Analysis of Metabolites in Stem Parasitic Plant Interactions: Interaction of Cuscuta–Momordica versus Cassytha–Ipomoea
Abstract
:1. Introduction
2. Results
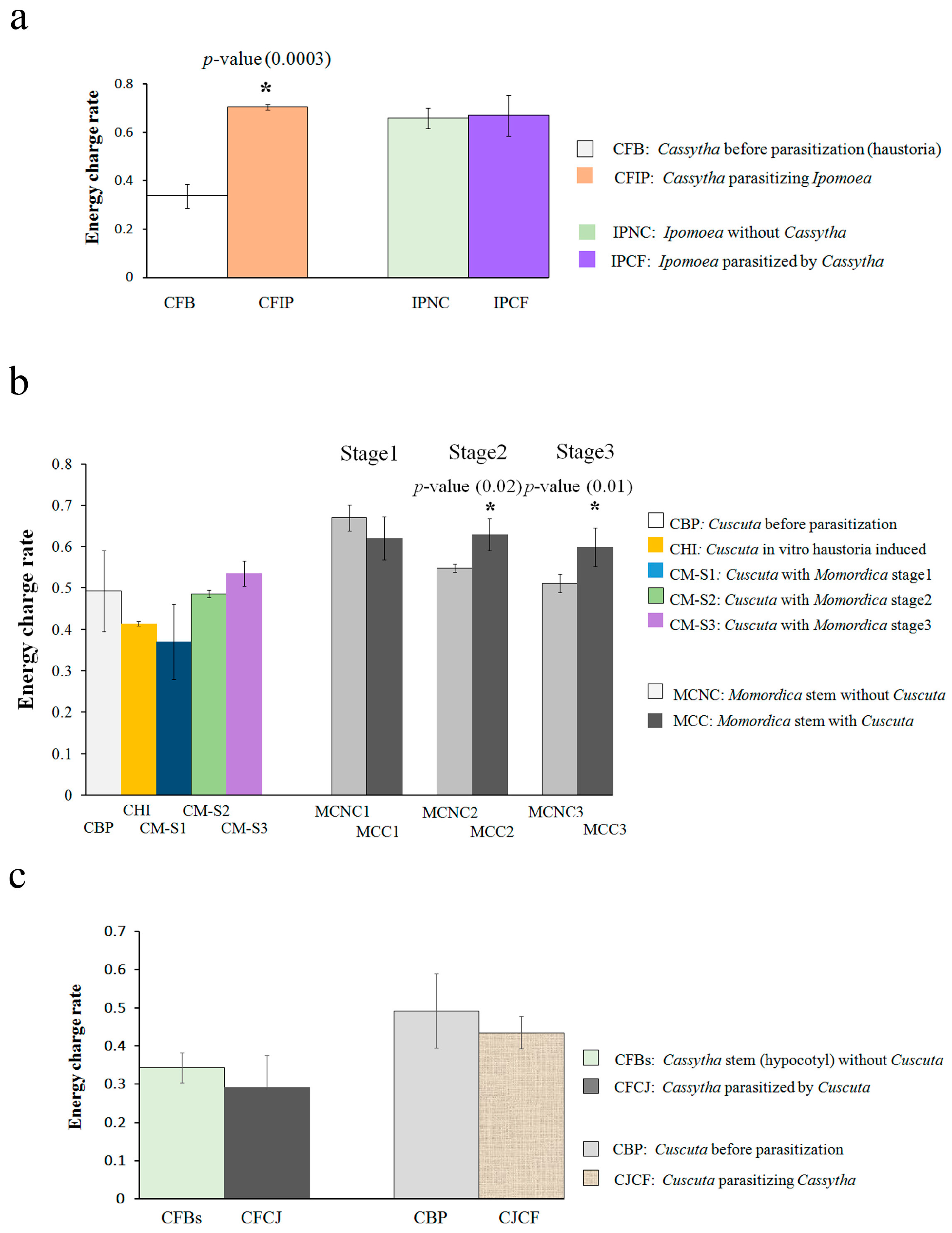
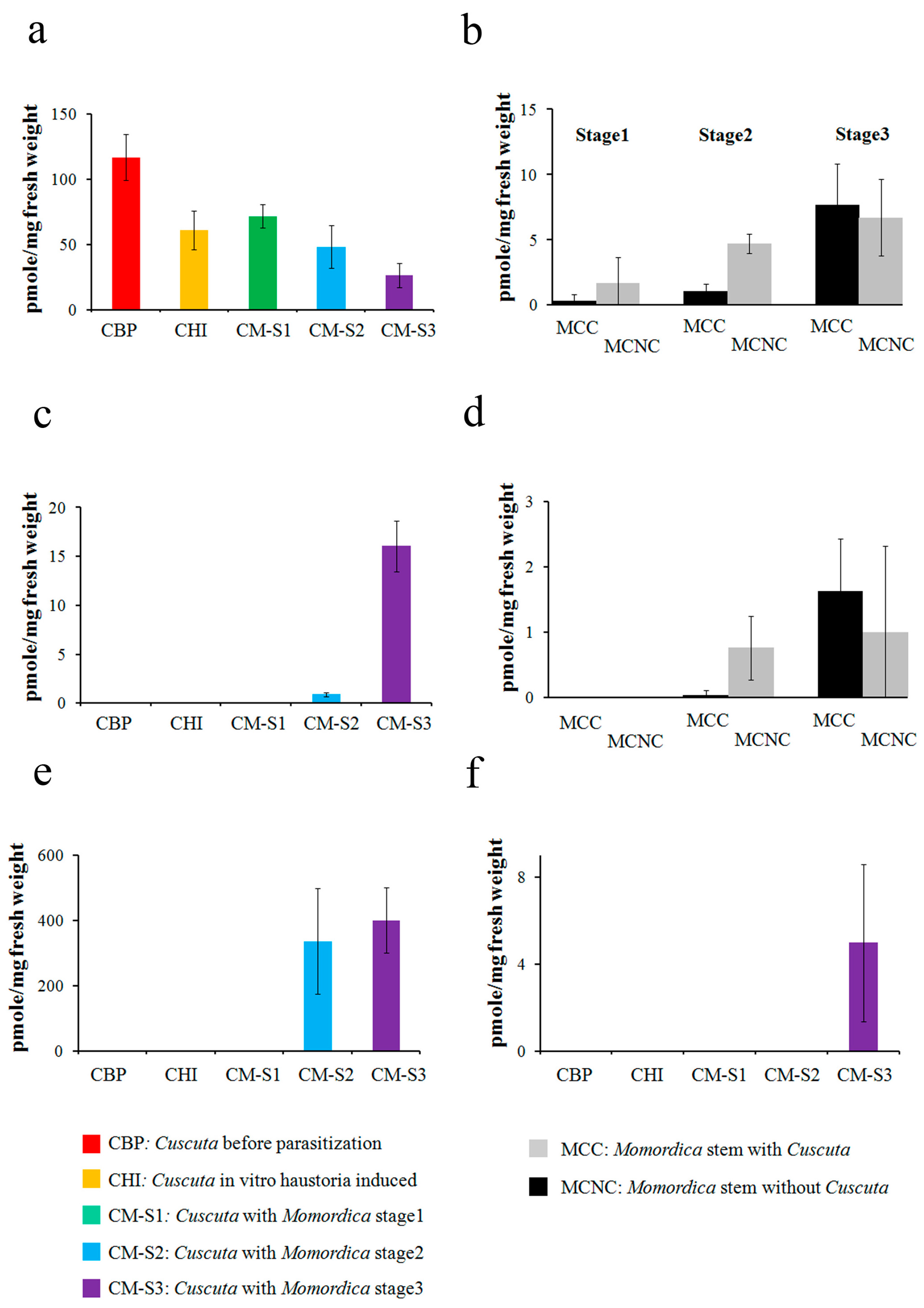
2.1. Energy Metabolite Analysis
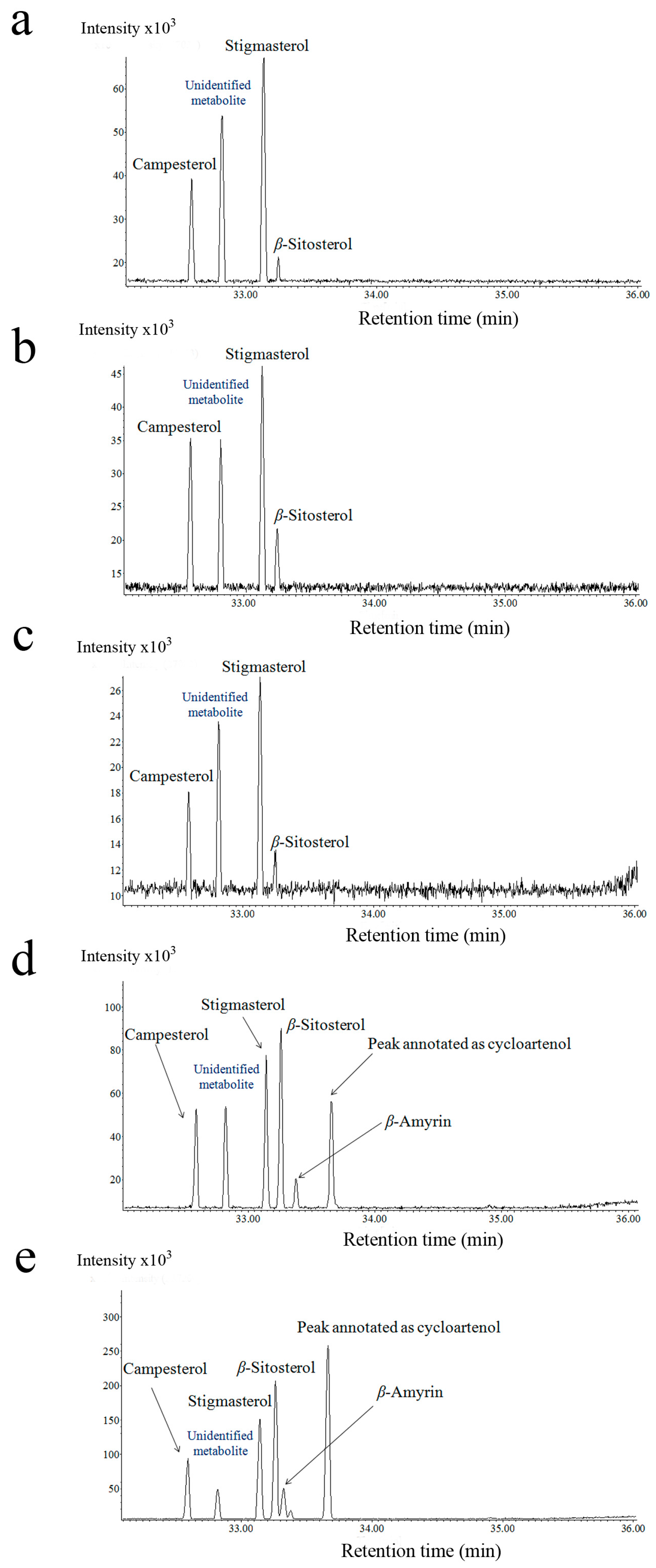
2.2. Steroid Profiling
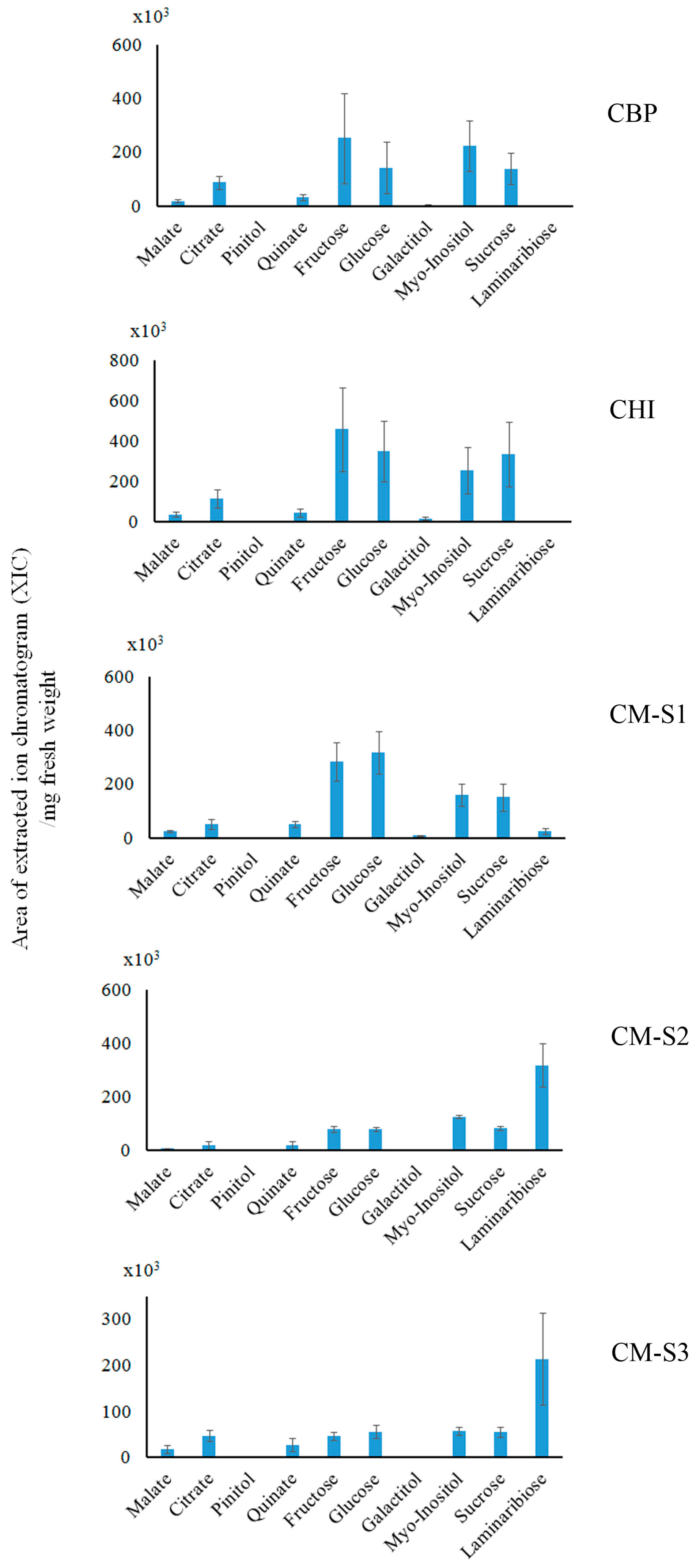
2.3. Saccharide (Polar Metabolite) Analysis by GC-MS
3. Discussion
3.1. Cassytha–Ipomoea Interaction
3.2. Cuscuta–Momordica Interaction
3.3. Cuscuta–Cassytha Interaction
3.4. Plant Interaction Comparison
4. Materials and Methods
4.1. Plant Culture and Sample Preparation
4.2. Extraction of Energy Metabolites, Polar Metabolites Analysis
4.3. Extraction of Steroids
4.4. UFLC Condition
4.5. GC/MS Condition
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ADP | Adenosine diphosphate |
| AMP | Adenosine monophosphate |
| ATP | Adenosine triphosphate |
| EI | Electron ionization |
| FAME | Fatty acid methyl ester |
| FI | Field ionization |
| GC-MS | Gas chromatography-mass spectrometry |
| HCl | Hydrochloric acid |
| HR | Hypersensitive response |
| ODS | Octadecyl-silica |
| PAD | Photo diode array detector |
| SAR | Systemic acquired resistance |
| TCA | Tricarboxylic acid cycle |
| TFA | Trifluoro acetic acid |
| UFLC | Ultra-high performance liquid chromatography |
References
- Jørgensen, H. Parasitic Flowering Plants; Brill: Leiden, The Netherlands, 2008; p. 421. [Google Scholar]
- Tada, Y.; Sugai, M.; Furuhashi, K. Haustoria of Cuscuta japonica, a holoparasitic flowering plant, are induced by cooperative effect of far-red light and tactile stimuli. Plant Cell Physiol. 1996, 37, 1049–1053. [Google Scholar] [CrossRef]
- Furuhashi, T.; Furuhashi, K.; Weckwerth, W. The Parasitic Mechanism of the Holostemparasitic Plant Cuscuta. J. Plant Interact. 2011, 6, 207–219. [Google Scholar] [CrossRef]
- Weber, J.Z. A taxonomic revision of Cassytha (Lauraceae) in Australia. J. Adel. Bot. Gard. 1981, 3, 187–262. [Google Scholar]
- McNeal, J.R.; Arumugunathan, K.; Kuehl, J.V.; Boore, J.L.; de Pamphilis, C.W. Systematics and plastid genome evolution of the cryptically photosynthetic parasitic plant genus Cuscuta (Convolvulaceae). BMC Biol. 2007, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- McLuckie, J. Studies in parasitism. A contribution to the physiology of genus Cassytha. Proc. Linn. Soc. NSW 1924, 49, 55–79. [Google Scholar]
- Kokubugata, G.; Nakamura, K.; Forster, P.I.; Wilson, G.W.; Holland, A.E.; Hirayama, Y.; Yokota, M. Cassytha pubescens and C. glabella (Lauraceae) are not disjunctly distributed between Australia and the Ryukyu Archipelago of Japan—Evidence from morphological and molecular data. Aust. Syst. Bot. 2012, 25, 364–373. [Google Scholar] [CrossRef]
- Hibberd, J.M.; Jeschke, W.D. Solute flux into parasitic plants. J. Exp. Bot. 2001, 52, 2043–2049. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Ruyter-Spira, C.; Bouwmeester, H. The interaction between strigolactones and other plant hormones in the regulation of plant development. Front. Plant Sci. 2013, 4, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; Ichihashi, Y.; Farhi, M.; Zumstein, K.; Townsley, B.; David-Schwartz, R.; Sinha, N.R. De novo assembly and characterization of the transcriptome of the parasitic weed dodder identifies genes associated with plant parasitism. Plant Physiol. 2014, 166, 1186–1199. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, T.; Kojima, M.; Sakakibara, H.; Fukushima, A.; Hirai, M.Y.; Furuhashi, K. Morphological and Plant Hormonal Changes during Parasitization by Cuscuta japonica on Momordica charantia. J. Plant Interact. 2014, 9, 220–232. [Google Scholar] [CrossRef]
- Kokubugata, G.; Yokota, M. Host Specificity of Cassytha filiformis and C. pergracilis (Lauraceae) in the Ryukyu Archipelago. Bull. Natl. Mus. Nat. Sci. Ser. B 2012, 38, 47–53. [Google Scholar]
- Chen, M.Y.; Tsai, J.L.; Liao, G.I. The parasitism of Cassytha filiformis in Taiwan. Q. J. Chin. For. 1992, 24, 75–86. [Google Scholar]
- Birschwilks, M.; Sauer, N.; Scheel, D.; Neumann, S. Arabidopsis thaliana is a susceptible host plant for the holoparasite Cuscuta spec. Planta 2007, 226, 1231–1241. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, M.; Kim, G.; Westwood, J.H. RNA trafficking in parasitic plant systems. Front. Plant Sci. 2012, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Pradet, A.; Raymond, P. Adenine nucleotide ratios and adenylate energy charge in energy metabolism. Annu. Rev. Plant Physiol. 1983, 34, 199–224. [Google Scholar] [CrossRef]
- Richter, A.; Popp, M. The physiological importance of accumulation of cyclitols in Visum album L. New Phytol. 1992, 121, 431–438. [Google Scholar] [CrossRef]
- Wanek, W.; Richter, A. L-Idiol: NAD 5-oxidoreductase in Viscum album: Utilization of host-derived sorbitol. Plant Physiol. Biochem. 1993, 31, 205–211. [Google Scholar]
- Rohmer, M.; Ourisson, G.; Benveniste, P.; Bimpson, T. Sterol biosynthesis in heterotrophic plant parasite. Phytochemistry 1975, 14, 727–730. [Google Scholar] [CrossRef]
- Albrechta, C.; Boutrot, F.; Segonzac, C.; Schwessinger, B.; Gimenez-Ibanez, S.; Chinchilla, D.; Rathjen, J.P.; de Vries, S.C.; Zipfel, C. Brassinosteroids inhibit pathogen-associated molecular pattern–triggered immune signaling independent of the receptor kinase BAK1. Proc. Natl. Acad. Sci. USA 2012, 109, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Duhalt, R.; Arredondo-Vega, B.O. Haloadaptation of the green alga Botryococcus braunii (race a). Phytochemistry 1991, 30, 2919–2925. [Google Scholar] [CrossRef]
- Abubacker, M.N.; Prince, M.; Hariharan, Y. Histochemical and biochemical studies of parasite–host interaction of Cassytha filiformis Linn. and Zizyphus jujuba Lamk. Curr. Sci. 2005, 89, 2156–2159. [Google Scholar]
- Kadkade, P.G.; Lujan, C.; Rolz, C. Studies on the distribution of phytosterols in Dioscorea species tubers. Z. Naturforsch. 1983, 38C, 488–489. [Google Scholar]
- Benveniste, P. Sterol biosynthesis. Annu. Rev. Plant Physiol. 1986, 37, 275–308. [Google Scholar] [CrossRef]
- Furuhashi, T.; Nakamura, T.; Fragner, L.; Roustan, V.; Schon, V.; Weckwerth, W. Biodiesel and poly-unsaturated fatty acids production from algae and crop plants—A rapid and comprehensive workflow for lipid analysis. Biotechnol. J. 2016, 11, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Edewor, T.I.; Owa, S.O.; Ologan, A.O.; Akinfemi, F. Quantitative determination of the saponin content and GC-MS study of the medicinal plant Cassytha filiformis (linn.) leaves. J. Coast. Life Med. 2016, 4, 154–156. [Google Scholar] [CrossRef]
- Lozano-Duran, R.; Zipfel, C. Trade-off between growth and immunity: Role of brassinosteroids. Trends Plant Sci. 2015, 20, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Shen, H.; Chen, H.; Li, L.; Hu, X.; Xu, X.; Ye, W.; Wang, Z. The morphology and anatomy of the haustoria of the holoparasitic angiosperm Cuscuta campestris. Pak. J. Bot. 2011, 43, 1853–1859. [Google Scholar]
- Streeter, J.G.; Lohnes, D.G.; Fioritto, R.J. Patterns of pinitol accumulation in soybean plants and relationships to drought tolerance. Plant Cell Environ. 2001, 24, 429–438. [Google Scholar] [CrossRef]
- Krasensky, J.; Jonak, C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J. Exp. Bot. 2012, 63, 1593–1608. [Google Scholar] [CrossRef] [PubMed]
- Murthy, P.S.; Manonmani, H.K. Physico-chemical, antioxidant and antimicrobial properties of Indian monsooned coffee. Eur. Food Res. Technol. 2009, 229, 645–650. [Google Scholar] [CrossRef]
- Furuhashi, T.; Fragner, L.; Furuhashi, K.; Valledor, L.; Sun, X.; Weckwerth, W. Metabolite Changes with Induction of Cuscuta Haustorium and Translocation from Host Plants. J. Plant Interact. 2012, 7, 84–93. [Google Scholar] [CrossRef]
- Loescher, W.H. Physiology and metabolism of sugar alcohols in higher plants. Physiol. Plant. 1987, 70, 553–557. [Google Scholar] [CrossRef]
- Plouvier, V. The distribution of aliphatic polyols and cyclitols. In Chemical Plant Taxonomy; Swain, T., Ed.; Academic Press Inc.: New York, NY, USA, 1963; pp. 359–388. [Google Scholar]
- Noiraud, N.; Maurousset, L.; Lemoine, R. Transport of polyols in higher plants. Plant Physiol. Biochem. 2001, 39, 717–728. [Google Scholar] [CrossRef]
- Furuhashi, T.; Ishii, K.; Tanaka, K.; Weckwerth, W.; Nakamura, T. Fragmentation patterns of methyloxime-trimethylsilyl derivatives of constitutive mono- and disaccharide isomers analyzed by gas chromatography/field ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2015, 29, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, T.; Weckwerth, W. Introduction to Lipid (FAME) Analysis in Algae Using Gas Chromatography-Mass Spectrometry. In Handbook of Plant Metabolomics: Metabolite Profiling and Networking; Weckwerth, W., Kahl, G., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2013; pp. 215–225. [Google Scholar]
- Basconcillo, L.S.; McCarry, B.E. Comparison of three GC/MS methodologies for the analysis of fatty acids in Sinorhizobium meliloti: Development of a micro-scale, one-vial method. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 871, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Jiang, Y.; Luo, Y.; Jiang, W. A simple and rapid determination of ATP, ADP and AMP concentrations in pericarp tissue of litchi fruit by high performance liquid chromatography. Food Technol. Biotechnol. 2006, 44, 531–534. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furuhashi, T.; Nakamura, T.; Iwase, K. Analysis of Metabolites in Stem Parasitic Plant Interactions: Interaction of Cuscuta–Momordica versus Cassytha–Ipomoea. Plants 2016, 5, 43. https://doi.org/10.3390/plants5040043
Furuhashi T, Nakamura T, Iwase K. Analysis of Metabolites in Stem Parasitic Plant Interactions: Interaction of Cuscuta–Momordica versus Cassytha–Ipomoea. Plants. 2016; 5(4):43. https://doi.org/10.3390/plants5040043
Chicago/Turabian StyleFuruhashi, Takeshi, Takemichi Nakamura, and Koji Iwase. 2016. "Analysis of Metabolites in Stem Parasitic Plant Interactions: Interaction of Cuscuta–Momordica versus Cassytha–Ipomoea" Plants 5, no. 4: 43. https://doi.org/10.3390/plants5040043





