Dynamic Morphological Changes Induced By GM1 and Protein Interactions on the Surface of Cell-Sized Liposomes
Abstract
:1. Introduction
2. Results
2.1. Homogeneous Liposome
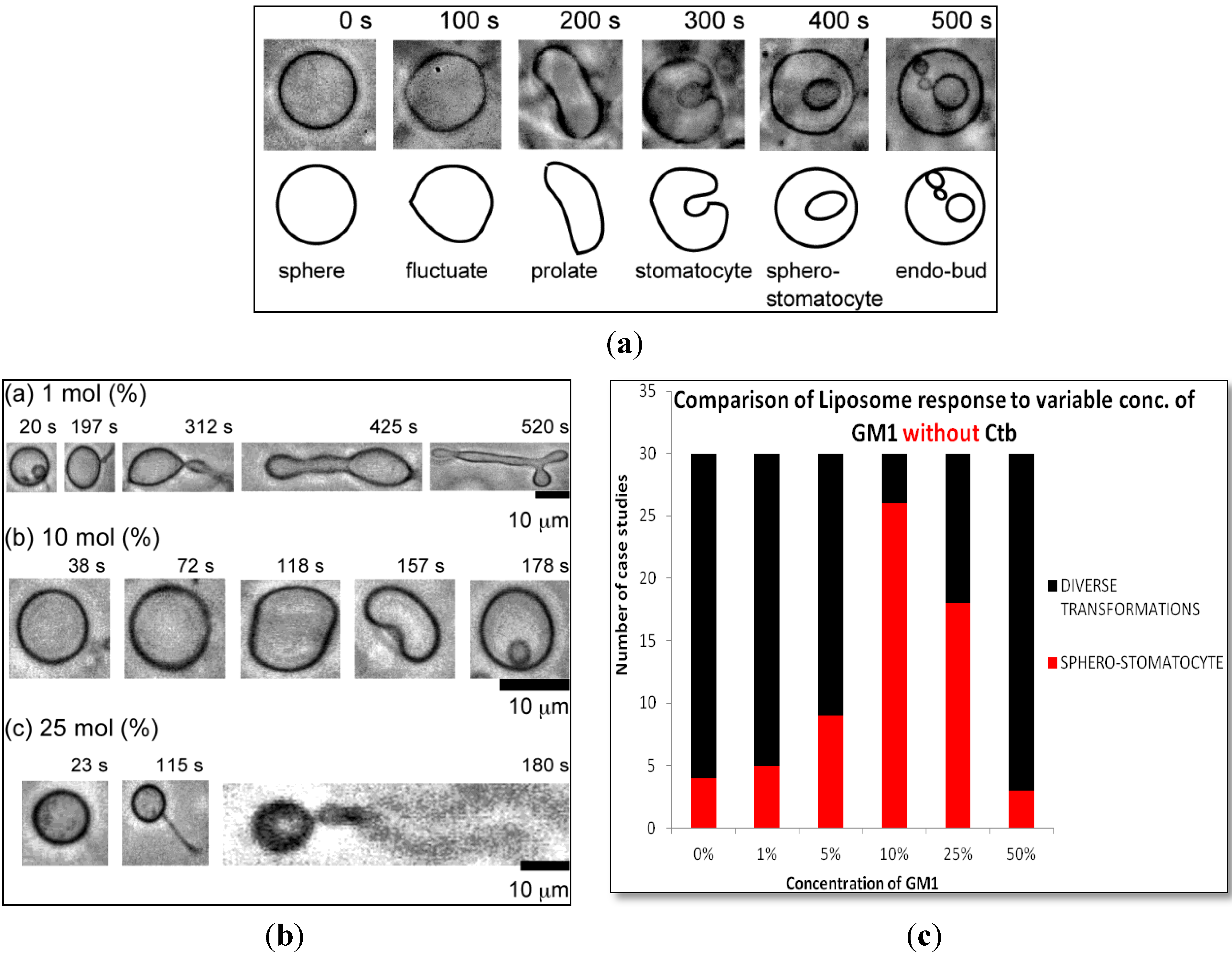
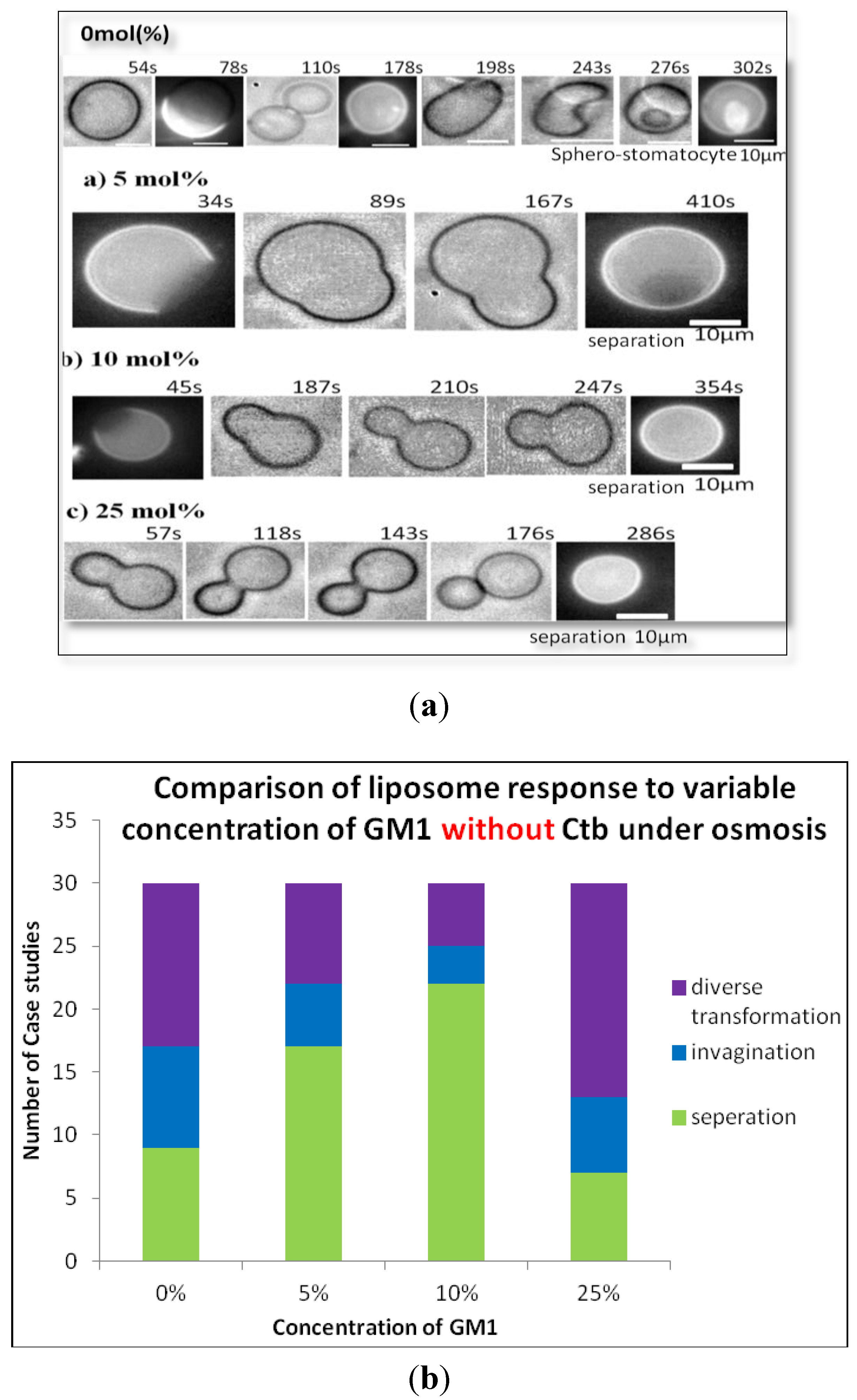
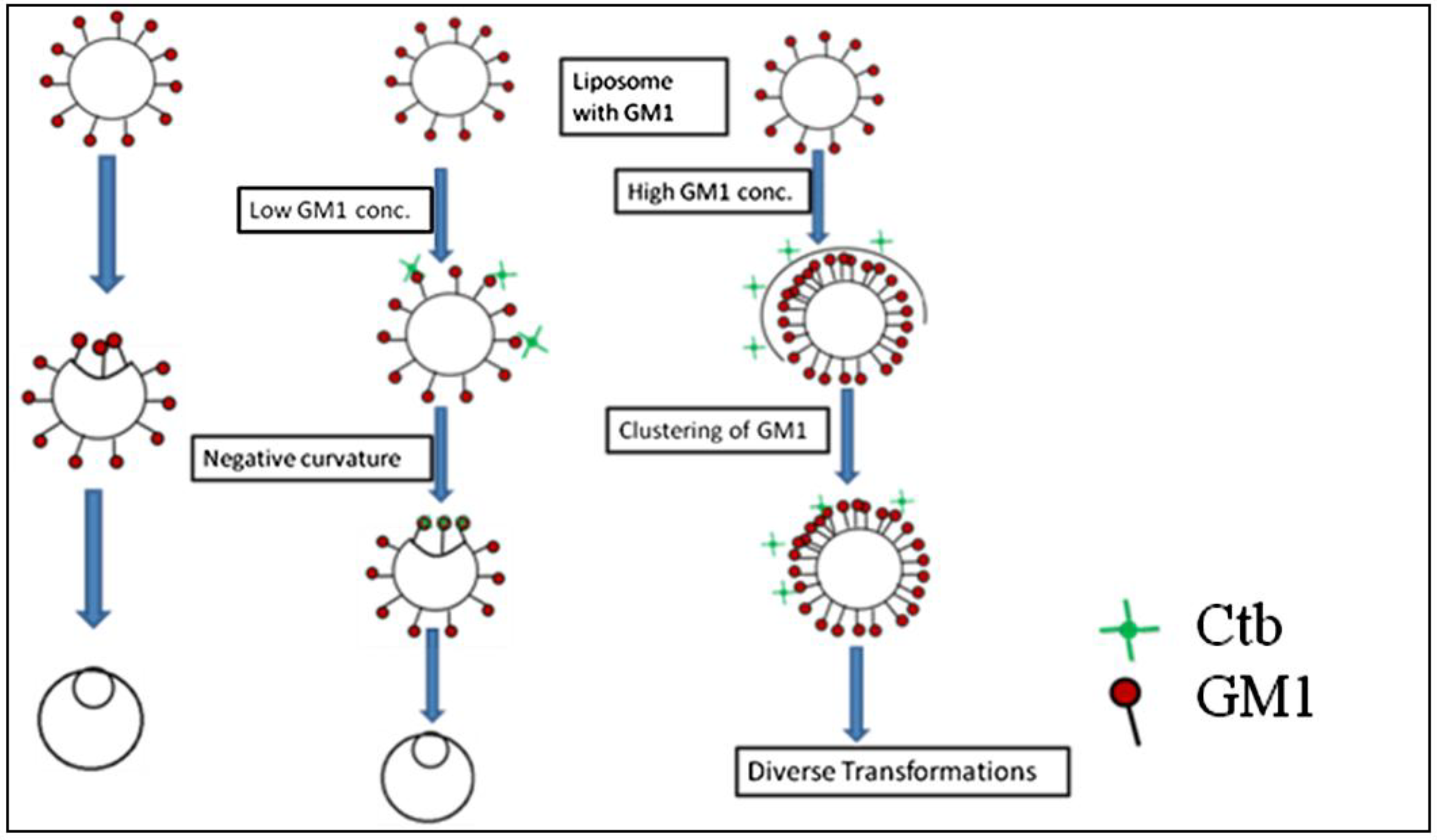
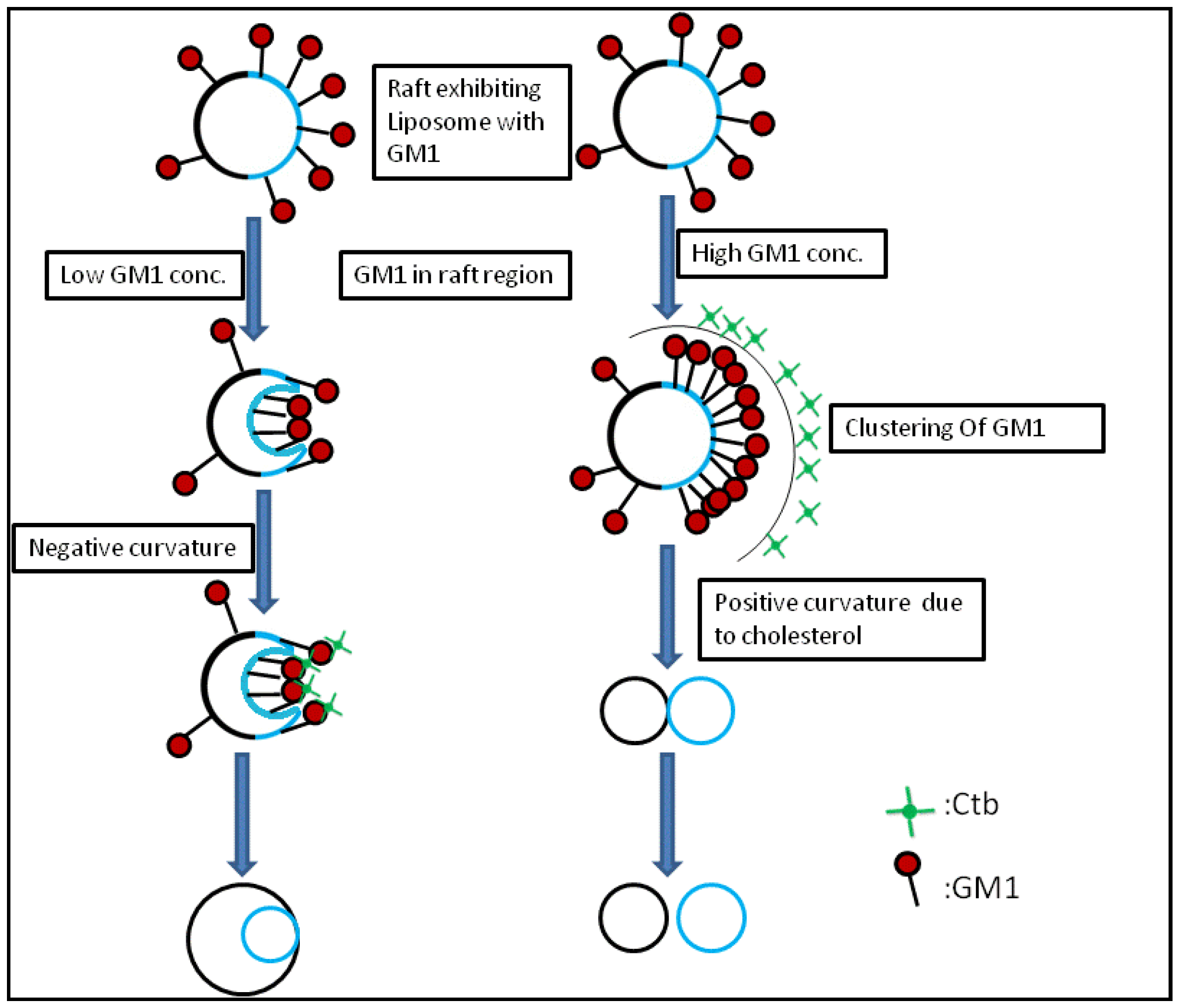
2.1.1. Homogeneous Liposome with Variable Concentration of GM1
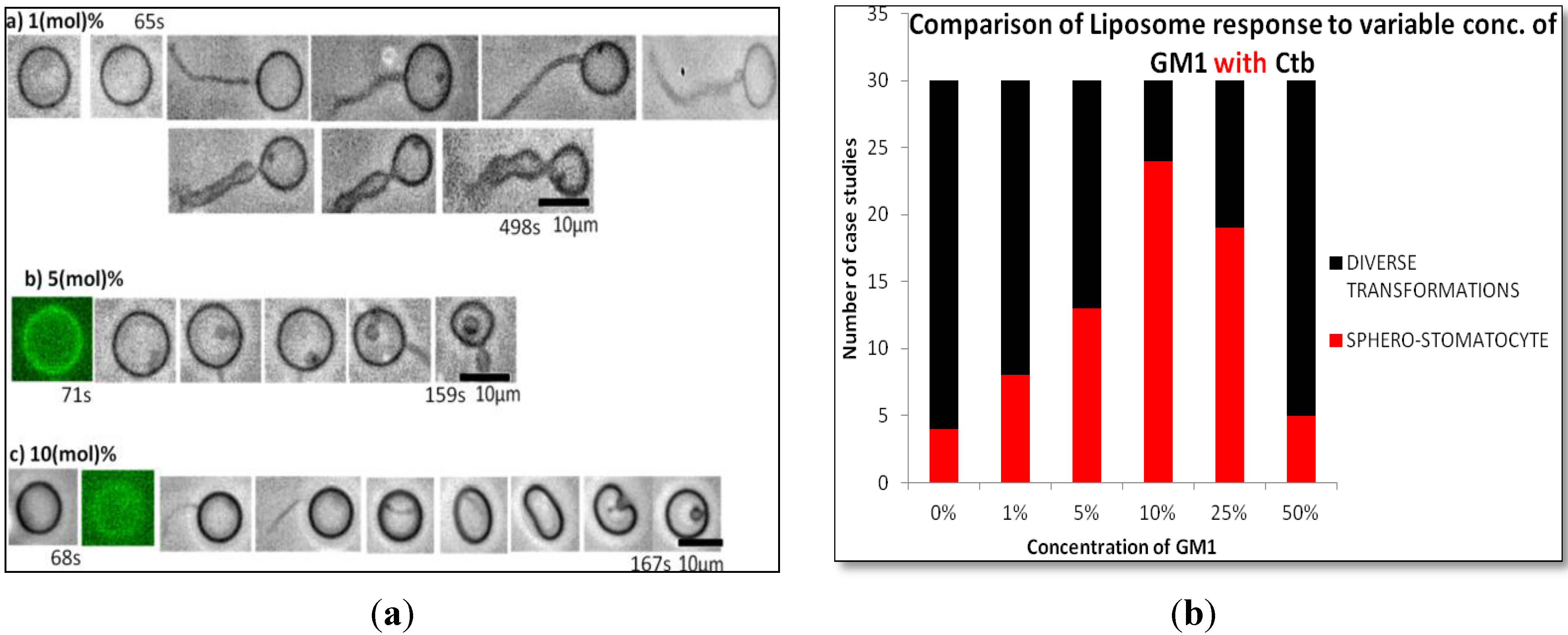
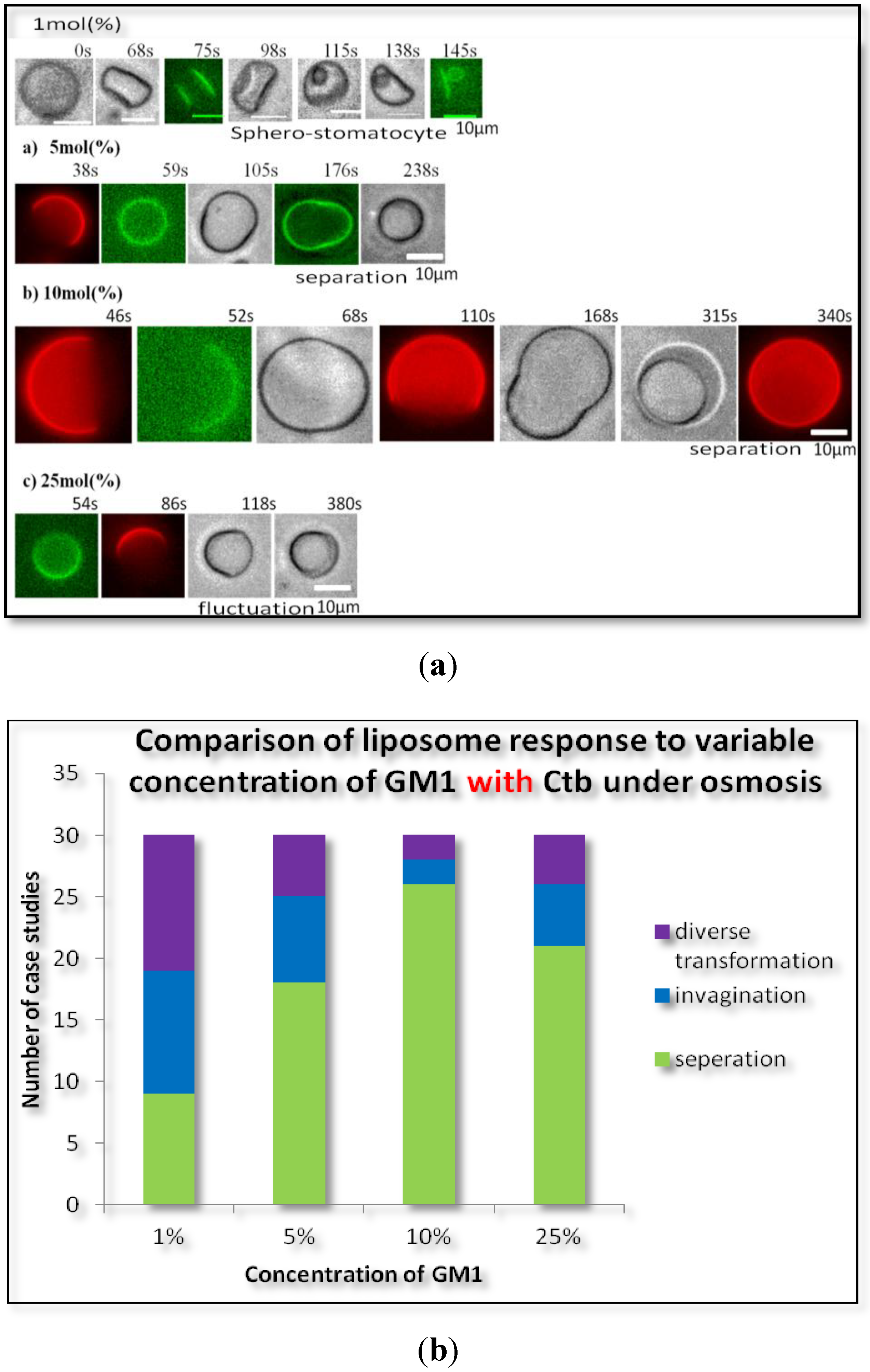
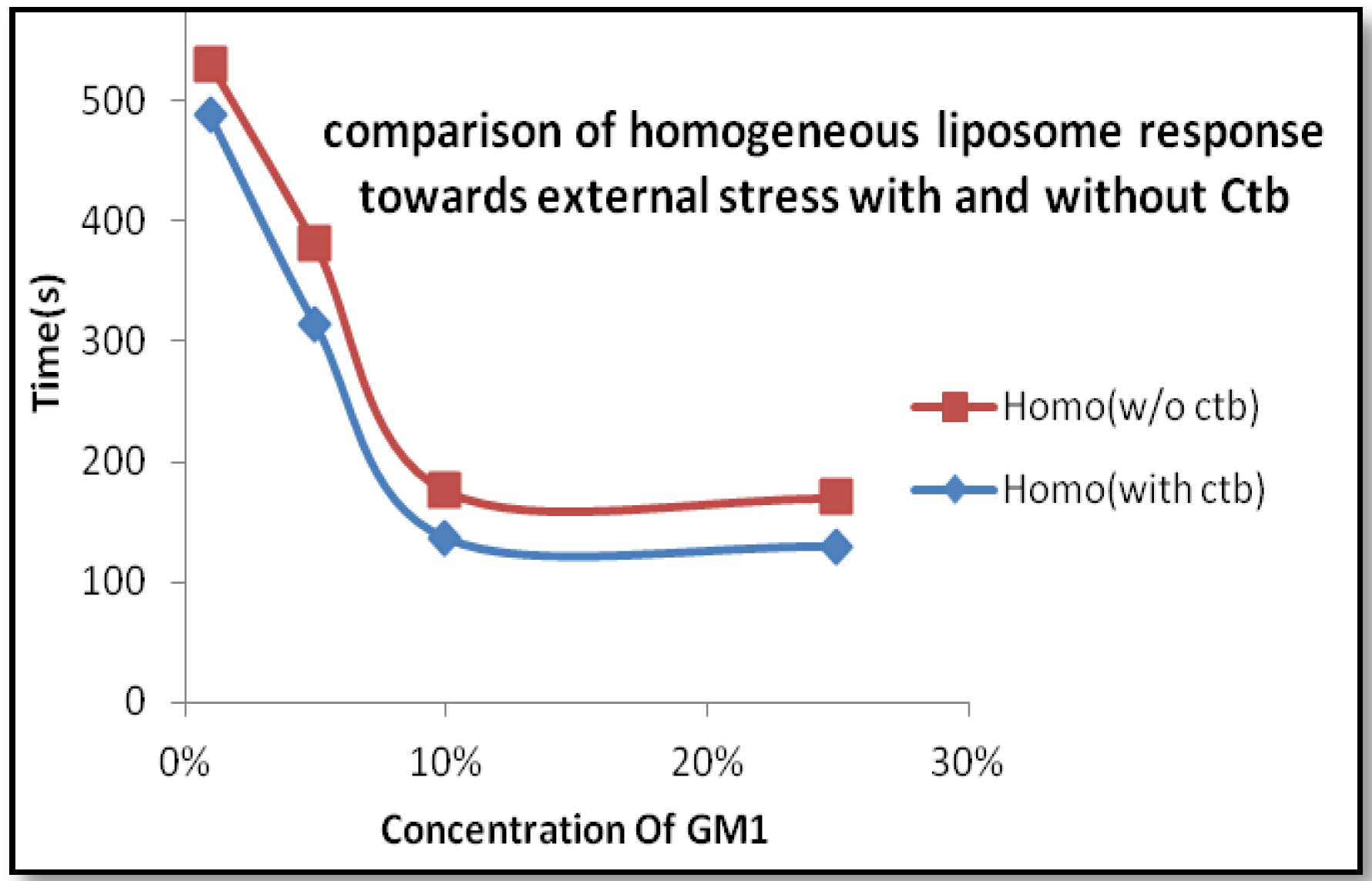
2.1.2. Homogeneous Liposome with Variable Concentration of GM1 with Ctb
2.2. Heterogeneous Liposome
- a.
- 5:5 (DOPC:DPPC) So/Ld phase;
- b.
- 4:4:2 (DOPC:DPPC:Cholesterol) Lo/Ld phase;
- c.
- 2:5:3 (DOPC:DPPC:Cholesterol) Lo/Ld phase.
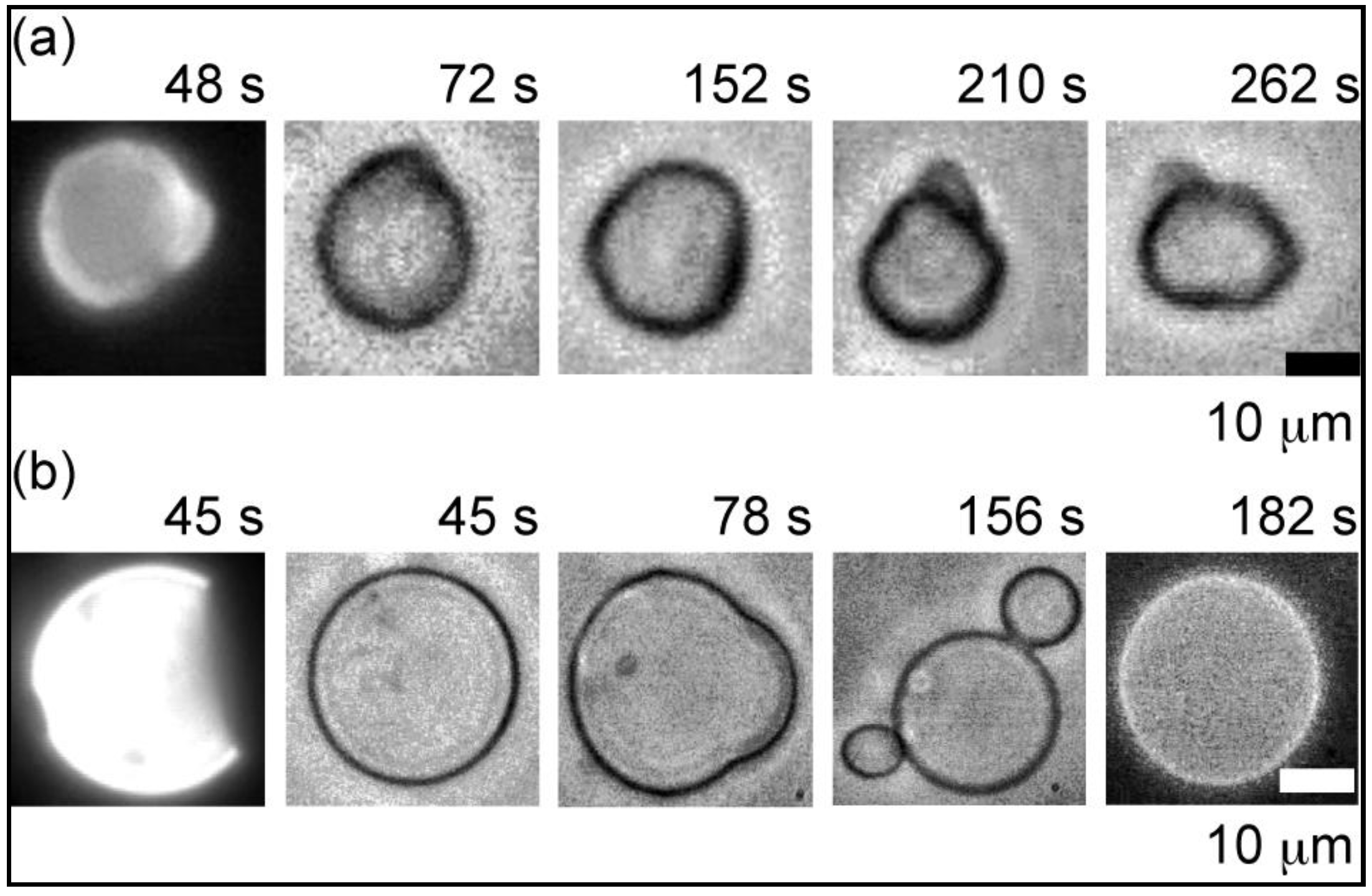
2.2.1. Heterogeneous Liposome with Variable Concentration of GM1
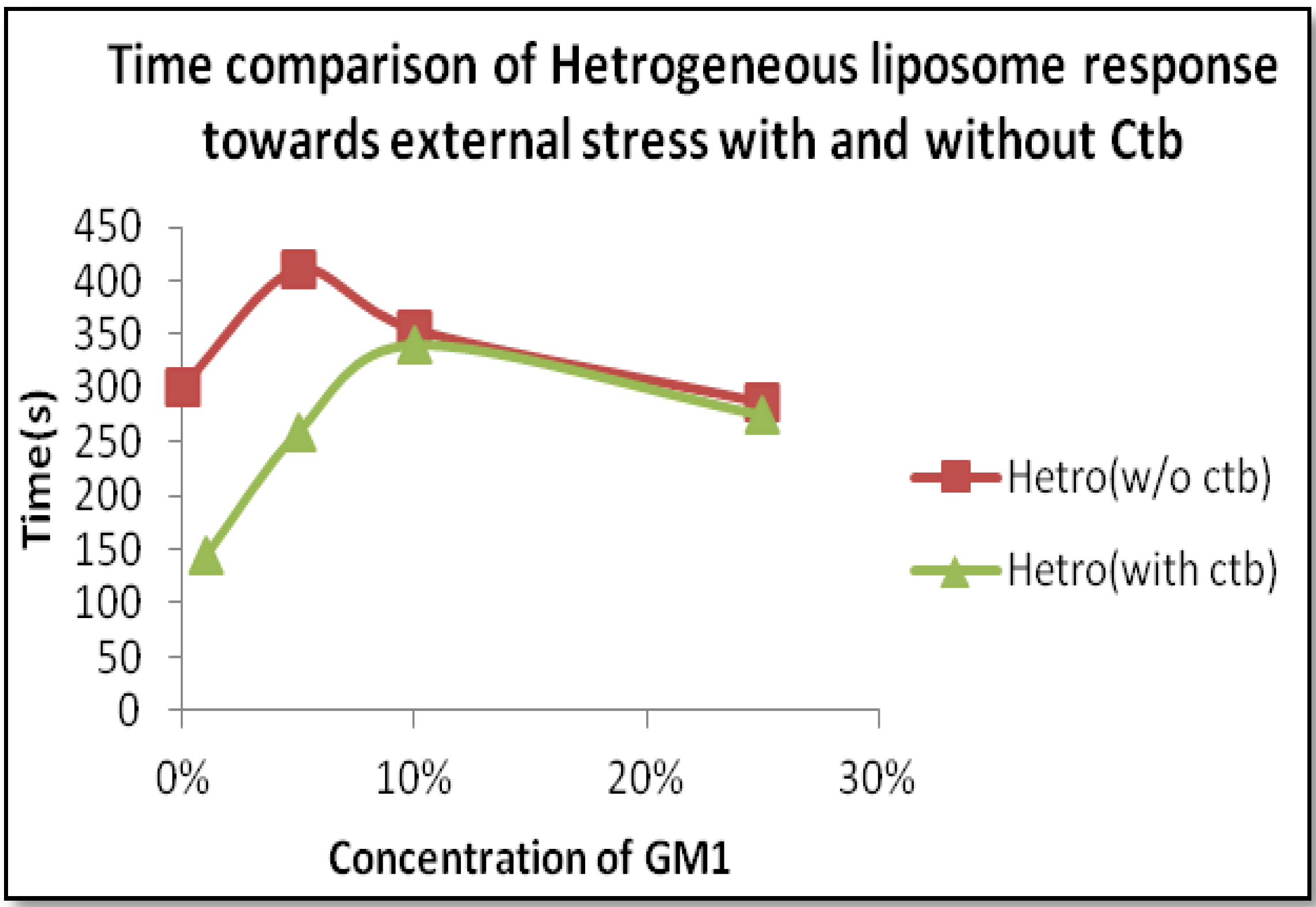
2.2.2. Heterogeneous Liposome with GM1-Ctb Interaction
3. Experimental Section
3.1. Materials
3.2. Preparation of Giant Unilamelar Vesicles (GUV)
3.3. Microscopic Observations
3.4. Induction of Osmotic Stress
4. Discussion
Acknowledgments
Conflict of Interest
References
- Spector, A.A.; Yorek, M.A. Membrane lipid composition and cellular function. J. Lipid Res. 1985, 26, 1015–1035. [Google Scholar]
- Bertoli, E.; Masserini, M.; Sonnino, S.; Ghidoni, R.; Cestaro, B.; Tettamanti, G. EPR studies on the fluidity and surface dynamics of egg phosphatidylcholine vesicles containing gangliosides. Biochim. Biophys. Acta Biomembr. 1981, 647, 196–202. [Google Scholar] [CrossRef]
- Hancock, J.F. Lipid rafts: Contentious only from simplistic standpoints. Nat. Rev. Mol. Cell Biol. 2006, 7, 456–462. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E.J. Structure and function of sphingolipids and cholesterol rich membrane rafts. J. Biol. Chem. 2000, 275, 17221–17224. [Google Scholar] [CrossRef]
- Simons, K.; Toomre, D. Lipid rafts and signal transduction. Nat. Rev. Mol. Cell Biol. 2000, 1, 31–39. [Google Scholar] [CrossRef]
- Harder, T.; Scheiffele, P.; Verkade, P.; Simons, K. Lipid domain structure of the plasma membrane revealed by the patching of cell membranes. J. Cell Biol. 1998, 41, 929–942. [Google Scholar] [CrossRef]
- Onai, T.; Hirai, M. Morphology transition of raft-model membrane induced by osmotic pressure: Formation of double layered vesicle structures similar to an endo- and/or exocytosis. J. Phys. Conf. Ser. 2010, 247. [Google Scholar] [CrossRef]
- Giocondi, M.; Besson, F.; Desset, P.; Milhiet, P.E.; Grimellec, C.L. Remodeling of ordered membrane domains by GP1—Anchored intestinal alkaline phosphatise. Langmuir 2007, 23, 9358–9364. [Google Scholar] [CrossRef]
- Terrel, J.; Yadava, P.; Castro, C.; Hughes, J. Liposome fluidity alters interaction between the ganglioside GM1 and cholera toxin B subunit. J. Liposome Res. 2008, 18, 21–29. [Google Scholar] [CrossRef]
- Nomura, F.; Inaba, T.; Ishikawa, S.; Nagata, M.; Takahashi, S.; Hotani, H.; Takiguchi, K. Microscopic observations reveal that fusogenic peptides induce liposome shrinkage prior to membrane fusion. Proc. Natl. Acad. Sci. USA 2004, 101, 3420–3425. [Google Scholar] [CrossRef]
- Frolov, V.A.J.; Chizmadzhev, Y.A.; Cohen, F.S.; Zimmerberg, J. Entropic traps in the kinetics of phase separation in multicomponent membranes stabilize nanodomains. Biophys. J. 2006, 91, 189–205. [Google Scholar] [CrossRef]
- Hamada, T.; Miura, Y.; Komatsu, Y.; Kishimoto, Y.; Vestergaard, M.; Takagi, M. Construction of asymmetric cell-sized lipid vesicles from lipid coated water in oil microdroplets. J. Phys. Chem. B 2008, 112, 14678–14681. [Google Scholar] [CrossRef]
- Yamada, N.L.; Hishida, M.; Seto, H.; Tsumoto, K.; Yoshimura, T. Unbinding transition induced by osmotic pressure in relation to unilamellar vesicle formation. Europhys. Lett. 2006, 80. [Google Scholar] [CrossRef]
- Hamada, T.; Miura, Y.; Ishii, K.; Araki, S.; Yoshikawa, K.; Vestergaard, M.; Takagi, M. Dynamic processes in endocytic transformation of a raft-exhibiting giant liposome. J. Phys. Chem. B 2007, 111, 10853–10857. [Google Scholar] [CrossRef]
- Seifert, U.; Berndl, K.; Lipowsky, R. Shape transformations of vesicles: Phase diagram for spontaneous-curvature and bilayer-coupling models. Phys. Rev. 1991, 44, 1182–1202. [Google Scholar] [CrossRef]
- Akimov, S.A.; Hlaponin, E.A.; Bashkirov, P.V.; Boldyrev, I.A.; Mikhaloyov, I.I.; Jelford, W.G.; Molotkovskaya, I.M. Ganglioside GM1 increases line tension at raft boundary in model membranes. J. Membr. Cell Biol. 2009, 3, 216–222. [Google Scholar]
- Mrówczyńska, L.; Lindqvist, C.; Iglič, A.; Hägerstrand, H. Spontaneous curvature of ganglioside GM1—Effect of cross-linking. Biochem. Biophys. Res. Commun. 2012, 422, 776–779. [Google Scholar] [CrossRef]
- Mori, K.; Mahmood, M.I.; Neya, S.; Matsuzaki, K.; Hoshino, T. Formation of GM1 ganglioside clusters on the lipid membrane containing sphingomyeline and cholesterol. J. Phys. Chem. B 2012, 116, 5111–5121. [Google Scholar] [CrossRef]
- Fukunaga, S.; Ueno, H.; Yamaguchi, T.; Yano, Y.; Hoshino, M.; Matsuzaki, K. GM1 cluster mediates formation of toxic Aβ fibrils by providing hydrophobic environments. Biochemistry 2012, 51, 8125–8131. [Google Scholar] [CrossRef]
- Matsubara, T.; Iijima, K.; Yamamoto, N.; Yanagisawa, K.; Sato, T. Density of GM1 in nanoclusters is a critical factor in the formation of a spherical assembly of amyloid β-protein on synaptic plasma membranes. Langmuir 2013, 29, 2258–2264. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dhingra, S.; Morita, M.; Yoda, T.; Vestergaard, M.C.; Hamada, T.; Takagi, M. Dynamic Morphological Changes Induced By GM1 and Protein Interactions on the Surface of Cell-Sized Liposomes. Materials 2013, 6, 2522-2533. https://doi.org/10.3390/ma6062522
Dhingra S, Morita M, Yoda T, Vestergaard MC, Hamada T, Takagi M. Dynamic Morphological Changes Induced By GM1 and Protein Interactions on the Surface of Cell-Sized Liposomes. Materials. 2013; 6(6):2522-2533. https://doi.org/10.3390/ma6062522
Chicago/Turabian StyleDhingra, Shruti, Masamune Morita, Tsuyoshi Yoda, Mun'delanji C. Vestergaard, Tsutomu Hamada, and Masahiro Takagi. 2013. "Dynamic Morphological Changes Induced By GM1 and Protein Interactions on the Surface of Cell-Sized Liposomes" Materials 6, no. 6: 2522-2533. https://doi.org/10.3390/ma6062522



