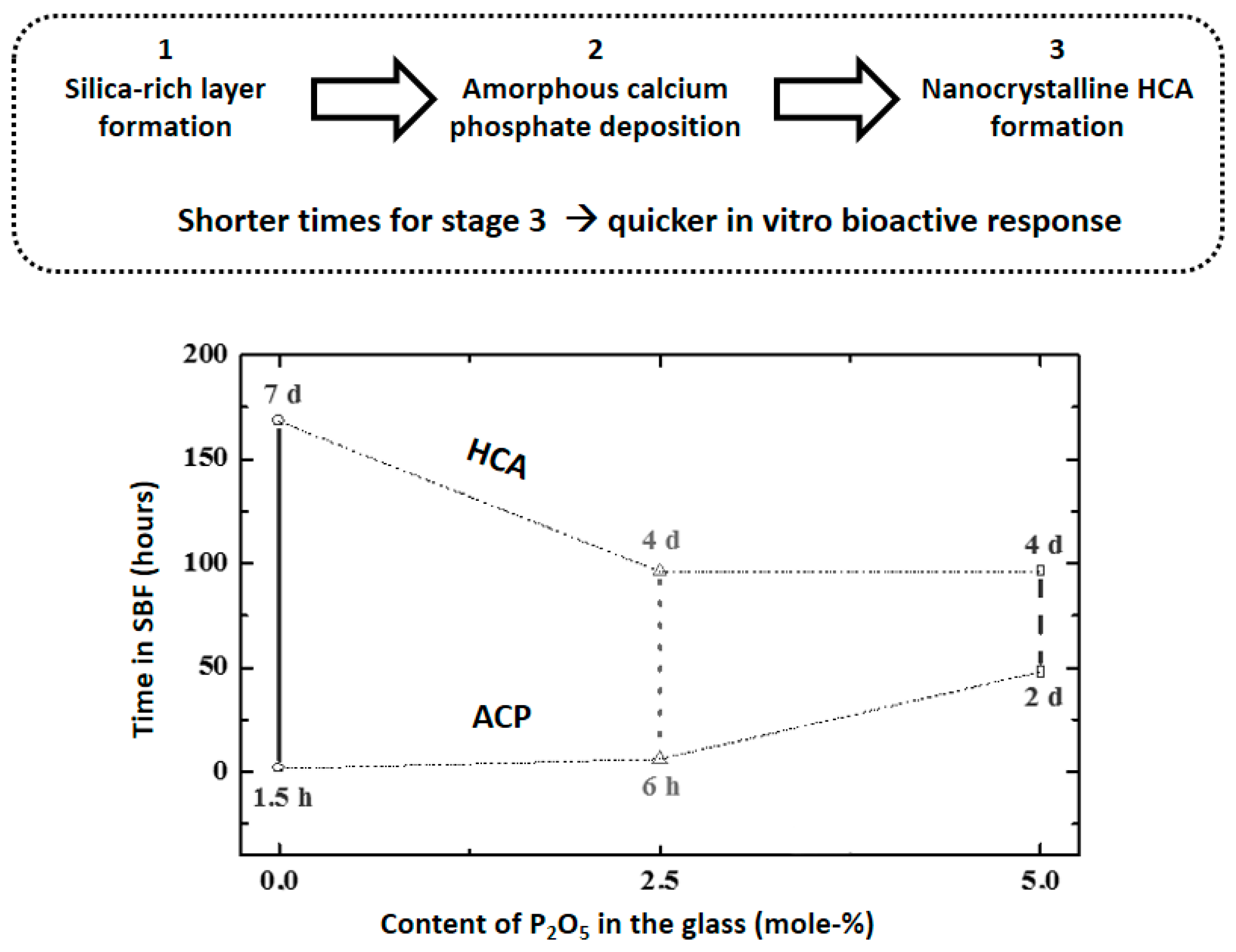
- Vallet-Regí, M.; Izquierdo-Barba, I.; Salinas, A.J. Influence of P2O5 on crystallinity of apatite formed in vitro on surface of bioactive glass. J. Biomed. Mater. Res. 1999, 46, 560–565. [Google Scholar] [CrossRef]
- Salinas, A.J.; Vallet-Regí, M.; Izquierdo-Barba, I. Biomimetic apatite deposition on calcium silicate gel glasses. J. Sol-Gel Sci. Technol. 2001, 21, 13–25. [Google Scholar] [CrossRef]
- Salinas, A.J.; Martin, A.I.; Vallet-Regí, M. Bioactivity of three CaO-P2O5-SiO2 sol-gel glasses. J. Biomed. Mater. Res. 2002, 61, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Dickens, B.; Schroeder, L.W.; Brown, W.E. Crystallographic studies of the role of Mg as a stabilizing impurity in beta-Ca3(PO4)2. I. The crystal structure of pure beta-Ca3(PO4)2. J. Solid State Chem. 1974, 10, 232–248. [Google Scholar] [CrossRef]
- Tilocca, A.; Cormack, A.N. Structural effects of phosphorus inclusion in bioactive silicate glasses. J. Phys. Chem. B 2007, 111, 14256–14264. [Google Scholar] [CrossRef] [PubMed]
- Tilocca, A. Structural models of bioactive glasses from molecular dynamics simulations, Proc. R. Soc. A 2009, 465, 1003–1027. [Google Scholar] [CrossRef]
- Aguiar, H.; Solla, E.L.; Serra, J.; Gonzalez, P.; Leon, B.; Almeida, N.; Cachinho, S.; Davim, E.J.C.; Correia, R.; Oliveira, J.M.; et al. Orthophosphate nanostructures in SiO2-P2O5-CaO-Na2O-MgO bioactive glasses. J. Non-Cryst. Solids 2008, 354, 4075–4080. [Google Scholar] [CrossRef]
- Aguiar, H.; Serra, J.; Gonzalez, P. Nanostructural Transitions in Bioactive Sol-Gel Silicate Glasses. Int. J. Appl. Ceram. Technol. 2011, 8, 511–522. [Google Scholar] [CrossRef]
- Malavasi, G.; Menabue, L.; Menziani, M.C.; Pedone, A.; Salinas, A.J.; Vallet-Regí, M. New insights into the bioactivity of SiO2-CaO and SiO2-CaO-P2O5 sol-gel glasses by molecular dynamics simulations. J. Sol-Gel Sci. Technol. 2013, 67, 208–219. [Google Scholar] [CrossRef]
- Cote, A.S.; Cormack, A.N.; Tilocca, A. Reactive molecular dynamics: An effective tool for modelling the sol–gel synthesis of bioglasses. J. Mater. Sci. 2017, 52, 9006–9013. [Google Scholar] [CrossRef]
- Salinas, A.J.; Vallet-Regí, M.; Toledo-Fernandez, J.A.; Mendoza-Serna, R.; Pinero, M.; Esquivias, L.; Ramirez-Castellanos, J.; Gonzalez-Calbet, J.M. Nanostructure and Bioactivity of Hybrid Aerogels. Chem. Mater. 2009, 21, 41–47. [Google Scholar] [CrossRef]
- Leonova, E.; Izquierdo-Barba, I.; Arcos, D.; Lopez-Noriega, A.; Hedin, N.; Vallet-Regì, M.; Edén, M. Multinuclear solid state NMR studies of ordered mesoporous bioactive glasses J. Phys. Chem. C 2008, 112, 5552–5562. [Google Scholar] [CrossRef]
- Gunawidjaja, P.N.; Mathew, R.; Lo, A.Y.H.; Izquierdo-Barba, I.; Garcia, A.; Arcos, D.; Vallet-Regì, M.; Edén, M. Local structures of mesoporous bioactive glasses and their surface alterations in vitro: Inferences from solid-state nuclear magnetic resonance. Philos. Trans. R. Soc. A 2012, 370, 1376–1399. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.; Turdean-Ionescu, C.; Stevensson, B.; Izquierdo-Barba, I.; Garcia, A.; Arcos, D.; Vallet-Regí, M.; Edén, M. Direct Probing of the Phosphate-Ion Distribution in Bioactive Silicate Glasses by Solid-State NMR: Evidence for Transitions between Random/Clustered Scenarios Chem. Mater. 2013, 25, 1877–1885. [Google Scholar] [CrossRef]
- Fayon, F.; Dueem, C.; Poumeyrol, T.; Allix, M.; Massiot, D. Evidence of Nanometric-Sized Phosphate Clusters in Bioactive Glasses as Revealed by Solid-State P-31 NMR. J. Phys. Chem. C 2013, 117, 2283–2288. [Google Scholar] [CrossRef]
- Stevensson, B.; Mathew, R.; Edén, M. Assessing the Phosphate Distribution in Bioactive Phosphosilicate Glasses by P-31 Solid-State NMR and Molecular Dynamics Simulations J. Phys. Chem. B 2014, 118, 8863–8876. [Google Scholar] [CrossRef] [PubMed]
- Gunawidjaja, P.N.; Izquierdo-Barba, I.; Mathew, R.; Jansson, K.; García, A.; Grins, J.; Arcos, D.; Vallet-Regí, M.; Edén, M. Quantifying apatite formation and cation leaching from mesoporous bioactive glasses in vitro: A SEM, solid-state NMR and powder XRD study. J. Mater. Chem. 2012, 22, 7214–7223. [Google Scholar] [CrossRef]
- Turdean-Ionescu, C.; Stevensson, B.; Grins, J.; Izquierdo-Barba, I.; García, A.; Arcos, D.; Vallet-Regí, M.; Edén, M. Composition-dependent in vitro apatite formation at mesoporous bioactive glass-surfaces quantified by solid-state NMR and powder XRD. RSC Adv. 2015, 5, 86061–86071. [Google Scholar] [CrossRef]
- Turean-Ionescu, C.; Stevensson, B.; Izquierdo-Barba, I.; García, A.; Arcos, D.; Vallet-Regì, M.; Edén, M. Surface Reactions of Mesoporous Bioactive Glasses Monitored by Solid-State NMR: Concentration Effects in Simulated Body Fluid. J. Phys. Chem. C 2016, 120, 4961–4974. [Google Scholar] [CrossRef]
- Mathew, R.; Turdean-Ionescu, C.; Yu, Y.; Stevensson, B.; Izquierdo-Barba, I.; García, A.; Arcos, D.; Vallet-Regì, M.; Edén, M. Proton Environments in Biomimetic Calcium Phosphates Formed from Mesoporous Bioactive CaO–SiO2–P2O5 Glasses in Vitro: Insights from Solid-State NMR. J. Phys. Chem. C 2017, 121, 13223–13238. [Google Scholar] [CrossRef] [PubMed]






© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vallet-Regi, M.; Salinas, A.J. Role of the Short Distance Order in Glass Reactivity. Materials 2018, 11, 415. https://doi.org/10.3390/ma11030415
Vallet-Regi M, Salinas AJ. Role of the Short Distance Order in Glass Reactivity. Materials. 2018; 11(3):415. https://doi.org/10.3390/ma11030415
Chicago/Turabian StyleVallet-Regi, María, and Antonio J. Salinas. 2018. "Role of the Short Distance Order in Glass Reactivity" Materials 11, no. 3: 415. https://doi.org/10.3390/ma11030415






