Influence of the Sonication Temperature on the Debundling Kinetics of Carbon Nanotubes in Propan-2-ol
Abstract
:1. Introduction
2. Experimental Details
3. Results
3.1. Effect of Powerful Horn Sonication on CNT Integrity and Debundling



3.2. Analysis of Bundling During Sonication
| Peak number from Figure 4 | Peak wave length (cm−1) |
|---|---|
| 1 (propan-2-ol) | 199 |
| CNT Peak 1 | 225 |
| CNT Peak 2 | 277 |
| CNT Peak 3 | 284 |

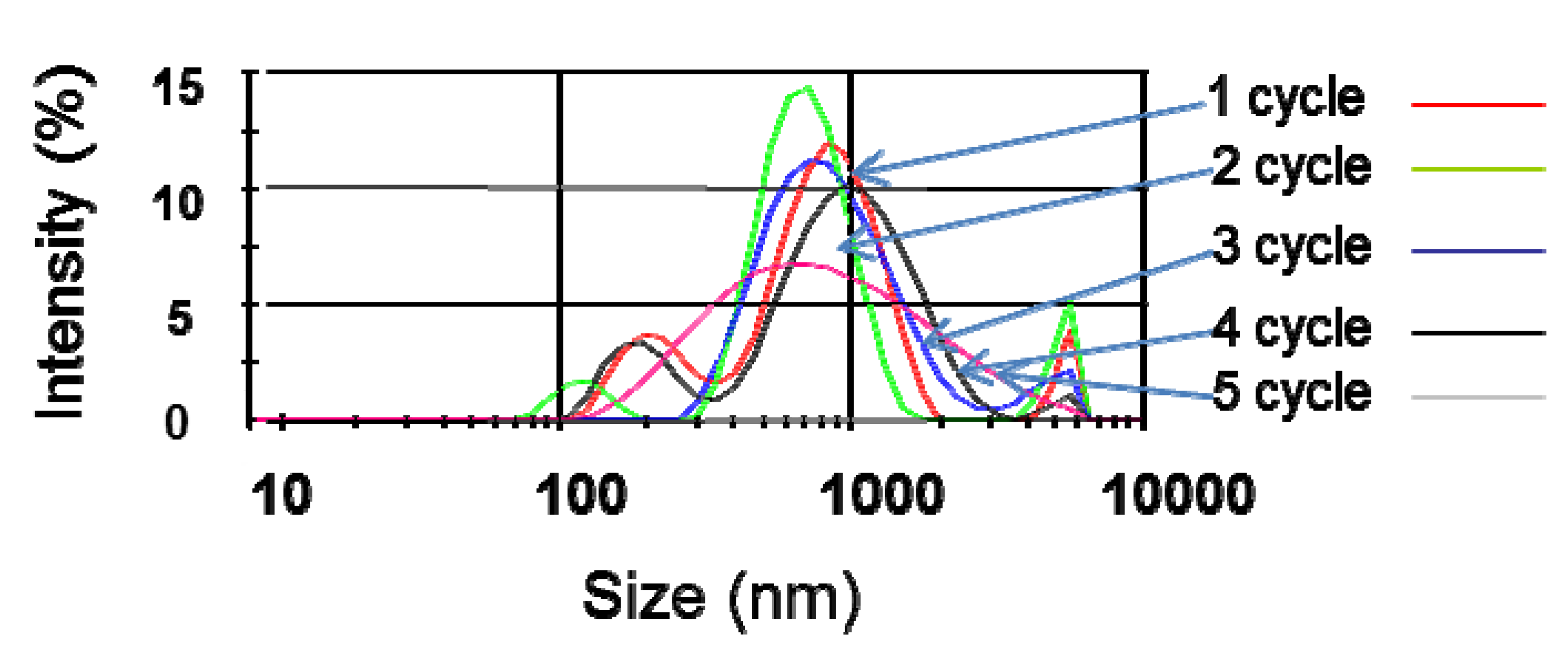
3.3. Stability and Dispersability of the Suspensions After Sonication at Higher Temperatures

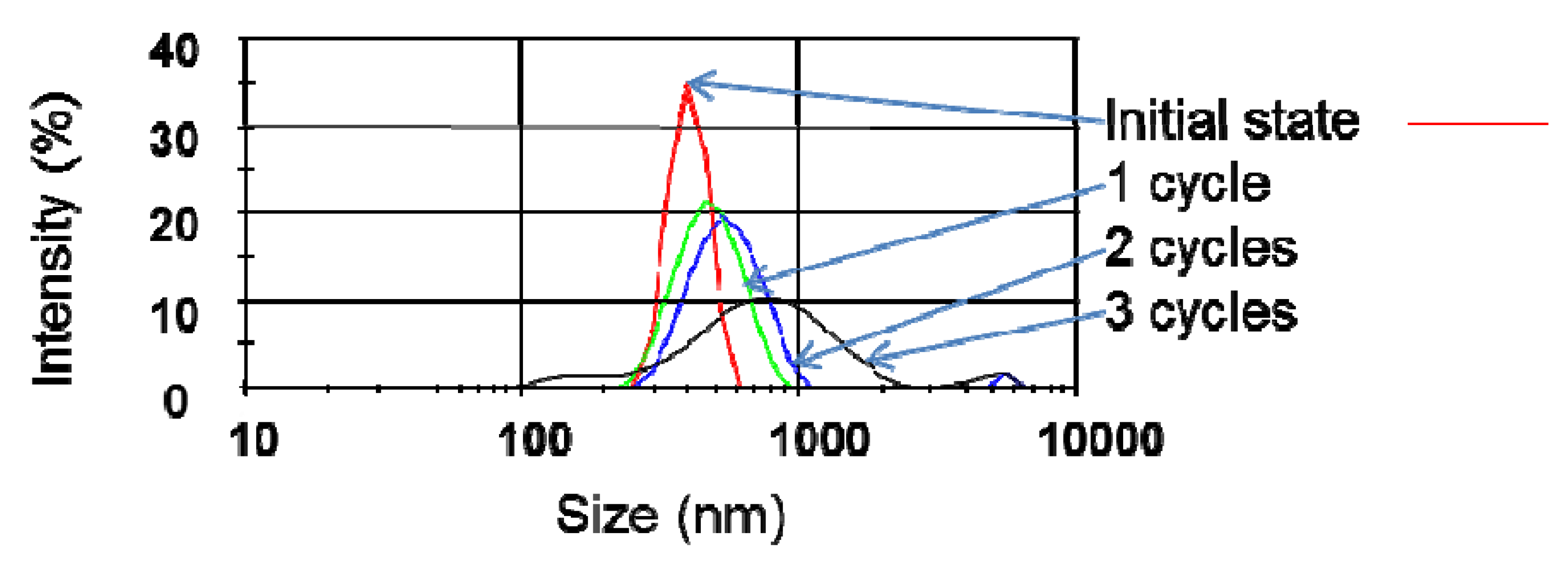

4. Discussion
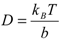
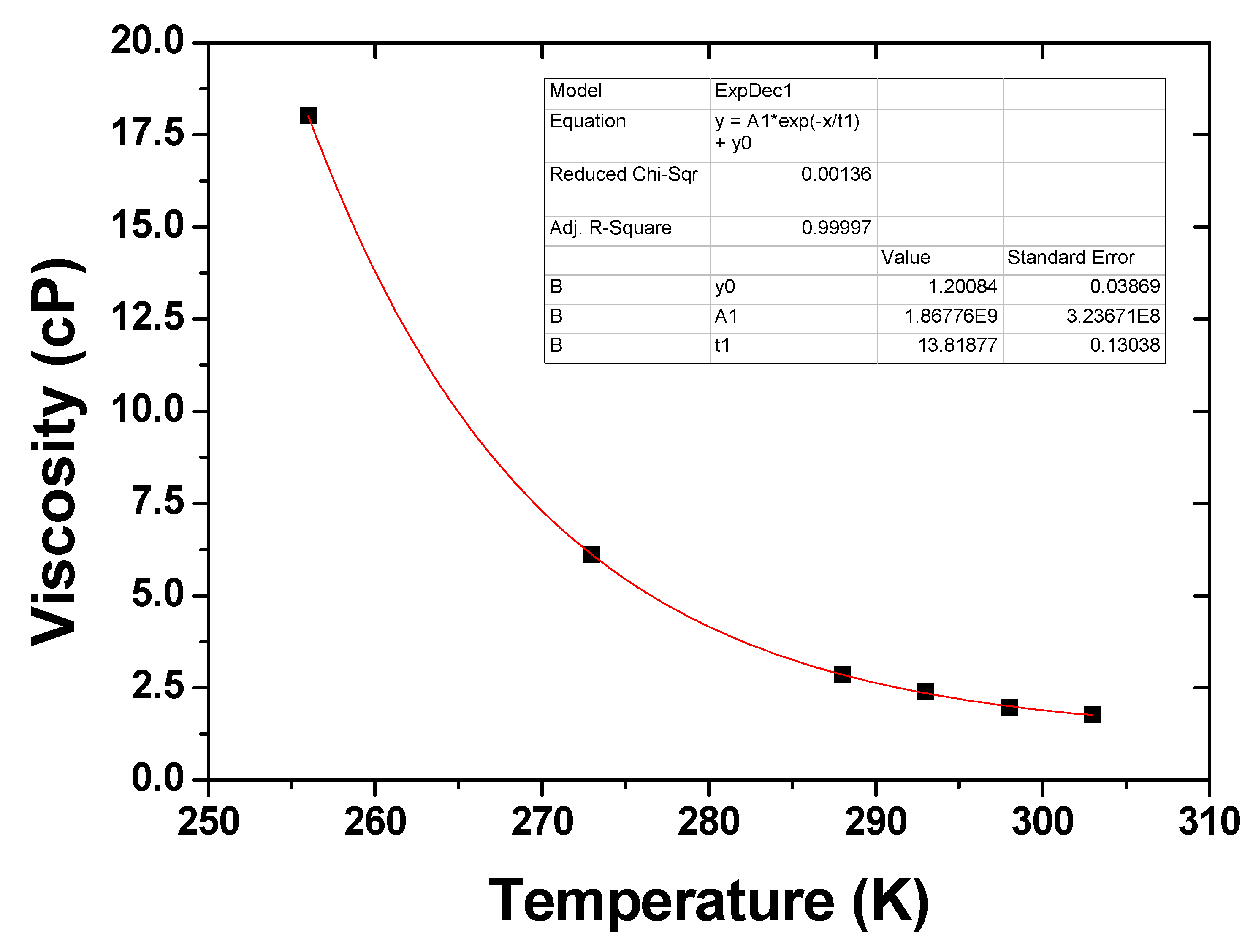
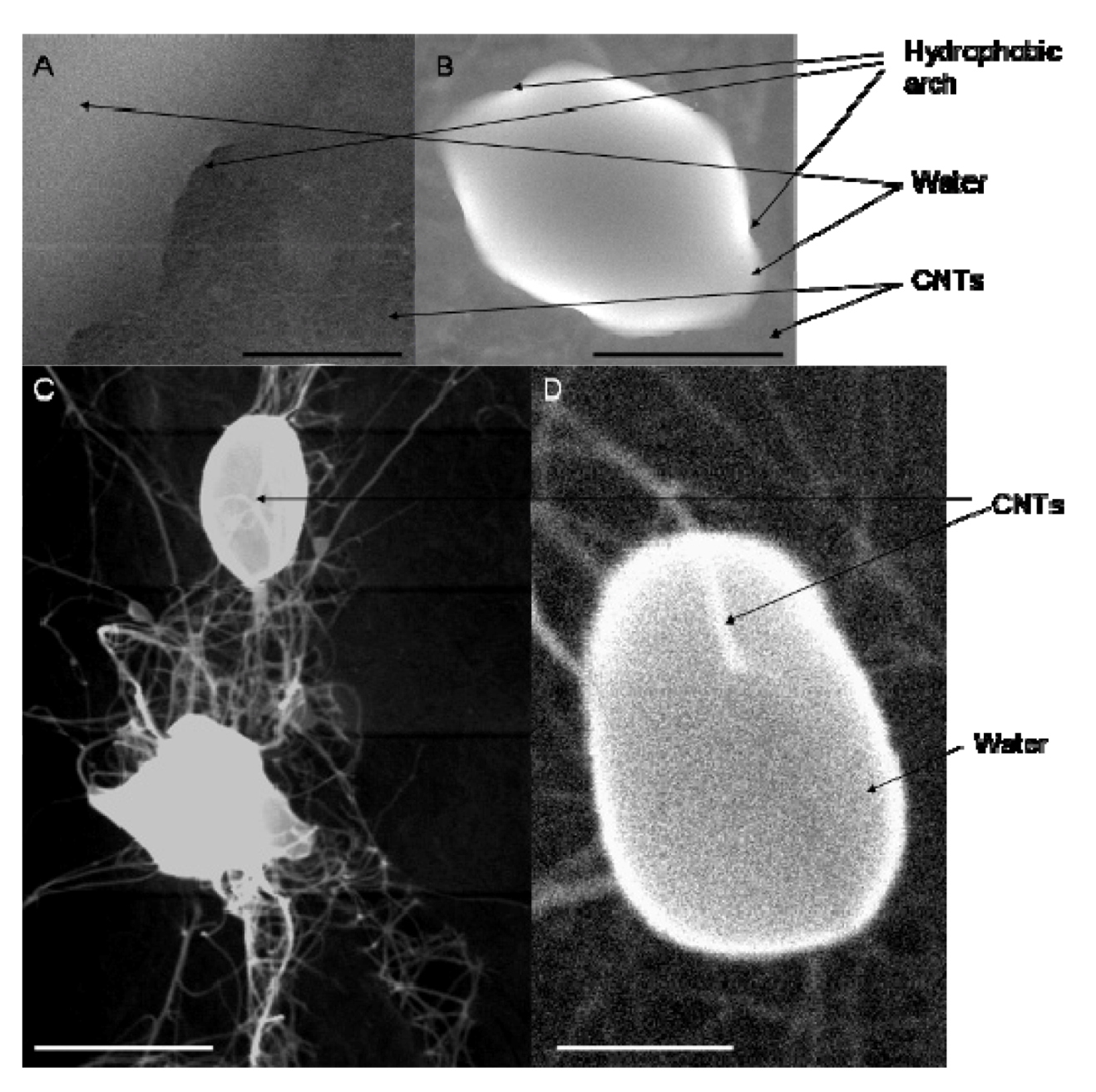
5. Conclusions
Acknowledgements
References
- Rosca, I.D.; Hoa, S.V. Method for reducing contact resistivity of carbon nanotube-containing epoxy adhesives for aerospace applications. Compos. Sci. Technol. 2011, 71, 95–100. [Google Scholar] [CrossRef]
- Yu, S.; Tong, M.N.; Critchlow, G. Use of carbon nanotubes reinforced epoxy as adhesives to join aluminum plates. Mater. Des. 2010, 31, S126–S129. [Google Scholar] [CrossRef] [Green Version]
- Pei, X.; Wang, J.; Wan, Q.; Kang, L.; Xiao, M.; Bao, H. Functionally graded carbon nanotubes/hydroxyapatite composite coating by laser cladding. Surf. Coat. Technol. 2011, 205, 4380–4387. [Google Scholar] [CrossRef]
- Chen, W.X.; Tu, J.P.; Gan, H.Y.; Xu, Z.D.; Wang, Q.G.; Lee, J.Y.; Liu, Z.L.; Zhang, X.B. Electroless preparation and tribological properties of ni-p-carbon nanotube composite coatings under lubricated condition. Surf. Coat. Technol. 2002, 160, 68–73. [Google Scholar] [CrossRef]
- Chen, W.X.; Tu, J.P.; Wang, L.Y.; Gan, H.Y.; Xu, Z.D.; Zhang, X.B. Tribological application of carbon nanotubes in a metal-based composite coating and composites. Carbon 2003, 41, 215–222. [Google Scholar] [CrossRef]
- Majumder, M.; Keis, K.; Zhan, X.; Meadows, C.; Cole, J.; Hinds, B.J. Enhanced electrostatic modulation of ionic diffusion through carbon nanotube membranes by diazonium grafting chemistry. J. Membr. Sci. 2008, 316, 89–96. [Google Scholar] [CrossRef]
- Dumée, L.F.; Sears, K.; Schütz, J.; Finn, N.; Huynh, C.; Hawkins, S.; Duke, M.; Gray, S. Characterization and evaluation of carbon nanotube bucky-paper membranes for direct contact membrane distillation. J. Membr. Sci. 2010, 351, 36–43. [Google Scholar] [CrossRef] [Green Version]
- Dumée, L.; Germain, V.; Sears, K.; Schütz, J.; Finn, N.; Duke, M.; Cerneaux, S.; Cornu, D.; Gray, S. Enhanced durability and hydrophobicity of carbon nanotube bucky paper membranes in membrane distillation. J. Membr. Sci. 2011, 376, 241–246. [Google Scholar] [CrossRef]
- Kim, M.; Park, Y.-B.; Okoli, O.I.; Zhang, C. Processing, characterization, and modeling of carbon nanotube-reinforced multiscale composites. Compos. Sci. Technol. 2009, 69, 335–342. [Google Scholar] [CrossRef]
- Khan, U.; Ryan, K.; Blau, W.J.; Coleman, J.N. The effect of solvent choice on the mechanical properties of carbon nanotube-polymer composites. Compos. Sci. Technol. 2007, 67, 3158–3167. [Google Scholar] [CrossRef]
- Dumee, L.; Hill, M.R.; Duke, M.; Velleman, L.; Sears, K.; Schutz, J.; Finn, N.; Gray, S. Activation of gold decorated carbon nanotube hybrids for targeted gas adsorption and enhanced catalytic oxidation. J. Mater. Chem. 2012, 22, 9374–9378. [Google Scholar]
- Cola, B.A.; Xu, J.; Fisher, T.S. Contact mechanics and thermal conductance of carbon nanotube array interfaces. Int. J. Heat Mass Transfer 2009, 52, 3490–3503. [Google Scholar] [CrossRef]
- Han, Z.; Fina, A. Thermal conductivity of carbon nanotubes and their polymer nanocomposites: A review. Prog. Polym. Sci. 2011, 36, 914–944. [Google Scholar]
- Spitalsky, Z.; Tasis, D.; Papagelis, K.; Galiotis, C. Carbon nanotube-polymer composites: Chemistry, processing, mechanical and electrical properties. Prog. Polym. Sci. 2010, 35, 357–401. [Google Scholar] [CrossRef]
- Köhler, A.R.; Som, C.; Helland, A.; Gottschalk, F. Studying the potential release of carbon nanotubes throughout the application life cycle. J. Clean. Product. 2008, 16, 927–937. [Google Scholar] [CrossRef]
- Bose, K.; Wood, R.J.K. Influence of load and speed on rolling micro-abrasion of cvd diamond and other hard coatings. Diam. Relat. Mater. 2003, 12, 753–756. [Google Scholar] [CrossRef]
- Kasaliwal, G.R.; Pegel, S.; Göldel, A.; Pötschke, P.; Heinrich, G. Analysis of agglomerate dispersion mechanisms of multiwalled carbon nanotubes during melt mixing in polycarbonate. Polymer 2010, 51, 2708–2720. [Google Scholar] [CrossRef]
- Ham, H.T.; Choi, Y.S.; Chung, I.J. An explanation of dispersion states of single-walled carbon nanotubes in solvents and aqueous surfactant solutions using solubility parameters. J. Colloid Interface Sci. 2005, 286, 216–223. [Google Scholar]
- Ma, P.-C.; Siddiqui, N.A.; Marom, G.; Kim, J.-K. Dispersion and functionalization of carbon nanotubes for polymer-based nanocomposites: A review. Compos. A 2010, 41, 1345–1367. [Google Scholar] [CrossRef]
- Xie, X.-L.; Mai, Y.-W.; Zhou, X.-P. Dispersion and alignment of carbon nanotubes in polymer matrix: A review. Mater. Sci. Eng. 2005, 49, 89–112. [Google Scholar] [CrossRef]
- Blagov, E.V.; Klimchitskaya, G.L.; Mostepanenko, V.M. Van der waals interaction between a microparticle and a single-walled carbon nanotube. Phys. Rev. B 2007, 75, 235413. [Google Scholar]
- Parsegian, V.A. Van der Waals Forces: A Handbook for Biologists, Chemists, Engineers, and Physicists; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Wei, C.; Srivastava, D.; Cho, K. Thermal expansion and diffusion coefficients of carbon nanotube-polymer composites. Nano Lett. 2002, 2, 647–650. [Google Scholar] [CrossRef]
- Hilding, J.; Grulke, E.A.; Zhang, Z.G.; Lockwood, F. Dispersion of carbon nanotubes in liquids. J. Dispers. Sci. Technol. 2003, 24, 1–41. [Google Scholar] [CrossRef]
- Cheng, Q.; Debnath, S.; Gregan, E.; Byrne, H.J. Effect of solvent solubility parameters on the dispersion of single-walled carbon nanotubes. J. Phys. Chem. C 2008, 112, 20154–20158. [Google Scholar]
- Gou, J.H. Single-walled nanotube bucky paper and nanocomposite. Polym. Int. 2006, 55, 1283–1288. [Google Scholar] [CrossRef]
- Lee, J.U.; Huh, J.; Kim, K.H.; Park, C.; Jo, W.H. Aqueous suspension of carbon nanotubes via non-covalent functionalization with oligothiophene-terminated poly(ethylene glycol). Carbon 2007, 45, 1051–1057. [Google Scholar] [CrossRef]
- Shaffer, M.S.P.; Fan, X.; Windle, A.H. Dispersion and packing of carbon nanotubes. Carbon 1998, 36, 1603–1612. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, L.; Sun, J.; Liu, Y.; Zheng, S.; Kajiura, H.; Li, Y.; Noda, K. An integrated route for purification, cutting and dispersion of single-walled carbon nanotubes. Chem. Phys. Lett. 2006, 432, 205–208. [Google Scholar] [CrossRef]
- Yu, J.; Grossiord, N.; Koning, C.E.; Loos, J. Controlling the dispersion of multi-wall carbon nanotubes in aqueous surfactant solution. Carbon 2007, 45, 618–623. [Google Scholar] [CrossRef]
- Vaisman, L.; Wagner, H.D.; Marom, G. The role of surfactants in dispersion of carbon nanotubes. Adv. Colloid Interface Sci. 2006, 128–130, 37–46. [Google Scholar] [CrossRef]
- Kernan, D.M.; Blau, W.J. Exploring the mechanisms of carbon-nanotube dispersion aggregation in a highly polar solvent. Europhys. Lett. 2008, 83, 66009. [Google Scholar] [CrossRef]
- Hui, C.; Harish, M.; Paul, S.; Jianhua, Z.; Xiong, L.; Jinhai, W.; Qun, H.; Saiful, I.K.; Lei, Z. Dispersion of carbon nanotubes and polymer nanocomposite fabrication using trifluoroacetic acid as a co-solvent. Nanotechnology 2007, 18, 415606. [Google Scholar] [CrossRef]
- Sun, Z.; Nicolosi, V.; Rickard, D.; Bergin, S.D.; Aherne, D.; Coleman, J.N. Quantitative evaluation of surfactant-stabilised single-walled carbon nanotubes:Dispersion quality and its correlation with zeta potential. J. Phys. Chem. C 2008, 112, 10692–10699. [Google Scholar] [CrossRef]
- Lin, T.; Bajapi, V.; Ji, T.; Dai, L. Chemistry of carbon nanotubes. Aust. J. Chem. 2003, 56, 635–651. [Google Scholar] [CrossRef]
- Priya, B.R.; Byrne, H.J. Investigation of sodium dodecyl benzene sulfonate assisted dispersion and debundling of single-walled carbon nanotubes. J. Phys. Chem. B 2008, 112, 332–337. [Google Scholar] [CrossRef]
- Nish, A.; Hwang, J.-J.; Doig, J.; Nicholas, R.J. Highly selective dispersion of single-walled carbon nanotubes using aromatic polymers. Nature 2007, 2, 640–646. [Google Scholar]
- Zheng, M.; Jagota, A.; Semke, E.D.; Diner, B.A.; Mclean, R.S.; Lustig, S.R.; Richardson, R.E.; Tassis, N.G. DNA-assisted dispersion and separation of carbon nanotubes. Nature 2003, 2, 338–342. [Google Scholar]
- Dumée, L.; Campbell, J.L.; Sears, K.; Schütz, J.; Finn, N.; Duke, M.; Gray, S. The impact of hydrophobic coating on the performance of carbon nanotube bucky-paper membranes in membrane distillation. Desalination 2011, 283, 64–67. [Google Scholar] [CrossRef]
- Dumée, L.F.; Gray, S.; Duke, M.; Sears, K.; Schütz, J.; Finn, N. The role of membrane surface energy on direct contact membrane distillation performance. Desalination 2013, in press.. [Google Scholar]
- Hou, P.-X.; Liu, C.; Cheng, H.-M. Purification of carbon nanotubes. Carbon 2008, 46, 2003–2025. [Google Scholar] [CrossRef]
- Park, T.-J.; Banerjee, S.; Hemraj-Benny, T.; Wong, S.S. Purification strategies and purity visualization for single-walled carbon nanotubes. J. Mater. Chem. 2006, 16, 141–154. [Google Scholar]
- Etika, K.C.; Jochum, F.D.; Theato, P.; Grunlan, J.C. Temperature controlled dispersion of carbon nanotubes in water with pyrene-functionalized poly(n-cyclopropylacrylamide). J. Am. Chem. Soc. 2009, 131, 13598–13599. [Google Scholar]
- Etika, K.C.; Cox, M.A.; Grunlan, J.C. Tailored dispersion of carbon nanotubes in water with ph-responsive polymers. Polymer 2010, 51, 1761–1770. [Google Scholar]
- Huynh, C.P.; Hawkins, S.C. Understanding the synthesis of directly spinnable carbon nanotube forests. Carbon 2010, 48, 1105–1115. [Google Scholar]
- Zhbanov, A.I.; Pogorelov, E.G.; Chang, Y.-C. Van der waals interaction between two crossed carbon nanotubes. ACS Nano 2010, 4, 5937–5945. [Google Scholar]
- Zarkova, L.; Hohm, U. Effective (n-6) lennard-jones potentials with temperature-dependent parameters introduced for accurate calculation of equilibrium and transport properties of ethene, propene, butene, and cyclopropane. J. Chem. Eng. Data 2009, 54, 1648–1655. [Google Scholar] [CrossRef]
- Jones, J.E. On the determination of molecular fields. II. From the equation of state of a gas. Proc. R. Soc. Lond. A 1924, 106, 463–477. [Google Scholar] [CrossRef]
- Esawi, A.M.K.; Farag, M.M. Carbon nanotube reinforced composites: Potential and current challenges. Mater. Des. 2007, 28, 2394–2401. [Google Scholar]
- O’Connell, M.J. Carbon Nanotubes: Properties and Applications; Taylor & Francis Group: Boca Raton, NW, USA, 2006. [Google Scholar]
- Agrawal, S.; Raghuveer, M.S.; Li, H.; Ramanath, G. Defect-induced electrical conductivity increase in individual multiwalled carbon nanotubes. Appl. Phys. Lett. 2007, 90, 193104–193103. [Google Scholar]
- Bernholc, J.; Brenner, D.; Buongiorno Nardelli, M.; Meunier, V.; Roland, C. Mechanical and electrical properties of nanotubes. Annu. Rev. Mater. Res. 2002, 32, 28. [Google Scholar]
- Rance, G.A.; Marsh, D.H.; Bourne, S.J.; Reade, T.J.; Khlobystov, A.N. Van der waals interactions between nanotubes and nanoparticles for controlled assembly of composite nanostructures. ACS Nano 2010, 4, 4920–4928. [Google Scholar]
- Rotkin, S.V.; Hess, K. Many-body terms in van der waals cohesion energy of nanotubes. J. Comput. Electron. 2002, 1, 323–326. [Google Scholar] [CrossRef]
- Lu, W.B.; Liu, B.; Wu, J.; Xiao, J.; Hwang, K.C.; Fu, S.Y.; Huang, Y. Continuum modeling of van der waals interactions between carbon nanotube walls. Appl. Phys. Lett. 2009, 94, 101917–101913. [Google Scholar]
- Urbina, A.; Miguel, C.; Delgado, J.L.; Langa, F.; Díaz-Paniagua, C.; Jiménez, M.; Batallán, F. Dynamics of functionalized single wall carbon nanotubes in solution studied by incoherent neutron scattering experiments. J. Phys. 2008, 20, 104208. [Google Scholar]
- Zheng, L.; Li, S.; Brody, J.P.; Burke, P.J. Manipulating nanoparticles in solution with electrically contacted nanotubes using dielectrophoresis. Langmuir 2004, 20, 8612–8619. [Google Scholar] [CrossRef]
- Pauzauskie, P.J.; Jamshidi, A.; Valley, J.K.; Satcher, J.J.H.; Wu, M.C. Parallel trapping of multiwalled carbon nanotubes with optoelectronic tweezers. Appl. Phys. Lett. 2009, 95, 113104–113103. [Google Scholar]
- Rossi, M.P.; Ye, H.; Gogotsi, Y.; Babu, S.; Ndungu, P.; Bradley, J.-C. Environmental scanning electron microscopy study of water in carbon nanopipes. Nano Lett. 2004, 4, 989–993. [Google Scholar]
- Checco, A.; Hofmann, T.; DiMasi, E.; Black, C.T.; Ocko, B.M. Morphology of air nanobubbles trapped at hydrophobic nanopatterned surfaces. Nano Lett. 2010, 10, 1354–1358. [Google Scholar] [CrossRef]
- Papastavrou, G.; Akari, S.; Möhwald, H. Interactions between hydrophilic and hydrophobic surfaces on microscopic scale and the influence of air bubbles as observed by scanning force microscopy in aqueous and alcoholic mediums. Europhys. Lett. 2000, 52, 551. [Google Scholar] [CrossRef]
- Krasowska, M.; Zawala, J.; Malysa, K. Air at hydrophobic surfaces and kinetics of three phase contact formation. Adv. Colloid Interface Sci. 2009, 147–148, 155–169. [Google Scholar] [CrossRef]
- Yoon, R.H.; Yordan, J.L. The critical rupture thickness of thin water films on hydrophobic surfaces. J. Colloid Interface Sci. 1991, 146, 565–572. [Google Scholar] [CrossRef]
- Pushkarova, R.A.; Horn, R.G. Surface forces measured between an air bubble and a solid surface in water. Colloids Surf. A 2005, 261, 147–152. [Google Scholar] [CrossRef]
- Dumée, L.F.; Sears, K.; Marmiroli, B.; Amenitsch, H.; Duan, X.; Lamb, R.; Buso, D.; Huynh, C.; Hawkins, S.; Kentish, S.; et al. A high volume and low damage route to hydroxyl functionalization of carbon nanotubes using hard X-ray lithography. Carbon 2013, 51, 430–434. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dumée, L.; Sears, K.; Schütz, J.; Finn, N.; Duke, M.; Gray, S. Influence of the Sonication Temperature on the Debundling Kinetics of Carbon Nanotubes in Propan-2-ol. Nanomaterials 2013, 3, 70-85. https://doi.org/10.3390/nano3010070
Dumée L, Sears K, Schütz J, Finn N, Duke M, Gray S. Influence of the Sonication Temperature on the Debundling Kinetics of Carbon Nanotubes in Propan-2-ol. Nanomaterials. 2013; 3(1):70-85. https://doi.org/10.3390/nano3010070
Chicago/Turabian StyleDumée, Ludovic, Kallista Sears, Jürg Schütz, Niall Finn, Mikel Duke, and Stephen Gray. 2013. "Influence of the Sonication Temperature on the Debundling Kinetics of Carbon Nanotubes in Propan-2-ol" Nanomaterials 3, no. 1: 70-85. https://doi.org/10.3390/nano3010070







