Combining Soil Immobilization and Dressing Techniques for Sustaining the Health of Metal-Contaminated Arable Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Samples and Remediation
2.2. Total Data Set (TDS) for Soil Health Assessment
2.3. Minimum Data Set (MDS) for Soil Health Assessment
2.4. Data Treatment and Statistics
3. Results and Discussion
3.1. Heavy Metals in Soil
3.2. Total Data Set for Soil Health Assessment
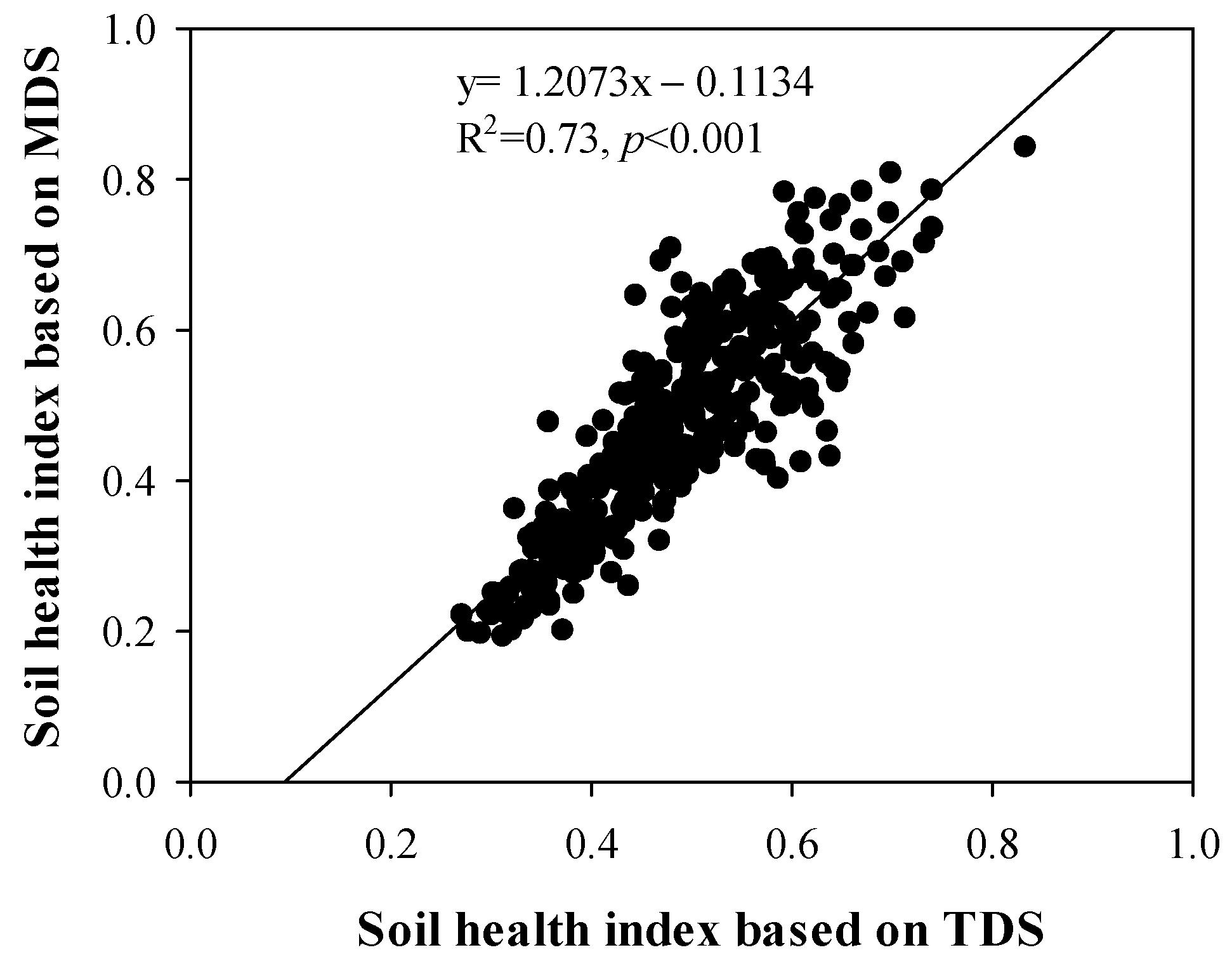
3.3. Selection of the Minimum Data Set (MDS)
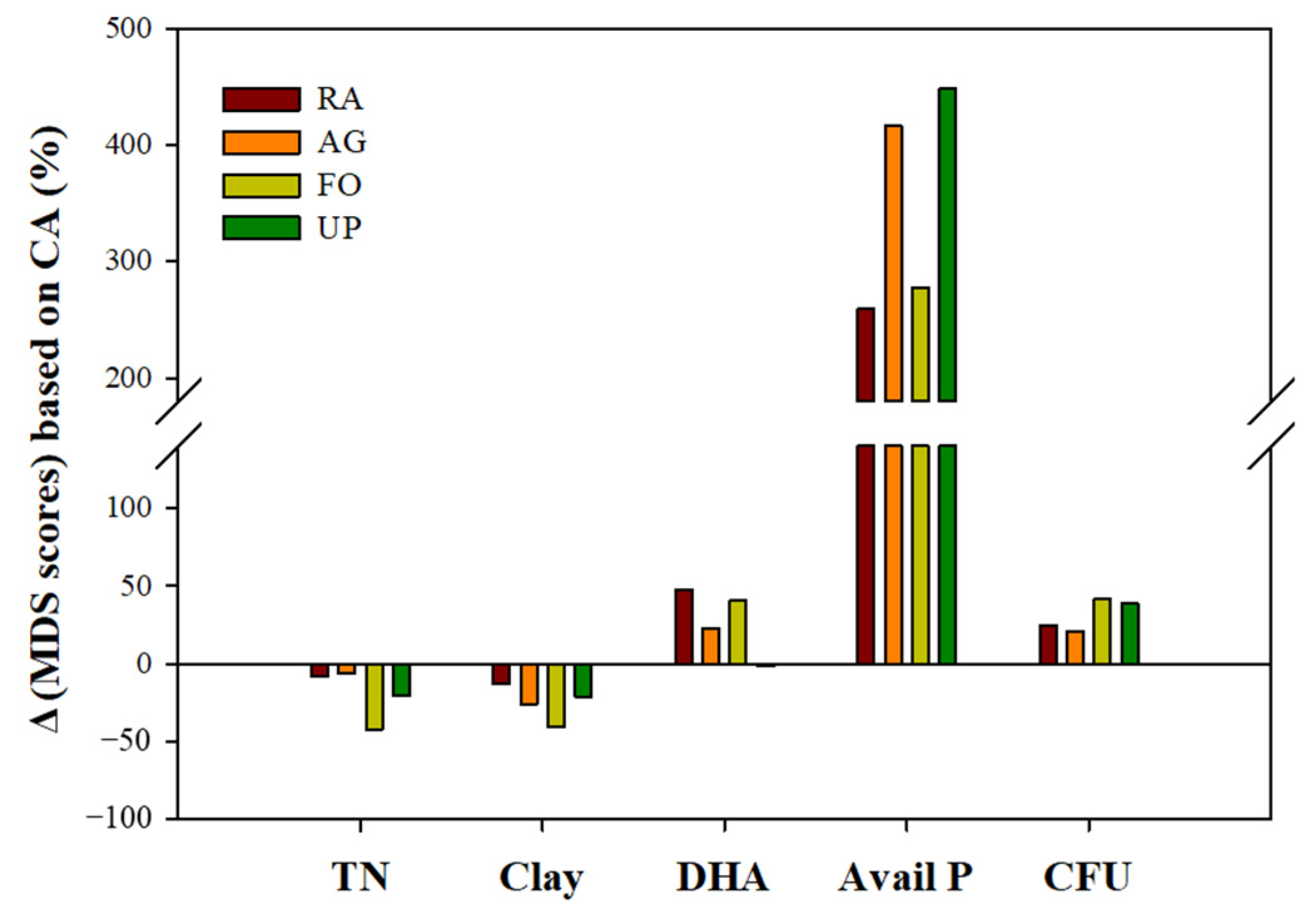
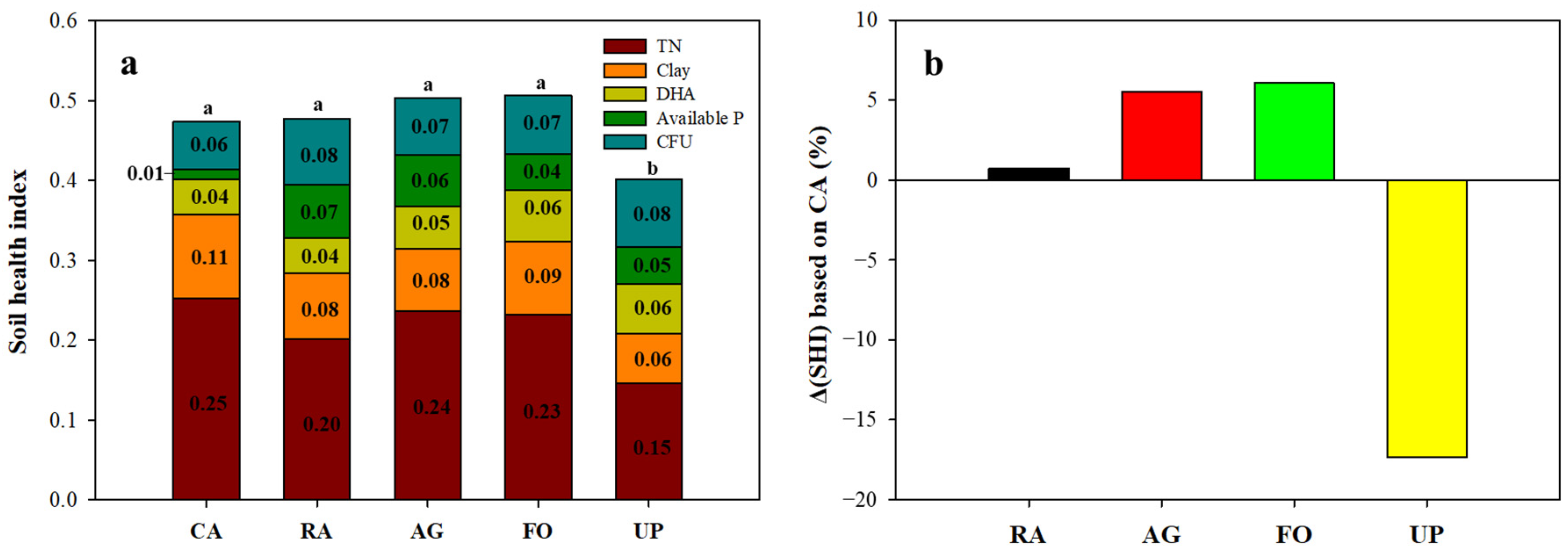
3.4. Scores of Soil Health Index (SHI)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adriano, D.C. Trace Elements in Terrestrial Environments: Biogeochemistry, Bioavailability and Risks of Metals, 2nd ed.; Springer: New York, NY, USA, 2001; pp. 1–796. [Google Scholar]
- Srivastava, V.; Sarkar, A.; Singh, S.; Singh, P.; de Araujo, A.S.F.; Singh, R.P. Agroecological responses of heavy metal pollution with special emphasis on soil health and plant performances. Front. Environ. Sci. 2017, 5, 64. [Google Scholar] [CrossRef]
- Lee, S.W.; Lee, B.T.; Kim, J.Y.; Kim, K.W.; Lee, J.S. Human risk assessment for heavy metals and As contamination in the abandoned metal mine areas, Korea. Environ. Monit. Assess. 2006, 119, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, S.S.; Singh, J.; Taneja, P.K.; Mandal, A. Remediation techniques for removal of heavy metals from the soil contaminated through different sources: A review. Environ. Sci. Pollut. Res. 2020, 27, 1319–1333. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Oh, S.J.; Kum, D.; Shin, M.; Kim, D.; Lee, S.S. Efficiency of heavy metal stabilizers in various soils. Korean J. Environ. Agric. 2021, 40, 231–238, (In Korean, with English abstract). [Google Scholar] [CrossRef]
- Rasafi, T.E.; Nouri, M.; Haddioui, A. Metals in mine wastes: Environmental pollution and soil remediation approaches—A review. Geos. Eng. 2017, 24, 157–172. [Google Scholar] [CrossRef]
- Giannis, A.; Gidarakos, E.; Skouta, A. Transport of cadmium and assessment of phytotoxicity after electrokinetic remediation. J. Environ. Manag. 2008, 86, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Zhao, D.; Wang, Q. An overview of field-scale studies on remediation of soil contaminated with heavy metals and metalloids: Technical progress over the last decade. Water Res. 2018, 147, 440–460. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.M.; Sung, K. Influence of washing treatment on the qualities of heavy metal-contaminated soil. Ecol. Eng. 2015, 81, 89–92. [Google Scholar] [CrossRef]
- Wang, Q.Y.; Zhou, D.M.; Cang, L.; Sun, T.R. Application of bioassays to evaluate a copper contaminated soil before and after a pilot-scale electrokinetic remediation. Environ. Pollut. 2009, 157, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Zupanc, V.; Kastelec, D.; Lestan, D.; Grcman, H. Soil physical characteristics after EDTA washing and amendment with inorganic and organic additives. Environ. Pollut. 2014, 186, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Kim, J.G.; Park, J.S.; Kim, M.S.; Owens, G.; Youn, G.H.; Lee, J.S. Immobilizer-assisted management of metal-contaminated agricultural soils for safer food production. J. Environ. Manag. 2012, 102, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Lwin, C.S.; Seo, B.H.; Kim, H.U.; Owens, G.; Kim, K.R. Application of soil amendments to contaminated soils for heavy metal immobilization and improved soil quality-a critical review. Soil Sci. Plant Nutr. 2018, 64, 156–167. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, K.R.; Kim, H.J.; Yoon, J.H.; Yang, J.E.; Ok, Y.S.; Owens, G.; Kim, K.H. Effect of biochar on heavy metal immobilization and uptake by lettuce (Lactuca sativa L.) in agricultural soil. Environ. Earth Sci. 2015; 74, 1249–1259. [Google Scholar]
- Awa, S.H.; Hadibarata, T. Removal of heavy metals in contaminated soil by phytoremediation mechanism: A review. Water Air Soil Pollut. 2020, 231, 47. [Google Scholar] [CrossRef]
- Liu, L.; Li, W.; Song, W.; Guo, M. Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci. Total Environ. 2018, 633, 206–219. [Google Scholar] [CrossRef] [PubMed]
- RoyChowdhury, A.; Datta, R.; Sarkar, D. Heavy metal pollution and remediation. In Green Chemistry; Török, B., Dransfield, T., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 3.10; pp. 359–373. [Google Scholar] [CrossRef]
- Yun, S.W.; Park, C.G.; Jeon, J.H.; Darnault, C.J.G.; Baveye, P.C.; Yu, C. Dissolution behavior of As and Cd in submerged paddy soil after treatment with stabilizing agents. Geoderma 2016, 270, 10–20. [Google Scholar] [CrossRef]
- Ma, Y.; Dong, B.; He, X.; Shi, Y.; Xu, M.; He, X.; Du, X.; Li, F. Quicklime-induced changes of soil properties: Implications for enhanced remediation of volatile chlorinated hydrocarbon contaminated soils via mechanical soil aeration. Chemosphere 2017, 173, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Choi, J.; Jeong, S.W. Changes in the health of metal-contaminated soil before and after stabilization and solidification. Environ. Pollut. 2023, 331, 121929. [Google Scholar] [CrossRef] [PubMed]
- Yuvaraj, A.; Thangaraj, R.; Karmegam, N.; Ravindran, B.; Chang, S.W.; Awasthi, M.K.; Kannan, S. Activation of biochar through exoenzymes prompted by earthworms for vermibiochar production: A viable resource recovery option for heavy metal contaminated soils and water. Chemosphere 2021, 278, 130458. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.S.; Adriano, D.C.; Mani, P.A.; Duraisamy, A. Immobilization and phytoavailability of cadmium in variable charge soils. II. Effect of lime addition. Plant Soil 2003, 251, 187–198. [Google Scholar] [CrossRef]
- EPA. The Use of Soil Amendments for Remediation, Revitalization, and Reuse (EPA 542-R-07-013); Environmental Protection Agency: Washington, DC, USA, 2007. [Google Scholar]
- Kumar, M.; Bolan, N.S.; Hoang, S.A.; Sawarkar, A.D.; Jasemizad, T.; Gao, B.; Keerthanan, S.; Padhye, L.P.; Singh, L.; Kumar, S.; et al. Remediation of soils and sediments polluted with polycyclic aromatic hydrocarbons: To immobilize, mobilize, or degrade? J. Hazard. Mater. 2021, 420, 126534. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.I.; Nam, S.H.; Kim, S.W.; Bajagain, R.; Jeong, S.W.; An, Y.J. Changes in soil properties after remediation influence the performance and survival of soil algae and earthworm. Ecotoxicol. Environ. Saf. 2019, 174, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Lee, J.H.; Jung, W.C.; Park, M.; Kim, M.S.; Lee, S.J.; Park, H. Changes in soil health with remediation of petroleum hydrocarbon contaminated soils using two different remediation technologies. Sustainability 2020, 12, 10078. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, S.O.; Lee, S.W.; Kim, M.S.; Park, H. Application of soil washing and thermal desorption for sustainable remediation and reuse of remediated soil. Sustainability 2021, 13, 12523. [Google Scholar] [CrossRef]
- Honeycutt, C.W.; Morgan, C.L.S.; Elias, P.; Doane, M.; Mesko, J.; Myers, R.; Odom, L.; Moebius-Clune, B.; Nichols, R. Soil health: Model programs in the USA. Front. Agric. Sci. Eng. 2020, 7, 356–361. [Google Scholar] [CrossRef]
- Lehmann, J.; Bossio, D.A.; Kögel-Knabner, I.; Rillig, M.C. The concept and future prospects of soil health. Nat. Rev. Earth Environ. 2020, 1, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Wang, C.; Liu, M.; Zhang, D.; Li, K.; Li, N. A new method for soil health assessment based on analytic hierarchy process and meta-analysis. Sci. Total Environ. 2019, 650, 2771–2777. [Google Scholar] [CrossRef] [PubMed]
- Buzzard, V.; Gil-Loaiza, J.; Grachet, N.G.; Talkington, H.; Youngerman, C.; Tfaily, M.M.; Meredith, L.K. Green infrastructure influences soil health: Biological divergence one year after installation. Sci. Total Environ. 2021, 801, 149644. [Google Scholar] [CrossRef] [PubMed]
- Karlen, D.L.; Ditzler, C.A.; Andrews, S.S. Soil quality: Why and how? Geoderma 2003, 114, 145–156. [Google Scholar] [CrossRef]
- Williams, H.; Colombi, T.; Keller, T. The influence of soil management on soil health: An on-farm study in southern Sweden. Geoderma 2020, 360, 114010. [Google Scholar] [CrossRef]
- Wulanningtyas, H.S.; Gong, Y.; Li, P.; Sakagami, N.; Nishiwaki, J.; Komatsuzaki, M. A cover crop and no-tillage system for enhancing soil health by increasing soil organic matter in soybean cultivation. Soil Tillage Res. 2021, 205, 104749. [Google Scholar] [CrossRef]
- Andrews, S.S.; Mitchell, J.P.; Mancinelli, R.; Karlen, D.L.; Hartz, T.K.; Horwath, W.R.; Pettygrove, G.S.; Scow, K.M.; Munk, D.S. On-farm assessment of soil quality in California’s Central Valley. Agron. J. 2001, 94, 12–23. [Google Scholar]
- Rinot, O.; Levy, G.J.; Steinberger, Y.; Svoray, T.; Eshel, G. Soil health assessment: A critical review of current methodologies and a proposed new approach. Sci. Total Environ. 2019, 648, 1484–1491. [Google Scholar] [CrossRef] [PubMed]
- Sintim, H.Y.; Bandopadhyay, S.; English, M.E.; Bary, A.I.; DeBruyn, J.M.; Schaeffer, S.M.; Miles, C.A.; Reganold, J.P.; Flury, M. Impacts of biodegradable plastic mulches on soil health. Agric. Ecosyst. Environ. 2019, 273, 36–49. [Google Scholar] [CrossRef]
- Bünemann, E.K.; Bongiorno, G.; Bai, Z.; Creamer, R.E.; De Deyn, G.; de Goede, R.; Fleskens, V.; Geissen, T.W.; Kuyper, P.; Mäder, M.; et al. Soil quality—A critical review. Soil Biol. Biochem. 2018, 120, 105–125. [Google Scholar] [CrossRef]
- Ministry of Environment. Soil Environment Conservation Act; Ministry of Environment: Sejong, Republic of Korea, 2019. [Google Scholar]
- Lee, S.H.; Ji, W.; Yang, H.J.; Kang, S.Y.; Jang, D.M. Reclamation of mine-degraded agricultural soils from metal mining: Lessons from 4 years of monitoring activity in Korea. Environ. Earth Sci. 2017, 76, 720. [Google Scholar] [CrossRef]
- NIAST (National Institute of Agricultural Science and Technology). Method of Soil and Plant Analysis; RDA: Suwon, Republic of Korea, 2000. (In Korean) [Google Scholar]
- Hong, Y.K.; Yoon, D.H.; Kim, J.W.; Chae, M.J.; Ko, B.K.; Kim, S.C. Ecological risk assessment of heavy metal-contaminated soil using the triad approach. J. Soils Sediments 2021, 21, 2732–2743. [Google Scholar] [CrossRef]
- Turan, V. Confident performance of chitosan and pistachio shell biochar on reducing Ni bioavailability in soil and plant plus improved the soil enzymatic activities, antioxidant defense system and nutritional quality of lettuce. Ecotoxicol. Environ. Saf. 2019, 183, 109594. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Arylsulfatase activivty of soil. Soil Sci. Soc. Am. J. 1970, 34, 225–229. [Google Scholar] [CrossRef]
- Eivazi, F.; Tabatabai, M. Factors affecting glucosidase and galactosidase activities in soils. Soil Biol. Biochem. 1990, 22, 891–897. [Google Scholar] [CrossRef]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Yi, Y.M.; Park, S.; Munster, C.; Kim, G.; Sung, K. Changes in ecological properties of petroleum oil-contaminated soil after low-temperature thermal desorption treatment. Water Air Soil Pollut. 2016, 227, 108. [Google Scholar] [CrossRef]
- Kang, S.S.; Roh, A.S.; Choi, S.C.; Kim, Y.S.; Kim, H.J.; Choi, M.T.; Ahn, B.K.; Kim, H.W.; Kim, H.K.; Park, J.H.; et al. Status and Changes in Chemical Properties of Paddy Soil in Korea. Korean J. Soil Sci. Fertil. 2012, 45, 968–972, (In Korean, with English abstract). [Google Scholar] [CrossRef]
- Kim, Y.H.; Kong, M.S.; Lee, E.J.; Lee, T.G.; Jung, G.B. Status and Changes in Chemical Properties of Upland Soil from 2001 to 2017 in Korea. Korean J. Environ. Agric. 2019, 38, 213–218, (In Korean, with English Abstract). [Google Scholar] [CrossRef]
- NIFoS (National Institute of Forest Science). Evaluation of the Forest Soil Properties Using Plant Response Methodology; National Institute of Forest Science: Seoul, Republic of Korea, 2018. (In Korean) [Google Scholar]
- Li, H.; Xu, H.; Zhou, S.; Yu, Y.; Li, H.; Zhou, C.; Chen, Y.; Li, Y.; Wang, M.; Wang, G. Distribution and transformation of lead in rice plants grown in contaminated soil amended with biochar and lime. Ecotoxicol. Environ. Saf. 2018, 165, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Aponte, H.; Meli, P.; Butler, B.; Paolini, J.; Matus, F.; Merino, C.; Cornejo, P.; Kuzyakov, Y. Meta-analysis of heavy metal effects on soil enzyme activities. Sci. Total Environ. 2020, 737, 139744. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Yu, L. Effects of Cd or/and Pb on soil enzyme activities and microbial community structure. Ecol. Eng. 2011, 37, 1889–1894. [Google Scholar] [CrossRef]
- Zhao, H.; Wu, L.; Zhu, S.; Sun, H.; Xu, C.; Fu, J.; Ning, T. Sensitivities of physical and chemical attributes of soil quality to different tillage management. Agronomy 2022, 12, 1153. [Google Scholar] [CrossRef]
- Tan, X.; Liu, Y.; Yan, K.; Wang, Z.; Lu, G.; He, Y.; He, W. Differences in the response of soil dehydrogenase activity to Cd contamination are determined by the different substrates used for its determination. Chemosphere 2017, 169, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Garau, G.; Castaldi, P.; Santona, L.; Deiana, P.; Melis, P. Influence of red mud, zeolite and lime on heavy metal immobilization, culturable heterotrophic microbial populations and enzyme activities in a contaminated soil. Geoderma 2007, 142, 47–57. [Google Scholar] [CrossRef]
- Oliveira, A.; Pampulha, M.E. Effects of long-term heavy metal contamination on soil microbial characteristics. J. Biosci. Sci. Bioeng. 2006, 102, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Dotaniya, M.L.; Pipalde, J.S. Soil enzymatic activities as influenced by lead and nickel concentrations in a Vertisol of Central India. Bull. Environ. Contam. Toxicol. 2018, 101, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Bååth, E.; Arnebrant, K. Growth rate and response of bacterial communities to pH in limed and ash treated forest soils. Soil Biol. Biochem. 1994, 26, 995–1001. [Google Scholar] [CrossRef]
- Deng, S.P.; Tabatabai, M.A. Effect of tillage and residue management on enzyme activities in soils: III. Phosphatases and arylsulfatase. Biol. Fertil. Soils 1997, 24, 141–146. [Google Scholar] [CrossRef]
- Chang, T.; Feng, G.; Paul, V.; Adeli, A.; Brooks, J.P.; Jenkins, J.N. Soil health assessment for different tillage and cropping systems to determine sustainable management practices in a humid region. Soil Tillage Res. 2023, 233, 105796. [Google Scholar] [CrossRef]
- Yu, P.; Liu, S.; Zhang, L.; Li, Q.; Zhou, D. Selecting the minimum data set and quantitative soil quality indexing of alkaline soils under different land uses in northeastern China. Sci. Total Environ. 2018, 616–617, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Dengiz, O. Soil quality index for paddy fields based on standard scoring functions and weight allocation method. Arch. Agron. Soil Sci. 2019, 66, 301–315. [Google Scholar] [CrossRef]
- Datta, A.; Gujre, N.; Gupta, D.; Agnihorti, R.; Mitra, S. Application of enzymes as a diagnostic tool for soils as affected by municipal solid wastes. J. Environ. Manag. 2021, 286, 112169. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.K.; Dubey, P.K.; Abhilash, P.C. Sustainable soil amendments for improving the soil quality, yield and nutrient content of Brassica juncea (L.) grown in different agroecological zones of eastern Uttar Pradesh, India. Soil Tillage Res. 2019, 195, 104418. [Google Scholar] [CrossRef]
- Vasu, D.; Singh, S.K.; Ray, S.K.; Duraisami, V.P.; Tiwary, P.; Chandran, P.; Nimkar, A.M.; Anantwar, S.G. Soil quality index (SQI) as a tool to evaluate crop productivity in semi-arid Deccan plateau, India. Geoderma 2016, 282, 70–79. [Google Scholar] [CrossRef]
- Juhos, K.; Czigány, S.; Madarász, B.; Ladányi, M. Interpretation of soil quality indicators for land suitability assessment—A multivariate approach for Central European arable soils. Ecol. Indic. 2019, 99, 261–272. [Google Scholar] [CrossRef]
- Lim, J.Y.; Bhuiyan, M.S.I.; Lee, S.B.; Lee, J.G.; Kim, P.J. Agricultural nitrogen and phosphorus balances of Korea and Japan: Highest nutrient surplus among OECD member countries. Environ. Pollut. 2021, 286, 117353. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Liu, G.; Huang, C.; Liu, Q. Soil quality assessment in Yellow River Delta: Establishing a minimum data set and fuzzy logic model. Geoderma 2019, 334, 82–89. [Google Scholar] [CrossRef]
- Chae, Y.; Cui, R.; Kim, S.W.; An, G.; Jeong, S.W.; An, Y.J. Exoenzyme activity in contaminated soils before and after soil washing: ß-glucosidase activity as a biological indicator of soil health. Ecotoxicol. Environ. Saf. 2017, 135, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhou, Q.; Huang, R.; Wu, K.; Li, Z. Contrasting impacts of mobilisation and immobilisation amendments on soil health and heavy metal transfer to food chain. Ecotoxicol. Environ. Saf. 2021, 209, 111836. [Google Scholar] [CrossRef] [PubMed]


| Properties | Soil Samples | ||||
|---|---|---|---|---|---|
| CA | RA | AG | FO | UP | |
| pH | 6.3 ± 0.8 a | 6.6 ± 0.8 a | 6.2 ± 0.9 a | 5.6 ± 1.0 b | 6.2 ± 1.3 a |
| EC (dS m−1) | 0.5 ± 0.2 a | 0.6 ± 0.9 a | 0.5 ± 0.3 a | 0.5 ± 0.3 a | 0.4 ± 0.4 a |
| CEC † (cmolc kg−1) | 18.3 ± 3.5 a | 12.2 ± 3.6 b | 11.9 ± 4.6 b | 13.3 ± 5.3 b | 9.0 ± 3.9 c |
| OM (g kg−1) | 44.0 ± 33.4 ab | 19.7 ± 15.0 c | 35.8 ± 17.3 b | 46.8 ± 29.6 a | 25.3 ± 13.8 c |
| Available P (mg kg−1) | 2062.5 ± 779.4 a | 267.1 ± 316.2 c | 487.6 ± 636.2 b | 104.3 ± 159.5 c | 96.2 ± 117.9 c |
| TN (%) | 0.14 ± 0.03 a | 0.11 ± 0.09 ab | 0.14 ± 0.13 a | 0.14 ± 0.12 a | 0.07 ± 0.07 b |
| BD (g cm−3) | 1.34 ± 0.09 b | 1.30 ± 0.20 b | 1.35 ± 0.16 b | 1.23 ± 0.23 c | 1.46 ± 0.17 a |
| Porosity (%) | 49.27 ± 3.3 b | 50.8 ± 7.4 ab | 49.3 ± 6.2 b | 53.5 ± 8.7 a | 45.2 ± 6.6 c |
| Water-stable aggregate (%) | 33.0 ± 5.6 b | 22.7 ± 13.4 c | 40.3 ± 20.6 b | 55.2 ± 22.6 a | 42.4 ± 22.0 b |
| Soil respiration (CO2 mg−1 kg−1 day−1) | 110.2 ± 58.5 a | 45.5 ± 26.9 c | 56.1 ± 31.4 bc | 71.0 ± 54.4 b | 57.2 ± 38.7 bc |
| Arylsulfatase (μmol PNP h−1 g−1) | 7.3 ± 3.9 a | 12.7 ± 22.5 a | 8.6 ± 9.0 a | 14.1 ± 17.9 a | 8.8 ± 11.9 a |
| Dehydrogenase (μg TPF g−1) | 10.6 ± 6.8 b | 11.2 ± 22.3 b | 23.3 ± 35.6 ab | 36.9 ± 50.0 a | 30.9 ± 38.6 ab |
| β-Glucosidase (μmol PNP h−1 g−1) | 2.7 ± 1.2 ab | 2.1 ± 2.4 b | 2.9 ± 1.9 ab | 3.5 ± 2.9 a | 2.3 ± 2.7 ab |
| Phosphatase (μmol PNP h−1 g−1) | 9.4 ± 4.0 c | 10.3 ± 6.3 c | 14.2 ± 7.5 b | 18.7 ± 7.6 a | 11.5 ± 7.4 bc |
| Urease (μg N g−1 2 h−1) | 81.8 ± 56.3 b | 78.2 ± 91.4 b | 121.4 ± 115.0 ab | 138.2 ± 96.3 a | 91.9 ± 106.7 ab |
| Bacterial colony-forming units (CFU × 106 g−1) | 1.9 ± 0.5 b | 23.2 ± 50.2 a | 9.9 ± 21.2 ab | 12.5 ± 28.2 ab | 18.9 ± 33.1 ab |
| Clay (%) | 18.2 ± 4.9 b | 25.3 ± 12.7 a | 16.1 ± 8.1 bc | 17.2 ± 7.2 b | 12.7 ± 5.4 c |
| Principal Component | PC 1 | PC 2 | PC 3 | PC 4 | PC 5 |
|---|---|---|---|---|---|
| Eigenvalue | 4.556 | 1.781 | 1.612 | 1.302 | 1.083 |
| Percent | 26.798 | 10.478 | 9.479 | 7.656 | 6.373 |
| Cumulative percent | 26.798 | 37.276 | 46.756 | 54.412 | 60.785 |
| Eigenvectors | |||||
| pH | −0.106 | 0.424 | 0.485 | 0.199 | 0.394 |
| EC | 0.307 | 0.215 | 0.008 | 0.207 | −0.072 |
| CEC † | 0.540 | 0.445 | 0.227 | 0.176 | 0.097 |
| OM | 0.696 | −0.223 | −0.100 | 0.328 | 0.091 |
| Available P | 0.202 | 0.269 | −0.265 | 0.700 | 0.053 |
| TN | 0.702 | 0.005 | −0.023 | 0.295 | −0.001 |
| BD | −0.692 | −0.282 | 0.489 | 0.305 | −0.164 |
| Porosity | 0.697 | 0.275 | −0.485 | −0.300 | 0.169 |
| Water-stable aggregate | 0.346 | −0.635 | −0.221 | −0.010 | 0.267 |
| Soil respiration | 0.639 | −0.073 | 0.350 | 0.180 | 0.018 |
| Arylsulfatase | 0.547 | 0.056 | 0.497 | −0.232 | 0.112 |
| Dehydrogenase | 0.426 | −0.278 | 0.551 | −0.207 | −0.071 |
| β-Glucosidase | 0.559 | 0.001 | 0.068 | −0.143 | −0.167 |
| Phosphatase | 0.659 | −0.262 | −0.082 | −0.098 | −0.127 |
| Urease | 0.625 | −0.149 | 0.177 | −0.070 | −0.194 |
| Bacterial colony-forming units | −0.128 | 0.047 | 0.074 | −0.249 | 0.753 |
| Clay | 0.106 | 0.706 | 0.034 | −0.308 | −0.331 |
| PC1 Variables | TN | OM | BD | Porosity | SR | PHA |
|---|---|---|---|---|---|---|
| Total nitrogen (TN) | 1 | - | - | - | - | - |
| Organic matter (OM) | 0.516 ** | 1 | - | - | - | - |
| Bulk density (BD) | −0.364 ** | −0.345 ** | 1 | - | - | - |
| Porosity | 0.368 ** | 0.351 ** | −0.996 ** | 1 | - | - |
| Soil respiration (SR) | 0.405 ** | 0.436 ** | −0.234 ** | 0.242 ** | 1 | - |
| Phosphatase (PHA) | 0.416 ** | 0.489 ** | −0.355 ** | 0.362 ** | 0.342 ** | 1 |
| PC2 Variables | Clay | WSA | ||||
| Clay | 1 | - | ||||
| Water-stable aggregate (WSA) | −0.358 ** | 1 | ||||
| PC3 Variables | DHA | ARS | ||||
| Dehydrogenase (DHA) | 1 | - | ||||
| Arylsulfatase (ARS) | 0.424 ** | 1 |
| Soil Health Indicators and Grouping | Soil Health Index (SHI) | Δ(SHI) Based on CA (%) ¶ | |||
|---|---|---|---|---|---|
| RA | CA | ||||
| MDS indicator | TN § (%) | >0.15 ‡ | 0.644 | 0.522 | 23.4 |
| <0.15 | 0.423 | 0.451 | −6.2 | ||
| Clay (%) | >24 | 0.476 | 0.474 | 0.3 | |
| 12~24 | 0.516 | 0.485 | 6.5 | ||
| <12 | 0.382 | 0.406 | −5.9 | ||
| Dehydrogenase (μg TPF g−1) | >15 | 0.589 | 0.510 | 15.5 | |
| <15 | 0.453 | 0.457 | −1.0 | ||
| Available P (mg kg−1) | >300 | 0.609 | 0.482 | 26.3 | |
| 50~300 | 0.550 | 0.419 | 31.2 | ||
| <50 | 0.453 | ND † | - | ||
| Bacterial colony-forming units (CFU × 106 g−1) | >1.6 | 0.480 | 0.466 | 3.0 | |
| <1.6 | 0.473 | 0.493 | −3.9 | ||
| Other chemical indicators | OM (g kg−1) | >30 | 0.565 | 0.484 | 16.7 |
| 15~30 | 0.546 | 0.481 | 13.5 | ||
| <15 | 0.395 | 0.406 | −2.8 | ||
| CEC (cmolc kg−1) | >15 | 0.526 | 0.493 | 27.7 | |
| <15 | 0.466 | 0.412 | 13.1 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, J.-H.; Lee, C.-G.; Park, B.-J.; Jeong, S.S.; Lee, Y.D.; Kirkham, M.B.; Kim, K.-R.; Yang, J.E.; Park, Y.-H.; Kim, S.C.; et al. Combining Soil Immobilization and Dressing Techniques for Sustaining the Health of Metal-Contaminated Arable Soils. Sustainability 2024, 16, 3227. https://doi.org/10.3390/su16083227
Yoon J-H, Lee C-G, Park B-J, Jeong SS, Lee YD, Kirkham MB, Kim K-R, Yang JE, Park Y-H, Kim SC, et al. Combining Soil Immobilization and Dressing Techniques for Sustaining the Health of Metal-Contaminated Arable Soils. Sustainability. 2024; 16(8):3227. https://doi.org/10.3390/su16083227
Chicago/Turabian StyleYoon, Jung-Hwan, Chan-Gyu Lee, Byung-Jun Park, Seok Soon Jeong, Young Don Lee, Mary Beth Kirkham, Kwon-Rae Kim, Jae E. Yang, Yong-Ha Park, Sung Chul Kim, and et al. 2024. "Combining Soil Immobilization and Dressing Techniques for Sustaining the Health of Metal-Contaminated Arable Soils" Sustainability 16, no. 8: 3227. https://doi.org/10.3390/su16083227
APA StyleYoon, J.-H., Lee, C.-G., Park, B.-J., Jeong, S. S., Lee, Y. D., Kirkham, M. B., Kim, K.-R., Yang, J. E., Park, Y.-H., Kim, S. C., & Kim, H. S. (2024). Combining Soil Immobilization and Dressing Techniques for Sustaining the Health of Metal-Contaminated Arable Soils. Sustainability, 16(8), 3227. https://doi.org/10.3390/su16083227






