Phenotypic Diversity of Litsea cubeba in Jiangxi China and the Identification of Germplasms with Desirable Characteristics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling Sites
2.2. Materials
2.3. Methods
2.3.1. Tree Growth
2.3.2. Fruit Traits
2.3.3. EO Characteristics
2.4. Statistical Analysis
3. Results
3.1. Phenotypic Variation Characteristics
3.1.1. Tree and Fruit Phenotypic Traits
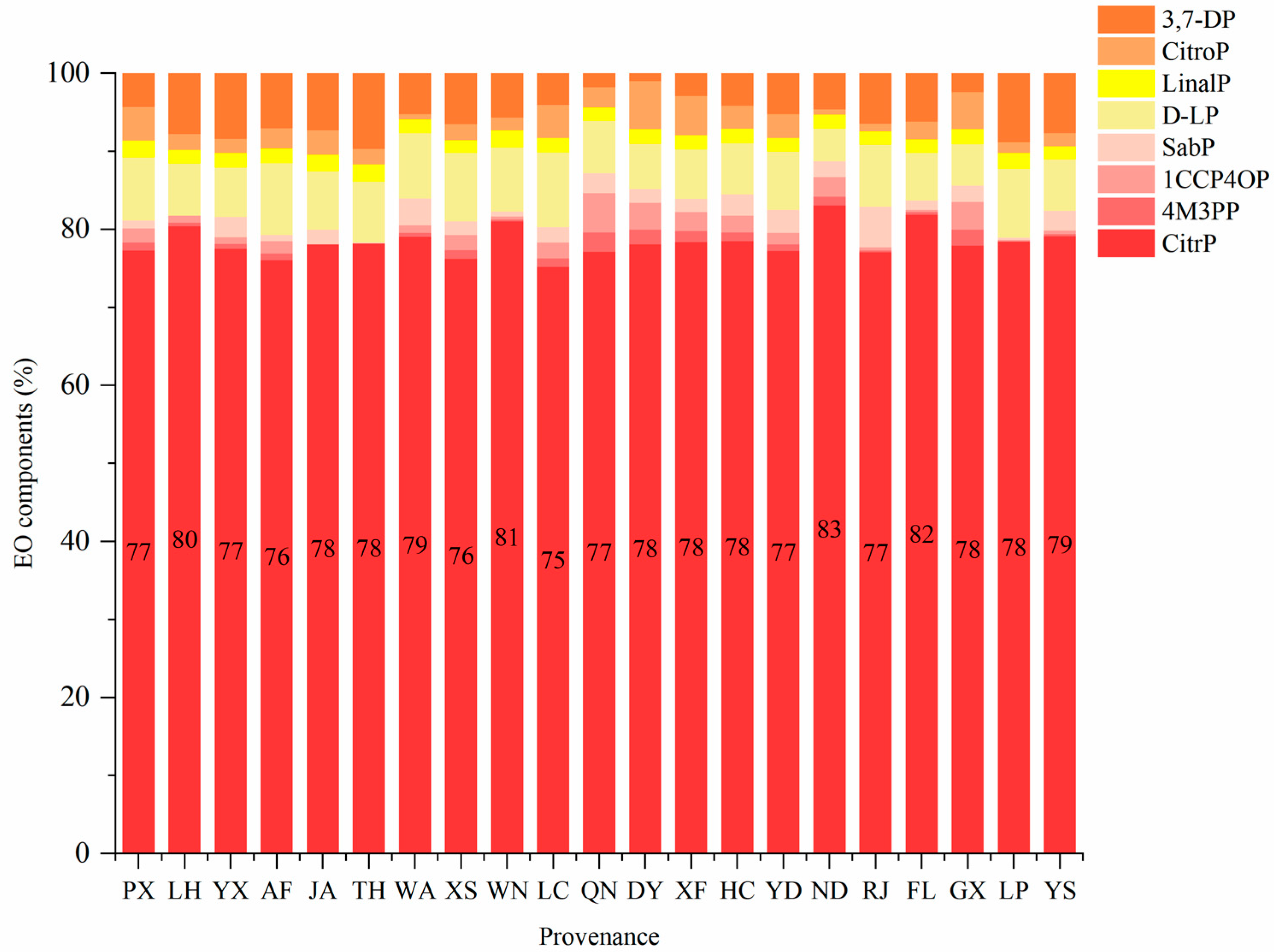
3.1.2. EO Characteristics
3.2. Correlation Analysis of Phenotypes and Geographical and Climatic Factors
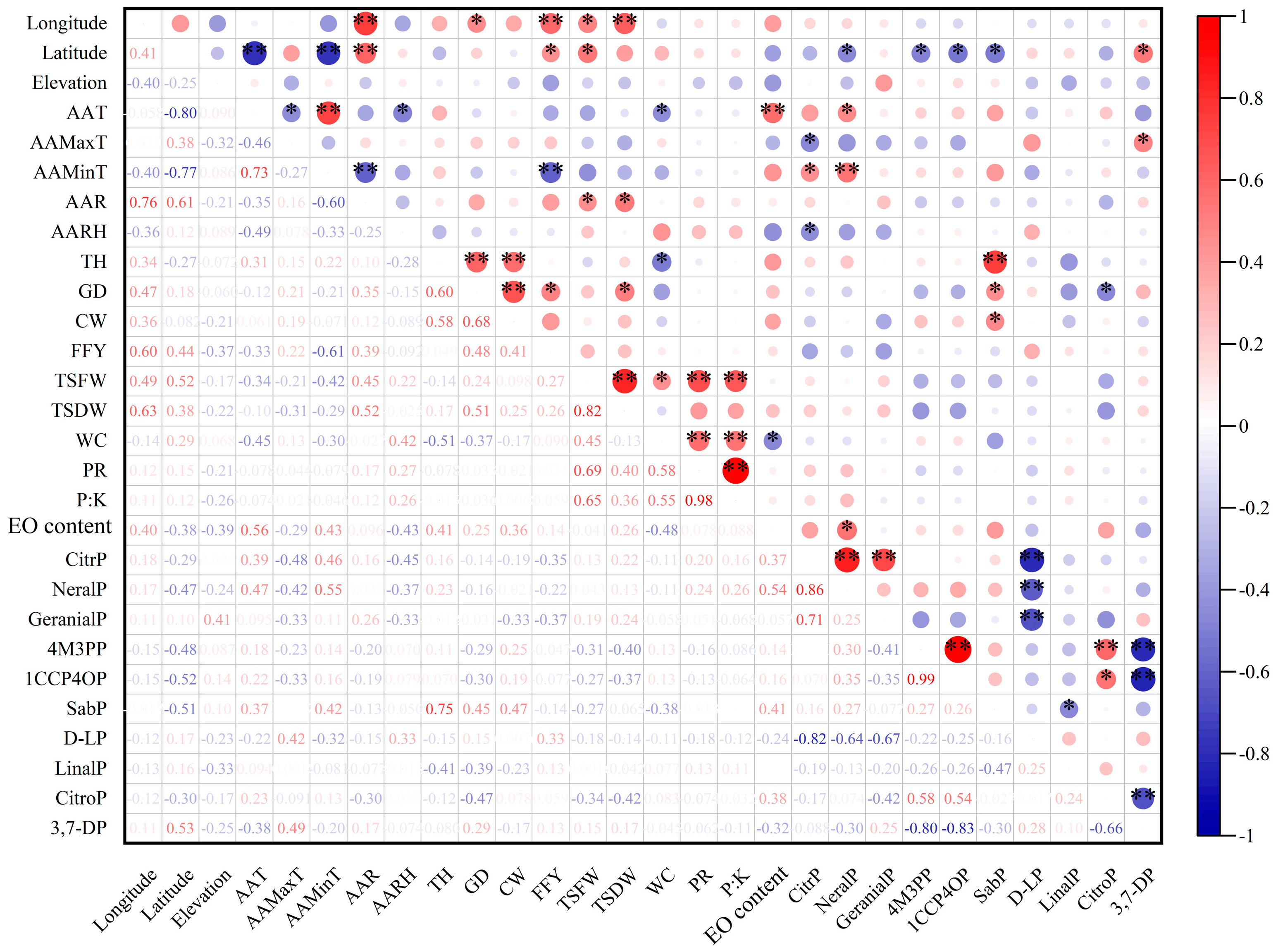
3.2.1. Correlation Analyses between Phenotypic Characteristics
3.2.2. Correlation Analysis between Geographical and Climatic Factors and Phenotypes
3.3. Path Analyses of FFY, EO Content and CitrP
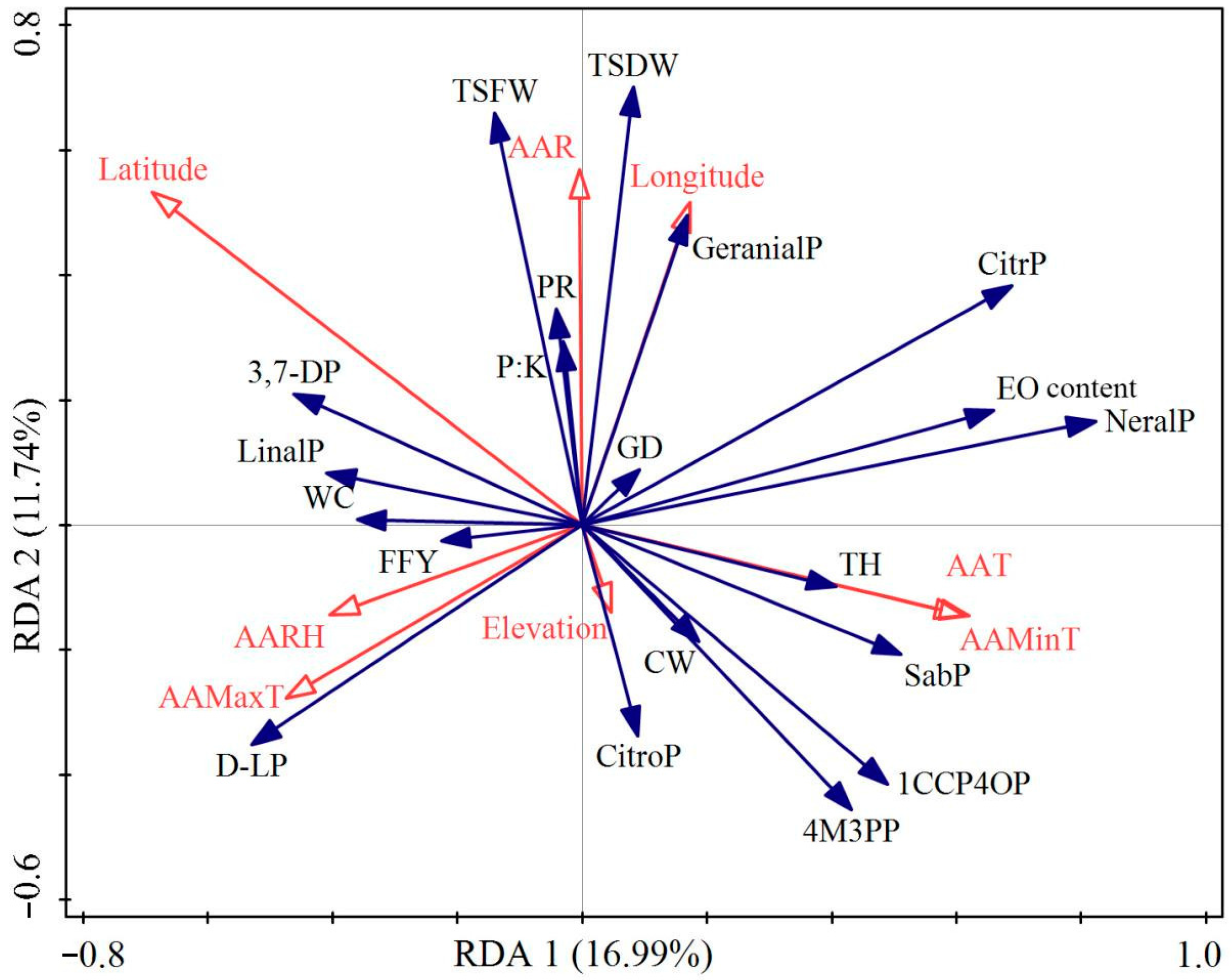
3.4. Redundancy Analysis
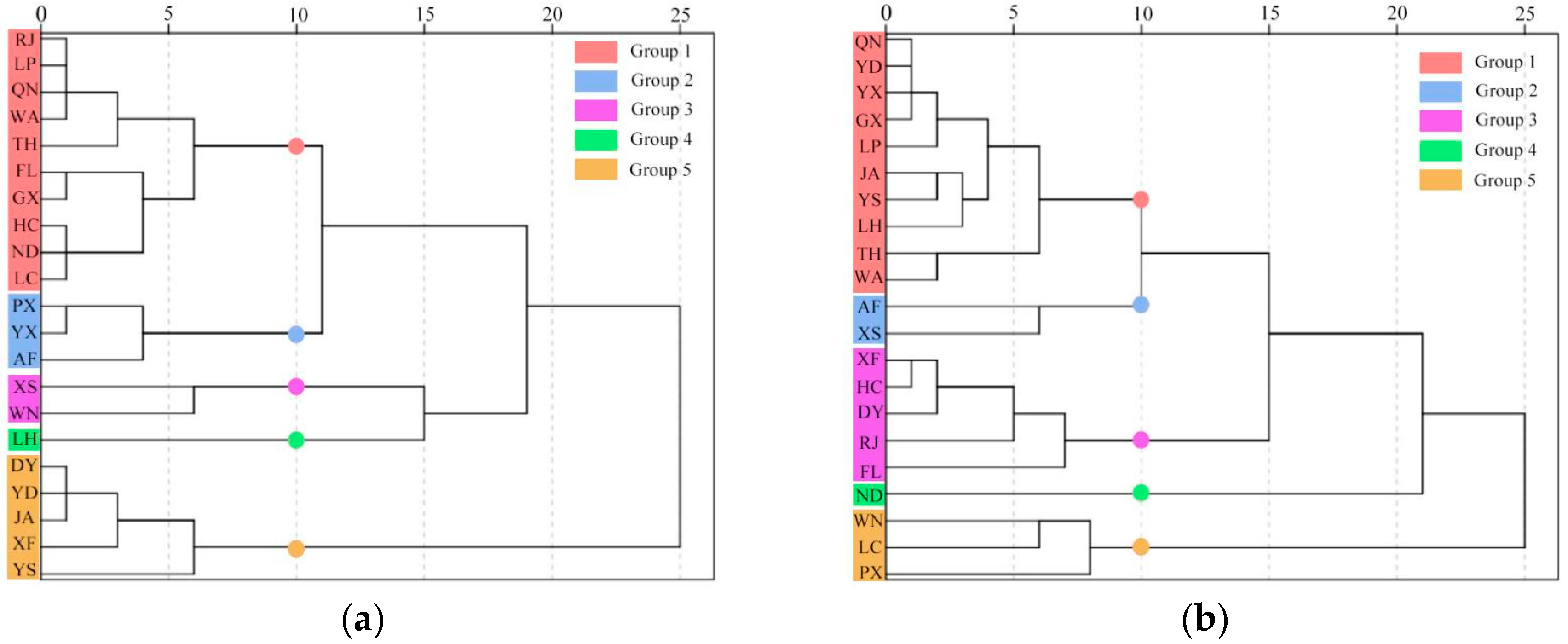
3.5. Identify Individual Trees and Provenances with Desirable Characteristic
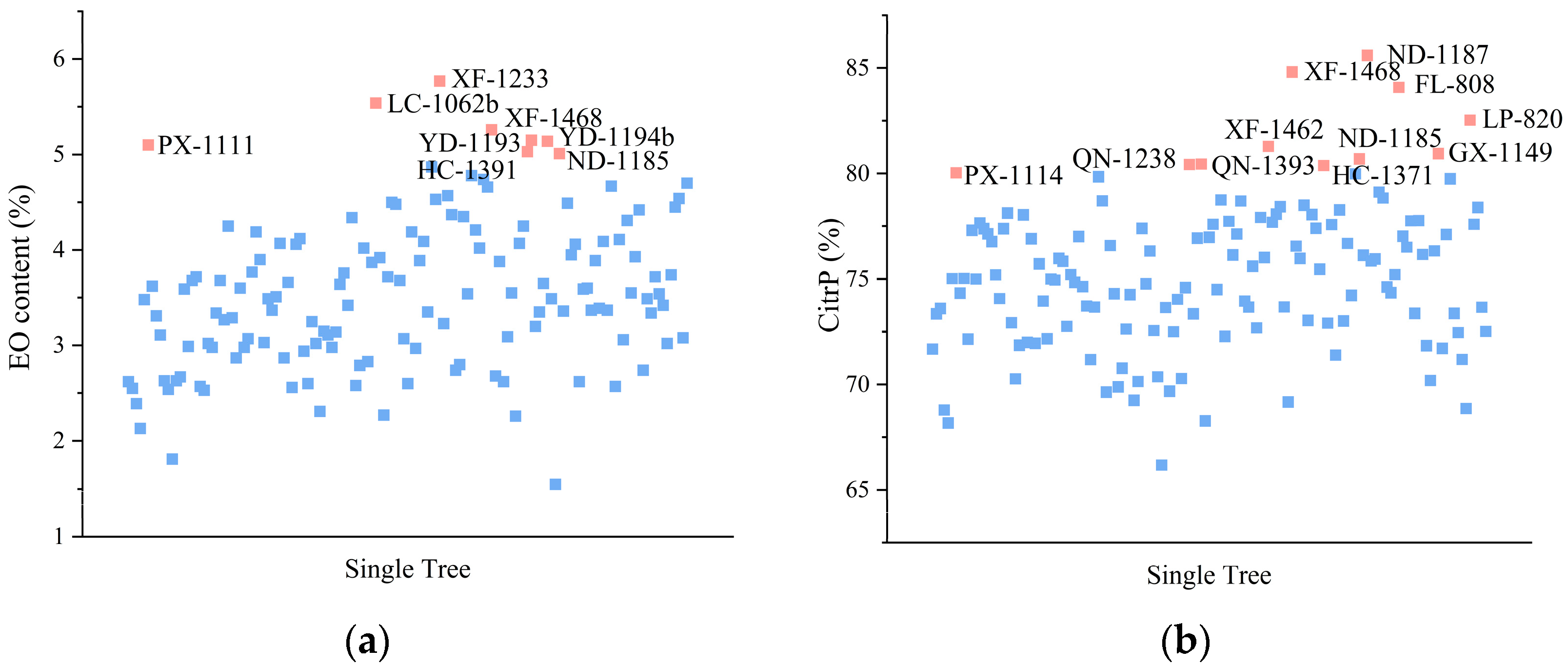
3.5.1. Identification of L. cubeba Trees
3.5.2. Identification of Individual Trees with Desirable Characteristics
3.5.3. Identification of Provenances with Desirable Traits
4. Discussion
4.1. Phenotypic Diversity
4.2. Geographic and Climate Factors Affecting the Phenotypes
4.3. Individual Trees and Provenances with Desirable Characteristics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Chen, W.J. A Study on the relationship between the change of outer color of Litsea cubeba fruit and its oil yield and quality. J. Henan Fores. Sci. Technol. 2001, 21, 33–34. [Google Scholar]
- Zhao, Y.; Chen, Y.; Gao, M.; Wu, L.; Wang, Y. Comparative investigation of key aroma terpenoids of Litsea cubeba essential oil by sensory, chromatographic, spectral and molecular studies. LWT-Food Sci. Technol. 2023, 176, 114519. [Google Scholar] [CrossRef]
- Pante, G.C.; Castro, J.C.; Lini, R.S.; Romoli, J.C.Z.; Almeida, R.T.R.; Garcia, F.P.; Nakamura, C.V.; Pilau, E.J.; Abreu Filho, B.A.; Machinski, M. Litsea cubeba essential oil: Chemical profile, antioxidant activity, cytotoxicity, effect against Fusarium verticillioides and fumonisins production. J. Environ. Sci. Health B 2021, 56, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Sattayakhom, A.; Songsamoe, S.; Yusakul, G.; Kalarat, K.; Matan, N.; Koomhin, P. Effects of Thai local ingredient odorants, Litsea cubeba and garlic essential oils, on brainwaves and moods. Molecules 2021, 26, 2939. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.C.; Chang, W.T.; Hsen, Y.C.; Chen, H.Y.; Chuang, C.H.; Lin, C.C.; Lee, M.S.; Lin, M.K. Immunosuppressive effect of Litsea cubeba L. essential oil on dendritic cell and contact hypersensitivity responses. Int. J. Mol. Sci. 2016, 17, 1319. [Google Scholar] [CrossRef]
- Lv, H.; Huo, S.; Zhao, L.; Zhang, H.; Liu, Y.; Liu, S.; Tani, A.; Wang, R. Preparation and application of cinnamon-Litsea cubeba compound essential oil microcapsules for peanut kernel postharvest storage. Food Chem. 2023, 415, 135734. [Google Scholar] [CrossRef]
- Xia, S.; Lin, H.; Zhu, P.; Wang, P.; Liao, S.; Chen, S.; Wang, Z.; Fan, G. Inhibitory effects of Litsea cubeba oil and its active components on aspergillus flavus. J. Food Qual. 2020, 2020, 8843251. [Google Scholar] [CrossRef]
- Li, X.; Li, G.; Shan, Y.; Zhu, X. Preparation, characterization, and antifungal property of the inclusion complex of Litsea cubeba essential oil/hydroxypropyl-β-cyclodextrin and its application in preservation of Shatang mandarin. J. Food Sci. 2022, 87, 4714–4724. [Google Scholar] [CrossRef]
- Dai, J.; Li, C.; Cui, H.; Lin, L. Unraveling the anti-bacterial mechanism of Litsea cubeba essential oil against E. coli O157:H7 and its application in vegetable juices. Int. J. Food Microbiol. 2021, 338, 108989. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, F.; Yin, Y.; Huang, P.; Jiang, Q.; Liu, Z.; Yin, Y.; Chen, J. Dietary Litsea cubeba essential oil supplementation improves growth performance and intestinal health of weaned piglets. Anim. Nutr. 2022, 13, 9–18. [Google Scholar] [CrossRef]
- Bampidis, V.; Azimonti, G.; Bastos, M.d.L.; Christensen, H.; Fašmon Durjava, M.; Kouba, M.; López, A.M.; López Puente, S.; Marcon, F.; Mayo, B.; et al. Safety and efficacy of a feed additive consisting of an essential oil from the fruits of Litsea cubeba (Lour.) Pers. (litsea berry oil) for use in all animal species (FEFANA asbl). EFSA J. 2021, 19, e06623. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhuang, X.; Wu, X.; Qiu, C.; Wang, Y. Sustainable valorization of Litsea cubeba (Lour.) Pers. residue as the new lauric oil source using alternative green extraction and refining methods. Foods 2022, 11, 2047. [Google Scholar] [CrossRef] [PubMed]
- Li, C.N.; Wang, Y.; Chen, S.X.; Wu, L.F.; Wang, Z.H. Comprehensive evaluation of drought resistance of 9 pioneer plants in western Guangdong mining area. J. Weat China Fores. Sci. 2022, 51, 97–105. (In Chinese) [Google Scholar] [CrossRef]
- Qiu, Y.; Yu, Y.; Lan, P.; Wang, Y.; Li, Y. An overview on total valorization of Litsea cubeba as a new woody oil plant resource toward a zero-waste biorefinery. Molecules 2021, 26, 3948. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, Y.; Han, X.; Si, L.; Wu, Q.; Lin, L. Biology and chemistry of Litsea cubeba, a promising industrial tree in China. J. Essent. Oil Res. 2013, 25, 103–111. [Google Scholar] [CrossRef]
- Zheng, Y.T.; Cai, Q.S.; Xv, L.C.; Li, C.Y. Present situation and development trend of exploitation for Litsea cubeba in Jiangxi Province. Jiangxi Fores. Sci. Tech. 2010, 2, 33–35. (In Chinese) [Google Scholar] [CrossRef]
- Zhuang, X.; Zhang, Z.; Wang, Y.; Li, Y. The effect of alternative solvents to n-hexane on the green extraction of Litsea cubeba kernel oils as new oil sources. Ind. Crops Prod. 2018, 126, 340–346. [Google Scholar] [CrossRef]
- Guo, Y.; Li, Y.; Li, Z.; Jiang, L.; Cao, X.; Gao, W.; Wang, J.; Luo, D.; Chen, F. Deep eutectic solvent-homogenate based microwave-assisted hydrodistillation of essential oil from Litsea cubeba (Lour.) Pers. fruits and its chemical composition and biological activity. J. Chromatogr. A 2021, 1646, 462089. [Google Scholar] [CrossRef]
- Qiu, Y.; Wang, Y.; Li, Y. Solvent-free microwave extraction of essential oils from Litsea cubeba (Lour.) Pers. at different harvesting times and their skin-whitening cosmetic potential. Antioxidants 2022, 11, 2398. [Google Scholar] [CrossRef]
- Hung, T.T.; Ngan, L.T.M.; Viet, H.; Hoang, N.V.M.; Hieu, T.T. Chemical composition and anti-Helicobacter pylori activity of essential oil from fresh fruits of Litsea cubeba (Lour.) Pers. J. Essent. Oil Res. 2023, 35, 207–219. [Google Scholar] [CrossRef]
- Si, L.; Chen, Y.; Han, X.; Zhan, Z.; Tian, S.; Cui, Q.; Wang, Y. Chemical composition of essential oils of Litsea cubeba harvested from its distribution areas in China. Molecules 2012, 17, 7057–7066. [Google Scholar] [CrossRef] [PubMed]
- Thielmann, J.; Murányi, P. Review on the chemical composition of Litsea cubeba essential oils and the bioactivity of its major constituents citral and limonene. J. Essent. Oil Res. 2019, 31, 361–378. [Google Scholar] [CrossRef]
- Dalimunthe, A.; Muhammad, M.; Rafi, M.; Syafma, V.M.; Hulwani, F.; Aprilliawati, I.; Satria, D. Antioxidant activity of N-hexane, Dichloromethane, Ethyl acetate, and Methanol extracts of Litsea cubeba Lour. barks. Rasayan J. Chem. 2023, 16, 642–647. [Google Scholar] [CrossRef]
- Gu, Z.Y.; Yang, L.; Chen, H.; Ran, M.H.; Yang, R.N. Analysis of essential oil in pericarp from 6 wild Litsea cubeba populations in Hunan Province. J. Chin. Cereals Oils Assoc. 2019, 34, 87–91. (In Chinese) [Google Scholar]
- Gao, M.; Chen, Y.; Wang, Y. Evaluation of the yields and chemical compositions of the essential oils of different Litsea cubeba varieties. J. Essent. Oil Bear. Plants. 2016, 19, 1888–1902. [Google Scholar] [CrossRef]
- Suwandhi, I.; Kusmana, C.; Suryani, A.; Tiryana, T. Litsea cubeba essential oil yield harvested from different habitat types on Mt. Papandayan, west Java, Indonesia. J. Math. Fundam. Sci. 2014, 46, 269–277. [Google Scholar] [CrossRef]
- Tian, S.P.; Wang, Y.D.; Chen, Y.C.; Han, X.J.; Zhan, Z.Y. Phenotypic diversity ofnatural Litsea cubeba population’s leaf and fruit traits. Chin. J. Ecol. 2012, 31, 1665–1672. (In Chinese) [Google Scholar] [CrossRef]
- Lan, G.Y.; Chen, J.Z.; Ma, Y.Z.; Liu, X.; Zhou, S.Y. Regulations of fruit development in Litsea cubeba and changes of its inclusions and essential oil. Non-Wood Forest Res. 2020, 38, 201–208. (In Chinese) [Google Scholar] [CrossRef]
- Tang, X.H. Provenance variation of fruit essential oil of Litsea cubeba and content of citral. Protec. Forest Sci. Technol. 2015, 35–37. Available online: https://www.en.cnki.com.cn/Article_en/CJFDTOTAL-FHLK201510011.htm (accessed on 6 December 2022). (In Chinese).
- Fan, G.; Ning, X.; Chen, S.; Zhong, L.; Guo, C.; Yang, Y.; Liu, J.; Tang, M.; Liao, G.; Wang, X.; et al. Differences in fruit yields and essential oil contents and composition among natural provenances of Litsea cubeba in China and their relationships with main habitat factors. Ind. Crops Prod. 2023, 194, 116285. [Google Scholar] [CrossRef]
- Munir, M.; Alhajhoj, M.R.; Sallam, A.-K.M.; Ghazzawy, H.S.; Al-Bahigan, A.M. Impact of pollination time of the day on the fruit, yield, and quality traits of date palm cultivar Khalas. J. Appl. Hortic. 2021, 23, 348–352. [Google Scholar] [CrossRef]
- Kattmah, G.; Mahdi Al-kalaby, J.E.; Al-Ibrahem, A.; Al-Rasheed, M.M. Evolution and characterization of olive genetic resources in Palmyra-Central Syria. Kufa J. Agric. Sci. 2019, 11, 77–89. Available online: https://search.emarefa.net/detail/BIM-895767. (accessed on 2 January 2023).
- Šmilauer, P.; Lepš, J. Multivariate Analysis of Ecological Data Using CANOCO 5, 2nd ed.; Cambridge University Press: Cambridge, UK, 2014; ISBN 978-11-3962-706-1. [Google Scholar]
- Hamil, S.; Bouchelouche, D.; Arab, S.; Alili, M.; Baha, M.; Arab, A. The relationship between zooplankton community and environmental factors of Ghrib Dam in Algeria. Environ. Sci. Pollut. R 2021, 28, 46592–46602. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, Y.; Si, Y.; Du, C.; Zhou, X.; Liu, M.; Ning, H.; Ye, P. Principal component analysis and comprehensive evaluation of saline-alkaline tolerance related traits of northern japonica rice. J. Nucl. Agric. Sci. 2020, 34, 1862–1871. (In Chinese) [Google Scholar] [CrossRef]
- Liu, D.L.; Wang, X.Q.; Li, W.S.; Li, J.J.; Tan, W.B.; Xing, W. Genetic diversity analysis of the phenotypic traits of 215 sugar beet germplasm resources. Sugar Tech. 2022, 24, 1790–1800. [Google Scholar] [CrossRef]
- Chikh-Rouhou, H.; Mezghani, N.; Mnasri, S.; Mezghani, N.; Garcés-Claver, A. Assessing the genetic diversity and population structure of a tunisian melon (Cucumis melo L.) collection using phenotypic traits and SSR molecular markers. Agronomy 2021, 11, 1121. [Google Scholar] [CrossRef]
- Lodolini, E.M.; Polverigiani, S.; Giorgi, V.; Famiani, F.; Neri, D. Time and type of pruning affect tree growth and yield in high-density olive orchards. Sci. Hortic. 2023, 311, 111831. [Google Scholar] [CrossRef]
- Plavcová, L.; Mészáros, M.; Šilhán, K.; Jupa, R. Relationships between trunk radial growth and fruit yield in apple and pear trees on size-controlling rootstocks. Ann. Bot. 2022, 130, 477–489. [Google Scholar] [CrossRef]
- Song, Q.N.; Xiao, Z.Y.; Xu, J.W.; Song, S.L.; Luan, F.G.; Liu, J.; Yang, Q.P. Influence of neighbor competition and tree attributes on the fruit production of Sinomanglietia glauca, an endangered tree species endemic to China. Plant Ecol. 2021, 222, 561–573. [Google Scholar] [CrossRef]
- Gao, M.; Chen, Y.C.; Wu, L.W.; Wang, Y.D. Changes in the profiles of yield, yield component, oil content, and citral content in Litsea cubeba (Lour.) Persoon following foliar fertilization with zinc and boron. Forests 2019, 10, 59. [Google Scholar] [CrossRef]
- Li, H.S.; Wang, Y.D.; Xv, G.B.; Chen, Y.C.; Wu, L.W.; Yu, M.Y.; Gao, M. Analysis of the genetic variation and stability of growth traits of Litsea cubeba young plantation. For. Res. 2018, 31, 168–175. (In Chinese) [Google Scholar] [CrossRef]
- Li, H.S.; Wang, Y.D.; Chen, Y.C.; Gao, M.; Wu, L.W.; Xv, G.B. Genetic variation analysis of growth and economic characteristics and superior family selection in Litsea cubeba. Non-Wood Forest Res. 2017, 35, 64–71. (In Chinese) [Google Scholar] [CrossRef]
- Yang, Y.-N.; Zheng, F.-P.; Yu, A.-N.; Sun, B.-G. Changes of the free and bound volatile compounds in Rubus corchorifolius L. f. fruit during ripening. Food Chem. 2019, 287, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, C.; Hartman, T.G. Decrease in fruit moisture content heralds and might launch the onset of ripening processes. J. Food Sci. 2012, 77, S365–S376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zheng, Y.; Fu, C.; Yang, H.; Liu, X.; Qiu, F.; Wang, X.; Wang, Z. Chemical variation and environmental influence on essential oil of Cinnamomum camphora. Molecules 2023, 28, 973. [Google Scholar] [CrossRef]
- Karami, A.; Khoushbakht, T.; Esmaeili, H.; Maggi, F. Essential oil chemical variability in Oliveria decumbens (Apiaceae) from different regions of iran and its relationship with environmental factors. Plants 2020, 9, 680. [Google Scholar] [CrossRef]
- Tripp, E.A.; Zhuang, Y.; Schreiber, M.; Stone, H.; Berardi, A.E. Evolutionary and ecological drivers of plant flavonoids across a large latitudinal gradient. Mol. Phylogenet. Evol. 2018, 128, 147–161. [Google Scholar] [CrossRef]
- Rahimmalek, M.; Heidari, E.F.; Ehtemam, M.H.; Mohammadi, S. Essential oil variation in Iranian Ajowan (Trachyspermum ammi (L.) Sprague) populations collected from different geographical regions in relation to climatic factors. Ind. Crops Prod. 2017, 95, 591–598. [Google Scholar] [CrossRef]
- Félix, M.-A. Phenotypic evolution with and beyond genome evolution. Curr. Top. Dev. Biol. 2016, 119, 291–347. [Google Scholar] [CrossRef]
- Halbritter, A.H.; Fior, S.; Keller, I.; Billeter, R.; Edwards, P.J.; Holderegger, R.; Karrenberg, S.; Pluess, A.R.; Widmer, A.; Alexander, J.M. Trait differentiation and adaptation of plants along elevation gradients. J. Evol. Biol. 2018, 31, 784–800. [Google Scholar] [CrossRef]
- Huang, C.S.; Xu, Y.; Zang, R.G. Variation patterns of functional trait moments along geographical gradients and their environmental determinants in the subtropical evergreen broadleaved forests. Front. Plant Sci. 2021, 12, 686965. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, S.; Liu, X.; Yu, H.; Yu, D.; Li, G.; Wang, L. Oil content, fatty acid composition and biodiesel properties among natural provenances of Siberian apricot (Prunus sibirica L.) from China. GCB Bioenergy 2021, 13, 112–132. [Google Scholar] [CrossRef]
- Guo, J.; Zhou, X.; Wang, T.L.; Wang, G.B.; Cao, F.L. Regulation of flavonoid metabolism in ginkgo leaves in response to different day-night temperature combinations. Plant. Physiol. Bioch. 2020, 147, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Marčetić, M.; Kovačević, N.; Lakušić, D.; Lakušić, B. Habitat-related variation in composition of the essential oil of Seseli rigidum Waldst. & Kit. (Apiaceae). Phytochemistry 2017, 135, 80–92. [Google Scholar] [CrossRef]
- Feng, X.; Zhang, W.; Wu, W.; Bai, R.; Kuang, S.; Shi, B.; Li, D. Chemical composition and diversity of the essential oils of Juniperus rigida along the elevations in Helan and Changbai Mountains and correlation with the soil characteristics. Ind. Crop. Prod. 2021, 159, 113032. [Google Scholar] [CrossRef]
- Melito, S.; Petretto, G.L.; Podani, J.; Foddai, M.; Maldini, M.; Chessa, M.; Pintore, G. Altitude and climate influence Helichrysum italicum subsp. microphyllum essential oils composition. Ind. Crop. Prod. 2016, 80, 242–250. [Google Scholar] [CrossRef]
- Dong, L.; Li, T.; Huang, W.Y.; Wang, B.; Xv, L.C.; Xv, L.A.; Wen, Q. Selection and comprehensive evaluation of superior individual plant in Camellia chekiangoleosa. J. Cent. South Univ. For. Technol. 2021, 41, 35–45. (In Chinese) [Google Scholar] [CrossRef]
- Gao, M.; Chen, Y.C.; Wu, L.W.; Wang, Y.D. Genetic variation of seedling growth of Litsea cubeba from different provenances/families. J. Tropi. Subtrop. Plants 2018, 26, 47–55. (In Chinese) [Google Scholar] [CrossRef]
- Wang, H.; Li, Y.; Li, Z.; Ma, R.; Bai, X.; Zhan, X.; Luo, K.; Su, R.; Li, X.; Xia, X.; et al. Inhibition of Cronobacter sakazakii by Litsea cubeba essential oil and the antibacterial mechanism. Foods 2022, 11, 3900. [Google Scholar] [CrossRef]
- Lin, B.; Liu, Q. Plasticity responses of 4 tree species in subalpine-coniferous-forest to different light regimes. J. Ecol. 2008, 28, 4665–4675. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Ning, X.; Liao, G.; Fan, G.; Shi, X.; Fu, D.; Wang, Z.; Chen, S.; Wang, J. Phenotypic Diversity of Litsea cubeba in Jiangxi China and the Identification of Germplasms with Desirable Characteristics. Forests 2023, 14, 2283. https://doi.org/10.3390/f14122283
Wang X, Ning X, Liao G, Fan G, Shi X, Fu D, Wang Z, Chen S, Wang J. Phenotypic Diversity of Litsea cubeba in Jiangxi China and the Identification of Germplasms with Desirable Characteristics. Forests. 2023; 14(12):2283. https://doi.org/10.3390/f14122283
Chicago/Turabian StyleWang, Xuefang, Xiaodan Ning, Guoxiang Liao, Guorong Fan, Xiaodeng Shi, Dan Fu, Zongde Wang, Shangxing Chen, and Jiawei Wang. 2023. "Phenotypic Diversity of Litsea cubeba in Jiangxi China and the Identification of Germplasms with Desirable Characteristics" Forests 14, no. 12: 2283. https://doi.org/10.3390/f14122283




