Magnetron-Sputtered Long-Term Superhydrophilic Thin Films for Use in Solid-State Cooling Devices
Abstract
:1. Introduction
2. Materials and Methods
2.1. Thin Films Preparation
2.2. Characterization
3. Results and Discussion
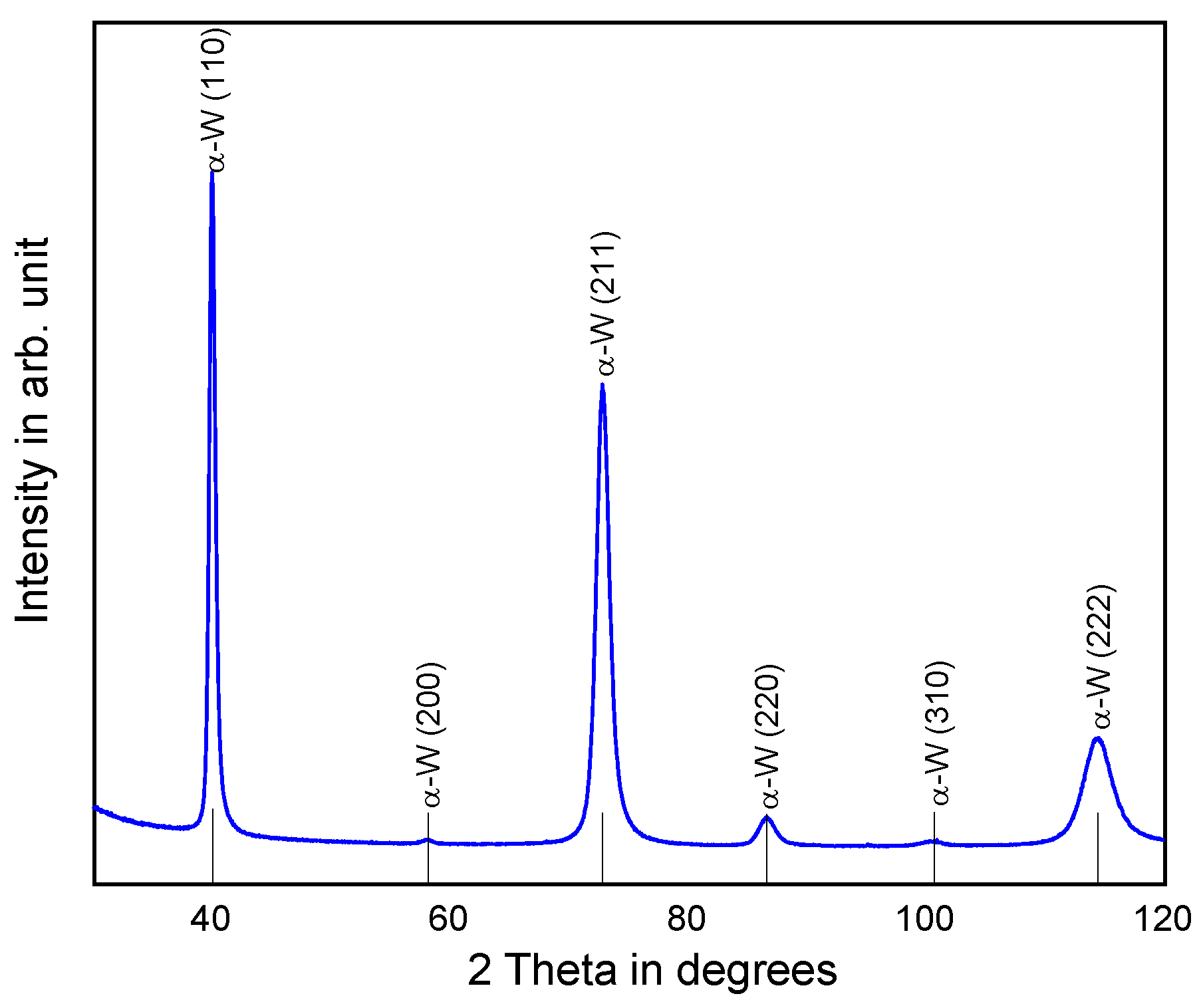
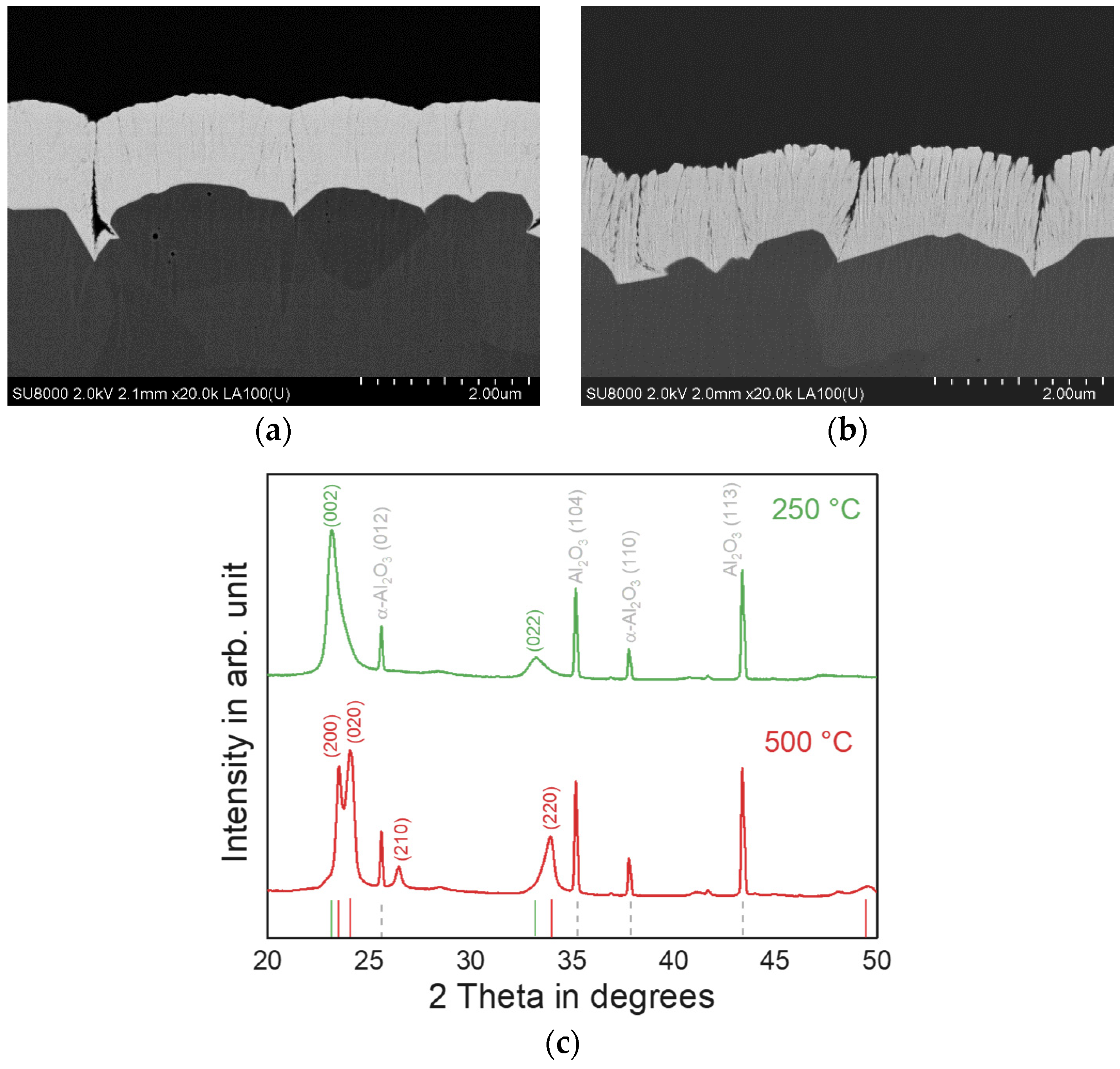
3.1. Tungsten Top Electrodes
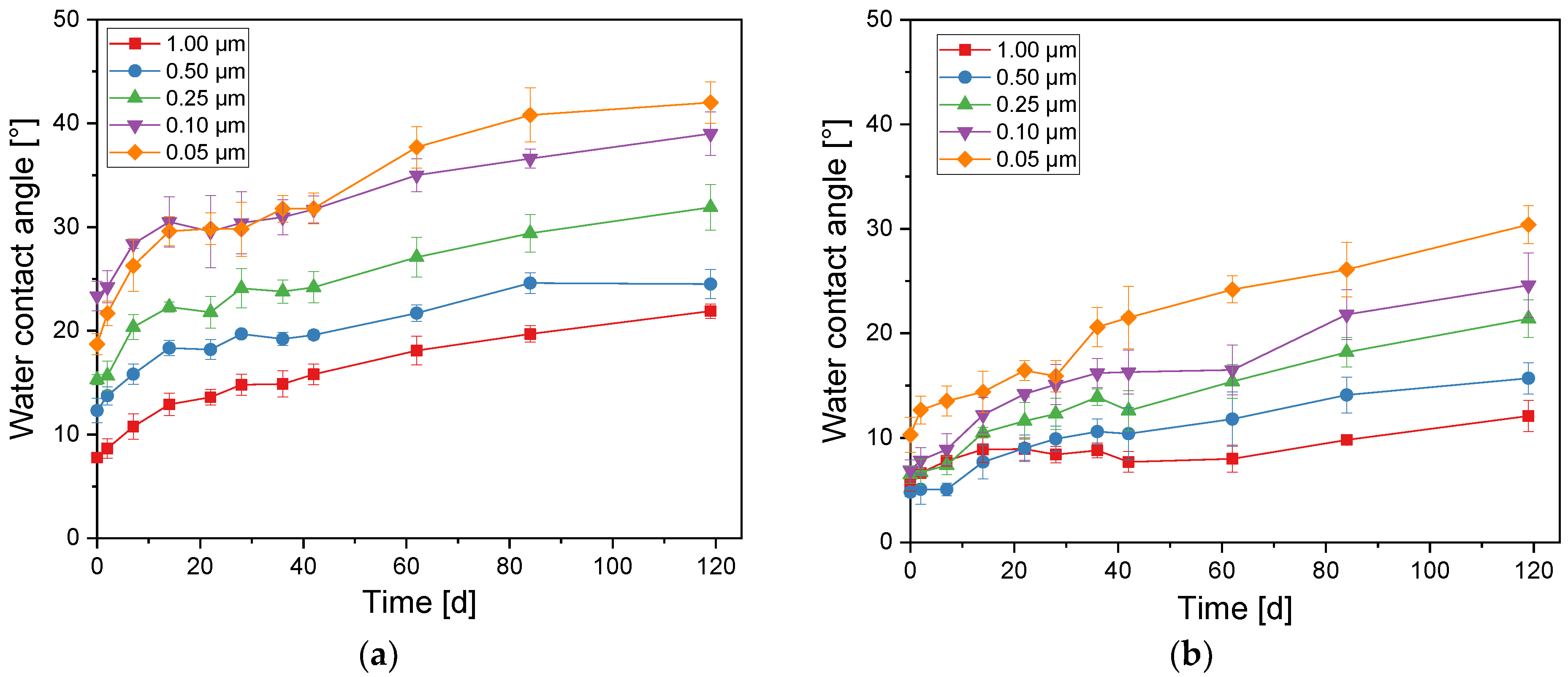
3.2. Tungsten Trioxide Thin Films
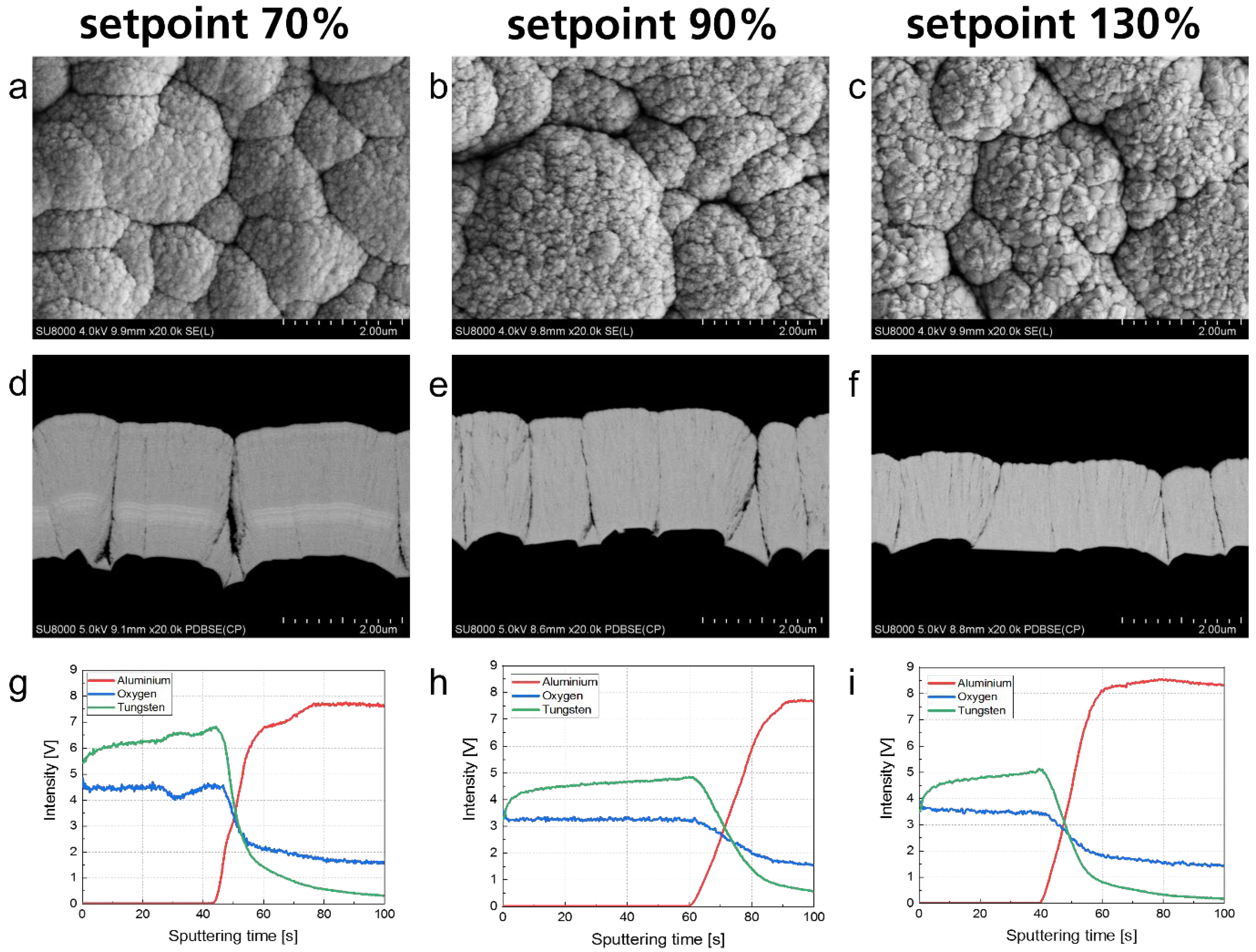
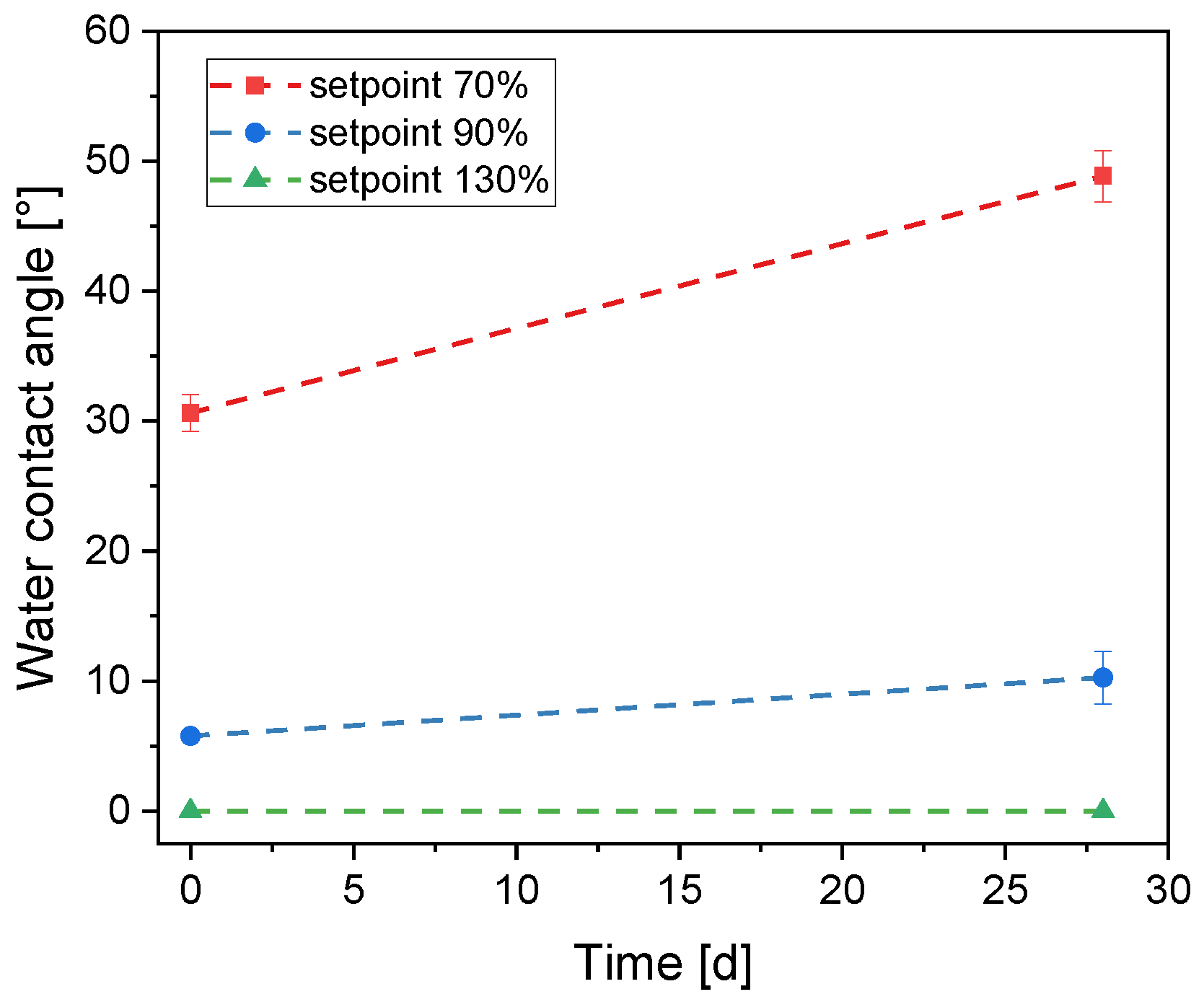
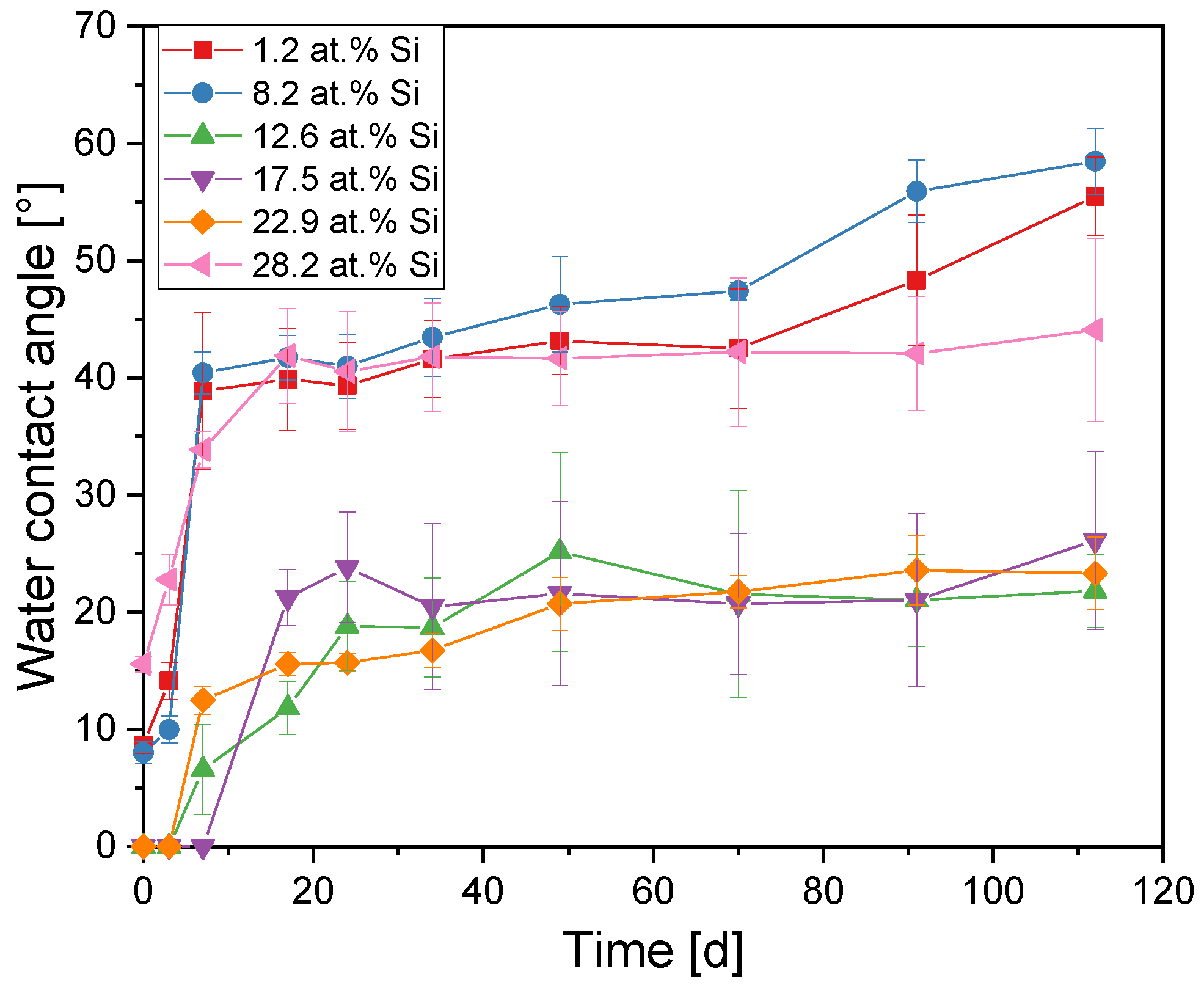
3.3. Titanium Dioxide–Silicon Dioxide Outer Layer
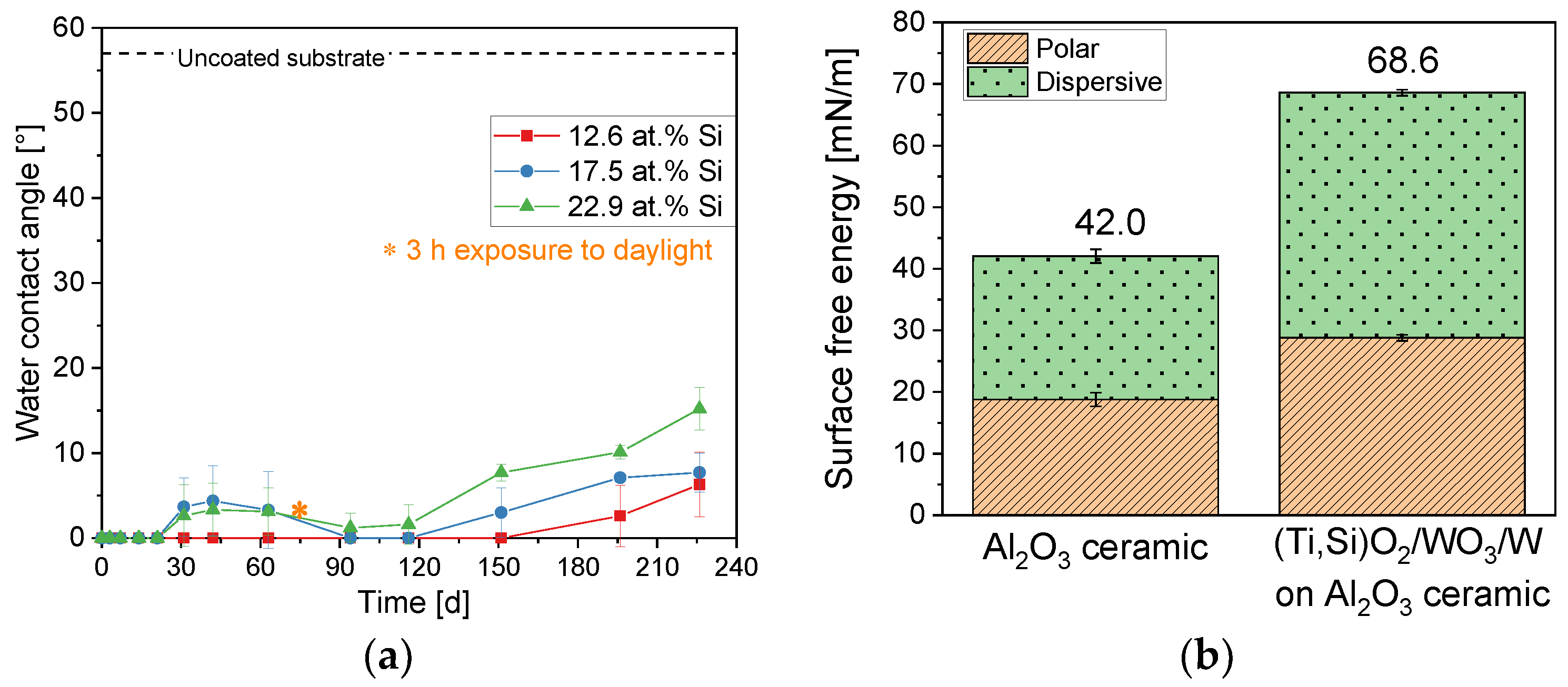
3.4. Multilayer System
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mönch, S.; Reiner, R.; Waltereit, P.; Basler, M.; Quay, R.; Gebhardt, S.; Molin, C.; Bach, D.; Binninger, R.; Bartholomé, K. How highly efficient power electronics transfers high electrocaloric material performance to heat pump systems. MRS Adv. 2023, 8, 787–796. [Google Scholar] [CrossRef]
- Zhu, S.; Xie, K.; Lin, Q.; Cao, R.; Qiu, F. Experimental determination of surface energy for high-energy surface: A review. Adv. Colloid Interface Sci. 2023, 315, 102905. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, N.; Barschilia, H.C.; Rajam, K.S. Effect of substrate roughness on the apparent surface free energy of sputter deposited superhydrophobic polytetrafluoroethylene coatings: A comparison of experimental data with different theoretical models. J. Appl. Phys. 2010, 108, 013505. [Google Scholar] [CrossRef]
- Young, T. An essay on the cohesion of fluids. Philos. Trans. R. Soc. Lond. 1805, 95, 65–87. [Google Scholar]
- Ramana, C.V.; Battu, A.K.; Dubey, P.; Lopez, G.A. Phase-control-enabled enhancement in hydrophilicity and mechanical toughness in nanocrystalline tungsten oxide films for energy-related applications. ACS Appl. Nano Mater. 2020, 3, 3264–3274. [Google Scholar] [CrossRef]
- Drelich, J.; Chibowski, E. Superhydrophilic and superwetting surfaces: Definition and mechanisms of control. Langmuir 2010, 26, 18621–18623. [Google Scholar] [CrossRef] [PubMed]
- Azimirad, R.; Naseri, N.; Akhavan, O.; Moshfegh, A.Z. Hydrophilicity variation of WO3 thin films with annealing temperature. J. Phys. D Appl. Phys. 2007, 40, 1134–1137. [Google Scholar] [CrossRef]
- Miyauchi, M.; Nakajima, A.; Watanabe, A.; Hashimoto, K. Photocatalysis and photoinduced hydrophilicity of various metal oxide thin films. Chem. Mater. 2002, 14, 2812–2816. [Google Scholar] [CrossRef]
- Kong, X.; Hu, Y.; Wang, X.; Pan, W. Effect of surface morphology on wettability conversion. J. Adv. Ceram. 2016, 5, 284–290. [Google Scholar] [CrossRef]
- Vrakatseli, V.E.; Kalarakis, A.N.; Kalampounias, A.G.; Amanatides, E.K.; Mataras, D.S. Glancing angle deposition effect on structure and light-induced wettability of RF-sputtered TiO2 thin films. Micromachines 2018, 9, 389. [Google Scholar] [CrossRef]
- Frach, P.; Glöß, D.; Metzner, C.; Modes, T.; Scheffel, B.; Zywitzki, O. Deposition of photocatalytic TiO2 layers by pulse magnetron sputtering and by plasma-activated evaporation. Vacuum 2006, 80, 679–683. [Google Scholar] [CrossRef]
- Fateh, R.; Dillert, R.; Bahnemann, D. Preparation and characterization of transparent hydrophilic photocatalytic TiO2/SiO2 thin films on polycarbonate. Langmuir 2013, 29, 3730–3739. [Google Scholar] [CrossRef]
- Adachi, T.; Latthe, S.S.; Gosavi, S.W.; Roy, N.; Suzuki, N.; Ikari, H.; Kato, K.; Katsumata, K.-I.; Nakata, K.; Furudate, M.; et al. Photocatalytic, superhydrophilic, self-cleaning TiO2 coating on cheap, light-weight, flexible polycarbonate substrates. Appl. Surf. Sci. 2018, 458, 917–923. [Google Scholar] [CrossRef]
- Yao, L.; He, J. Facile dip-coating approach to fabrication of mechanically robust hybrid thin films with high transmittance and durable superhydrophilicity. J. Mater. Chem. A 2014, 2, 6994–7003. [Google Scholar] [CrossRef]
- Bai, Z.; Hu, Y.; Yan, S.; Shan, W.; Wei, C. Preparation of mesoporous SiO2/Bi2O3/TiO2 superhydrophilic thin films and their surface self-cleaning properties. RSC Adv. 2017, 7, 1966–1974. [Google Scholar] [CrossRef]
- Chen, P.; Hu, Y.; Wei, C. Preparation of superhydrophilic mesoporous SiO2 thin films. App. Surf. Sci. 2012, 258, 4334–4338. [Google Scholar] [CrossRef]
- Hendi, A.; Al-Kuhaili, M.; Durrani, S.M.A.; Faiz, M.; Ul-Hamid, A.; Qurashi, A.; Khan, I. Modulation of the band gap of tungsten oxide thin films through mixing with cadmium telluride towards photovoltaic applications. Mater. Res. Bull. 2017, 87, 148–154. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, C.; Zhang, Z.; Tian, W.; Hui, B.; Zhang, J.; Zhang, K. Tungsten oxide polymorphs and their multifunctional applications. Adv. Colloid Interface Sci. 2022, 300, 102596. [Google Scholar] [CrossRef]
- Bandi, S.; Srivastav, A. Review: Oxygen-deficient tungsten oxides. J. Mater. Sci. 2021, 56, 6615–6644. [Google Scholar] [CrossRef]
- Zhang, R.; Ning, F.; Xu, S.; Zhou, L.; Shao, M.; Wei, M. Oxygen vacancy engineering of WO3 toward largely enhanced photoelectrochemical water splitting. Electrochim. Acta 2018, 274, 217–223. [Google Scholar] [CrossRef]
- Schiller, S.; Goedicke, K.; Reschke, J.; Kirchhoff, V.; Schneider, S.; Milde, F. Pulsed magnetron sputtering technology. Surf. Coat. Technol. 1993, 61, 331–337. [Google Scholar] [CrossRef]
- Billings, B.H.; Gray, D.E. American Institute of Physics Handbook, 3rd ed.; McGraw-Hill: New York, NY, USA, 1972. [Google Scholar]
- Ellmer, K. Magnetron sputtering of transparent conductive zinc oxide: Relation between the sputtering parameters and the electronic properties. J. Phys. D Appl. Phys. 2000, 33, R17. [Google Scholar] [CrossRef]
- Weil, M.; Schubert, W.-D. The Beautiful Colors of Tungsten Oxides; International Tungsten Industry Association: London, UK, 2013. [Google Scholar]
- Guillen, C. Polycrystalline WO3-x thin films obtained by reactive DC sputtering at room temperature. Materials 2023, 16, 1359. [Google Scholar] [CrossRef] [PubMed]
- Colovic, B.; Kisic, D.; Jokanovic, B.; Rakocevic, Z.; Nasov, I.; Petroska, A.; Jokanovic, V. Wetting properties of titanium oxides, oxynitrides and nitrides obtained by DC and pulsed magnetron sputtering and cathodic arc evaporation. Mater. Sci.-Pol. 2019, 37, 173–181. [Google Scholar] [CrossRef]
- Xia, Z.-J.; Wang, H.-L.; Su, Y.-F.; Tang, P.; Dai, M.-J.; Lin, H.-J.; Zhang, Z.-G.; Shi, Q. Enhanced electrochromic properties by improvement of crystallinity for sputtered WO3 film. Coatings 2020, 10, 577. [Google Scholar] [CrossRef]
- Gullapalli, S.K.; Vemuri, R.S.; Manciu, F.S.; Enriquez, J.L.; Ramana, C.V. Tungsten oxide (WO3) thin films for application in advanced energy systems. J. Vac. Sci. Technol. A 2010, 28, 824–828. [Google Scholar] [CrossRef]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical properties and electronic structure of amorphous Germanium. Physica Status Solidi 1966, 15, 627–637. [Google Scholar] [CrossRef]
- Zhao, Y.; Balasubramanyam, S.; Sinha, R.; Lavrijsen, R.; Verheijen, M.A.; Bol, A.A.; Bieberle-Hütter, A. Physical and chemical defects in WO3 thin films and their impact on photoelectrochemical water splitting. ACS Appl. Energy Mater. 2018, 1, 5887–5895. [Google Scholar] [CrossRef]
- Subrahmanyam, A.; Karuppasamy, A. Optical and electrochromic properties of oxygen sputtered tungsten oxide (WO3) thin films. Sol. Energy Mater Sol. Cells 2007, 91, 266–274. [Google Scholar] [CrossRef]
- Chien, D.M.; Viet, N.N.; Van, N.T.K.; Phong, N.T.P. Characteristics modification of TiO2 thin films by doping with silica and alumina for self-cleaning application. J. Exp. Nanosci. 2009, 4, 221–232. [Google Scholar] [CrossRef]
- Golovko, D.S.; Butt, H.-J.; Bonaccurso, E. Transition in the evaporation kinetics of water microdrops on hydrophilic surfaces. Langmuir 2008, 25, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.C.; Bonaccurso, E. Evaporation control of sessile water drops by soft viscoelastic surfaces. Soft Matter 2012, 8, 7875–7881. [Google Scholar] [CrossRef]
- Starov, V.M.; Kostvintsev, S.R.; Sobolev, V.D.; Velarde, M.G.; Zhdanov, S.A. Spreading of liquid drops over dry porous layers: Complete wetting case. J. Colloid Interface Sci. 2002, 252, 397–408. [Google Scholar] [CrossRef]
- Günay, A.A.; Kim, M.-K.; Yan, X.; Miljkovic, N.; Sett, S. Droplet evaporation dynamics on microstructured biphilic, hydrophobic, and smooth surfaces. Exp. Fluids 2021, 62, 153. [Google Scholar] [CrossRef]

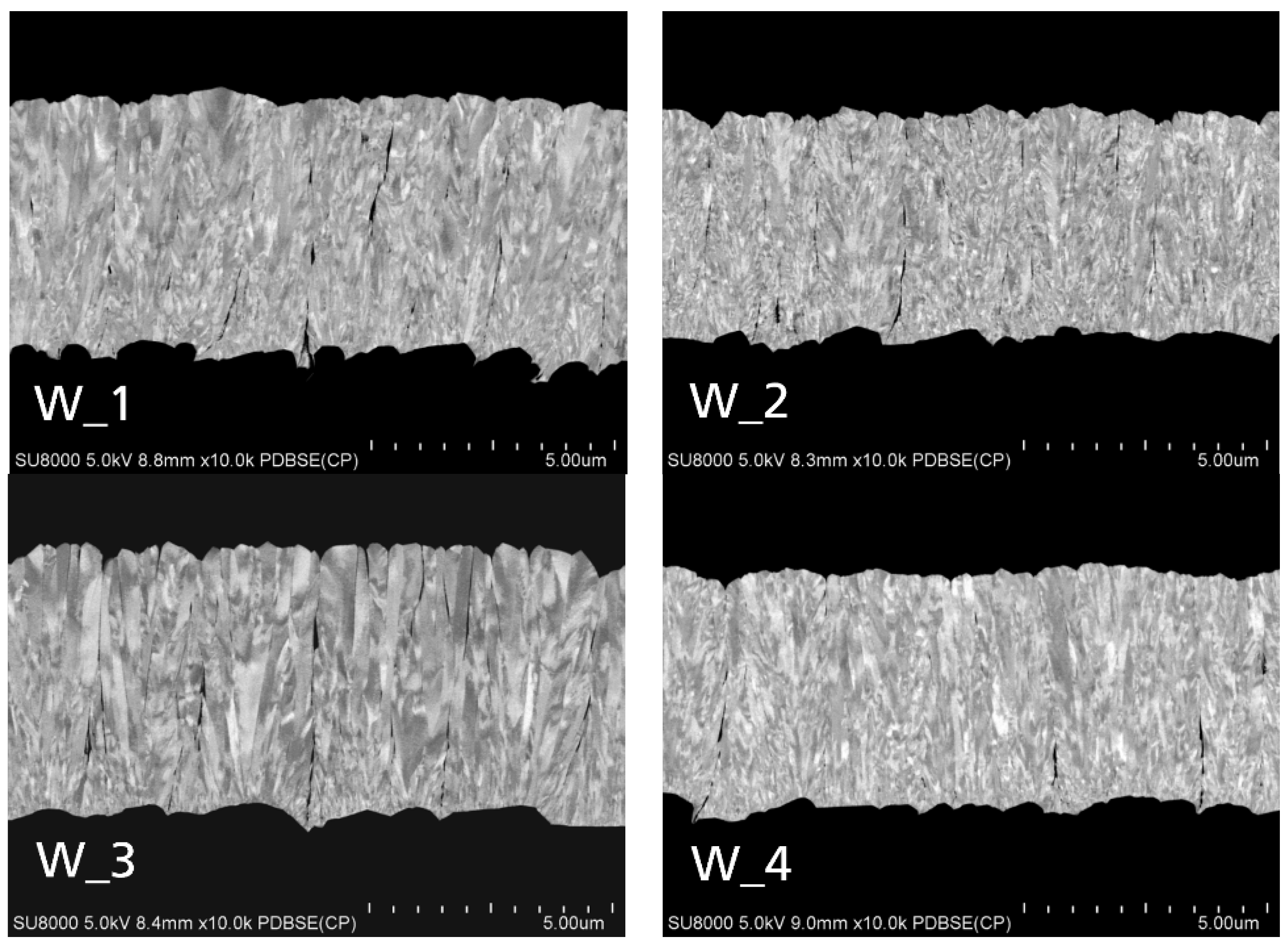





| Sample | Temperature | Pulse Frequency | Voltage | Thickness | Rate | Electrical Conductivity |
|---|---|---|---|---|---|---|
| No. | °C | kHz | V | nm | nm/m | MS/m |
| W_1 | 250 | 2.5 | 645 | 5200 | 86 | 4.1 |
| W_2 | 250 | 25 | 870 | 4600 | 76 | 3.1 |
| W_3 | 500 | 2.5 | 654 | 5500 | 91 | 3.6 |
| W_4 | 500 | 25 | 835 | 4800 | 80 | 3.4 |
| Sample | Temperature | Setpoint | O2 Flow 1 | Total Pressure | Thickness | Rate |
|---|---|---|---|---|---|---|
| No. | °C | % | sccm | Pa | nm | nm/min |
| WOx_1 | 250 | 70 | 145 | 1.21 | 2200 | 220 |
| WOx_2 | 250 | 90 | 140 | 1.28 | 2100 | 210 |
| WOx_3 | 250 | 130 | 120 | 1.40 | 1500 | 150 |
| WOx_4 | 250 | 130 | 120 | 1.40 | 1100 | 137 |
| WOx_5 | 500 | 130 | 190 | 1.80 | 1000 | 125 |
| Sample | Temperature | Si Pulse-on | Setpoint | O2 Flow 1 | Total Pressure | Thickness | Rate | Ti | Si | O |
|---|---|---|---|---|---|---|---|---|---|---|
| No. | °C | µs | % | sccm | Pa | nm | nm/m | at. % | at. % | at. % |
| TiSiO_1 | 250 | 5 | 10 | 29 | 1.09 | 1289 | 43 | 32.6 | 1.2 | 66.2 |
| TiSiO_2 | 250 | 6 | 10 | 23 | 1.09 | 955 | 32 | 26.3 | 8.2 | 65.5 |
| TiSiO_3 | 250 | 7 | 10 | 21 | 1.09 | 988 | 33 | 22.1 | 12.6 | 65.3 |
| TiSiO_4 | 250 | 8 | 10 | 28 | 1.09 | 1041 | 35 | 17.4 | 17.5 | 65.1 |
| TiSiO_5 | 250 | 6 | 15 | 20 | 1.10 | 610 | 20 | 11.9 | 22.9 | 65.2 |
| TiSiO_6 | 250 | 6 | 25 | 22 | 1.17 | 402 | 13 | 6.6 | 28.2 | 65.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrera, M.; Zywitzki, O.; Modes, T.; Fietzke, F. Magnetron-Sputtered Long-Term Superhydrophilic Thin Films for Use in Solid-State Cooling Devices. Coatings 2024, 14, 622. https://doi.org/10.3390/coatings14050622
Barrera M, Zywitzki O, Modes T, Fietzke F. Magnetron-Sputtered Long-Term Superhydrophilic Thin Films for Use in Solid-State Cooling Devices. Coatings. 2024; 14(5):622. https://doi.org/10.3390/coatings14050622
Chicago/Turabian StyleBarrera, Maria, Olaf Zywitzki, Thomas Modes, and Fred Fietzke. 2024. "Magnetron-Sputtered Long-Term Superhydrophilic Thin Films for Use in Solid-State Cooling Devices" Coatings 14, no. 5: 622. https://doi.org/10.3390/coatings14050622
APA StyleBarrera, M., Zywitzki, O., Modes, T., & Fietzke, F. (2024). Magnetron-Sputtered Long-Term Superhydrophilic Thin Films for Use in Solid-State Cooling Devices. Coatings, 14(5), 622. https://doi.org/10.3390/coatings14050622






