Perspective: Contribution of Epstein–Barr virus (EBV) Reactivation to the Carcinogenicity of Nasopharyngeal Cancer Cells
Abstract
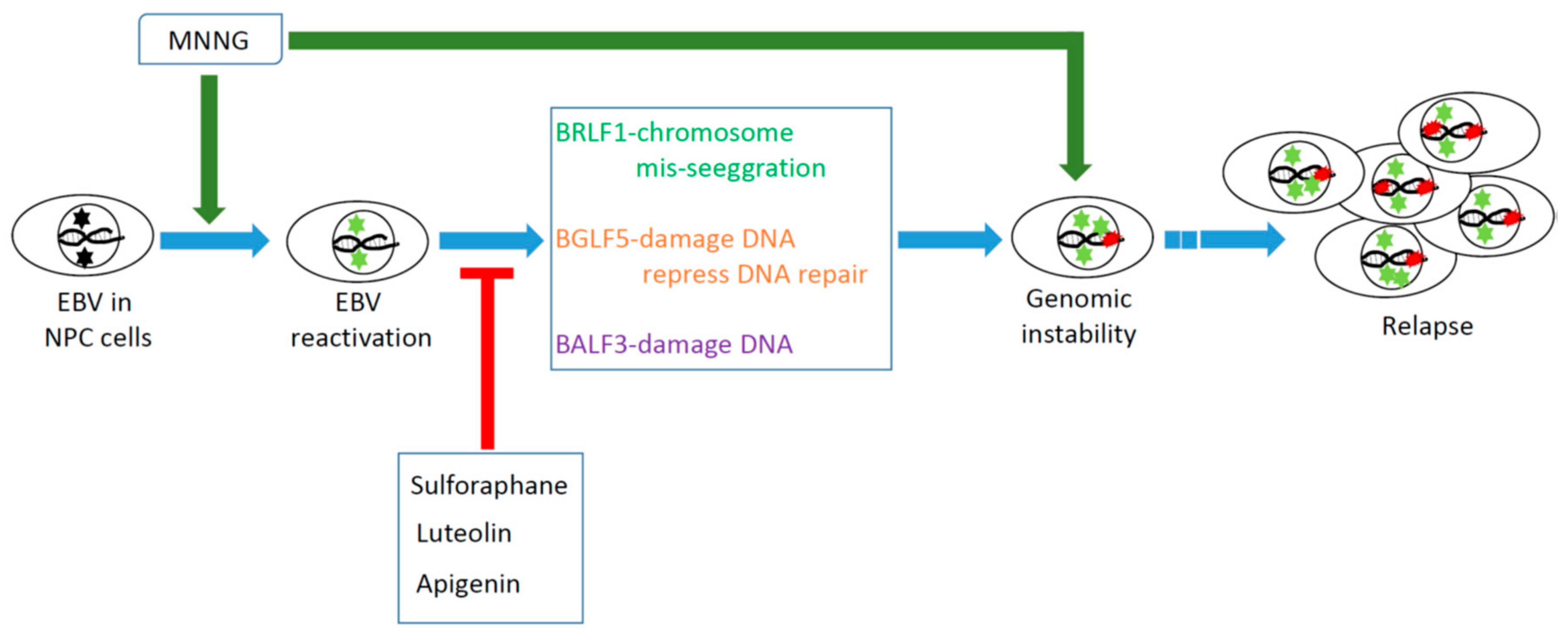
:1. Introduction
2. Reactivation of EBV Has a Significant Carcinogenic Effect on the Genomic Instability (GI) and Tumorigenesis of NPC Cells
3. BGLF5 is the Strongest Inducer of Micronuclei Formation and DNA Damage
4. BALF3 Mediates Genome Instability and Progressive Malignancy in NPC Cells
5. BRLF1 Induces Genomic Instability and Progressive Malignancy in NPC Cells
6. EBV Reactivation by Chemical Carcinogens May Contribute to the Carcinogenesis of NPC Cells
7. Inhibition of EBV Reactivation May Help Prevent the Malignant Progression of NPC Cells
8. Perspective
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hildesheim, A.; Levine, P.H. Etiology of nasopharyngeal carcinoma: A review. Epidemiol. Rev. 1993, 15, 466–485. [Google Scholar] [CrossRef] [PubMed]
- McDermott, A.L.; Dutt, S.N.; Watkinson, J.C. The aetiology of nasopharyngeal carcinoma. Clin. Otolaryngol. Allied Sci. 2001, 26, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.C.; Chen, J.Y.; Liu, M.Y.; Yang, H.I.; Hsu, M.M.; Chen, C.J.; Yang, C.S. Serologic markers of Epstein-Barr virus infection and nasopharyngeal carcinoma in Taiwanese men. N. Engl. J. Med. 2001, 345, 1877–1882. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.M.; Liu, Z.; Jia, W.H.; Huang, Q.H.; Liu, Q.; Guo, X.; Hong, M.H. Fluctuations of Epstein-Barr virus serological antibodies and risk for nasopharyngeal carcinoma: A prospective screening study with a 20-year follow-up. PLoS ONE 2011, 6, e19100. [Google Scholar] [CrossRef] [PubMed]
- Ji, M.F.; Wang, D.K.; Yu, Y.L.; Guo, Y.Q.; Liang, J.S.; Cheng, W.M.; Chua, D.T.T. Sustained elevation of Epstein-Barr virus antibody levels preceding clinical onset of nasopharyngeal carcinoma. Br. J. Cancer 2007, 96, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Young, L.S.; Rickinson, A.B. Epstein-Barr virus: 40 years on. Nat. Rev. 2004, 4, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Elgui de Oliveira, D.; Muller-Coan, B.G.; Pagano, J.S. Viral carcinogenesis beyond malignant transformation: EBV in the progression of human cancers. Trends Microbiol. 2016, 24, 649–664. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y. EBV based cancer prevention and therapy in nasopharyngeal carcinoma. Precis. Oncol. 2017, 1, 10. [Google Scholar] [CrossRef]
- Hong, G.K.; Gulley, M.L.; Feng, W.H.; Delecluse, H.J.; Holley-Guthrie, E.; Kenney, S.C. Epstein-Barr virus lytic infection contributes to lymphoproliferative disease in a SCID mouse model. J. Virol. 2005, 79, 13993–14003. [Google Scholar] [CrossRef] [PubMed]
- Stuart, A.D.; Stewart, J.P.; Arrand, J.R.; Mackett, M. The Epstein-Barr virus encoded cytokine viral interleukin-10 enhances transformation of human B lymphocytes. Oncogene 1995, 11, 1711–1719. [Google Scholar] [PubMed]
- Wang, Q.; Tsao, S.W.; Ooka, T.; Nicholls, J.M.; Cheung, H.W.; Fu, S.; Wang, X. Anti-apoptotic role of BARF1 in gastric cancer cells. Cancer Lett. 2006, 238, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, S.; Hu, J.; Luo, X.; Li, N.; Bode, A.M.; Cao, Y. Epstein-Barr virus lytic reactivation regulation and its pathogenic role in carcinogenesis. Int. J. Biol. Sci. 2016, 12, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Henle, W.; Ho, J.H.; Henle, G.; Chau, J.C.; Kwan, H.C. Nasopharyngeal carcinoma: Significance of changes in Epstein-Barr virus-related antibody patterns following therapy. Int. J. Cancer 1977, 20, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Heddle, J.A.; Cimino, M.C.; Hayashi, M.; Romagna, F.; Shelby, M.D.; Tucker, J.D.; MacGregor, J.T. Micronuclei as an index of cytogenetic damage: Past, present, and future. Environ. Mol. Mutagen. 1991, 18, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Tung, C.H.; Huang, Y.T.; Lu, J.; Chen, J.Y.; Tsai, C.H. Requirement for cell-to-cell contact in Epstein-Barr virus infection of nasopharyngeal carcinoma cells and keratinocytes. J. Virol. 1999, 73, 8857–8866. [Google Scholar] [PubMed]
- Lin, C.T.; Wong, C.I.; Chan, W.Y.; Tzung, K.W.; Ho, J.K.; Hsu, M.M.; Chuang, S.M. Establishment and characterization of two nasopharyngeal carcinoma cell lines. Lab. Investig. J. Tech. Methods Pathol. 1990, 62, 713–724. [Google Scholar]
- Fang, C.Y.; Lee, C.H.; Wu, C.C.; Chang, Y.T.; Yu, S.L.; Chou, S.P.; Tsai, C.H. Recurrent chemical reactivations of EBV promotes genome instability and enhances tumor progression of nasopharyngeal carcinoma cells. Int. J. Cancer 2009, 124, 2016–2025. [Google Scholar] [CrossRef] [PubMed]
- Rickinson, A.B.; Kieff, E. Epstein-Barr Virus, 4th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2001. [Google Scholar]
- Wu, C.C.; Liu, M.T.; Chang, Y.T.; Fang, C.Y.; Chou, S.P.; Liao, H.W.; Chen, Y.L. Epstein-Barr virus DNase (BGLF5) induces genomic instability in human epithelial cells. Nucleic Acids Res. 2010, 38, 1932–1949. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.H.; Wu, M.C.; Wu, C.C.; Chen, Y.C.; Lin, S.F.; Hsu, J.T.A.; Chen, J.Y. Epstein-Barr virus BALF3 has nuclease activity and mediates mature virion production during the lytic cycle. J. Virol. 2014, 88, 4962–4975. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.H.; Wu, C.C.; Fang, C.Y.; Yu, S.L.; Hsu, H.Y.; Chow, Y.H.; Chen, J.Y. Epstein-Barr virus BALF3 mediates genomic instability and progressive malignancy in nasopharyngeal carcinoma. Oncotarget 2014, 5, 8583–8601. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y.; Wu, C.C.; Cheng, Y.J.; Chou, S.P.; Jiang, Y.J.; Chu, K.C.; Chen, J.Y. Epstein-Barr virus BRLF1 induces genomic instability and progressive malignancy in nasopharyngeal carcinoma cells. Oncotarget 2017, 8, 78948–78964. [Google Scholar] [CrossRef] [PubMed]
- Mirvish, S.S. Role of N-nitroso compounds (NOC) and N-nitrosation in etiology of gastric, esophageal, nasopharyngeal and bladder cancer and contribution to cancer of known exposures to NOC. Cancer Lett. 1995, 93, 17–48. [Google Scholar] [CrossRef]
- Huang, S.Y.; Fang, C.Y.; Wu, C.C.; Tsai, C.H.; Lin, S.F.; Chen, J.Y. Reactive oxygen species mediate Epstein-Barr virus reactivation by N-methyl-N′-nitro-N-nitrosoguanidine. PLoS ONE 2013, 8, e84919. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y.; Fang, C.Y.; Tsai, C.H.; Chang, Y.; Takada, K.; Hsu, T.Y.; Chen, J.Y. N-methyl-N′-nitro-N-nitrosoguanidine induces and cooperates with 12-O-tetradecanoylphorbol-1,3-acetate/sodium butyrate to enhance Epstein-Barr virus reactivation and genome instability in nasopharyngeal carcinoma cells. Chem. Biol. Interact. 2010, 188, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.Y.; Huang, S.Y.; Wu, C.C.; Hsu, H.Y.; Chou, S.P.; Tsai, C.H.; Chen, J.Y. The synergistic effect of chemical carcinogens enhances Epstein-Barr virus reactivation and tumor progression of nasopharyngeal carcinoma cells. PLoS ONE 2012, 7, e44810. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Chuang, H.Y.; Lin, C.Y.; Chen, Y.J.; Tsai, W.H.; Fang, C.Y.; Chen, J.Y. Inhibition of Epstein-Barr virus reactivation in nasopharyngeal carcinoma cells by dietary sulforaphane. Mol. Carcinog. 2013, 52, 946–958. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Fang, C.Y.; Hsu, H.Y.; Chen, Y.J.; Chou, S.P.; Huang, S.Y.; Chen, J.Y. Luteolin inhibits Epstein-Barr virus lytic reactivation by repressing the promoter activities of immediate-early genes. Antivir. Res. 2016, 132, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Fang, C.Y.; Cheng, Y.J.; Hsu, H.Y.; Chou, S.P.; Huang, S.Y.; Chen, J.Y. Inhibition of Epstein-Barr virus reactivation by the flavonoid apigenin. J. Biomed. Sci. 2017, 24, 2. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Fang, C.Y.; Hsu, H.Y.; Chuang, H.Y.; Cheng, Y.J.; Chen, Y.J.; Tsai, C.H. EBV reactivation as a target of luteolin to repress NPC tumorigenesis. Oncotarget 2016, 7, 18999–19017. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-C.; Fang, C.-Y.; Huang, S.-Y.; Chiu, S.-H.; Lee, C.-H.; Chen, J.-Y. Perspective: Contribution of Epstein–Barr virus (EBV) Reactivation to the Carcinogenicity of Nasopharyngeal Cancer Cells. Cancers 2018, 10, 120. https://doi.org/10.3390/cancers10040120
Wu C-C, Fang C-Y, Huang S-Y, Chiu S-H, Lee C-H, Chen J-Y. Perspective: Contribution of Epstein–Barr virus (EBV) Reactivation to the Carcinogenicity of Nasopharyngeal Cancer Cells. Cancers. 2018; 10(4):120. https://doi.org/10.3390/cancers10040120
Chicago/Turabian StyleWu, Chung-Chun, Chih-Yeu Fang, Sheng-Yen Huang, Shih-Hsin Chiu, Chia-Huei Lee, and Jen-Yang Chen. 2018. "Perspective: Contribution of Epstein–Barr virus (EBV) Reactivation to the Carcinogenicity of Nasopharyngeal Cancer Cells" Cancers 10, no. 4: 120. https://doi.org/10.3390/cancers10040120




