Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields
Abstract
:1. Introduction
- food and cosmetic packaging
- solar energy, especially organic photovoltaics
- automotive structural parts
2. Processing
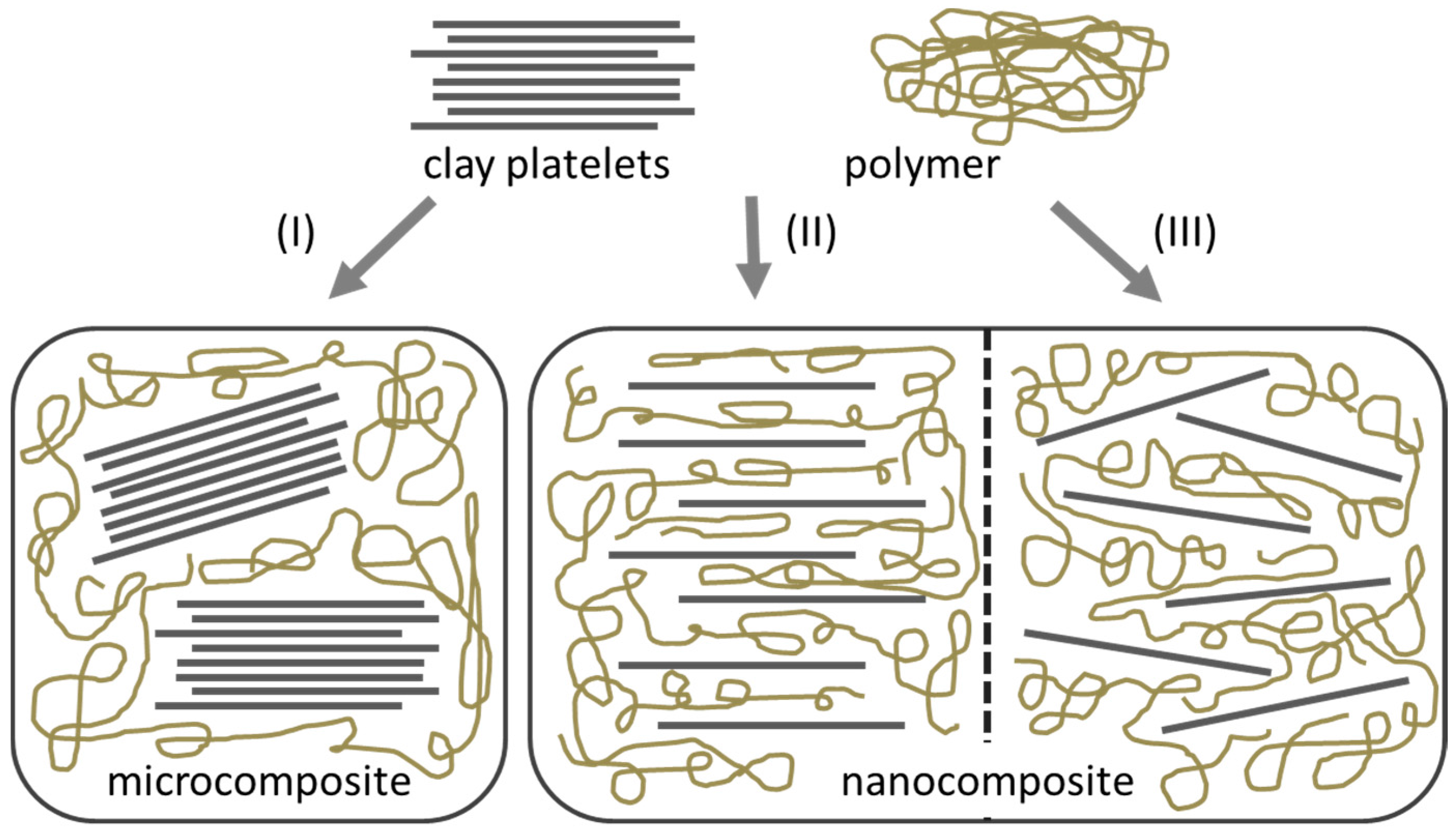
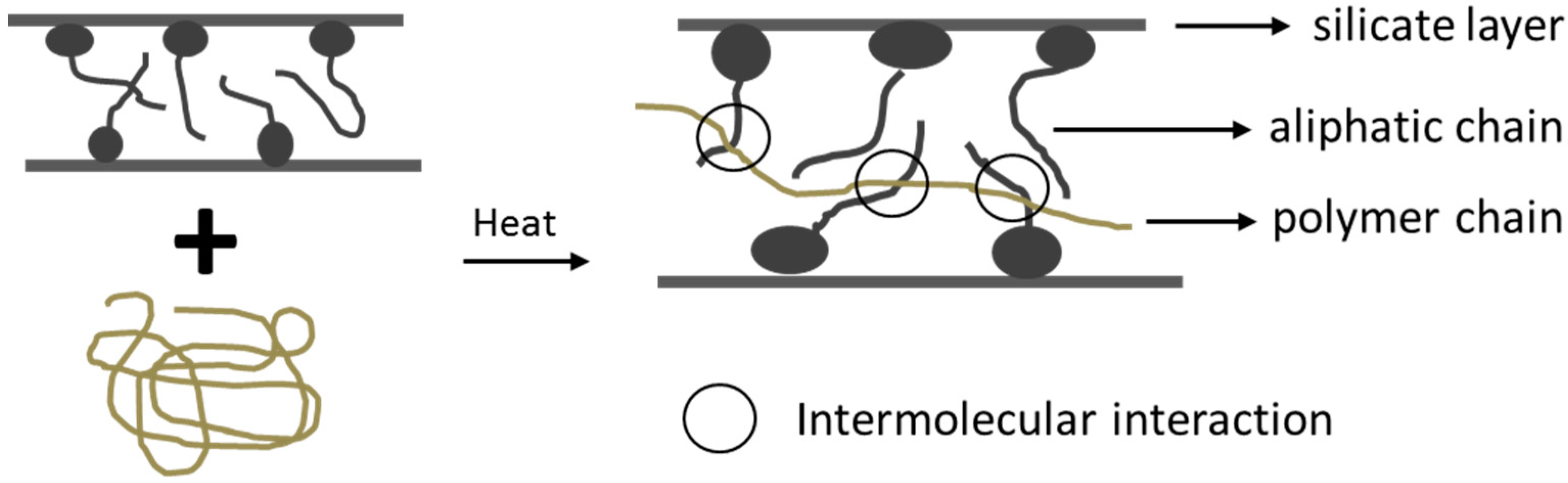
2.1. Nanocomposites
- Intercalation of the polymer
- In-situ intercalative polymerization
- Melt intercalation
- Direct mixture of polymer and particulates
- Template synthesis
- In-situ polymerization
- Sol-gel process
- similar to equal surface energy of polymer and particle surface
- low agglomeration energy
- low polymer viscosity
- high mixing efficiency in the process.
2.1.1. Wet Chemical Processing
2.1.2. Thermoplastic Processing
2.2. Nanodeposits
2.2.1. Common Processing
2.2.2. Electrospraying
2.3. Limitations and Process Improvements
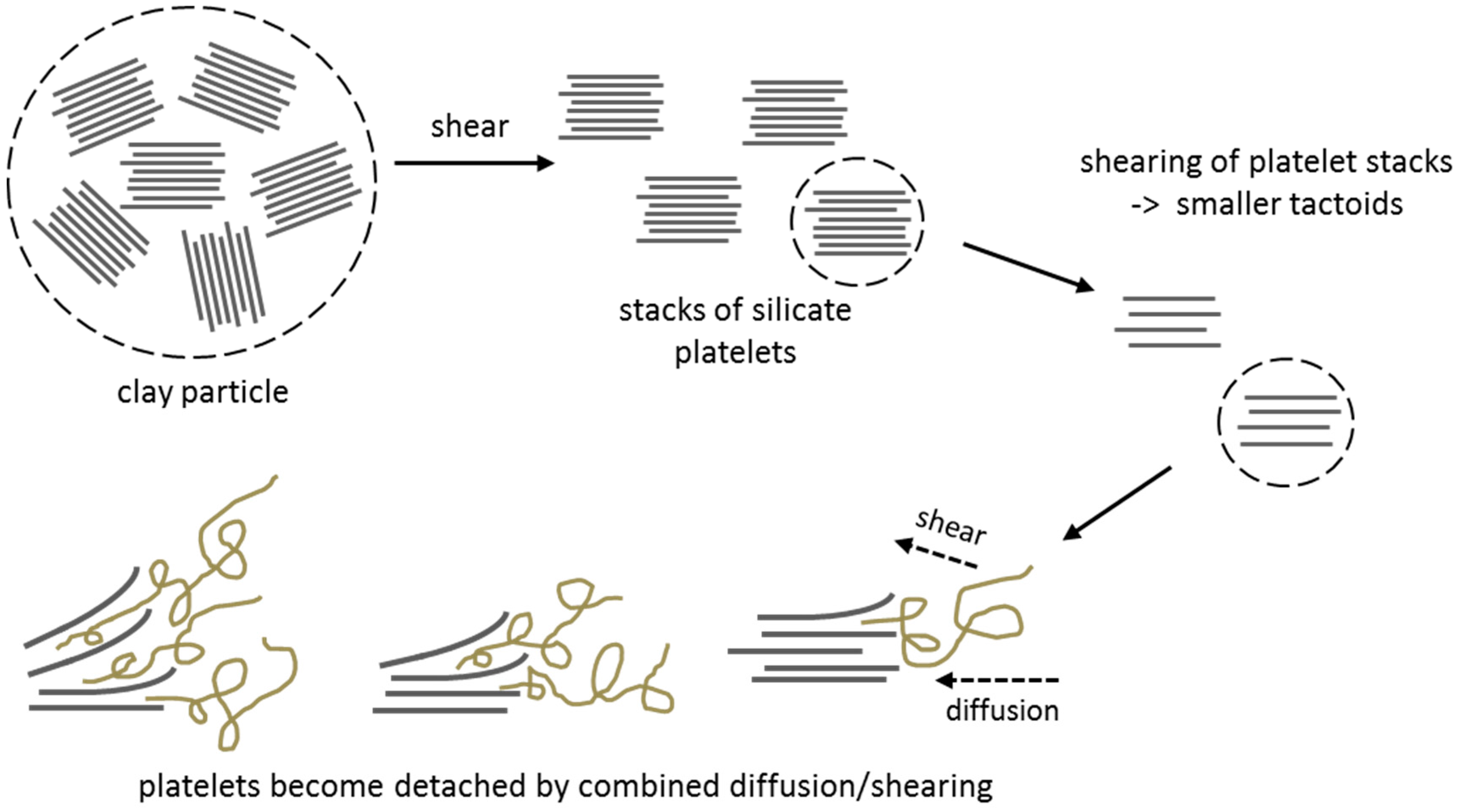
2.3.1. Dispersion Quality and Reaggregation
Particle Surface Modification
Ultrasonic Oscillations
Mechanical Alloying
3. Material Properties
3.1. Nanocomposites
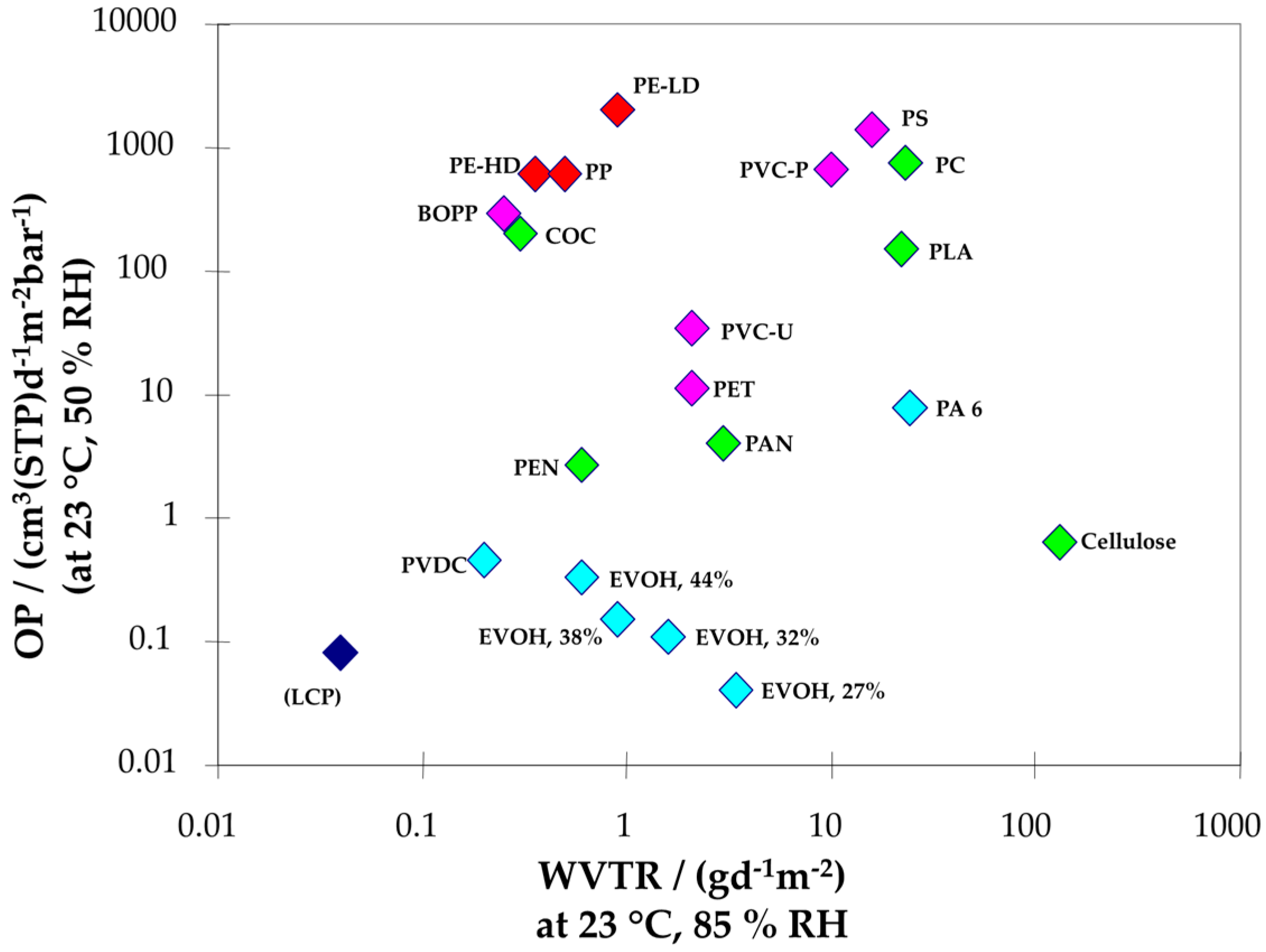
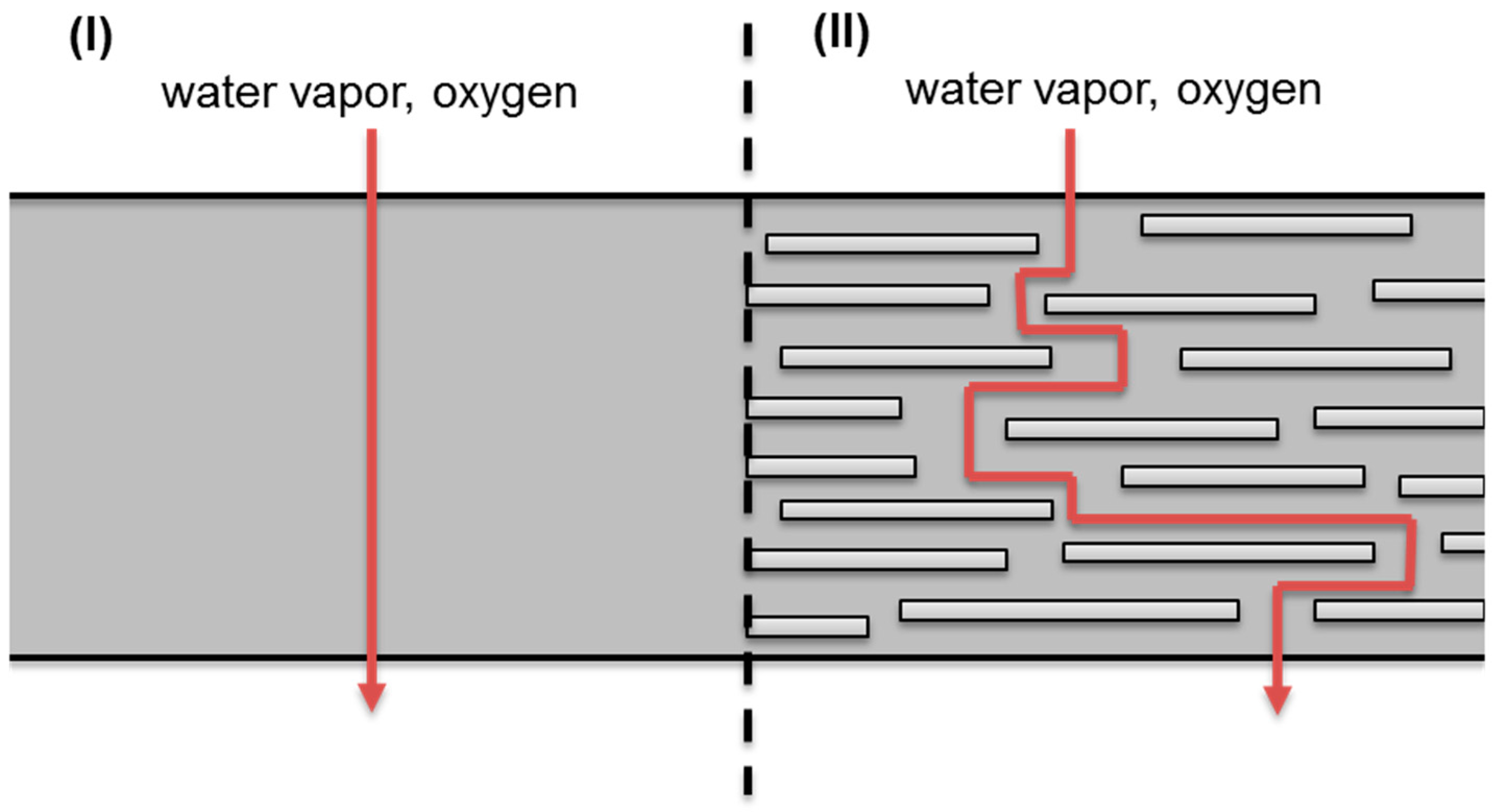
3.1.1. Barrier Properties
- Structural characteristics and polarity of the polymeric chains
- Hydrogen bonding features and other intermolecular interactions
- Polydispersity and molecular weight
- Degree of cross-linking or branching
- Synthesis method and processing technology
- Crystallinity
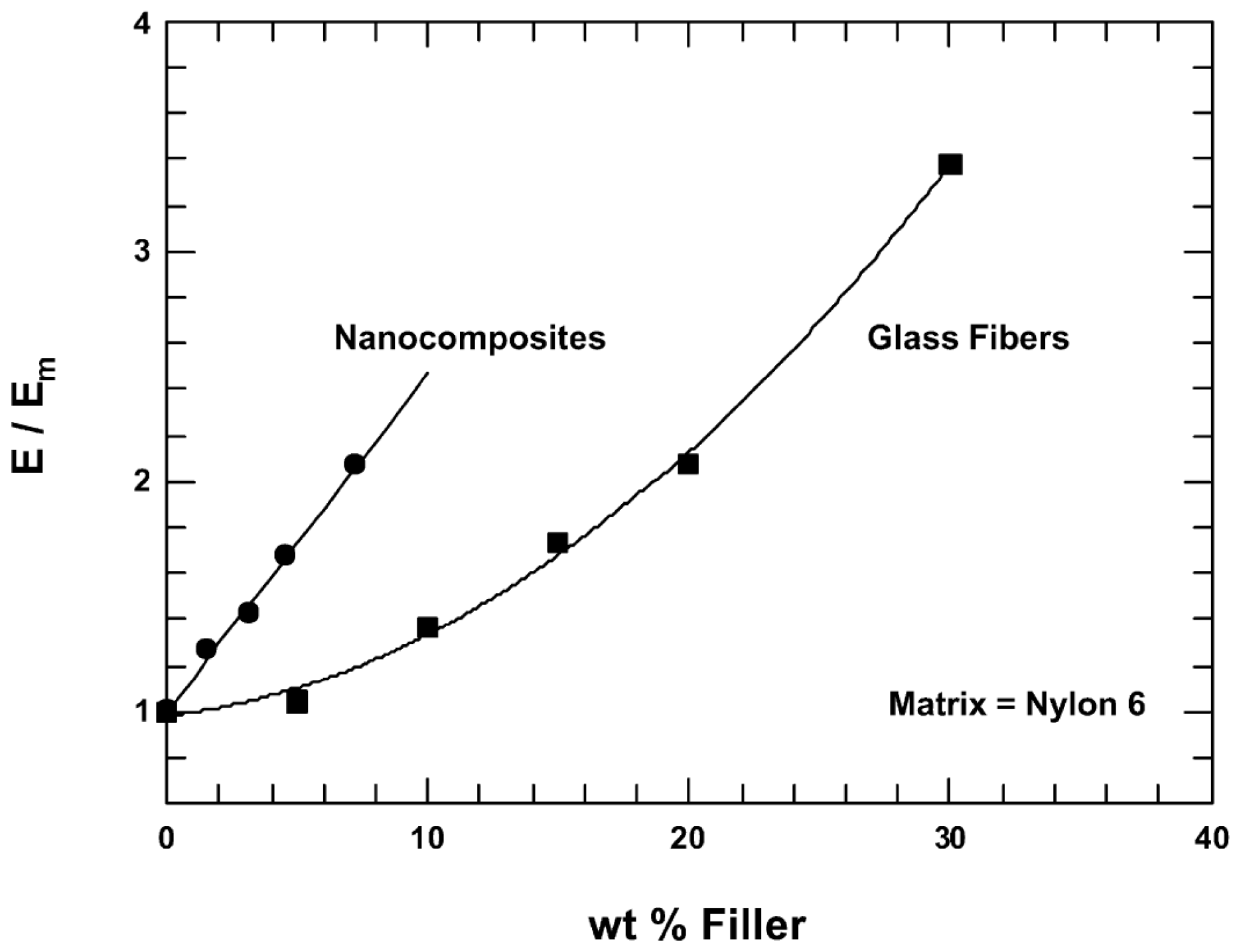
3.1.2. Mechanical Properties: Reinforcement and Light Weighting vs. Conventional Composites
3.1.3. Viscosity = Processability vs. Mechanical Properties
3.1.4. Polymer Blend Compatibilization
3.1.5. Flammability Resistance
3.1.6. Electrical Properties—Electronics
3.1.7. Microwave Absorbing Property
3.2. Nanodeposits
3.2.1. Repellence to Selected Liquids
3.2.2. Self-Cleaning through Photocatalysis
4. Applications of Polymer Nanocomposites
4.1. Packaging
4.1.1. Barrier Materials
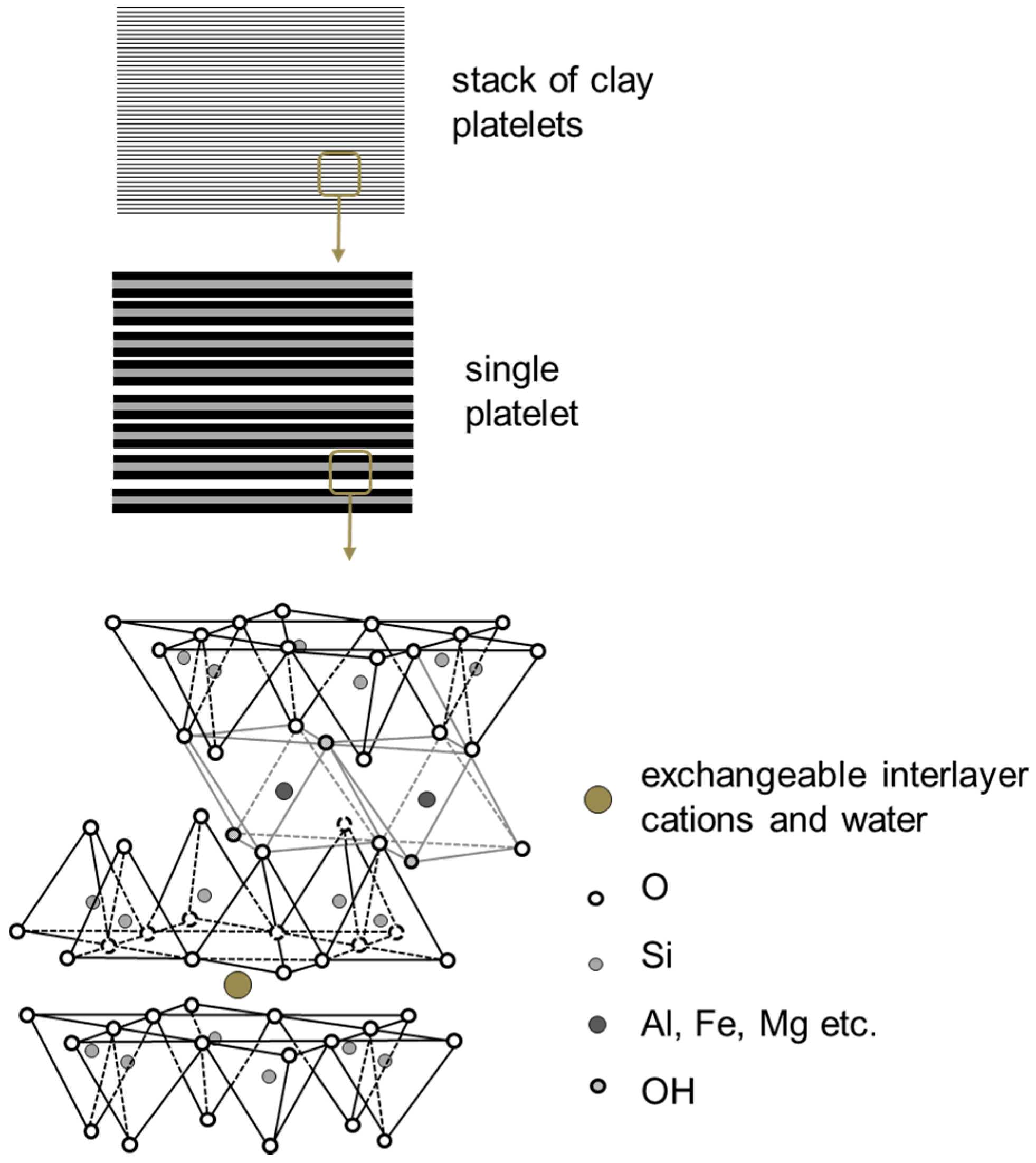
Clay Nanocomposites
Graphene Based Nanocomposites
Other Nanoparticles with Potential in Packaging Applications
4.1.2. Easy-to-Empty Features
4.2. Solar Panels
4.3. Automotive Parts
5. Nanosafety
- There are potential hazards to human health and the environment from certain types and forms of nanoparticles, but not all, and this is largely influenced by their composition and morphology;
- There is a paucity of knowledge about whether and how these potential hazards manifest as actual risks to human and environmental health, through exposure, and their significance;
- The absence of data makes it challenging for manufacturers, suppliers and users to have well-informed and effective risk management processes in compliance with their regulatory obligations.
- good information about the hazardous nature of materials;
- good information about the effectiveness of control approaches;
- convenient and accessible ways to monitor exposure.
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- International Organization for Standardization (ISO). Nanotechnologies—Vocabulary. In Part 4: Nanostructured Materials; International Organization for Standardization: Geneva, Switzerland, 2011. [Google Scholar]
- Schmid, M.; Benz, A.; Stinga, C.; Samain, D.; Zeyer, K.P. Fundamental investigations regarding barrier properties of grafted PVOH layers. Int. J. Polym. Sci. 2012, 2012, 637837. [Google Scholar] [CrossRef]
- Schmid, M.; Sängerlaub, S.; Miesbauer, O.; Jost, V.; Werthan, J.; Stinga, C.; Samain, D.; Stramm, C.; Noller, K.; Müller, K. Water repellence and oxygen and water vapor barrier of pvoh-coated substrates before and after surface esterification. Polymers 2014, 6, 2764–2783. [Google Scholar] [CrossRef]
- Hosokawa, M.; Nogi, K.; Naito, M.; Yokoyama, T. Nanoparticle Technology Handbook; Elsevier Science: Oxford, UK, 2007. [Google Scholar]
- International Organization for Standardization (ISO). Nanotechnologies—Vocabulary. In Part 2: Nano-Objects; International Organization for Standardization: Geneva, Switzerland, 2015. [Google Scholar]
- Okada, A.; Usuki, A. Twenty years of polymer-clay nanocomposites. Macromol. Mater. Eng. 2006, 291, 1449–1476. [Google Scholar] [CrossRef]
- Bugnicourt, E. Development of Sub-Micro Structured Composites based on an Epoxy Matrix and Pyrogenic Silica: Mechanical Behavior Related to the Interactions and Morphology at Multi-Scale. Ph.D. Thesis, Intelligence and National Security Alliance (INSA), Villeurbanne, France, 2005. [Google Scholar]
- Paul, D.R.; Robeson, L.M. Polymer nanotechnology: Nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef]
- McAdam, C.P.; Hudson, N.E.; Liggat, J.J.; Pethrick, R.A. Synthesis and characterization of nylon 6/clay nanocomposites prepared by ultrasonication and in situ polymerization. J. Appl. Polym. Sci. 2008, 108, 2242–2251. [Google Scholar] [CrossRef]
- Lee, S.; Lee, B.; Kim, B.J.; Park, J.; Yoo, M.; Bae, W.K.; Char, K.; Hawker, C.J.; Bang, J.; Cho, J. Free-Standing nanocomposite multilayers with various length scales, adjustable internal structures, and functionalities. J. Am. Chem. Soc. 2009, 131, 2579–2587. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Carraro, G.; Warwick, M.E.A.; Kaunisto, K.; Gasparotto, A.; Gombac, V.; Sada, C.; Turner, S.; Van Tendeloo, G.; Maccato, C.; et al. Fe2O3-TiO2 nanosystems by a hybrid PE-CVD/ALD approach: Controllable synthesis, growth mechanism, and photocatalytic properties. CrystEngComm 2015, 17, 6219–6226. [Google Scholar] [CrossRef]
- Sinha Ray, S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar] [CrossRef]
- Bertuoli, P.T.; Piazza, D.; Scienza, L.C.; Zattera, A.J. Preparation and characterization of montmorillonite modified with 3-aminopropyltriethoxysilane. Appl. Clay Sci. 2014, 87, 46–51. [Google Scholar] [CrossRef]
- Huskić, M.; Žigon, M.; Ivanković, M. Comparison of the properties of clay polymer nanocomposites prepared by montmorillonite modified by silane and by quaternary ammonium salts. Appl. Clay Sci. 2013, 85, 109–115. [Google Scholar] [CrossRef]
- Usuki, A.; Kojima, Y.; Kawasumi, M.; Okada, A.; Fukushima, Y.; Kurauchi, T.; Kamigaito, O. Synthesis of nylon 6-clay hybrid. J. Mater. Res. 1993, 8, 1179–1184. [Google Scholar] [CrossRef]
- Messersmith, P.B.; Giannelis, E.P. Synthesis and characterization of layered silicate-epoxy nanocomposites. Chem. Mater. 1994, 6, 1719–1725. [Google Scholar] [CrossRef]
- Xia, H.S.; Wang, Q. Preparation of conductive polyaniline/nanosilica particle composites through ultrasonic irradiation. J. Appl. Polym. Sci. 2003, 87, 1811–1817. [Google Scholar] [CrossRef]
- Costantino, A.; Pettarin, V.; Viana, J.; Pontes, A.; Pouzada, A.; Frontini, P. Microstructure of PP/clay nanocomposites produced by shear induced injection moulding. Proc. Mater. Sci. 2012, 1, 34–43. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, J.B.; Liu, X.Y.; Hu, Q.L.; Huang, J.G. Antibacterial hybrid materials fabricated by nanocoating of microfibril bundles of cellulose substance with titania/chitosan/silver-nanoparticle composite films. J. Mater. Chem. B 2013, 1, 3477–3485. [Google Scholar] [CrossRef]
- Si, Y.F.; Zhu, H.; Chen, L.W.; Jiang, T.; Guo, Z.G. A multifunctional transparent superhydrophobic gel nanocoating with self-healing properties. Chem. Commun. 2015, 51, 16794–16797. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Mannen, S.; Cain, A.C.; Grunlan, J.C. Flame retardant polymer/clay layer-by-layer assemblies on cotton fabric. Abstr. Pap. Am. Chem. Soc. 2011, 241. [Google Scholar]
- Holder, K.M.; Spears, B.R.; Huff, M.E.; Priolo, M.A.; Harth, E.; Grunlan, J.C. Stretchable gas barrier achieved with partially hydrogen-bonded multilayer nanocoating. Macromol. Rapid Commun. 2014, 35, 960–964. [Google Scholar] [CrossRef] [PubMed]
- Joshi, M.; Khanna, R.; Shekhar, R.; Jha, K. Chitosan nanocoating on cotton textile substrate using layer-by-layer self-assembly technique. J. Appl. Polym. Sci. 2011, 119, 2793–2799. [Google Scholar] [CrossRef]
- Rahman, A.; Ashraf, A.; Xin, H.; Tong, X.; Sutter, P.; Eisaman, M.D.; Black, C.T. Sub-50-nm self-assembled nanotextures for enhanced broadband antireflection in silicon solar cells. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.V.; Paddon, P.A. Nanotechnology impact on the automotive industry. Recent Patents Nanotechnol. 2014, 8, 181–199. [Google Scholar] [CrossRef]
- Arora, A.; Padua, G.W. Review: Nanocomposites in food packaging. J. Food Sci. 2010, 75, R43–R49. [Google Scholar] [CrossRef] [PubMed]
- Mihindukulasuriya, S.D.F.; Lim, L.T. Nanotechnology development in food packaging: A review. Trends Food Sci. Technol. 2014, 40, 149–167. [Google Scholar] [CrossRef]
- Anand, S.; Paxson, A.T.; Dhiman, R.; Smith, J.D.; Varanasi, K.K. Enhanced condensation on lubricant-impregnated nanotextured surfaces. ACS Nano 2012, 6, 10122–10129. [Google Scholar] [CrossRef] [PubMed]
- Sims, L.; Egelhaaf, H.J.; Hauch, J.A.; Kogler, F.R.; Steim, R. Plastic solar cells. Compr. Renew. Energy 2012, 1, 439–480. [Google Scholar]
- Solar Cells That Can Face almost Any Direction and Keep Themselves Clean. Available online: http://m.phys.org/news/2015-12-solar-cells.html (accessed on 18 February 2016).
- Alexandre, M.; Dubois, P. Polymer-Layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R-Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Kamigaito, O. What can be improved by nanometer composites? J. Jpn. Soc. Powder Powder Metall. 1991, 38, 315–321. [Google Scholar] [CrossRef]
- Vaia, R.A.; Giannelis, E.P. Polymer melt intercalation in organically-modified layered silicates: Model predictions and experiment. Macromolecules 1997, 30, 8000–8009. [Google Scholar] [CrossRef]
- Vaia, R.A.; Giannelis, E.P. Lattice model of polymer melt intercalation in organically-modified layered silicates. Macromolecules 1997, 30, 7990–7999. [Google Scholar] [CrossRef]
- Tomasko, D.L.; Han, X.M.; Liu, D.H.; Gao, W.H. Supercritical fluid applications in polymer nanocomposites. Curr. Opin. Solid State Mater. Sci. 2003, 7, 407–412. [Google Scholar] [CrossRef]
- Carrado, K.A.; Xu, L.Q. In situ synthesis of polymer-clay nanocomposites from silicate gels. Chem. Mater. 1998, 10, 1440–1445. [Google Scholar] [CrossRef]
- Mbhele, Z.H.; Salemane, M.G.; van Sittert, C.G.C.E.; Nedeljkovic, J.M.; Djokovic, V.; Luyt, A.S. Fabrication and characterization of silver-polyvinyl alcohol nanocomposites. Chem. Mater. 2003, 15, 5019–5024. [Google Scholar] [CrossRef]
- Park, S.S.; Bernet, N.; de la Roche, S.; Hahn, H.T. Processing of iron oxide-epoxy vinyl ester nanocomposites. J. Compos. Mater. 2003, 37, 465–476. [Google Scholar] [CrossRef]
- Xu, X.L.; Yin, Y.D.; Ge, X.W.; Wu, H.K.; Zhang, Z.C. Gamma-Radiation synthesis of poly(acrylic acid) metal nanocomposites. Mater. Lett. 1998, 37, 354–358. [Google Scholar] [CrossRef]
- Gangopadhyay, R.; De, A. Conducting polymer nanocomposites: A brief overview. Chem. Mater. 2000, 12, 608–622. [Google Scholar] [CrossRef]
- Tracton, A.A. Coatings Technology Handbook, 3rd ed.; CRC Press: Boca Ratom, FL, USA, 2005. [Google Scholar]
- Embuscado, M.; Huber, K.C. Edible Films and Coatings for Food Applications; Springer: New York, NY, USA, 2009. [Google Scholar]
- Rastogi, V.; Samyn, P. Bio-Based coatings for paper applications. Coatings 2015, 5, 887. [Google Scholar] [CrossRef]
- Domininghaus, H.; Eyerer, P.; Elsner, P.; Hirth, T. Kunststoffe: Eigenschaften und Anwendungen; mit 240 Tabellen; Springer-Verlag GmbH: Heidelberg, Germany, 2008. [Google Scholar]
- Braun, D. Kunststofftechnik für Einsteiger; Hanser: Munich, Germany, 2003. [Google Scholar]
- Kaiser, W. Kunststoffchemie für Ingenieure; Hanser: Munich, Germany, 2007. [Google Scholar]
- Dennis, H.R.; Hunter, D.L.; Chang, D.; Kim, S.; White, J.L.; Cho, J.W.; Paul, D.R. Effect of melt processing conditions on the extent of exfoliation in organoclay-based nanocomposites. Polymer 2001, 42, 9513–9522. [Google Scholar] [CrossRef]
- Chavarria, F.; Shah, R.K.; Hunter, D.L.; Paul, D.R. Effect of melt processing conditions on the morphology and properties of nylon 6 nanocomposites. Polym. Eng. Sci. 2007, 47, 1847–1864. [Google Scholar] [CrossRef]
- Lapshin, S.; Swain, S.K.; Isayev, A.I. Ultrasound aided extrusion process for preparation of polyolefin-clay nanocomposites. Polym. Eng. Sci. 2008, 48, 1584–1591. [Google Scholar] [CrossRef]
- Fornes, T.D.; Yoon, P.J.; Keskkula, H.; Paul, D.R. Nylon 6 nanocomposites: The effect of matrix molecular weight. Polymer 2001, 42, 9929–9940. [Google Scholar] [CrossRef]
- Villmow, T.; Pegel, S.; Potschke, P.; Wagenknecht, U. Influence of injection molding parameters on the electrical resistivity of polycarbonate filled with multi-walled carbon nanotubes. Compos. Sci. Technol. 2008, 68, 777–789. [Google Scholar] [CrossRef]
- Chandra, A.; Kramschuster, A.J.; Hu, X.; Turng, L.S. Effect of injection molding parameters on the electrical conductivity of polycarbonate/carbon nanotube nanocomposites. In Proceedings of the Annual Technical Conference (ANTEC), Cincinnati, OH, USA, 6–11 May 2007; pp. 2171–2175. [Google Scholar]
- Stan, F.; Sandu, L.I.; Fetecau, C. Effect of processing parameters and strain rate on mechanical properties of carbon nanotube-filled polypropylene nanocomposites. Compos. Part B Eng. 2014, 59, 109–122. [Google Scholar] [CrossRef]
- Rios, P.F.; Ophir, A.; Kenig, S.; Efrati, R.; Zonder, L.; Popovitz-Biro, R. Impact of injection-molding processing parameters on the electrical, mechanical, and thermal properties of thermoplastic/carbon nanotube nanocomposites. J. Appl. Polym. Sci. 2011, 120, 70–78. [Google Scholar] [CrossRef]
- Schmid, M.; Dallmann, K.; Bugnicourt, E.; Cordoni, D.; Wild, F.; Lazzeri, A.; Noller, K. Properties of whey-protein-coated films and laminates as novel recyclable food packaging materials with excellent barrier properties. Int. J. Polym. Sci. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- De Azeredo, H.M.C. Nanocomposites for food packaging applications. Food Res. Int. 2009, 42, 1240–1253. [Google Scholar] [CrossRef]
- Alberto, R. Fundamentos de la Tecnología de Extrusión de Película Cast. Available online: http://www.plastico.com/ (accessed on 4 February 2016).
- Coltelli, M.-B.; Wild, F.; Bugnicourt, E.; Cinelli, P.; Lindner, M.; Schmid, M.; Weckel, V.; Müller, K.; Rodriguez, P.; Staebler, A. State of the art in the development and properties of protein-based films and coatings and their applicability to cellulose based products: An extensive review. Coatings 2016, 6, 1. [Google Scholar] [CrossRef]
- Caruso, R.A.; Antonietti, M. Sol-Gel nanocoating: An approach to the preparation of structured materials. Chem. Mater. 2001, 13, 3272–3282. [Google Scholar] [CrossRef]
- Anghel, I.; Holban, A.M.; Andronescu, E.; Grumezescu, A.M.; Chifiriuc, M.C. Efficient surface functionalization of wound dressings by a phytoactive nanocoating refractory to candida albicans biofilm development. Biointerphases 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Gurzawska, K.; Svava, R.; Yu, Y.H.; Haugshoj, K.B.; Dirscherl, K.; Levery, S.B.; Byg, I.; Damager, I.; Nielsen, M.W.; Jorgensen, B.; et al. Osteoblastic response to pectin nanocoating on titanium surfaces. Mater. Sci. Eng. C Mater. 2014, 43, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Mattioli-Belmonte, M.; Cometa, S.; Ferretti, C.; Iatta, R.; Trapani, A.; Ceci, E.; Falconi, M.; De Giglio, E. Characterization and cytocompatibility of an antibiotic/chitosan/cyclodextrins nanocoating on titanium implants. Carbohydr. Polym. 2014, 110, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yang, S.H.; Lee, J.; Ko, E.H.; Hong, D.; Choi, I.S. Nanocoating of single cells: From maintenance of cell viability to manipulation of cellular activities. Adv. Mater. 2014, 26, 2001–2010. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Y.; Singh, D.; Sirkar, K.K.; Zhu, J.T.; Pfeffer, R. Continuous polymer nanocoating on silica nanoparticles. Langmuir 2014, 30, 7804–7810. [Google Scholar] [CrossRef] [PubMed]
- Smietana, M.; Bock, W.J.; Szmidt, J.; Pickrell, G.R. Nanocoating enhanced optical fiber sensors. Ceram. Trans. 2010, 222, 275–286. [Google Scholar]
- Chen, M.M.; Sun, X.; Qiao, Z.J.; Ma, Q.Q.; Wang, C.Y. Anatase-TiO2 nanocoating of Li4Ti5O12 nanorod anode for lithium-ion batteries. J. Alloy Compd. 2014, 601, 38–42. [Google Scholar] [CrossRef]
- Hamedi, G.H.; Nejad, F.M. Using energy parameters based on the surface free energy concept to evaluate the moisture susceptibility of hot mix asphalt. Road Mater. Pavement 2015, 16, 239–255. [Google Scholar] [CrossRef]
- Hsieh, C.T.; Chang, B.S.; Lin, J.Y. Improvement of water and oil repellency on wood substrates by using fluorinated silica nanocoating. Appl. Surf. Sci. 2011, 257, 7997–8002. [Google Scholar] [CrossRef]
- Zhang, T.; Yan, H.; Peng, M.; Wang, L.; Ding, H.; Fang, Z. Construction of flame retardant nanocoating on ramie fabric via layer-by-layer assembly of carbon nanotube and ammonium polyphosphate. Nanoscale 2013, 5, 3013–3021. [Google Scholar] [CrossRef] [PubMed]
- Langowski, H.-C. Permeation of gases and condensable substances through monolayer and multilayer structures. In Plastic Packaging; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 297–347. [Google Scholar]
- Bishop, C. Vacuum Deposition onto Webs, Films and Foils; Elsevier Science: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Fahlteich, J. Transparente Hochbarriereschichten auf Flexiblen Substraten. Ph.D. Thesis, Technische Universität Chemnitz, Chemnitz, Germany, 2010. [Google Scholar]
- Torrisi, V.; Ruffino, F. Metal-Polymer nanocomposites: (co-)evaporation/(co)sputtering approaches and electrical properties. Coatings 2015, 5, 378. [Google Scholar] [CrossRef]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecules 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Yoon, Y.S.; Park, H.Y.; Lim, Y.C.; Choi, K.G.; Lee, K.C.; Park, G.B.; Lee, C.J.; Moon, D.G.; Han, J.I.; Kim, Y.B.; et al. Effects of parylene buffer layer on flexible substrate in organic light emitting diode. Thin Solid Films 2006, 513, 258–263. [Google Scholar] [CrossRef]
- Kääriäinen, T.; Cameron, D.; Kääriäinen, M.L.; Sherman, A. Atomic Layer Deposition: Principles, Characteristics, and Nanotechnology Applications; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Lewis, J.S.; Weaver, M.S. Thin-Film permeation-barrier technology for flexible organic light-emitting devices. IEEE J. Sel. Top. Quantum Electron. 2004, 10, 45–57. [Google Scholar] [CrossRef]
- Miesbauer, O.; Kucukpinar, E.; Kiese, S.; Carmi, Y.; Noller, K.; Langowski, H.C. Studies on the barrier performance and adhesion strength of novel barrier films for vacuum insulation panels. Energy Build. 2014, 85, 597–603. [Google Scholar] [CrossRef]
- Miesbauer, O.; Schmidt, M.; Langowski, H.-C. Stofftransport durch Schichtsysteme aus Polymeren und dünnen anorganischen Schichten. Vak. Forsch. Prax. 2008, 20, 32–40. [Google Scholar] [CrossRef]
- Langowski, H.-C. Flexible materialien mit ultrahohen barriereeigenschaften. Flexible ultra high barrier materials. Vak. Forsch. Prax. 2002, 14, 297–302. [Google Scholar] [CrossRef]
- Jaworek, A. Micro- and nanoparticle production by electrospraying. Powder Technol. 2007, 176, 18–35. [Google Scholar] [CrossRef]
- Wu, Y.Q.; Clark, R.L. Electrohydrodynamic atomization: A versatile process for preparing materials for biomedical applications. J. Biomat. Sci.-Polym. E 2008, 19, 573–601. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; Pardo, P.; Martinez-Sanz, M.; Lopez-Rubio, A.; Lagaron, J.M. Combining polyhydroxyalkanoates with nanokeratin to develop novel biopackaging structures. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Khan, S.; Doh, Y.H.; Khan, A.; Rahman, A.; Choi, K.H.; Kim, D.S. Direct patterning and electrospray deposition through ehd for fabrication of printed thin film transistors. Curr. Appl. Phys. 2011, 11, S271–S279. [Google Scholar] [CrossRef]
- Pérez-Masiá, R.; Lagaron, J.M.; López-Rubio, A. Surfactant-Aided electrospraying of low molecular weight carbohydrate polymers from aqueous solutions. Carbohydr. Polym. 2014, 101, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Kango, S.; Kalia, S.; Celli, A.; Njuguna, J.; Habibi, Y.; Kumar, R. Surface modification of inorganic nanoparticles for development of organic-inorganic nanocomposites-a review. Prog. Polym. Sci. 2013, 38, 1232–1261. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Galy, J.; Gérard, J.; Boué, F.; Barthel, H. Structural investigations of pyrogenic silica-epoxy composites by small-angle neutron scattering and transmission electron microscopy. Polymer 2006, 48, 949–958. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Kehoe, T.; Latorre, M.; Serrano, C.; Philippe, S.; Schmid, M. Recent prospects in the inline monitoring of nanocomposites and nanocoatings by optical technologies. Nanomaterials 2016, 6, 150. [Google Scholar] [CrossRef] [PubMed]
- Mittal, V. Optimization of Polymer Nanocomposite Properties; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009. [Google Scholar]
- Rong, M.Z.; Zhang, M.Q.; Ruan, W.H. Surface modification of nanoscale fillers for improving properties of polymer nanocomposites: A review. Mater. Sci. Technol. 2006, 22, 787–796. [Google Scholar] [CrossRef]
- Fekete, E.; Pukánszky, B.; Tóth, A.; Bertóti, I. Surface modification and characterization of particulate mineral fillers. J. Colloid Interface Sci. 1990, 135, 200–208. [Google Scholar] [CrossRef]
- Lin, H.; Isayev, A.I. Ultrasonic treatment of polypropylene, polyamide 6, and their blends. J. Appl. Polym. Sci. 2006, 102, 2643–2653. [Google Scholar] [CrossRef]
- Kumar, R.V.; Koltypin, Y.; Palchik, O.; Gedanken, A. Preparation and characterization of nickel-polystyrene nanocomposite by ultrasound irradiation. J. Appl. Polym. Sci. 2002, 86, 160–165. [Google Scholar] [CrossRef]
- Isayev, A.I.; Hong, C.K.; Kim, K.J. Continuous mixing and compounding of polymer/filler and polymer/polymer mixtures with the aid of ultrasound. Rubber Chem. Technol. 2003, 76, 923–947. [Google Scholar] [CrossRef]
- Ullah, M.; Ali, M.E.; Abd Hamid, S.B. Surfactant-Assisted ball milling: A novel route to novel materials with controlled nanostructure—A review. Rev. Adv. Mater. Sci. 2014, 37, 1–14. [Google Scholar]
- Fecht, H.J.; Hellstern, E.; Fu, Z.; Johnson, W.L. Nanocrystalline metals prepared by high-energy ball milling. Metall. Trans. 1990, 21, 2333–2337. [Google Scholar] [CrossRef]
- Nandhini, R.; Mini, P.A.; Avinash, B.; Nair, S.V.; Subramanian, K.R.V. Supercapacitor electrodes using nanoscale activated carbon from graphite by ball milling. Mater. Lett. 2012, 87, 165–168. [Google Scholar] [CrossRef]
- Cho, K.; Chang, H.; Kil, D.S.; Kim, B.-G.; Jang, H.D. Synthesis of dispersed CaCO3 nanoparticles by the ultrafine grinding. J. Ind. Eng. Chem. 2009, 15, 243–246. [Google Scholar] [CrossRef]
- Zhang, D.L. Processing of advanced materials using high-energy mechanical milling. Prog. Mater. Sci. 2004, 49, 537–560. [Google Scholar] [CrossRef]
- Mark, J.E.; Jiang, C.Y.; Tang, M.Y. Simultaneous curing and filling of elastomers. Macromolecules 1984, 17, 2613–2616. [Google Scholar] [CrossRef]
- Wen, J.Y.; Wilkes, G.L. Organic/inorganic hybrid network materials by the sol-gel approach. Chem. Mater. 1996, 8, 1667–1681. [Google Scholar] [CrossRef]
- Huang, H.-H.; Orler, B.; Wilkes, G.L. Ceramers: Hybrid Materials Incorporating Polymeric/Oligomeric Species with Inorganic Glasses Utilizing a Sol-Gel Process. Polym. Bull. 1985, 14, 557–564. [Google Scholar] [CrossRef]
- Fischer, H. Polymer nanocomposites: From fundamental research to specific applications. Mat. Sci. Eng. C 2003, 23, 763–772. [Google Scholar] [CrossRef]
- The Royal Society. Nanoscience and Nanotechnologies: Opportunities and Uncertainties; The Royal Society & The Royal Academy of Engineering: London, UK, 2004. [Google Scholar]
- Oriakhi, C. Nano sandwiches. Chem. Br. 1998, 34, 59–62. [Google Scholar]
- Usuki, A.; Kawasumi, M.; Kojima, Y.; Okada, A.; Kurauchi, T.; Kamigaito, O. Swelling behavior of montmorillonite cation exchanged for omega-amino acids by epsilon-caprolactam. J. Mater. Res. 1993, 8, 1174–1178. [Google Scholar] [CrossRef]
- Kuchibhatla, S.V.N.T.; Karakoti, A.S.; Bera, D.; Seal, S. One dimensional nanostructured materials. Prog. Mater. Sci. 2007, 52, 699–913. [Google Scholar] [CrossRef]
- Li, C.; Bai, H.; Shi, G.Q. Conducting polymer nanomaterials: Electrosynthesis and applications. Chem. Soc. Rev. 2009, 38, 2397–2409. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.D.; Li, D.; Kaner, R.B. One-Dimensional conducting polymer nanostructures: Bulk synthesis and applications. Adv. Mater. 2009, 21, 1487–1499. [Google Scholar] [CrossRef]
- MacDiarmid, A.G. “Synthetic metals”: A novel role for organic polymers (nobel lecture). Angew. Chem. Int. Ed. 2001, 40, 2581–2590. [Google Scholar] [CrossRef]
- Heeger, A.J. Semiconducting polymers: The third generation. Chem. Soc. Rev. 2010, 39, 2354–2371. [Google Scholar] [CrossRef] [PubMed]
- Manias, E. Nanocomposites: Stiffer by design. Nat. Mater. 2007, 6, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Muncke, J. Food Packaging Materials. Available online: http://www.foodpackagingforum.org/food-packaging-health/food-packaging-materials (accessed on 4 February 2016).
- Yam, K.L. The Wiley Encyclopedia of Packaging Technology, 3rd ed.; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Hammann, F.; Schmid, M. Determination and quantification of molecular interactions in protein films: A review. Materials 2014, 7, 7975–7996. [Google Scholar] [CrossRef]
- Zink, J.; Wyrobnik, T.; Prinz, T.; Schmid, M. Physical, chemical and biochemical modifications of protein-based films and coatings: An extensive review. Int. J. Mol. Sci. 2016, 17, 1376. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Reichert, K.; Hammann, F.; Stäbler, A. Storage time-dependent alteration of molecular interaction-property relationships of whey protein isolate-based films and coatings. J. Mater. Sci. 2015, 50, 4396–4404. [Google Scholar] [CrossRef]
- Schmid, M.; Zillinger, W.; Müller, K.; Sängerlaub, S. Permeation of water vapour, nitrogen, oxygen and carbon dioxide through whey protein isolate based films and coatings—Permselectivity and activation energy. Food Packag. Shelf Life 2015, 6, 21–29. [Google Scholar] [CrossRef]
- Zhang, Z.; Britt, I.J.; Tung, M.A. Oxygen ingress in plastic retortable packages during thermal processing and storage. J. Plast. Film Sheeting 1998, 14, 287–307. [Google Scholar]
- Yeo, J.H.; Lee, C.H.; Park, C.-S.; Lee, K.-J.; Nam, J.-D.; Kim, S.W. Rheological, morphological, mechanical, and barrier properties of pp/evoh blends. Adv. Polym. Technol. 2001, 20, 191–201. [Google Scholar] [CrossRef]
- Lagaron, J.M.; Gimenez, E.; Gavara, R.; Saura, J.J. Study of the influence of water sorption in pure components and binary blends of high barrier ethylene-vinyl alcohol copolymer and amorphous polyamide and nylon-containing ionomer. Polymer 2001, 42, 9531–9540. [Google Scholar] [CrossRef]
- Reig, C.S.; Lopez, A.D.; Ramos, M.H.; Ballester, V.A.C. Nanomaterials: A map for their selection in food packaging applications. Packag. Technol. Sci. 2014, 27, 839–866. [Google Scholar] [CrossRef]
- Vladimirov, V.; Betchev, C.; Vassiliou, A.; Papageorgiou, G.; Bikiaris, D. Dynamic mechanical and morphological studies of isotactic polypropylene/fumed silica nanocomposites with enhanced gas barrier properties. Compos. Sci. Technol. 2006, 66, 2935–2944. [Google Scholar] [CrossRef]
- Kim, J.Y.; Han, S.-I.; Kim, S.H. Crystallization behaviors and mechanical properties of poly(ethylene 2,6-naphthalate)/multiwall carbon nanotube nanocomposites. Polym. Eng. Sci. 2007, 47, 1715–1723. [Google Scholar] [CrossRef]
- Chen, Y.; Cao, X.; Chang, P.R.; Huneault, M.A. Comparative study on the films of poly(vinyl alcohol)/pea starch nanocrystals and poly(vinyl alcohol)/native pea starch. Carbohydr. Polym. 2008, 73, 8–17. [Google Scholar] [CrossRef]
- Azeredo, H.M.C.; Mattoso, L.H.C.; Wood, D.; Williams, T.G.; Avena-Bustillos, R.J.; McHugh, T.H. Nanocomposite edible films from mango puree reinforced with cellulose nanofibers. J. Food Sci. 2009, 74, N31–N35. [Google Scholar] [CrossRef] [PubMed]
- Bilbao-Sáinz, C.; Avena-Bustillos, R.J.; Wood, D.F.; Williams, T.G.; McHugh, T.H. Composite edible films based on hydroxypropyl methylcellulose reinforced with microcrystalline cellulose nanoparticles. J. Agric. Food Chem. 2010, 58, 3753–3760. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Chen, Y.; Chang, P.R.; Stumborg, M.; Huneault, M.A. Green composites reinforced with hemp nanocrystals in plasticized starch. J. Appl. Polym. Sci. 2008, 109, 3804–3810. [Google Scholar] [CrossRef]
- Podsiadlo, P.; Choi, S.Y.; Shim, B.; Lee, J.; Cuddihy, M.; Kotov, N.A. Molecularly engineered nanocomposites: Layer-by-layer assembly of cellulose nanocrystals. Biomacromolecules 2005, 6, 2914–2918. [Google Scholar] [CrossRef] [PubMed]
- De Moura, M.R.; Aouada, F.A.; Avena-Bustillos, R.J.; McHugh, T.H.; Krochta, J.M.; Mattoso, L.H.C. Improved barrier and mechanical properties of novel hydroxypropyl methylcellulose edible films with chitosan/tripolyphosphate nanoparticles. J. Food Eng. 2009, 92, 448–453. [Google Scholar] [CrossRef]
- De Moura, M.R.; Lorevice, M.V.; Mattoso, L.H.C.; Zucolotto, V. Highly stable, edible cellulose films incorporating chitosan nanoparticles. J. Food Sci. 2011, 76, N25–N29. [Google Scholar] [CrossRef] [PubMed]
- Choudalakis, G.; Gotsis, A.D. Permeability of polymer/clay nanocomposites: A review. Eur. Polym. J. 2009, 45, 967–984. [Google Scholar] [CrossRef]
- Duncan, T.V. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.E. Models for the permeability of filled polymer systems. J. Macromol. Sci. 1967, 1, 929–942. [Google Scholar] [CrossRef]
- Lape, N.K.; Nuxoll, E.E.; Cussler, E.L. Polydisperse flakes in barrier films. J. Membr. Sci. 2004, 236, 29–37. [Google Scholar] [CrossRef]
- Gusev, A.A.; Lusti, H.R. Rational design of nanocomposites for barrier applications. Adv. Mater. 2001, 13, 1641–1643. [Google Scholar] [CrossRef]
- Fredrickson, G.H.; Bicerano, J. Barrier properties of oriented disk composites. J. Chem. Phys. 1999, 110, 2181–2188. [Google Scholar] [CrossRef]
- Bharadwaj, R.K. Modeling the barrier properties of polymer-layered silicate nanocomposites. Macromolecules 2001, 34, 9189–9192. [Google Scholar] [CrossRef]
- Sun, L.; Boo, W.J.; Clearfield, A.; Sue, H.J.; Pham, H.Q. Barrier properties of model epoxy nanocomposites. J. Membr. Sci. 2008, 318, 129–136. [Google Scholar] [CrossRef]
- Picard, E.; Gauthier, H.; Gérard, J.F.; Espuche, E. Influence of the intercalated cations on the surface energy of montmorillonites: Consequences for the morphology and gas barrier properties of polyethylene/montmorillonites nanocomposites. J. Colloid Interface Sci. 2007, 307, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Fornes, T.; Paul, D. Modeling properties of nylon 6/clay nanocomposites using composite theories. Polymer 2003, 44, 4993–5013. [Google Scholar] [CrossRef]
- Sen, S.; Thomin, J.D.; Kumar, S.K.; Keblinski, P. Molecular underpinnings of the mechanical reinforcement in polymer nanocomposites. Macromolecules 2007, 40, 4059–4067. [Google Scholar] [CrossRef]
- Vaia, R.; Giannelis, E. Liquid crystal polymer nanocomposites: Direct intercalation of thermotropic liquid crystalline polymers into layered silicates. Polymer 2001, 42, 1281–1285. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Galy, J.; Gérard, J.-F.; Barthel, H. Effect of sub-micron silica fillers on the mechanical performances of epoxy-based composites. Polymer 2007, 48, 1596–1605. [Google Scholar] [CrossRef]
- Hunter, D.L.; Kamena, K.W.; Paul, D.R. Processing and properties of polymers modified by clays. MRS Bull. 2007, 32, 323–327. [Google Scholar] [CrossRef]
- Hotta, S.; Paul, D. Nanocomposites formed from linear low density polyethylene and organoclays. Polymer 2004, 45, 7639–7654. [Google Scholar] [CrossRef]
- Jang, J.S.; Bouveret, B.; Suhr, J.; Gibson, R.F. Combined numerical/experimental investigation of particle diameter and interphase effects on coefficient of thermal expansion and young’s modulus of SiO2/epoxy nanocomposites. Polym. Compos. 2012, 33, 1415–1423. [Google Scholar] [CrossRef]
- Yu, S.; Yang, S.; Cho, M. Multi-Scale modeling of cross-linked epoxy nanocomposites. Polymer 2009, 50, 945–952. [Google Scholar] [CrossRef]
- Choi, J.; Yu, S.; Yang, S.; Cho, M. The glass transition and thermoelastic behavior of epoxy-based nanocomposites: A molecular dynamics study. Polymer 2011, 52, 5197–5203. [Google Scholar] [CrossRef]
- Tsai, J.-L.; Tzeng, S.-H. Characterizing mechanical properties of particulate nanocomposites using micromechanical approach. J. Compos. Mater. 2008, 42, 2345–2361. [Google Scholar] [CrossRef]
- Yang, S.; Cho, M. Scale bridging method to characterize mechanical properties of nanoparticle/polymer nanocomposites. Appl. Phys. Lett. 2008, 93, 043111. [Google Scholar] [CrossRef]
- Liu, H.; Brinson, L.C. Reinforcing efficiency of nanoparticles: A simple comparison for polymer nanocomposites. Compos. Sci. Technol. 2008, 68, 1502–1512. [Google Scholar] [CrossRef]
- Yang, B.; Hwang, Y.; Lee, H. Elastoplastic modeling of polymeric composites containing randomly located nanoparticles with an interface effect. Compos. Struct. 2013, 99, 123–130. [Google Scholar] [CrossRef]
- Goertzen, W.K.; Kessler, M. Thermal expansion of fumed silica/cyanate ester nanocomposites. J. Appl. Polym. Sci. 2008, 109, 647–653. [Google Scholar] [CrossRef]
- Yu, S.; Yang, S.; Cho, M. Multiscale modeling of cross-linked epoxy nanocomposites to characterize the effect of particle size on thermal conductivity. J. Appl. Phys. 2011, 110, 124302. [Google Scholar] [CrossRef]
- Shin, H.; Yang, S.; Chang, S.; Yu, S.; Cho, M. Multiscale homogenization modeling for thermal transport properties of polymer nanocomposites with kapitza thermal resistance. Polymer 2013, 54, 1543–1554. [Google Scholar] [CrossRef]
- Kratochvíla, J.; Boudenne, A.; Krupa, I. Effect of filler size on thermophysical and electrical behavior of nanocomposites based on expanded graphite nanoparticles filled in low-density polyethylene matrix. Polym. Compos. 2013, 34, 149–155. [Google Scholar] [CrossRef]
- Harton, S.E.; Kumar, S.K.; Yang, H.; Koga, T.; Hicks, K.; Lee, H.; Mijovic, J.; Liu, M.; Vallery, R.S.; Gidley, D.W. Immobilized polymer layers on spherical nanoparticles. Macromolecules 2010, 43, 3415–3421. [Google Scholar] [CrossRef]
- Choi, J.; Yang, S.; Yu, S.; Shin, H.; Cho, M. Method of scale bridging for thermoelasticity of cross-linked epoxy/sic nanocomposites at a wide range of temperatures. Polymer 2012, 53, 5178–5189. [Google Scholar] [CrossRef]
- Qiao, R.; Brinson, L.C. Simulation of interphase percolation and gradients in polymer nanocomposites. Compos. Sci. Technol. 2009, 69, 491–499. [Google Scholar] [CrossRef]
- Hadden, C.; Jensen, B.; Bandyopadhyay, A.; Odegard, G.; Koo, A.; Liang, R. Molecular modeling of epon-862/graphite composites: Interfacial characteristics for multiple crosslink densities. Compos. Sci. Technol. 2013, 76, 92–99. [Google Scholar] [CrossRef]
- Buryachenko, V.; Roy, A.; Lafdi, K.; Anderson, K.; Chellapilla, S. Multi-Scale mechanics of nanocomposites including interface: Experimental and numerical investigation. Compos. Sci. Technol. 2005, 65, 2435–2465. [Google Scholar] [CrossRef]
- Buryan, O.; Novikov, V. Modeling of the interphase of polymer-matrix composites: Determination of its structure and mechanical properties. Mech. Compos. Mater. 2002, 38, 187–190. [Google Scholar] [CrossRef]
- Bhuiyan, M.A.; Pucha, R.V.; Karevan, M.; Kalaitzidou, K. Tensile modulus of carbon nanotube/polypropylene composites—A computational study based on experimental characterization. Comput. Mater. Sci. 2011, 50, 2347–2353. [Google Scholar] [CrossRef]
- Bhuiyan, M.A.; Pucha, R.V.; Worthy, J.; Karevan, M.; Kalaitzidou, K. Defining the lower and upper limit of the effective modulus of CNT/polypropylene composites through integration of modeling and experiments. Compos. Struct. 2013, 95, 80–87. [Google Scholar] [CrossRef]
- Ayatollahi, M.; Shadlou, S.; Shokrieh, M. Multiscale modeling for mechanical properties of carbon nanotube reinforced nanocomposites subjected to different types of loading. Compos. Struct. 2011, 93, 2250–2259. [Google Scholar] [CrossRef]
- Shokrieh, M.M.; Rafiee, R. On the tensile behavior of an embedded carbon nanotube in polymer matrix with non-bonded interphase region. Compos. Struct. 2010, 92, 647–652. [Google Scholar] [CrossRef]
- Arrighi, V.; McEwen, I.; Qian, H.; Prieto, M.S. The glass transition and interfacial layer in styrene-butadiene rubber containing silica nanofiller. Polymer 2003, 44, 6259–6266. [Google Scholar] [CrossRef]
- Yang, S.; Yu, S.; Ryu, J.; Cho, J.-M.; Kyoung, W.; Han, D.-S.; Cho, M. Nonlinear multiscale modeling approach to characterize elastoplastic behavior of CNT/polymer nanocomposites considering the interphase and interfacial imperfection. Int. J. Plast. 2013, 41, 124–146. [Google Scholar] [CrossRef]
- Mortezaei, M.; Famili, M.H.N.; Kokabi, M. The role of interfacial interactions on the glass-transition and viscoelastic properties of silica/polystyrene nanocomposite. Compos. Sci. Technol. 2011, 71, 1039–1045. [Google Scholar] [CrossRef]
- Fragiadakis, D.; Pissis, P.; Bokobza, L. Glass transition and molecular dynamics in poly (dimethylsiloxane)/silica nanocomposites. Polymer 2005, 46, 6001–6008. [Google Scholar] [CrossRef]
- Yang, S.; Cho, M. A scale-bridging method for nanoparticulate polymer nanocomposites and their nondilute concentration effect. Appl. Phys. Lett. 2009, 94, 223104. [Google Scholar] [CrossRef]
- Loo, Y.-L.; Register, R.A.; Ryan, A.J. Polymer crystallization in 25-nm spheres. Phys. Rev. Lett. 2000, 84, 4120. [Google Scholar] [CrossRef] [PubMed]
- Loo, Y.-L.; Register, R.A.; Ryan, A.J. Modes of crystallization in block copolymer microdomains: Breakout, templated, and confined. Macromolecules 2002, 35, 2365–2374. [Google Scholar] [CrossRef]
- Santana, O.O.; Müller, A.J. Homogeneous nucleation of the dispersed crystallisable component of immiscible polymer blends. Polym. Bull. 1994, 32, 471–477. [Google Scholar] [CrossRef]
- Woo, E.; Huh, J.; Jeong, Y.G.; Shin, K. From homogeneous to heterogeneous nucleation of chain molecules under nanoscopic cylindrical confinement. Phys. Rev. Lett. 2007, 98, 136103. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, W.; Yang, H.; Su, Z. Crystallization and orientation of syndiotactic polystyrene in nanorods. Macromolecules 2007, 40, 4244–4249. [Google Scholar] [CrossRef]
- Shin, K.; Woo, E.; Jeong, Y.G.; Kim, C.; Huh, J.; Kim, K.-W. Crystalline structures, melting, and crystallization of linear polyethylene in cylindrical nanopores. Macromolecules 2007, 40, 6617–6623. [Google Scholar] [CrossRef]
- Di Maio, E.; Iannace, S.; Sorrentino, L.; Nicolais, L. Isothermal crystallization in pcl/clay nanocomposites investigated with thermal and rheometric methods. Polymer 2004, 45, 8893–8900. [Google Scholar] [CrossRef]
- Zhang, Q.X.; Yu, Z.Z.; Yang, M.; Ma, J.; Mai, Y.W. Multiple melting and crystallization of nylon-66/montmorillonite nanocomposites. J. Polym. Sci. Part B 2003, 41, 2861–2869. [Google Scholar] [CrossRef]
- Mu, B.; Wang, Q.; Wang, H.; Jian, L. Nonisothermal crystallization kinetics of nylon 66/montmorillonite nanocomposites. J. Macromol. Sci. Part B 2007, 46, 1093–1104. [Google Scholar] [CrossRef]
- Nam, J.Y.; Sinha Ray, S.; Okamoto, M. Crystallization behavior and morphology of biodegradable polylactide/layered silicate nanocomposite. Macromolecules 2003, 36, 7126–7131. [Google Scholar] [CrossRef]
- Lincoln, D.M.; Vaia, R.A.; Krishnamoorti, R. Isothermal crystallization of nylon-6/montmorillonite nanocomposites. Macromolecules 2004, 37, 4554–4561. [Google Scholar] [CrossRef]
- Li, L.; Li, C.Y.; Ni, C.; Rong, L.; Hsiao, B. Structure and crystallization behavior of nylon 66/multi-walled carbon nanotube nanocomposites at low carbon nanotube contents. Polymer 2007, 48, 3452–3460. [Google Scholar] [CrossRef]
- Phang, I.Y.; Pramoda, K.; Liu, T.; He, C. Crystallization and melting behavior of polyester/clay nanocomposites. Polym. Int. 2004, 53, 1282–1289. [Google Scholar] [CrossRef]
- Wu, D.; Zhou, C.; Fan, X.; Mao, D.; Bian, Z. Morphology, crystalline structure and isothermal crystallization kinetics of polybutylene terephthalate/montmorillonite nanocomposites. Polym. Polym. Compos. 2005, 13, 61–71. [Google Scholar]
- Bilotti, E.; Fischer, H.; Peijs, T. Polymer nanocomposites based on needle-like sepiolite clays: Effect of functionalized polymers on the dispersion of nanofiller, crystallinity, and mechanical properties. J. Appl. Polym. Sci. 2008, 107, 1116–1123. [Google Scholar] [CrossRef]
- Xu, D.; Wang, Z. Role of multi-wall carbon nanotube network in composites to crystallization of isotactic polypropylene matrix. Polymer 2008, 49, 330–338. [Google Scholar] [CrossRef]
- Homminga, D.; Goderis, B.; Dolbnya, I.; Reynaers, H.; Groeninckx, G. Crystallization behavior of polymer/montmorillonite nanocomposites. Part I. Intercalated poly (ethylene oxide)/montmorillonite nanocomposites. Polymer 2005, 46, 11359–11365. [Google Scholar] [CrossRef]
- Chen, E.C.; Wu, T.M. Isothermal and nonisothermal crystallization kinetics of nylon 6/functionalized multi-walled carbon nanotube composites. J. Polym. Sci. Part B 2008, 46, 158–169. [Google Scholar] [CrossRef]
- Harrats, C.; Groeninckx, G. Features, questions and future challenges in layered silicates clay nanocomposites with semicrystalline polymer matrices. Macromol. Rapid Commun. 2008, 29, 14–26. [Google Scholar] [CrossRef]
- Guérin, G.; Prud’Homme, R.E. Thickness dependence of free-standing thin films. J. Polym. Sci. Part B 2007, 45, 10–17. [Google Scholar] [CrossRef]
- Rittigstein, P.; Torkelson, J.M. Polymer-Nanoparticle interfacial interactions in polymer nanocomposites: Confinement effects on glass transition temperature and suppression of physical aging. J. Polym. Sci. Part B 2006, 44, 2935–2943. [Google Scholar] [CrossRef]
- Pluta, M.; Jeszka, J.; Boiteux, G. Polylactide/montmorillonite nanocomposites: Structure, dielectric, viscoelastic and thermal properties. Eur. Polym. J. 2007, 43, 2819–2835. [Google Scholar] [CrossRef]
- Siegel, R.W. Exploring mesoscopia: The bold new world of nanostructures. Phys. Today 1993, 46. [Google Scholar] [CrossRef]
- Mayo, M.J.; Siegel, R.W.; Narayanasamy, A.; Nix, W.D. Mechanical properties of nanophase TiO2 as determined by nanoindentation. J. Mater. Res. 1990, 5, 1073–1082. [Google Scholar] [CrossRef]
- Mackay, M.E.; Tuteja, A.; Duxbury, P.M.; Hawker, C.J.; Van Horn, B.; Guan, Z.; Chen, G.; Krishnan, R. General strategies for nanoparticle dispersion. Science 2006, 311, 1740–1743. [Google Scholar] [CrossRef] [PubMed]
- Tuteja, A.; Mackay, M.E.; Hawker, C.J.; Van Horn, B. Effect of ideal, organic nanoparticles on the flow properties of linear polymers: Non-einstein-like behavior. Macromolecules 2005, 38, 8000–8011. [Google Scholar] [CrossRef]
- Merkel, T.; Freeman, B.; Spontak, R.; He, Z.; Pinnau, I.; Meakin, P.; Hill, A. Ultrapermeable, reverse-selective nanocomposite membranes. Science 2002, 296, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Kharchenko, S.B.; Douglas, J.F.; Obrzut, J.; Grulke, E.A.; Migler, K.B. Flow-Induced properties of nanotube-filled polymer materials. Nat. Mater. 2004, 3, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Krishnamoorti, R. Nonlinear viscoelastic properties of layered-silicate-based intercalated nanocomposites. Macromolecules 2003, 36, 4443–4451. [Google Scholar] [CrossRef]
- Jain, S.; Goossens, H.; Picchioni, F.; Magusin, P.; Mezari, B.; van Duin, M. Synthetic aspects and characterization of polypropylene-silica nanocomposites prepared via solid-state modification and sol-gel reactions. Polymer 2005, 46, 6666–6681. [Google Scholar] [CrossRef]
- Li, Y.; Shimizu, H. Novel morphologies of poly (phenylene oxide)(PPO)/polyamide 6 (PA6) blend nanocomposites. Polymer 2004, 45, 7381–7388. [Google Scholar] [CrossRef]
- Khatua, B.; Lee, D.J.; Kim, H.Y.; Kim, J.K. Effect of organoclay platelets on morphology of nylon-6 and poly (ethylene-ran-propylene) rubber blends. Macromolecules 2004, 37, 2454–2459. [Google Scholar] [CrossRef]
- Si, M.; Araki, T.; Ade, H.; Kilcoyne, A.; Fisher, R.; Sokolov, J.C.; Rafailovich, M.H. Compatibilizing bulk polymer blends by using organoclays. Macromolecules 2006, 39, 4793–4801. [Google Scholar] [CrossRef]
- Vo, L.T.; Giannelis, E.P. Compatibilizing poly(vinylidene fluoride)/nylon-6 blends with nanoclay. Macromolecules 2007, 40, 8271–8276. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, H.; Fu, Q. Kinetics-Controlled compatibilization of immiscible polypropylene/polystyrene blends using nano-SiO2 particles. Polymer 2004, 45, 1913–1922. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Q.; Fu, Q. Compatibilization of immiscible poly(propylene)/polystyrene blends using clay. Macromol. Rapid Commun. 2003, 24, 231–235. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Road Vehicles, and Tractors and Machinery for Agriculture and Forestry—Determination of Burning Behaviour of Interior Materials; International Organization for Standardization: Geneva, Switzerland, 2013; Volume ISO 3795:1989, p. 6. [Google Scholar]
- Stevens, M.; Bugnicourt, E.; Seignan, J. Harmonised fr regulations accelerate composites growth in public transportation. Reinf. Plast. 2008, 52, 40–41. [Google Scholar] [CrossRef]
- Intrater, J. Flame Retardant Polymer Nanocomposites; Morgan, A., Wilkie, C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007. [Google Scholar]
- Horrocks, A.R.; Kandola, B.K.; Nazare, S.; Padbury, S. Nanoclay-Flame retardant interactions in thermoplastic and thermosetting polymers. In MODEST; the MoDeSt Society: Villeurbanne, France, 2004. [Google Scholar]
- Zanetti, M.; Lomakin, S.; Camino, G. Polymer layered silicate nanocomposites. Macromol. Mater. Eng. 2000, 279, 1–9. [Google Scholar] [CrossRef]
- Hsiue, G.H.; Liu, Y.L.; Liao, H.H. Flame-Retardant epoxy resins: An approach from organic-inorganic hybrid nanocomposites. J. Polym. Sci. Part A 2001, 39, 986–996. [Google Scholar] [CrossRef]
- Ndoro, T.V.; Böhm, M.C.; Müller-Plathe, F. Interface and interphase dynamics of polystyrene chains near grafted and ungrafted silica nanoparticles. Macromolecules 2011, 45, 171–179. [Google Scholar] [CrossRef]
- Ndoro, T.V.; Voyiatzis, E.; Ghanbari, A.; Theodorou, D.N.; Böhm, M.C.; Müller-Plathe, F. Interface of grafted and ungrafted silica nanoparticles with a polystyrene matrix: Atomistic molecular dynamics simulations. Macromolecules 2011, 44, 2316–2327. [Google Scholar] [CrossRef]
- Liu, J.; Wu, Y.; Shen, J.; Gao, Y.; Zhang, L.; Cao, D. Polymer-Nanoparticle interfacial behavior revisited: A molecular dynamics study. Phys. Chem. Chem. Phys. 2011, 13, 13058–13069. [Google Scholar] [CrossRef] [PubMed]
- Ragesh, P.; Ganesh, V.A.; Naira, S.V.; Nair, A.S. A review on ‘self-cleaning and multifunctional materials’. J. Mater. Chem. A 2014, 2, 14773–14797. [Google Scholar] [CrossRef]
- Kota, A.K.; Choi, W.; Tuteja, A. Superomniphobic surfaces: Design and durability. MRS Bull. 2013, 38, 383–390. [Google Scholar] [CrossRef]
- Tuteja, A.; Choi, W.; Ma, M.L.; Mabry, J.M.; Mazzella, S.A.; Rutledge, G.C.; McKinley, G.H.; Cohen, R.E. Designing superoleophobic surfaces. Science 2007, 318, 1618–1622. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, B.; Jung, Y.C. Micro- and nanoscale characterization of hydrophobic and hydrophilic leaf surfaces. Nanotechnology 2006, 17, 2758–2772. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Barthlott, W. Diversity of structure, morphology and wetting of plant surfaces. Soft Matter 2008, 4, 1943–1963. [Google Scholar] [CrossRef]
- Ensikat, H.J.; Ditsche-Kuru, P.; Neinhuis, C.; Barthlott, W. Superhydrophobicity in perfection: The outstanding properties of the lotus leaf. Beilstein J. Nanotechnol. 2011, 2, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Yan, Y.Y. Characterisation of surface wettability based on nanoparticles. Nanoscale 2012, 4, 2202–2218. [Google Scholar] [CrossRef] [PubMed]
- Leng, B.X.; Shao, Z.Z.; de With, G.; Ming, W.H. Superoleophobic cotton textiles. Langmuir 2009, 25, 2456–2460. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.T.; Wu, F.L.; Chen, W.Y. Contact angle hysteresis and work of adhesion of oil droplets on nanosphere stacking layers. J. Phys. Chem. C 2009, 113, 13683–13688. [Google Scholar] [CrossRef]
- Wang, L.F.; Yang, S.Y.; Wang, J.; Wang, C.F.; Chen, L. Fabrication of superhydrophobic TPU film for oil-water separation based on electrospinning route. Mater. Lett. 2011, 65, 869–872. [Google Scholar] [CrossRef]
- Yang, H.T.; Jiang, P. Scalable fabrication of superhydrophobic hierarchical colloidal arrays. J. Colloid Interface Sci. 2010, 352, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Y.; Chen, S.; Tian, Y.; Feng, C.; Chen, L. Facile transformation of a native polystyrene (PS) film into a stable superhydrophobic surface via sol-gel process. Chem. Mater. 2008, 20, 1233–1235. [Google Scholar] [CrossRef]
- Gould, P. Eu funding bonanza begins. Mater. Today 2003, 6, 44–49. [Google Scholar] [CrossRef]
- Mats, L.; Bramwell, A.; Dupont, J.; Liu, G.J.; Oleschuk, R. Electrowetting on superhydrophobic natural (colocasia) and synthetic surfaces based upon fluorinated silica nanoparticles. Microelectron. Eng. 2015, 148, 91–97. [Google Scholar] [CrossRef]
- Perl, E.E.; McMahon, W.E.; Farrell, R.M.; DenBaars, S.P.; Speck, J.S.; Bowers, J.E. Surface structured optical coatings with near-perfect broadband and wide-angle antireflective properties. Nano Lett. 2014, 14, 5960–5964. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Du, M.; Zheng, Z.; Song, Y.H.; Zheng, Q. A facile, multifunctional, transparent, and superhydrophobic coating based on a nanoscale porous structure spontaneously assembled from branched silica nanoparticles. Adv. Mater. Interfaces 2015, 2. [Google Scholar] [CrossRef]
- Jiao, L.Y.; Zhang, L.; Wang, X.R.; Diankov, G.; Dai, H.J. Narrow graphene nanoribbons from carbon nanotubes. Nature 2009, 458, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yan, L.L.; Li, Q.; Wang, C.F.; Chen, S. Controllable synthesis of new polymerizable macrosurfactants via cctp and raft techniques and investigation of their performance in emulsion polymerization. Langmuir 2010, 26, 1724–1733. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.L.; Gao, D. Transparent superhydrophobic and highly oleophobic coatings. Faraday Discuss. 2010, 146, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Cha, N.G.; Echegoyen, Y.; Kim, T.H.; Park, J.G.; Busnaina, A.A. Convective assembly and dry transfer of nanoparticles using hydrophobic/hydrophilic monolayer templates. Langmuir 2009, 25, 11375–11382. [Google Scholar] [CrossRef] [PubMed]
- Lassiaz, S.; Galarneau, A.; Trens, P.; Labarre, D.; Mutin, H.; Brunel, D. Organo-Lined alumina surface from covalent attachment of alkylphosphonate chains in aqueous solution. New J. Chem. 2010, 34, 1424–1435. [Google Scholar] [CrossRef]
- Sas, I.; Gorga, R.E.; Joines, J.A.; Thoney, K.A. Literature review on superhydrophobic self-cleaning surfaces produced by electrospinning. J. Polym. Sci. Part B 2012, 50, 824–845. [Google Scholar] [CrossRef]
- Menini, R.; Farzaneh, M. Production of superhydrophobic polymer fibers with embedded particles using the electrospinning technique. Polym. Int. 2008, 57, 77–84. [Google Scholar] [CrossRef]
- Asmatulu, R.; Ceylan, M.; Nuraje, N. Study of superhydrophobic electrospun nanocomposite fibers for energy systems. Langmuir 2011, 27, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.J.; Wu, J.; Xu, Z.K. Electrospraying/electrospinning of poly(γ-stearyl-l-glutamate): Formation of surfaces with superhydrophobicity. Chin. J. Polym. Sci. 2009, 27, 115–120. [Google Scholar] [CrossRef]
- Singh, A.; Steely, L.; Allcock, H.R. Poly[bis(2,2,2-trifluoroethoxy)phosphazene] superhydrophobic nanofibers. Langmuir 2005, 21, 11604–11607. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Sanz, M.; Lopez-Rubio, A.; Lagaron, J.M. High-Barrier coated bacterial cellulose nanowhiskers films with reduced moisture sensitivity. Carbohydr. Polym. 2013, 98, 1072–1082. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Mammen, L.; Butt, H.J.; Vollmer, D. Candle soot as a template for a transparent robust superamphiphobic coating. Science 2012, 335, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.; Kim, M.; Kim, D.H. Candle-Based process for creating a stable superhydrophobic surface. Carbon 2014, 68, 583–596. [Google Scholar] [CrossRef]
- Huang, Y.H. Hydrophobic and Lipophobic Coating Material. U.S. Patent 20120219801 A1, 30 August 2012. [Google Scholar]
- Valpey, R.S.; Jones, M.A. Process and Composition for Producing Self-Cleaning Surfaces from Aqueous Systems. International Patent WO2004037944, 27 March 2004. [Google Scholar]
- Hu, J.; Zou, H.; Liu, G.; Li, F.; Lin, S.; Zhang, G.; Tu, Y.; Hu, M. Stable and Transparent Superhydrophobic Coating or Super Amphiphobic Its Preparation Method and Application. Chinese Patent CN103436138 B, 28 October 2015. [Google Scholar]
- Kanagasabapathy, S.; Baumgart, R.J.; Dituro, M.A.; Su, W.C.; Lockwood, F.E. Hydrophobic Self-Cleaning Coating Composition. International Patent WO2008153687 A3, 29 January 2009. [Google Scholar]
- Zischka, M.; Spanring, J.; Reischl, M. Method for Producing a Coated Packaging Material, and Packaging Material Having at least One Barrier Layer for Hydrophobic Compounds. International Patent WO2012168433 A1, 13 December 2012. [Google Scholar]
- Lee, J.H.; Jo, K.S.; Lee, S.W.; Park, M.Y.; Lee, S.E.; Lee, K.W.; Kim, J.H.; Lim, H.K.; Lee, S.S. Food Packing Material Having Hydrophobicity, Manufacturing Method and Mold Thereof. International Patent WO2013012123 A1, 24 January 2013. [Google Scholar]
- Carew, D.; Hamilton, S.P. Hydrophobic Packaging Material including a Sulphite Salt. International Patent WO2008025085 A1, 6 March 2008. [Google Scholar]
- Lindgren, E.; Larsson, K.; Sundstrand, S.; Andersson, A. Packaging Material Process for Producing Same and Use Thereof. U.S. Patent 5603997 A, 18 February 1997. [Google Scholar]
- Sajot, N.; Pollacchi, B.; Sellak, S. Method of Gluing Hydrophobic and Oleophobic Substrates Which Are Intended for Packaging. U.S. Patent 20080221247 A1, 11 September 2008. [Google Scholar]
- Beydoun, D.; Amal, R.; Low, G.; McEvoy, S. Role of nanoparticles in photocatalysis. J. Nanopart. Res. 1999, 1, 439–458. [Google Scholar] [CrossRef]
- Yoon, H.; Joshi, B.; Na, S.-H.; Yoon, S.S. Antibacterial activity and photocatalysis of electrosprayed titania films. J. Electrochem. Soc. 2012, 159, H823–H827. [Google Scholar] [CrossRef]
- Tudor, I.A.; Petriceanu, M.; Piticescu, R.-R.; Piticescu, R.M.; Predescu, C. Hydrothermal synthesis of doped zno and tio2 nanomaterials: Oppurtunities for textile applications. UPB Sci. Bull. Ser. B 2014, 76, 207–215. [Google Scholar]
- Serrano, C.; Bugnicourt, E.; Niculescu, C.; Ghituleasa, C.; Dumitrescu, I.; Sobetkii, A.; Mocioiu, A.M.; Petriceanu, M.; Piticescu, R.M. Manucoat. Integrated manufacturing process of active textiles by deposition of doped TiO2. In Proceedings of the ANNIC 2015 Applied Nanotechnology and Nanoscience International Conference, Paris, France, 5–7 November 2015. [Google Scholar]
- Cantarella, M.; Sanz, R.; Buccheri, M.A.; Ruffino, F.; Rappazzo, G.; Scalese, S.; Impellizzeri, G.; Romano, L.; Privitera, V. Immobilization of nanomaterials in pmma composites for photocatalytic removal of dyes, phenols and bacteria from water. J. Photochem. Photobiol. A 2016, 321, 1–11. [Google Scholar] [CrossRef]
- Hu, K.; Kulkarni, D.D.; Choi, I.; Tsukruk, V.V. Graphene-Polymer nanocomposites for structural and functional applications. Prog. Polym. Sci. 2014, 39, 1934–1972. [Google Scholar] [CrossRef]
- Yano, K.; Usuki, A.; Okada, A. Synthesis and properties of polyimide-clay hybrid films. J. Polym. Sci. Part A 1997, 35, 2289–2294. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Ng, P.K.W. Natural biopolymer-based nanocomposite films for packaging applications. Crit. Rev. Food Sci. Nutr. 2007, 47, 411–433. [Google Scholar] [CrossRef] [PubMed]
- Nazarenko, S.; Meneghetti, P.; Julmon, P.; Olson, B.G.; Qutubuddin, S. Gas barrier of polystyrene montmorillonite clay nanocomposites: Effect of mineral layer aggregation. J. Polym. Sci. Part B 2007, 45, 1733–1753. [Google Scholar] [CrossRef]
- Choi, W.J.; Kim, H.J.; Yoon, K.H.; Kwon, O.H.; Hwang, C.I. Preparation and barrier property of poly(ethylene terephthalate)/clay nanocomposite using clay-supported catalyst. J. Appl. Polym. Sci. 2006, 100, 4875–4879. [Google Scholar] [CrossRef]
- Sanchez-Garcia, M.D.; Gimenez, E.; Lagaron, J.M. Novel pet nanocomposites of interest in food packaging applications and comparative barrier performance with biopolyester nanocomposites. J. Plast. Film Sheeting 2007, 23, 133–148. [Google Scholar] [CrossRef]
- Lagaron, J.M.; Cabedo, L.; Cava, D.; Feijoo, J.L.; Gavara, R.; Gimenez, E. Improving packaged food quality and safety. Part 2: Nanocomposites. Food Addit. Contam. 2005, 22, 994–998. [Google Scholar] [CrossRef] [PubMed]
- Thellen, C.; Orroth, C.; Froio, D.; Ziegler, D.; Lucciarini, J.; Farrell, R.; D’Souza, N.A.; Ratto, J.A. Influence of montmorillonite layered silicate on plasticized poly(l-lactide) blown films. Polymer 2005, 46, 11716–11727. [Google Scholar] [CrossRef]
- Sinha Ray, S.; Yamada, K.; Okamoto, M.; Fujimoto, Y.; Ogami, A.; Ueda, K. New polylactide/layered silicate nanocomposites. 5. Designing of materials with desired properties. Polymer 2003, 44, 6633–6646. [Google Scholar] [CrossRef]
- Osman, M.A.; Rupp, J.E.P.; Suter, U.W. Gas permeation properties of polyethylene-layered silicate nanocomposites. J. Mater. Chem. 2005, 15, 1298–1304. [Google Scholar] [CrossRef]
- Lotti, C.; Isaac, C.S.; Branciforti, M.C.; Alves, R.M.V.; Liberman, S.; Bretas, R.E.S. Rheological, mechanical and transport properties of blown films of high density polyethylene nanocomposites. Eur. Polym. J. 2008, 44, 1346–1357. [Google Scholar] [CrossRef]
- Dadbin, S.; Noferesti, M.; Frounchi, M. Oxygen barrier LDPE/LLDPE/organoclay nano-composite films for food packaging. Macromol. Symp. 2008, 274, 22–27. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Bunch, J.S.; Verbridge, S.S.; Alden, J.S.; van der Zande, A.M.; Parpia, J.M.; Craighead, H.G.; McEuen, P.L. Impermeable atomic membranes from graphene sheets. Nano Lett. 2008, 8, 2458–2462. [Google Scholar] [CrossRef] [PubMed]
- Leenaerts, O.; Partoens, B.; Peeters, F.M. Graphene: A perfect nanoballoon. Appl. Phys. Lett. 2008, 93, 193107. [Google Scholar] [CrossRef]
- Yoo, B.M.; Shin, H.J.; Yoon, H.W.; Park, H.B. Graphene and graphene oxide and their uses in barrier polymers. J. Appl. Polym. Sci. 2014, 131, 39628. [Google Scholar] [CrossRef]
- Huang, P.Y.; Ruiz-Vargas, C.S.; van der Zande, A.M.; Whitney, W.S.; Levendorf, M.P.; Kevek, J.W.; Garg, S.; Alden, J.S.; Hustedt, C.J.; Zhu, Y.; et al. Grains and grain boundaries in single-layer graphene atomic patchwork quilts. Nature 2011, 469, 389–392. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Suk, J.W.; Piner, R.D.; An, J.; Ruoff, R.S. Mechanical properties of monolayer graphene oxide. ACS Nano 2010, 4, 6557–6564. [Google Scholar] [CrossRef] [PubMed]
- Compton, O.C.; Kim, S.; Pierre, C.; Torkelson, J.M.; Nguyen, S.T. Crumpled graphene nanosheets as highly effective barrier property enhancers. Adv. Mater. 2010, 22, 4759–4763. [Google Scholar] [CrossRef] [PubMed]
- Kuila, T.; Bose, S.; Mishra, A.K.; Khanra, P.; Kim, N.H.; Lee, J.H. Effect of functionalized graphene on the physical properties of linear low density polyethylene nanocomposites. Polym. Test. 2012, 31, 31–38. [Google Scholar] [CrossRef]
- Shim, S.H.; Kim, K.T.; Lee, J.U.; Jo, W.H. Facile method to functionalize graphene oxide and its application to poly(ethylene terephthalate)/graphene composite. ACS Appl. Mater. Interfaces 2012, 4, 4184–4191. [Google Scholar] [CrossRef] [PubMed]
- Al-Jabareen, A.; Al-Bustami, H.; Harel, H.; Marom, G. Improving the oxygen barrier properties of polyethylene terephthalate by graphite nanoplatelets. J. Appl. Polym. Sci. 2013, 128, 1534–1539. [Google Scholar] [CrossRef]
- Pinto, A.M.; Cabral, J.; Tanaka, D.A.P.; Mendes, A.M.; Magalhães, F.D. Effect of incorporation of graphene oxide and graphene nanoplatelets on mechanical and gas permeability properties of poly(lactic acid) films. Polym. Int. 2013, 62, 33–40. [Google Scholar] [CrossRef]
- Song, P.a.; Yu, Y.; Zhang, T.; Fu, S.; Fang, Z.; Wu, Q. Permeability, viscoelasticity, and flammability performances and their relationship to polymer nanocomposites. Ind. Eng. Chem. Res. 2012, 51, 7255–7263. [Google Scholar] [CrossRef]
- NANOMASTER. Graphene based thermoplastic masterbatches for conventional and additive manufactoring processes. In Nanomaster Periodic Report Summary; Community Research and Development Information Service: Lodon, UK, 2013. [Google Scholar]
- Casiraghi, C.; Robertson, J.; Ferrari, A.C. Diamond-Like carbon for data and beer storage. Mater. Today 2007, 10, 44–53. [Google Scholar] [CrossRef]
- Castillo, L.; López, O.; López, C.; Zaritzky, N.; García, M.A.; Barbosa, S.; Villar, M. Thermoplastic starch films reinforced with talc nanoparticles. Carbohydr. Polym. 2013, 95, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. Use of the electrohydrodynamic process to develop active/bioactive bilayer films for food packaging applications. Food Hydrocoll. 2016, 55, 11–18. [Google Scholar] [CrossRef]
- Rao, S.; Upadhyay, J.; Das, R. Manufacturing and characterization of multifunctional polymer-reduced graphene oxide nanocomposites. In Fillers and Reinforcements for Advanced Nanocomposites; Woodhead Publishing: Camebridge, UK, 2015; pp. 157–232. [Google Scholar]
- Wen, P.; Zhu, D.H.; Feng, K.; Liu, F.J.; Lou, W.Y.; Li, N.; Zong, M.H.; Wu, H. Fabrication of electrospun polylactic acid nanofilm incorporating cinnamon essential oil/β-cyclodextrin inclusion complex for antimicrobial packaging. Food Chem. 2016, 196, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; López-Rubio, A.; Sentandreu, E.; Lagaron, J.M. Development of multilayer corn starch-based food packaging structures containing β-carotene by means of the electro-hydrodynamic processing. Starch/Staerke 2015, 68, 603–610. [Google Scholar] [CrossRef]
- Schmidt, M.C.; Loibl, F.; Muller, M.; Oehr, C.; Hirth, T. The emptying behavior of highly viscous liquids. Part I: Polymeric surfaces and plasma coatings. J. Adhes. Sci. Technol. 2012, 26, 2449–2467. [Google Scholar] [CrossRef]
- Bellanger, H.; Darmanin, T.; Taffin de Givenchy, E.; Guittard, F. Chemical and physical pathways for the preparation of superoleophobic surfaces and related wetting theories. Chem. Rev. 2014, 114, 2694–2716. [Google Scholar] [CrossRef] [PubMed]
- Nishino, T.; Meguro, M.; Nakamae, K.; Matsushita, M.; Ueda, Y. The lowest surface free energy based on –CF3 alignment. Langmuir 1999, 15, 4321–4323. [Google Scholar] [CrossRef]
- Teshima, K.; Sugimura, H.; Inoue, Y.; Takai, O.; Takano, A. Ultra-Water-Repellent poly(ethylene terephthalate) substrates. Langmuir 2003, 19, 10624–10627. [Google Scholar] [CrossRef]
- Teshima, K.; Sugimura, H.; Inoue, Y.; Takai, O.; Takano, A. Transparent ultra water-repellent poly(ethylene terephthalate) substrates fabricated by oxygen plasma treatment and subsequent hydrophobic coating. Appl. Surf. Sci. 2005, 244, 619–622. [Google Scholar] [CrossRef]
- LiquiGlide. Available online: http://liquiglide.com (accessed on 23 Februay 2016).
- Smith, J.D.; Dhiman, R.; Anand, S.; Reza-Garduno, E.; Cohen, R.E.; McKinley, G.H.; Varanasi, K.K. Droplet mobility on lubricant-impregnated surfaces. Soft Matter 2013, 9, 1772–1780. [Google Scholar] [CrossRef]
- Loibl, F.; Schmidt, M.C.; Auer-Seidl, A.; Kirchner, C.; Holtz, C.; Muller, K.; Stramm, C.; Langowski, H.C. The emptying behaviour of highly viscous liquids. Part II: Development of test methods and evaluation of untreated and coated films. J. Adhes. Sci. Technol. 2012, 26, 2469–2503. [Google Scholar] [CrossRef]
- Merck. Merck Presents Organic Photovoltaic Materials at Expo 2015 in Milan; Merck KGaA: Darmstadt, Germany, 2015. [Google Scholar]
- Belectric. Gray Modules: New Dimension in Organic Photovoltaics for Buildings; BELECTRIC OPV GmbH: Nuremberg, Germany, 2015. [Google Scholar]
- European Commission DG, Environment News Alert Service. Science for Environment Policy; SCU, Ed.; the University of the West of England: Bristol, UK, 2015. [Google Scholar]
- Brongersma, M.L.; Cui, Y.; Fan, S. Light management for photovoltaics using high-index nanostructures. Nat. Mater. 2014, 13, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Shrotriya, V.; Huang, J.; Yao, Y.; Moriarty, T.; Emery, K.; Yang, Y. High-Efficiency solution processable polymer photovoltaic cells by self-organization of polymer blends. Nat. Mater. 2005, 4, 864–868. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, S.H.; Lee, H.H.; Lee, K.; Ma, W.; Gong, X.; Heeger, A.J. New architecture for high-efficiency polymer photovoltaic cells using solution-based titanium oxide as an optical spacer. Adv. Mater. 2006, 18, 572–576. [Google Scholar] [CrossRef]
- Wang, E.; Wang, L.; Lan, L.; Luo, C.; Zhuang, W.; Peng, J.; Cao, Y. High-Performance polymer heterojunction solar cells of a polysilafluorene derivative. Appl. Phys. Lett. 2008, 92, 33307. [Google Scholar] [CrossRef]
- Reese, M.O.; Morfa, A.J.; White, M.S.; Kopidakis, N.; Shaheen, S.E.; Rumbles, G.; Ginley, D.S. Pathways for the degradation of organic photovoltaic p3ht: Pcbm based devices. Sol. Energy Mater. Sol. C 2008, 92, 746–752. [Google Scholar] [CrossRef]
- Pei, J.; Tao, J.; Zhou, Y.; Dong, Q.; Liu, Z.; Li, Z.; Chen, F.; Zhang, J.; Xu, W.; Tian, W. Efficiency enhancement of polymer solar cells by incorporating a self-assembled layer of silver nanodisks. Sol. Energy Mater. Sol. C 2011, 95, 3281–3286. [Google Scholar] [CrossRef]
- Chen, F.-C.; Wu, J.-L.; Lee, C.-L.; Hong, Y.; Kuo, C.-H.; Huang, M.H. Plasmonic-Enhanced polymer photovoltaic devices incorporating solution-processable metal nanoparticles. Appl. Phys. Lett. 2009, 95, 013305. [Google Scholar] [CrossRef]
- Tang, Z.; Liu, Q.; Chen, Q.; Khatri, I.; Shirai, H. Plasmonic-Enhanced crystalline silicon/organic heterojunction cells by incorporating gold nanoparticles. Phys. Status Solidi A 2014, 211, 1179–1183. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Self-Cleaning and next generation anti-fog surfaces and coatings. Macromol. Rapid Commun. 2008, 29, 455–466. [Google Scholar] [CrossRef]
- Gray, A. Self-Cleaning Hydrophobic Nano Coating for Solar Panel Glass Generating Great Results for Nanoshell. Available online: http://www.pv-magazine.com/services/press-releases/details/beitrag/self-cleaning-hydrophobic-nano-coating-for-solar-panel-glass-generating-great-results-for-nanoshell_100005577/#ixzz3Sr9qqsGv (accessed on 18 February 2016).
- Friday, L. Self-Cleaning System Boosts Efficiency of Solar Panels. Available online: http://www.bu.edu/today/2014/self-cleaning-system-boosts-efficiency-of-solar-panels/ (accessed on 18 February 2016).
- Bullis, K. Self-Cleaning Solar Panels. Available online: http://www.technologyreview.com/news/420524/self-cleaning-solar-panels/ (accessed on 18 February 2016).
- Zhao, X.Y.; Yang, W.W.; Lie, C.; Wang, X.Z.; Lim, S.L.; Qi, D.C.; Wang, R.; Gao, Z.Q.; Mi, B.X.; Chen, Z.K.; et al. Effects of damkholer number of evaporation on the morphology of active layer and the performance of organic heterojunction solar cells fabricated by electrospray method. Sol. Energy Mater. Sol. C 2015, 134, 140–147. [Google Scholar] [CrossRef]
- Yoon, H.; Mali, M.G.; Kim, M.W.; Al-Deyab, S.S.; Yoon, S.S. Electrostatic spray deposition of transparent tungsten oxide thin-film photoanodes for solar water splitting. Catal. Today 2016, 260, 89–94. [Google Scholar] [CrossRef]
- Liu, J.; Fu, X.; Cao, D.P.; Mao, L.; Wang, J.; Mu, D.H.; Mi, B.X.; Zhao, B.M.; Gao, Z.Q. Stacked graphene-TiO2 photoanode via electrospray deposition for highly efficient dye-sensitized solar cells. Organ. Electron. Phys. Mater. Appl. 2015, 23, 158–163. [Google Scholar] [CrossRef]
- Ranjgar, A.; Norouzi, R.; Zolanvari, A.; Sadeghi, H. Characterization and optical absorption properties of plasmonic nanostructured thin films. Armen. J. Phys. 2013, 6, 198–203. [Google Scholar]
- Pavlidou, S.; Papaspyrides, C. A review on polymer-layered silicate nanocomposites. Prog. Polym. Sci. 2008, 33, 1119–1198. [Google Scholar] [CrossRef]
- Kotal, M.; Bhowmick, A.K. Polymer nanocomposites from modified clays: Recent advances and challenges. Prog. Polym. Sci. 2015, 51, 127–187. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Nanotechnologies—Nanomaterial Risk Evaluation; International Organization for Standardization: Geneva, Switzerland, 2011; Volume ISO/TR 13121:2011, p. 58. [Google Scholar]
- Poland, C.A.; Read, S.A.K.; Varet, J.; Carse, G.; Christensen, F.M.; Hankin, S.M. Dermal Absorption of Nanomaterials; Danish Ministry of the Environment: Copenhagen, Denmark, 2013. [Google Scholar]
- Störmer, A.; Bott, J.; Kemmer, D.; Franz, R. Critical review of the migration potential of nanoparticles in food contact plastics. Trends Food Sci. Technol. 2017, 63, 39–50. [Google Scholar] [CrossRef]
- Duncan, T.V. Release of engineered nanomaterials from polymer nanocomposites: The effect of matrix degradation. ACS Appl. Mater. Interfaces 2015, 7, 20–39. [Google Scholar] [CrossRef] [PubMed]
- Duncan, T.V.; Pillai, K. Release of engineered nanomaterials from polymer nanocomposites: Diffusion, dissolution, and desorption. ACS Appl. Mater. Interfaces 2015, 7, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Heitbrink, W.A.; Lo, L.-M.; Farwick, D.R. Evaluation of Enclosing Hood and Downflow Room for Nanocomposite Manufacturing; EPHB-356-16a; U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention: Cincinnati, OH, USA, 2012.
- Heitbrink, W.; Lo, L.-M.; Garcia, A. Case Study: Particle Emissions from the Processes of Machining Nanocomposites; EPHB 356-19a; National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 2013.
- Nowack, B.; David, R.M.; Fissan, H.; Morris, H.; Shatkin, J.A.; Stintz, M.; Zepp, R.; Brouwer, D. Potential release scenarios for carbon nanotubes used in composites. Environ. Int. 2013, 59, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Froggett, S.J.; Clancy, S.F.; Boverhof, D.R.; Canady, R.A. A review and perspective of existing research on the release of nanomaterials from solid nanocomposites. Part. Fibre Toxicol. 2014, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Methner, M.; Crawford, C.; Geraci, C. Evaluation of the potential airborne release of carbon nanofibers during the preparation, grinding, and cutting of epoxy-based nanocomposite material. J. Occup. Environ. Hyg. 2012, 9, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Ogura, I.; Kotake, M.; Shigeta, M.; Uejima, M.; Saito, K.; Hashimoto, N.; Kishimoto, A. Potential release of carbon nanotubes from their composites during grinding. J. Phys. Conf. Ser. 2013, 429, 012049. [Google Scholar] [CrossRef]
- Bello, D.; Wardle, B.L.; Yamamoto, N.; Guzman DeVilloria, R.; Garcia, E.J.; Hart, A.J.; Ahn, K.; Ellenbecker, M.J.; Hallock, M. Exposure to nanoscale particles and fibers during machining of hybrid advanced composites containing carbon nanotubes. J. Nanopart. Res. 2009, 11, 231–249. [Google Scholar] [CrossRef]
- Lo, L.-M.; Dunn, K.H.; Hammond, D.; Marlow, D.; Topmiller, J.; Tsai, C.S.J.; Ellenbecker, M.; Huang, C.-C. Evaluation of Engineering Controls in a Manufacturing Facility Producing Carbon Nanotube-Based Products; EPHB-356-13a; U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention: Cincinnati, OH, USA, 2012.
- Bello, D.; Wardle, B.L.; Zhang, J.; Yamamoto, N.; Santeufemio, C.; Hallock, M.; Virji, M.A. Characterization of exposures to nanoscale particles and fibers during solid core drilling of hybrid carbon nanotube advanced composites. Int. J. Occup. Environ. Health 2010, 16, 434–450. [Google Scholar] [CrossRef] [PubMed]
- Wohlleben, W.; Brill, S.; Meier, M.W.; Mertler, M.; Cox, G.; Hirth, S.; Von Vacano, B.; Strauss, V.; Treumann, S.; Wiench, K.; et al. On the lifecycle of nanocomposites: Comparing released fragments and their in vivo hazards from three release mechanisms and four nanocomposites. Small 2011, 7, 2384–2395. [Google Scholar] [CrossRef] [PubMed]
- Schlagenhauf, L.; Buerki-Thurnherr, T.; Kuo, Y.Y.; Wichser, A.; Nüesch, F.; Wick, P.; Wang, J. Carbon nanotubes released from an epoxy-based nanocomposite: Quantification and particle toxicity. Environ. Sci. Technol. 2015, 49, 10616–10623. [Google Scholar] [CrossRef] [PubMed]
- Schlagenhauf, L.; Kianfar, B.; Buerki-Thurnherr, T.; Kuo, Y.Y.; Wichser, A.; Nüesch, F.; Wick, P.; Wang, J. Weathering of a carbon nanotube/epoxy nanocomposite under uv light and in water bath: Impact on abraded particles. Nanoscale 2015, 7, 18524–18536. [Google Scholar] [CrossRef] [PubMed]
- Cena, L.G.; Peters, T.M. Characterization and control of airborne particles emitted during production of epoxy/carbon nanotube nanocomposites. J. Occup. Environ. Hyg. 2011, 8, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Nguyen, T.; Pellegrin, B.; Holbrook, R.D.; Zhao, M.; Chin, J. Fate of graphene in polymer nanocomposite exposed to UV radiation. J. Phys. 2011, 304, 012063. [Google Scholar] [CrossRef]
- Wohlleben, W.; Meier, M.W.; Vogel, S.; Landsiedel, R.; Cox, G.; Hirth, S.; Tomović, Z. Elastic CNT-polyurethane nanocomposite: Synthesis, performance and assessment of fragments released during use. Nanoscale 2013, 5, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.F., Jr.; Wu, Z.; Mitra, S.; Shaw, P.K.; Holian, A. Effect of MWCNT size, carboxylation, and purification on in vitro and in vivo toxicity, inflammation and lung pathology. Part. Fibre Toxicol. 2013, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Ma-Hock, L.; Strauss, V.; Treumann, S.; Küttler, K.; Wohlleben, W.; Hofmann, T.; Gröters, S.; Wiench, K.; van Ravenzwaay, B.; Landsiedel, R. Comparative inhalation toxicity of multi-wall carbon nanotubes, graphene, graphite nanoplatelets and low surface carbon black. Part. Fibre Toxicol. 2013, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Grosse, Y.; Loomis, D.; Guyton, K.Z.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Scoccianti, C.; Mattock, H.; et al. Carcinogenicity of fluoro-edenite, silicon carbide fibres and whiskers, and carbon nanotubes. Lancet Oncol. 2014, 15, 1427–1428. [Google Scholar] [CrossRef]
- Koponen, I.K.; Jensen, K.A.; Schneider, T. Sanding dust from nanoparticle-containing paints: Physical characterisation. J. Phys. 2009, 151, 012048. [Google Scholar] [CrossRef]
| Model | Filler Type | Particle Geometry | Formulas | Reference |
|---|---|---|---|---|
| Nielsen | Ribbon a |  | (P0/P)(1 − φ) = 1 + αφ/2 | [134] |
| Cussler (Regular array) | Ribbon a |  | (P0/P)(1 − φ) = (1 + αφ)2/4 | [135] |
| Cussler (Random array) | Ribbon a |  | (P0/P)(1 − φ) = (1 + αφ/3)2 | [135] |
| Gusev and Lusti | Disk b |  | (P0/P)(1 − φ) = exp[(αφ/3.47)0.71] | [136] |
| Fredrickson and Bicerano | Disk b |  | (P0/P)(1 − φ) = 4(1 + x + 0.1245x2)/(2 + x)2 where x = αφ/2ln(α/2) | [137] |
| Bharadwaj | Disk b |  | (P0/P)(1 − φ) = 1 + 0.667αφ(S + (1/2)) where S = orientation factor (from −1/2 to 1) | [138] |
| Polymer | Nanofiller | Tg Change | References |
|---|---|---|---|
| Polystyrene | SWCNT | 3 | [157] |
| Polycarbonate | SiC (0.5–1.5 wt %) (20–60 nm particles) | No change | [159] |
| Poly(vinyl chloride) | Exfoliated clay (MMT) (<10 wt %) | 1 to 3 | [160] |
| Poly(dimethyl siloxane) | Silica (2–3 nm) | 10 | [161] |
| Poly(propylene carbonate) | Nanoclay (4 wt %) | 13 | [162] |
| Poly(methyl methacrylate) | Nanoclay (2.5–15.1 wt %) | 4–13 | [163] |
| Polyimide | MWCNT (0.25–6.98 wt %) | 4 to 8 | [164] |
| Polystyrene | Nanoclay (5 wt %) | 6.7 | [165] |
| Natural rubber | Nanoclay (5 wt %) | 3 | [167] |
| Poly(butylene terephthalate) | Mica (3 wt %) | 6 | [168] |
| Polylactide | Nanoclay (3 wt %) | 1 to 4 | [158] |
| Polymer Matrix | Filler | Clay (wt %) | P(O2) | P(H2O) | References |
|---|---|---|---|---|---|
| PS | Modified montmorillonite | 16.7 | 2.8 | [264] | |
| PET | Modified montmorillonite | 5 | 15.6 | 1.2 | [265] |
| Modified montmorillonite | 5 | 2.23 | 1.15 | [266] | |
| EVOH | Kaolinite | 5 | 3–4 | 1.2 | [267] |
| PLA | Montmorillonite | 5 | 1.16 | 1.21 | [266] |
| Modified montmorillonite | 5 | 1.2–1.9 | 1.7–2 | [268] | |
| Mica | 4 | 2.8 | [269] | ||
| PHB | Kaolinite | 5 | 1.26 | 1.06 | [266] |
| HDPE | Modified montmorillonite | 4 | 1.2–1.7 | [270] | |
| Modified montmorillonite | 5 | 2.8–2.9 | 1.8–2.4 | [271] | |
| LDPE | Modified montmorillonite | 4.76 | 2.2 | [272] |
| Polymer Matrix | Type of Graphene | Preparation Method | Maximum Fraction | Reference |
|---|---|---|---|---|
| PS | GO | Melting | 2.27 vol % | [280] |
| LLPDE | Functionalized graphene | Solution mixing | 5 wt % | [281] |
| PET | Functionalized GO | Solution mixing | 3 wt% | [282] |
| Reduced GO | Melting | 1.5 wt% | [283] | |
| PLA | GO, graphene | Solution mixing | 0.6 wt% | [284] |
| PP | Reduced GO | Melting | 1 wt % | [285] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Müller, K.; Bugnicourt, E.; Latorre, M.; Jorda, M.; Echegoyen Sanz, Y.; Lagaron, J.M.; Miesbauer, O.; Bianchin, A.; Hankin, S.; Bölz, U.; et al. Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields. Nanomaterials 2017, 7, 74. https://doi.org/10.3390/nano7040074
Müller K, Bugnicourt E, Latorre M, Jorda M, Echegoyen Sanz Y, Lagaron JM, Miesbauer O, Bianchin A, Hankin S, Bölz U, et al. Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields. Nanomaterials. 2017; 7(4):74. https://doi.org/10.3390/nano7040074
Chicago/Turabian StyleMüller, Kerstin, Elodie Bugnicourt, Marcos Latorre, Maria Jorda, Yolanda Echegoyen Sanz, José M. Lagaron, Oliver Miesbauer, Alvise Bianchin, Steve Hankin, Uwe Bölz, and et al. 2017. "Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields" Nanomaterials 7, no. 4: 74. https://doi.org/10.3390/nano7040074








