Carbon Nanotubes: Solution for the Therapeutic Delivery of siRNA?
Abstract
:1. Introduction
1.1. Therapeutic Uses of Carbon Nanotubes
1.2. Toxicity
1.3. PK and Biodistribution
1.4. SWCNT Delivery of siRNA
2. Results and Discussion
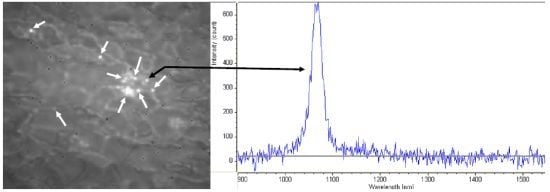
2.1. Processing of SWCNT Samples for Biological Evaluation




2.2. Transfection of siRNA/SWCNT Complexes


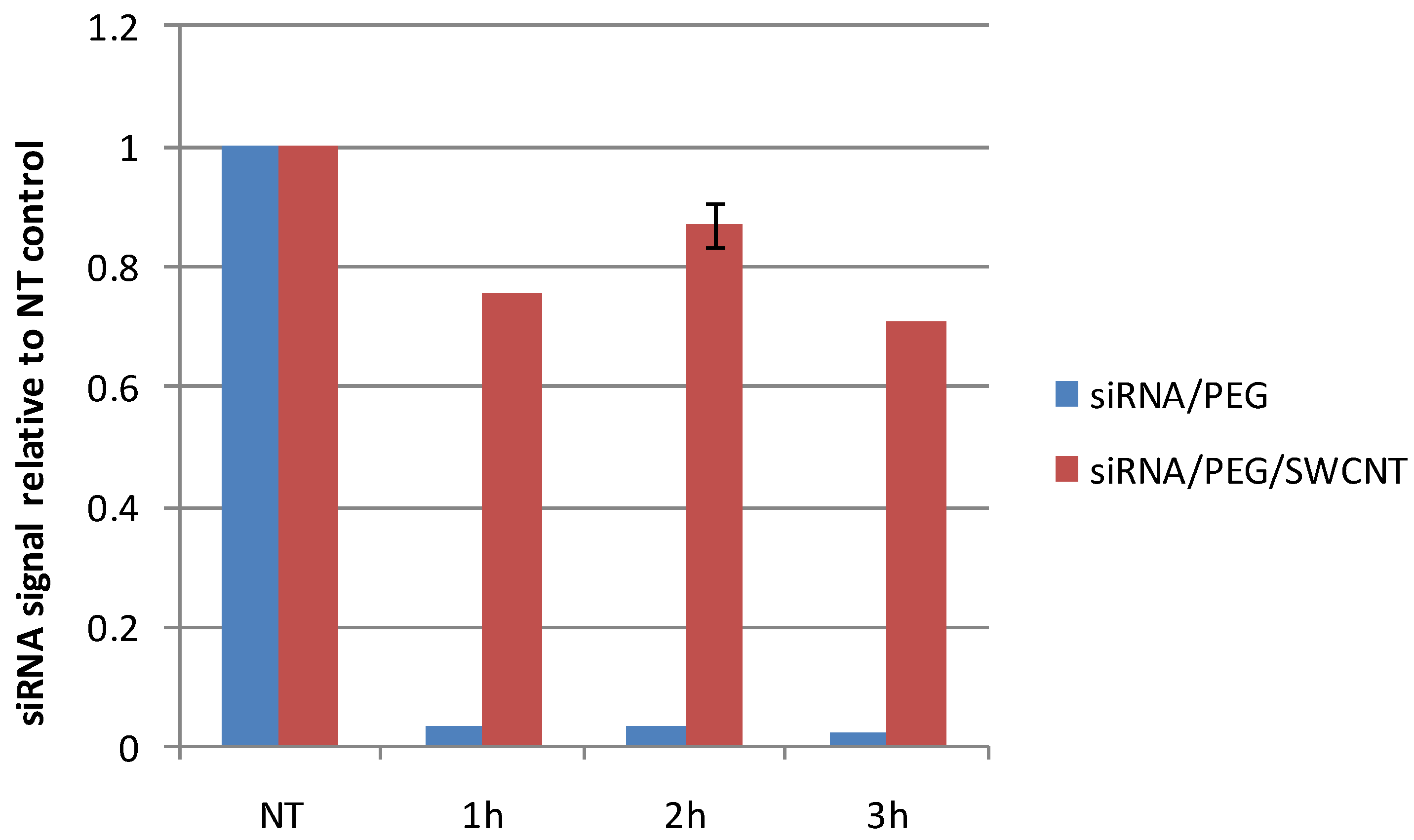
2.3. Stability of siRNA/SWCNT Complexes
2.4. Biological Activity

 normal mouse range). NT = non-treatment control; S = SWCNT control; E = siEGFR/SWCNT; K = siKRAS/SWCNT; E K = siEGFR/siKRAS/SWCNT. Subset of data is shown; (d) No weight loss was observed in animals treated weekly or biweekly (not shown).
normal mouse range). NT = non-treatment control; S = SWCNT control; E = siEGFR/SWCNT; K = siKRAS/SWCNT; E K = siEGFR/siKRAS/SWCNT. Subset of data is shown; (d) No weight loss was observed in animals treated weekly or biweekly (not shown).
 normal mouse range). NT = non-treatment control; S = SWCNT control; E = siEGFR/SWCNT; K = siKRAS/SWCNT; E K = siEGFR/siKRAS/SWCNT. Subset of data is shown; (d) No weight loss was observed in animals treated weekly or biweekly (not shown).
normal mouse range). NT = non-treatment control; S = SWCNT control; E = siEGFR/SWCNT; K = siKRAS/SWCNT; E K = siEGFR/siKRAS/SWCNT. Subset of data is shown; (d) No weight loss was observed in animals treated weekly or biweekly (not shown).

2.5. Manipulating the Pharmacokinetic Properties of SWCNT


3. Experimental Section
3.1. Preparation of SWCNT Solutions and SWCNT Complexes with siRNA
3.2. siRNA/PEG/SWCNT Complex Preparation
3.2.1. Atomic Force Microscopy (AFM)
3.2.2. Dynamic Light Scattering (DLS) and Zeta Potential
3.3. In Vitro Stability of siRNA/SWCNT Complexes
3.4. In Vitro Cellular Uptake Studies
3.5. In Vivo Analyses of SWCNT and siRNA/SWCNT Complexes
3.5.1. Toxicity Evaluation of SWCNT with no siRNA Payload
3.5.2. Pharmacokinetic Analyses of siRNA/PEG/SWCNT Solutions
3.5.3. Biodistribution and Elimination
3.6. Antitumor and Toxicity Evaluation of siEGFR/siKRAS/ SWCNT Payloads
3.6.1. Preparation of siEGFR/SWCNT or siKRAS/SWCNT and siEGFR/siKRAS/SWCNT
3.6.2. Animal Treatment Schedule, Blood Draws and Tissue Harvesting
3.6.3. Tissue Analyses
4. Conclusions
Acknowledgments
References
- Liu, Z.; Chen, K.; Davis, C.; Sherlock, S.; Cao, Q.; Chen, X.; Dai, H. Drug delivery with carbon nanotubes for in vivo cancer treatment. Cancer Res. 2008, 68, 6652–6660. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Tabakman, S.; Welsher, K.; Dai, H. Carbon nanotubes in biology and medicine: In vitro and in vivo detection, imaging and drug delivery. Nano Res. 2009, 2, 85–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Fan, A.C.; Rakhra, K.; Sherlock, S.; Goodwin, A.; Chen, X.; Yang, Q.; Felsher, D.W.; Dai, H. Supramolecular stacking of doxorubicin on carbon nanotubes for in vivo cancer therapy. Angew. Chem. Int. Ed. Engl. 2009, 48, 7668–7672. [Google Scholar] [CrossRef] [PubMed]
- Kostarelos, K.; Bianco, A.; Prato, M. Promises, facts and challenges for carbon nanotubes in imaging and therapeutics. Nat. Nanotechnol. 2009, 4, 627–633. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, A.; Villa, C.H.; Holland, J.P.; Sprinkle, S.R.; May, C.; Lewis, J.S.; Scheinberg, D.A.; McDevitt, M.R. Imaging and treating tumor vasculature with targeted radiolabeled carbon nanotubes. Int. J. Nanomedicine 2010, 5, 783–802. [Google Scholar] [PubMed]
- Han, Z.J.; Ostrikov, K.K.; Tan, C.M.; Tay, B.K.; Peel, S.A. Effect of hydrophilicity of carbon nanotube arrays on the release rate and activity of recombinant human bone morphogenetic protein-2. Nanotechnology 2011, 22. [Google Scholar] [CrossRef]
- Beg, S.; Rizwan, M.; Sheikh, A.M.; Hasnain, M.S.; Anwer, K.; Kohli, K. Advancement in carbon nanotubes: Basics, biomedical applications and toxicity. J. Pharm. Pharmacol. 2011, 63, 141–163. [Google Scholar] [CrossRef] [PubMed]
- Pantarotto, D.; Singh, R.; McCarthy, D.; Erhardt, M.; Briand, J.P.; Prato, M.; Kostarelos, K.; Bianco, A. Functionalized carbon nanotubes for plasmid DNA gene delivery. Angew. Chem. Int. Ed. Engl. 2004, 43, 5242–5246. [Google Scholar] [CrossRef] [PubMed]
- Bartholomeusz, G.; Cherukuri, P.; Kingston, J.; Cognet, L.; Lemos, R.; Leeuw, T.K.; Gumbiner-Russo, L.; Weisman, R.B.; Powis, G. In vivo therapeutic silencing of hypoxia-inducible factor 1 α (HIF-1α) Using single-walled carbon nanotubes noncovalently coated with siRNA. Nano Res. 2009, 2, 279–291. [Google Scholar] [CrossRef]
- Pastorin, G. Crucial functionalizations of carbon nanotubes for improved drug delivery: A valuable option? Pharm. Res. 2009, 26, 746–769. [Google Scholar] [CrossRef] [PubMed]
- Ke, P.C. Carbon nanomaterials in biological systems. J. Phys. Condens. Matter 2007, 19. [Google Scholar] [CrossRef]
- Yang, R.; Yang, X.; Zhang, Z.; Zhang, Y.; Wang, S.; Cai, Z.; Jia, Y.; Ma, Y.; Zheng, C.; Lu, Y.; Roden, R.; Chen, Y. Single-walled carbon nanotubes-mediated in vivo and in vitro delivery of siRNA into antigen-presenting cells. Gene Ther. 2006, 13, 1714–1723. [Google Scholar] [CrossRef] [PubMed]
- Kam, N.W.; Liu, Z.; Dai, H. Functionalization of carbon nanotubes via cleavable disulfide bonds for efficient intracellular delivery of siRNA and potent gene silencing. J. Am. Chem. Soc. 2005, 127, 12492–12493. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Winters, M.; Holodniy, M.; Dai, H. siRNA delivery into human T cells and primary cells with carbon-nanotube transporters. Angew. Chem. Int. Ed. Engl. 2007, 46, 2023–2027. [Google Scholar] [CrossRef] [PubMed]
- Podesta, J.E.; Al-Jamal, K.T.; Herrero, M.A.; Tian, B.; Ali-Boucetta, H.; Hegde, V.; Bianco, A.; Prato, M.; Kostarelos, K. Antitumor activity and prolonged survival by carbon-nanotube-mediated therapeutic siRNA silencing in a human lung xenograft model. Small 2009, 5, 1176–1185. [Google Scholar] [CrossRef] [PubMed]
- Al-Jamal, K.T.; Gherardini, L.; Bardi, G.; Nunes, A.; Guo, C.; Bussy, C.; Herrero, M.A.; Bianco, A.; Prato, M.; Kostarelos, K.; et al. Functional motor recovery from brain ischemic insult by carbon nanotube-mediated siRNA silencing. Proc. Natl. Acad. Sci. USA 2011, 108, 10952–109527. [Google Scholar] [CrossRef] [PubMed]
- Bhirde, A.A.; Patel, V.; Gavard, J.; Zhang, G.; Sousa, A.A.; Masedunskas, A.; Leapman, R.D.; Weigert, R.; Gutkind, J.S.; Rusling, J.F. Targeted killing of cancer cells in vivo and in vitro with EGF-directed carbon nanotube-based drug delivery. ACS Nano 2009, 3, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Lin, G.; Lu, Q.; Meng, L.; Shen, X.; Dong, L.; Fu, C.; Zhang, X. Targeted therapy of SMMC-7721 liver cancer in vitro and in vivo with carbon nanotubes based drug delivery system. J. Colloid. Interface Sci. 2012, 365, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Guven, A.; Rusakova, I.A.; Lewis, M.T.; Wilson, L.J. Cisplatin@US-tube carbon nanocapsules for enhanced chemotherapeutic delivery. Biomaterials 2012, 33, 1455–1461. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Phillips, J.A.; Liu, H.; Yang, R.; Tan, W. Carbon nanotubes protect DNA strands during cellular delivery. ACS Nano 2008, 2, 2023–2028. [Google Scholar] [CrossRef] [PubMed]
- Welsher, K.; Liu, Z.; Sherlock, S.P.; Robinson, J.T.; Chen, Z.; Daranciang, D.; Dai, H. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat. Nanotechnol. 2009, 4, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Welsher, K.; Sherlock, S.P.; Dai, H. Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc. Natl. Acad. Sci. USA 2011, 108, 8943–8948. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Tisch, U.; Haick, H. Detection of nonpolar molecules by means of carrier scattering in random networks of carbon nanotubes: Toward diagnosis of diseases via breath samples. Nano Lett. 2009, 9, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Heller, D.A.; Kalbacova, M.; Kim, J.H.; Zhang, J.; Boghossian, A.A.; Maheshri, N.; Strano, M.S. Detection of single-molecule H2O2 signalling from epidermal growth factor receptor using fluorescent single-walled carbon nanotubes. Nat. Nanotechnol. 2010, 5, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Heller, D.A.; Jin, H.; Martinez, B.M.; Patel, D.; Miller, B.M.; Yeung, T.K.; Jena, P.V.; Hobartner, C.; Ha, T.; Silverman, S.K.; et al. Multimodal optical sensing and analyte specificity using single-walled carbon nanotubes. Nat. Nanotechnol. 2009, 4, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Barone, P.W.; Strano, M.S. Reversible control of carbon nanotube aggregation for a glucose affinity sensor. Angew. Chem. Int. Ed. Engl. 2006, 45, 8138–8141. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lu, F.; Tu, Y.; Ren, Z. Glucose biosensors based on carbon nanotube nanoelectrode ensembles. Nano Lett. 2004, 4, 191–195. [Google Scholar] [CrossRef]
- Ghosh, S.; Dutta, S.; Gomes, E.; Carroll, D.; D’Agostino, R., Jr.; Olson, J.; Guthold, M.; Gmeiner, W.H. Increased heating efficiency and selective thermal ablation of malignant tissue with DNA-encased multiwalled carbon nanotubes. ACS Nano 2009, 3, 2667–2673. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Xing, D.; Ou, Z.; Wu, B.; Resasco, D.E.; Chen, W.R. Cancer photothermal therapy in the near-infrared region by using single-walled carbon nanotubes. J. Biomed. Opt. 2009, 14. [Google Scholar] [CrossRef]
- Robinson, J.T.; Welsher, K.; Tabakman, S.M.; Sherlock, S.P.; Wang, H.; Luong, R.; Dai, H. High performance In vivo near-IR (>1 mum) imaging and photothermal cancer therapy with carbon nanotubes. Nano Res. 2010, 3, 779–793. [Google Scholar] [CrossRef] [PubMed]
- Tasis, D.; Tagmatarchis, N.; Georgakilas, V.; Prato, M. Soluble carbon nanotubes. Chemistry 2003, 9, 4000–4008. [Google Scholar] [CrossRef] [PubMed]
- Maynard, A.D.; Baron, P.A.; Foley, M.; Shvedova, A.A.; Kisin, E.R.; Castranova, V. Exposure to carbon nanotube material: Aerosol release during the handling of unrefined single-walled carbon nanotube material. J. Toxicol. Environ. Health. A 2004, 67, 87–107. [Google Scholar] [CrossRef] [PubMed]
- Huczko, A.; Lange, H.; Bystrzejewski, M.; Baranowski, P.; Grubek-Jaworska, H.; Nejman, P.; Przybylowski, T.; Czuminska, K.; Glapinski, J.; Walton, D.R.M.; et al. Pulmonary toxicity of 1-D nanocarbon materials. Fuller. Nanotub. Carbon Nanostructures 2005, 13, 141–145. [Google Scholar] [CrossRef]
- Lam, C.W.; James, J.T.; McCluskey, R.; Hunter, R.L. Pulmonary toxicity of single-wall carbon nanotubes in mice 7 and 90 days after intratracheal instillation. Toxicol. Sci. 2004, 77, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Warheit, D.B.; Laurence, B.R.; Reed, K.L.; Roach, D.H.; Reynolds, G.A.; Webb, T.R. Comparative pulmonary toxicity assessment of single-wall carbon nanotubes in rats. Toxicol. Sci. 2004, 77, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Shvedova, A.A.; Kisin, E.R.; Mercer, R.; Murray, A.R.; Johnson, V.J.; Potapovich, A.I.; Tyurina, Y.Y.; Gorelik, O.; Arepalli, S.; Schwegler-Berry, D.; et al. Unusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in mice. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, L698–L708. [Google Scholar] [CrossRef] [PubMed]
- Huczko, A.; Lange, H. Carbon nanotubes: Experimental evidence for a null risk of skin irritation and allergy. Fuller. Sci. Technol. 2001, 9, 247–250. [Google Scholar] [CrossRef]
- Yokoyama, A.; Sato, Y.; Nodasaka, Y.; Yamamoto, S.; Kawasaki, T.; Shindoh, M.; Kohgo, T.; Akasaka, T.; Uo, M.; Watari, F.; et al. Biological behavior of hat-stacked carbon nanofibers in the subcutaneous tissue in rats. Nano Lett. 2005, 5, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Yokoyama, A.; Shibata, K.; Akimoto, Y.; Ogino, S.; Nodasaka, Y.; Kohgo, T.; Tamura, K.; Akasaka, T.; Uo, M.; et al. Influence of length on cytotoxicity of multi-walled carbon nanotubes against human acute monocytic leukemia cell line THP-1 in vitro and subcutaneous tissue of rats in vivo. Mol. Biosyst. 2005, 1, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Kostarelos, K. The long and short of carbon nanotube toxicity. Nat. Biotechnol. 2008, 26, 774–776. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, A.; Villa, C.H.; Bander, E.; Rey, D.A.; Bergkvist, M.; Batt, C.A.; Manova-Todorova, K.; Deen, W.M.; Scheinberg, D.A.; McDevitt, M.R. Paradoxical glomerular filtration of carbon nanotubes. Proc. Natl. Acad. Sci. USA 2010, 107, 12369–12374. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Davis, C.; Cai, W.; He, L.; Chen, X.; Dai, H. Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2008, 105, 1410–1415. [Google Scholar] [CrossRef] [PubMed]
- Salvador-Morales, C.; Flahaut, E.; Sim, E.; Sloan, J.; Green, M.L.; Sim, R.B. Complement activation and protein adsorption by carbon nanotubes. Mol. Immunol. 2006, 43, 193–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamad, I.; Christy Hunter, A.; Rutt, K.J.; Liu, Z.; Dai, H.; Moein Moghimi, S. Complement activation by PEGylated single-walled carbon nanotubes is independent of C1q and alternative pathway turnover. Mol. Immunol. 2008, 45, 3797–3803. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.W.; James, J.T.; McCluskey, R.; Arepalli, S.; Hunter, R.L. A review of carbon nanotube toxicity and assessment of potential occupational and environmental health risks. Crit. Rev. Toxicol. 2006, 36, 189–217. [Google Scholar] [CrossRef] [PubMed]
- Sayes, C.M.; Liang, F.; Hudson, J.L.; Mendez, J.; Guo, W.; Beach, J.M.; Moore, V.C.; Doyle, C.D.; West, J.L.; Billups, W.E.; et al. Functionalization density dependence of single-walled carbon nanotubes cytotoxicity in vitro. Toxicol. Lett. 2006, 161, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, L.; Ali-Boucetta, H.; Herrero, M.A.; Pastorin, G.; Bianco, A.; Prato, M.; Kostarelos, K. Tissue histology and physiology following intravenous administration of different types of functionalized multiwalled carbon nanotubes. Nanomedicine 2008, 3, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Pantarotto, D.; Lacerda, L.; Pastorin, G.; Klumpp, C.; Prato, M.; Bianco, A.; Kostarelos, K. Tissue biodistribution and blood clearance rates of intravenously administered carbon nanotube radiotracers. Proc. Natl. Acad. Sci. USA 2006, 103, 3357–3362. [Google Scholar] [CrossRef] [PubMed]
- Schipper, M.L.; Nakayama-Ratchford, N.; Davis, C.R.; Kam, N.W.; Chu, P.; Liu, Z.; Sun, X.; Dai, H.; Gambhir, S.S. A pilot toxicology study of single-walled carbon nanotubes in a small sample of mice. Nat. Nanotechnol. 2008, 3, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Cherukuri, P.; Gannon, C.J.; Leeuw, T.K.; Schmidt, H.K.; Smalley, R.E.; Curley, S.A.; Weisman, R.B. Mammalian pharmacokinetics of carbon nanotubes using intrinsic near-infrared fluorescence. Proc. Natl. Acad. Sci. USA 2006, 103, 18882–18886. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-T.; Guo, W.; Lin, Y.; Deng, X.-Y.; Wang, H.-F.; Sun, H.-F.; Liu, Y.-F.; Wang, X.; Wang, W.; Chen, M.; et al. Biodistribution of pristine single-walled carbon nanotubes in vivo. J. Phys. Chem. C 2007, 111, 17761–17764. [Google Scholar] [CrossRef]
- Cai, D.; Kempa, K.; Ren, Z.; Carnahan, D.; Chiles, T.C. Nanospearing—Biomolecule delivery and its biocompatibility. Nanomater. Appl. Med. Biol. 2008, 81–92. [Google Scholar]
- Zhang, X.; Meng, L.; Wang, X.; Lu, Q. Preparation and cellular uptake of pH-dependent fluorescent single-wall carbon nanotubes. Chemistry 2010, 16, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Gaur, U.; Sahoo, S.K.; De, T.K.; Ghosh, P.C.; Maitra, A.; Ghosh, P.K. Biodistribution of fluoresceinated dextran using novel nanoparticles evading reticuloendothelial system. Int. J. Pharm. 2000, 202, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Harrington, K.J.; Mohammadtaghi, S.; Uster, P.S.; Glass, D.; Peters, A.M.; Vile, R.G.; Stewart, J.S. Effective targeting of solid tumors in patients with locally advanced cancers by radiolabeled pegylated liposomes. Clin. Cancer. Res. 2001, 7, 243–254. [Google Scholar] [PubMed]
- Liu, X.; Tao, H.; Yang, K.; Zhang, S.; Lee, S.T.; Liu, Z. Optimization of surface chemistry on single-walled carbon nanotubes for in vivo photothermal ablation of tumors. Biomaterials 2011, 32, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Tsyboulski, D.A.; Bachilo, S.M.; Weisman, R.B. Versatile visualization of individual single-walled carbon nanotubes with near-infrared fluorescence microscopy. Nano Lett. 2005, 5, 975–979. [Google Scholar] [CrossRef] [PubMed]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kirkpatrick, D.L.; Weiss, M.; Naumov, A.; Bartholomeusz, G.; Weisman, R.B.; Gliko, O. Carbon Nanotubes: Solution for the Therapeutic Delivery of siRNA? Materials 2012, 5, 278-301. https://doi.org/10.3390/ma5020278
Kirkpatrick DL, Weiss M, Naumov A, Bartholomeusz G, Weisman RB, Gliko O. Carbon Nanotubes: Solution for the Therapeutic Delivery of siRNA? Materials. 2012; 5(2):278-301. https://doi.org/10.3390/ma5020278
Chicago/Turabian StyleKirkpatrick, D. Lynn, Michelle Weiss, Anton Naumov, Geoffrey Bartholomeusz, R. Bruce Weisman, and Olga Gliko. 2012. "Carbon Nanotubes: Solution for the Therapeutic Delivery of siRNA?" Materials 5, no. 2: 278-301. https://doi.org/10.3390/ma5020278
APA StyleKirkpatrick, D. L., Weiss, M., Naumov, A., Bartholomeusz, G., Weisman, R. B., & Gliko, O. (2012). Carbon Nanotubes: Solution for the Therapeutic Delivery of siRNA? Materials, 5(2), 278-301. https://doi.org/10.3390/ma5020278