Microgastrinae (Hymenoptera: Braconidae) in the Forest State of Artikutza (Navarra: Spain): Diversity and Community Structure
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Area
2.2. Sampling Design
- M-1: On the edge of a clearing.
- M-2: In open forest.
- M-3: Under a mature beech tree in an area mostly populated with pine trees.
- H-1: Under a large beech tree.
- H-2: Near a small clearing and surrounded by a copse of beech shrubs.
- H-3: Under a shade of a mature beech tree on a landing.
| Trap | Hill orientation | North Pole orientation | Axis trap orientation | Altitude | Hill gradient |
|---|---|---|---|---|---|
| M-1 | NE-SW | N230E | N-S | 611 m | 12° |
| M-2 | NE-SW | N210E | N-S | 631 m | 15° |
| M-3 | NE-SW | N216E | N-S | 652 m | 20° |
| H-1 | NW-SW | N235E | N-S | 576 m | 19° |
| H-2 | NW-SW | N210E | N-S | 595 m | 18° |
| H-3 | NW-SW | N242E | N-S | 620 m | 9° |
2.3. Diversity and Community Structure
3. Results and Discussion
| Species | Trap number | Total Specimens | |||||||
|---|---|---|---|---|---|---|---|---|---|
| M-1 | M-2 | M-3 | H-1 | H-2 | H-3 | ΣM | ΣH | Total | |
| Choeras dorsalis | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Choeras hedymeles | 5 | 10 | 18 | 10 | 10 | 6 | 33 | 26 | 59 |
| Choeras parasitellae | 3 | 0 | 0 | 0 | 0 | 1 | 3 | 1 | 4 |
| Cotesia ancilla | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Cotesia chares | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Cotesia jucundus | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Cotesia sp1 | 4 | 0 | 0 | 0 | 0 | 0 | 4 | 0 | 4 |
| Dolichogenidea celsus | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 |
| Dolichogenidea laevigatoides | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 2 | 2 |
| Dolichogenidea laevigatus | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 2 | 2 |
| Dolichogenidea sp1 | 3 | 3 | 5 | 2 | 2 | 0 | 11 | 4 | 15 |
| Dolichogenidea sp2 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Dolichogenidea varifemur | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 2 | 2 |
| Glyptapanteles aliphera | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Glyptapanteles fulvipes | 2 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 |
| Glyptapanteles mygdonia | 0 | 1 | 3 | 1 | 0 | 4 | 4 | 5 | 9 |
| Glyptapanteles porthetriae | 2 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 |
| Glyptapanteles vitripennis | 4 | 9 | 4 | 1 | 2 | 1 | 17 | 4 | 21 |
| Microgaster sp1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Microplitis marshalii | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Paroplitis macrocephalus | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Paroplitis wesmaeli | 1 | 0 | 0 | 0 | 1 | 2 | 1 | 3 | 4 |
| Pholetesor circumscriptus | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Pholetesor sp1 | 3 | 0 | 1 | 0 | 1 | 1 | 4 | 2 | 6 |
| Pholetesor sp2 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Protapanteles anchisiades | 5 | 2 | 2 | 0 | 0 | 0 | 9 | 0 | 9 |
| Protapanteles sp1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Total specimens | 37 | 28 | 37 | 18 | 18 | 16 | 102 | 52 | 154 |
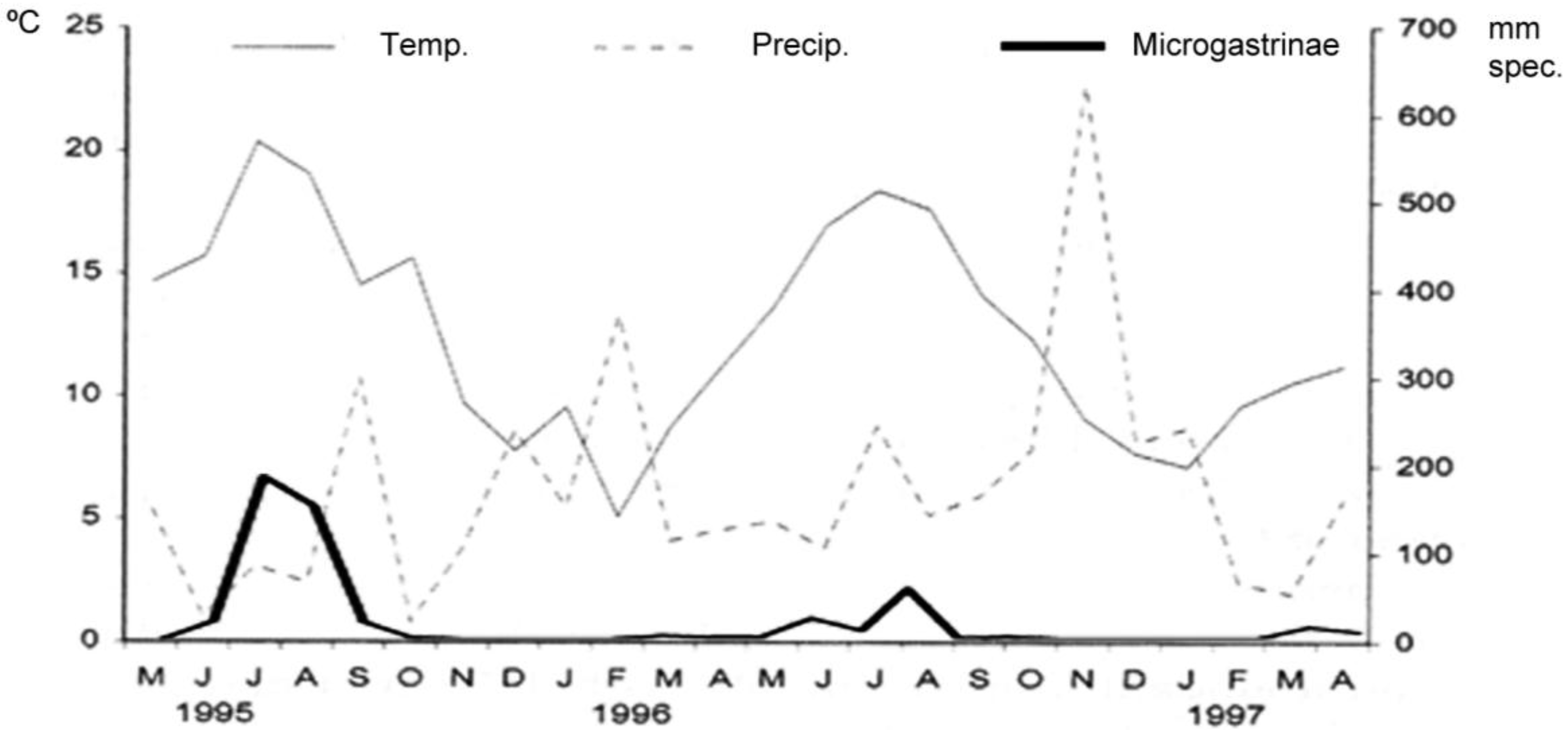
3.1. Alpha Diversity
| Data | Trap number | Total | ||||||
|---|---|---|---|---|---|---|---|---|
| M-1 | M-2 | M-3 | H-1 | H-2 | H-3 | ΣM | ΣH | |
| Species | 15 | 8 | 10 | 7 | 7 | 7 | 23 | 11 |
| Specimens | 37 | 28 | 37 | 18 | 18 | 16 | 102 | 52 |
| Shannon I. | 2.537 | 1.636 | 1.711 | 1.453 | 1.453 | 1.667 | 2.346 | 2.174 |
| Simpson I. | 0.090 | 0.252 | 0.280 | 0.345 | 0.345 | 0.234 | 0.159 | 0.281 |
| Margalef I. | 3.877 | 2.101 | 2.492 | 2.076 | 2.076 | 2.164 | 4.760 | 2.530 |
3.2. Beta Diversity
| M-1 | M-2 | M-3 | H-1 | H-2 | H-3 | ||
| M-1 | 0.211 | 0.250 | 0.158 | 0.294 | 0.294 | ||
| M-2 | 0.789 | 0.385 | 0.364 | 0.250 | 0.250 | ||
| M-3 | 0.750 | 0.615 | 0.308 | 0.308 | 0.308 | Jaccard I. | |
| H-1 | 0.842 | 0.636 | 0.818 | 0.400 | 0.273 | ||
| H-2 | 0.706 | 0.750 | 0.818 | 0.600 | 0.556 | ||
| H-3 | 0.706 | 0.750 | 0.818 | 0.545 | 0.444 | ||
| Complementarity | |||||||
3.3. Gamma Diversity
3.4. Community Structure
| Log-series | Log-normal | Broken-stick | ||||
|---|---|---|---|---|---|---|
| Class | Exp freq. | Obs freq. | Exp freq. | Obs freq. | Exp freq. | Obs freq. |
| 0 | - | - | 3.41 | 0 | - | - |
| 1 | 12.35 | 15 | 16.93 | 15 | 7.63 | 15 |
| 2 | 4.01 | 4 | 4.01 | 4 | 4.79 | 4 |
| 3 | 3.41 | 0 | 1.65 | 0 | 5.12 | 0 |
| 4 | 2.19 | 2 | 0.36 | 2 | 2.91 | 2 |
| 5 | 0.87 | 1 | 0.04 | 1 | 0.47 | 1 |
| 6 | 0.15 | 1 | 0 | 1 | 0.01 | 1 |
| χ2 = 8.963 | P = 0.111 | χ2 = 1031.5 | P = 1.399·10−219 | χ2 = 211.26 | P = 1.104·10−43 | |
| Log-series | Log-normal | Broken-stick | ||||
|---|---|---|---|---|---|---|
| Class | Exp freq. | Obs freq. | Exp freq. | Obs freq. | Exp freq. | Obs freq. |
| 0 | - | - | 0.19 | 0 | - | - |
| 1 | 5.78 | 6 | 6.38 | 6 | 3.26 | 6 |
| 2 | 1.90 | 3 | 3.05 | 3 | 2.27 | 3 |
| 3 | 1.66 | 1 | 1.31 | 1 | 8.46 | 1 |
| 4 | 1.11 | 0 | 0.24 | 0 | 16.92 | 0 |
| 5 | 0.478 | 1 | 0.02 | 1 | 33.85 | 1 |
| χ2 = 5.188 | P = 0.269 | χ2 =56.343 | P = 6.91·10−11 | χ2 = 57.91 | P = 7.96·10−12 | |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Shaw, M.R.; Hochberg, M.E. The neglect of Parasitic Hymenoptera in insect conservation strategies: The British fauna as a prime example. J. Insect Conservat. 2001, 5, 253–263. [Google Scholar] [CrossRef]
- Shaw, M.R.; Huddleston, T. Classification and biology of Braconidae wasps (Hymenoptera: Braconidae). In Hand-Books for the Identification of British Insects; Royal Entomology Society of London: London, UK, 1991; Volume 7, pp. 1–126. [Google Scholar]
- Martínez de Murguía, L. La taxocenosis de Hymenoptera en Artikutza (Navarra). Boletín de la SEA 2002, 31, 227–337. (in Spanish). [Google Scholar]
- Martínez de Murgía, L.; Vázquez, M.A.; Nieves-Aldrey, J.L. The families of Hymenoptera (Insecta) in an heterogeneous acidofilous forest in Artikutza (Navarra, Spain). Frustula Entomol. 2001, 24, 81–98. [Google Scholar]
- Samways, M.J. Insect Conservation Biology; Chapman & Hall: London, UK, 1994; p. 358. [Google Scholar]
- Sharkey, M.J. Hymenoptera of the world. An identification guide to families. Agric. Can. Publ. 1993, 3, 362–395. [Google Scholar]
- Scatolini, D.; Penteado-Dias, A.M. A fauna de Braconidae (Hymenoptera) como bioindicaroda do grau de preservação de duas localidades do Estado do Paraná. Rev. Bras. Ecol. 1997, 1, 84–87. (in Portuguese). [Google Scholar]
- González, J.A.; Ruíz, D.B. Los bracónidos (Hymenoptera: Braconidae) como grupo parámetro de biodiversidad en las selvas deciduas del trópico: Una discusión acerca de su posible uso. Acta Zool. Mexic. 2000, 79, 43–56. (in Spanish). [Google Scholar]
- Matthews, W. Biology of Braconidae. Ann. Rev. Ent. 1974, 19, 15–32. [Google Scholar] [CrossRef]
- Lasalle, J. Parasitic Hymenoptera, Biological Control and Diversity; C.A.B. International: Wallingford, UK, 1993; p. 348. [Google Scholar]
- Torenzan-Silingardi, H.M. Predatory behavior of Pachodynerus brevithorax (Hymenoptera: Vespidae, Eumeninae) on endophytic herbivore beetles in the Brazilian tropic savanna. Sociobiology 2011, 57, 181–189. [Google Scholar]
- Triplehorn, C.A.; Johnson, N.F. Borror and Delong’s Introduction to the Study of Insects; Thompson Brooks: San Francisco, CA, USA, 2005. [Google Scholar]
- Wharton, R.A.; Marsh, P.M.; Sharkey, M.J. Manual of the New World Genera in the Family Braconidae (Hymenoptera); The International Society of Hymenopterists: Washington, DC, USA, 1997; p. 439. [Google Scholar]
- Elpino-Campos, A.; Del-Claro, K.; Prezoto, F. Diversity of social wasps (Hymenoptera: Vespidae) in Cerrado fragments of Uberlandia, Minas Gerais state, Brazil. Neotrop. Entomol. 2007, 36, 685–692. [Google Scholar] [CrossRef]
- Whitfield, J.B.; Mardulyn, P.; Austin, A.D.; Dowton, M. Phylogenetic analysis of relationships among microgastrine braconid wasp genera based on data from the 16S, COI and 28S genes and morphology. Syst. Entomol. 2002, 27, 337–359. [Google Scholar] [CrossRef]
- Rodriguez, J.J.; Fernández-Triana, J.L.; Smith, M.A.; Janzen, D.H.; Hallwachs, W.; Erwin, T.L.; Whitfield, J.B. Extrapolations from field studies and known faunas converge on dramatically increased estimates of global microgastrine parasitoid wasp species richness (Hymenoptera: Braconidae). Insect Conserv. Diver. 2013, 6, 530–536. [Google Scholar] [CrossRef]
- Alex Smith, M.; Fernández-Triana, J.L.; Eveleigh, E.; Gómez, J.; Guclu, C.; Hallwachs, W.; Hebert, P.D.N.; Hrcek, J.; Huber, J.T.; Janzen, D.; et al. DNA barcoding and the taxonomy of Microgastrinae wasps (Hymenoptera, Braconidae): Impacts after eight years and nearly 20,000 sequences. Mol. Ecol. Resour. 2012, 13, 168–176. [Google Scholar]
- Fernández-Flores, S.; Fernández-Triana, J.L.; Martínez, J.J.; Zaldívar-Riverón, A. DNA barcoding species inventory of Microgastrinae wasps (Hymenoptera, Braconidae) from a Mexican tropical dry forest. Mol. Ecol. Resour. 2013, in press. [Google Scholar]
- Stahlhut, J.K.; Fernández-Triana, J.; Adamowicz, S.J.; Buck, M.; Goulet, H.; Hebert, P.D.N.; Huber, J.T.; Merilo, M.T.; Sheffield, C.S.; Woodcock, T.; et al. DNA barcoding reveals diversity of Hymenoptera and the dominance of parasitoids in a sub-Arctic environment. BMC Ecol. 2013, 13, 2. [Google Scholar] [CrossRef]
- Whitfield, J.B. Subfamily Microgastrinae. In Manual of the New World Genera of the Family Braconidae (Hymenoptera); The International Society of Hymenopterists: Washington, DC, USA, 1997; pp. 339–371. [Google Scholar]
- Cirelli, K.R.; Penteado-Dias, A.M. Fenologia dos Braconidae (Hymenoptera: Ichneumonidea) da Área de Proteçao Ambiental (APA) de Descalvado, SP. Rev. Bras. Entomol. 2003, 47, 99–105. (in Portuguese). [Google Scholar] [CrossRef]
- Briceño, R.; Torres, D.; Zaldívar-Riverón, A. Primer reporte de la familia Braconidae (Hymenoptera: Ichneumonoidea) en los parques Cerro Saroche y Terepaima, Estado Lara, Venezuela. Bioagro 2009, 21, 223–226. (in Spanish). [Google Scholar]
- Nieves-Aldrey, J.L.; Del Castillo, C. Ensayo preliminary sobre la captura de insectos por medio de una trampa Malaise en la Sierra de Guadarrama (España) con especial referencia a los himenópteros (Insecta, Hymenoptera). Ecología 1991, 5, 383–403. (in Spanish). [Google Scholar]
- Pujade-Villar, J. Resultados preliminaries obtenidos a partir de una trampa Malaise situada en una zona mediterránea pirenaica. Pirineos 1996, 147–148, 61–80. (in Spanish). [Google Scholar] [CrossRef]
- Segade, C.; Ros-Farré, P.; Algarra, A.; Ventura, D.; Pujade-Villar, J. Estudio comparativo de las capturas de realizadas con trampa Malaise en Andorra con especial atención a los himenópteros (Hymenoptera). Zapateri 1997, 7, 71–82. (in Spanish). [Google Scholar]
- Ros-Farré, P.; Pujade-Villar, J. Estudio mediante una trampa Malaise de la comunidad de cinípedos cecidógenos e inquilinos de Santa Coloma, Andorra (Hymenoptera, Cynipidae). Ecología 1998, 12, 441–454. (in Spanish). [Google Scholar]
- González, J.A.; Tomé, M.A.M.; Gayubo, S.F.; Torres, F. Himenópteros aculeados capturados mediante trampas Malaise en un sector arenoso de la Submeseta Norte (España) (Hymenoptera, Aculeata). Nouv. Rev. Entomol. 2000, 17, 337–353. (in Spanish). [Google Scholar]
- Tomé, M.A.; González, J.A.; Gayubo, S.F.; Torres, F. Estudio comparative sobre la eficacia de captura de insectos (Arthtopoda, Hexapoda) mediante trampas Malaise, en un biotopo arenoso de la submeseta Norte (España). Boletín Real Soc. Española Hist. Nat. Sección Biol. 2001, 96, 231–241. (in Spanish). [Google Scholar]
- Falcó-Garí, J.V.; Oltra-Moscardó, M.T.; Moreno-Marí, J.; Pujade-Villar, J.; Jiménez-Peydró, R. Fenología de los bracónidos (Hymenoptera, Braconidae) del Pirineo andorrano. Pirineos 2006, 191, 111–132. (in Spanish). [Google Scholar]
- Jiménez-Peydró, R.; Peris-Felipo, F.J. Diversity and community structure of Opiinae (Hymenoptera: Braconidae) in the forest estate of Artikutza (Spain). Fla. Entomol. 2011, 94, 472–479. [Google Scholar] [CrossRef]
- Peris-Felipo, F.J.; Jiménez-Peydró, R. Biodiversity within the subfamily Alysiinae (Hymenoptera, Braconidae) in the Natural Park Peñas de Aya (Spain). Rev. Bras. Entomol. 2011, 55, 406–410. [Google Scholar] [CrossRef]
- Mason, W.R.M. The polyphyletic nature of Apanteles Forster (Hymenoptera: Braconidae): A phylogeny and reclassification of Microgastrinae. Mem. Entomol. Soc. Can. 1981, 115, 1–147. [Google Scholar] [CrossRef]
- Papp, J. New Apanteles Föerster species from Hungary (Hymenoptera: Braconidae). An. Hist. Nat. Mus. Hung. 1972, 64, 335–345. [Google Scholar]
- Papp, J. Three new Apanteles Föerster species forma Hungary (Hymenoptera: Braconidae). An. Hist. Mus. Nat. Hung. 1973, 65, 287–304. [Google Scholar]
- Papp, J. New Apanteles Föerster species from Hungary (Hymenoptera: Braconidae) III. Ann. Hist. Nat. Mus. Nat. Hung. 1974, 66, 325–337. [Google Scholar]
- Papp, J. On the classification of the species Apanteles Foerster with special respect to the species living in Hungary (Hymenoptera: Braconidae). Állattani Közlemények 1974, 1, 86–100. [Google Scholar]
- Papp, J. A study on the Systematics of Braconidae. Folia Ento. Hungary 1974, 27, 109–133. [Google Scholar]
- Papp, J. A survey of the European Species of Apanteles Foerster. (Hymenoptera: Braconidae: Microgastrinae), I. The species group. Ann. Hist. Nat. Mus. Nat. Hung. 1976, 68, 251–274. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster (Hymenoptera: Braconidae: Microgastrinae), II. The laevigatus-groups, 1. Ann. Hist. Nat. Hung. 1978, 70, 265–301. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster (Hymenoptera: Braconidae: Microgastrinae), III. The laevigatus-groups, 2. Ann. Hist. Nat. Hung. 1979, 71, 235–250. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster. (Hymenoptera: Braconidae: Microgastrinae), IV. The lineipes-, obscurus-, and ater-groups. Ann. Hist. Nat. Hung. 1980, 72, 241–272. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster (Hymenoptera: Braconidae: Microgastrinae), V. The lacteus-, longipalpis-, ultor-, butalidis-, and vipio- groups. Ann. Hist. Nat. Hung. 1981, 73, 263–291. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster (Hymenoptera: Braconidae: Microgastrinae), VI. The laspeyresiella-, merula-, falcatus-, and validus- groups. Ann. Hist. Nat. Hung. 1982, 74, 255–267. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Föerster (Hymenoptera: Braconidae: Microgastrinae), VIII. The metacarpalis-, formosus-, popularis-, and suevus-groups. Ann. Hist. Nat. Mus. Nat. Hung. 1984, 76, 265–295. [Google Scholar]
- Papp, J. Contributions to the Braconid fauna of Hungary. VI. Microgastrinae (Hymenoptera: Braconidae). Fol. Ent. Hung. 1984, 2, 157–168. [Google Scholar]
- Papp, J. Paleartic species of Microgaster Latreille (Microplitis Föerster) with description of seven new species (Hymenptera: Braconidae: Microgastrinae). Entomol. Abh. 1984, 47, 95–140. [Google Scholar]
- Papp, J. A survey of the European species of Apanteles Foerster (Hymenoptera: Braconidae: Microgastrinae) IX. The glomeratus-groups, 1. Ann. Hist. Nat. Mus. Nat. Hung. 1986, 78, 225–247. [Google Scholar]
- Tobias, V.I.; Jakimavicius, A.B.; Kiriya, I.G. Family Braconidae. In Keys to the Insects of the European Part if the USSR; III. Part V; Medvedev, G.S, Ed.; Nauca Publisher: Leningrad, USSR, 1986; p. 507, (In Russian, English translation in 1995). [Google Scholar]
- Moreno, C. Métodos para medir la biodiversidad; (in Spanish). Sociedad Entomológica Aragonesa: Zaragoza, Spain, 2001; p. 84, (MGT-Manuales y Tesis SEA). [Google Scholar]
- Magurran, A.E. Ecological Diversity and Its Measurement; Chapman and Hall: London, UK, 1991; p. 179. [Google Scholar]
- Villarreal, H.; Alvarez, H.M.; Córdoba, S.; Escobar, F.; Fagua, G.; Gast, F.; Mendoza, H.; Ospina, M.; Umaña, A.M. Insectos. In Manual de métodos para el desarrollo de inventarios de biodiversidad; (in Spanish). Instituto de Investigación de Recursos Biológicos Alexander von Humboldt: Bogotá, Colombia, 2004; pp. 149–184. [Google Scholar]
- Schluter, D.; Ricklefs, R.E. Species diversity: An introduction to the problem. In Species Diversity in Ecological Communities; Ricklefs, R.E., Schluter, D., Eds.; Univ. Chicago Press: Chicago, IL, USA, 1993; pp. 1–10. [Google Scholar]
- Soares, S.A.; Antonialli-Junior, W.F.; Lima-Juniors, S.E. Diversidade de formigas epigéicas (Hymenoptera, Formicidae) em dois ambientes no Centro-Oeste do Brasil. Rev. Bras. Entomol. 2010, 54, 76–81. (in Portuguese). [Google Scholar] [CrossRef]
- Zar, J.H. Biostadistical Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 1999; p. 663. [Google Scholar]
- Benavent-Corai, J.; Arregui-Roig, M.; Oltra-Moscardó, M.T.; Jiménez-Peydró, R. Evolución anual de la comunidade de bracónidos en el Parque Natural de la Font Roja (Hymenoptera: Braconidae). Iberis 2005, 3, 23–32. (in Spanish). [Google Scholar]
- Smith, M.A.; Rodríguez, J.J.; Whitfield, J.B.; Deans, A.R.; Janzen, D.H.; Hallwachs, W.; Hebert, P.D.N. Extreme diversity of tropical parasitoid wasps exposed by iterative integration of natural history, DNA barcoding, morphology, and collections. Proc. Natl. Acad. Sci. USA 2008, 105, 12359–12364. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pérez-Rodríguez, J.; Oltra-Moscardó, T.; Peris-Felipo, F.J.; Jiménez-Peydró, R. Microgastrinae (Hymenoptera: Braconidae) in the Forest State of Artikutza (Navarra: Spain): Diversity and Community Structure. Insects 2013, 4, 493-505. https://doi.org/10.3390/insects4030493
Pérez-Rodríguez J, Oltra-Moscardó T, Peris-Felipo FJ, Jiménez-Peydró R. Microgastrinae (Hymenoptera: Braconidae) in the Forest State of Artikutza (Navarra: Spain): Diversity and Community Structure. Insects. 2013; 4(3):493-505. https://doi.org/10.3390/insects4030493
Chicago/Turabian StylePérez-Rodríguez, Jesica, Teresa Oltra-Moscardó, Francisco Javier Peris-Felipo, and Ricardo Jiménez-Peydró. 2013. "Microgastrinae (Hymenoptera: Braconidae) in the Forest State of Artikutza (Navarra: Spain): Diversity and Community Structure" Insects 4, no. 3: 493-505. https://doi.org/10.3390/insects4030493
APA StylePérez-Rodríguez, J., Oltra-Moscardó, T., Peris-Felipo, F. J., & Jiménez-Peydró, R. (2013). Microgastrinae (Hymenoptera: Braconidae) in the Forest State of Artikutza (Navarra: Spain): Diversity and Community Structure. Insects, 4(3), 493-505. https://doi.org/10.3390/insects4030493




