Preparation, Functionalization, Modification, and Applications of Nanostructured Gold: A Critical Review
Abstract
:1. Introduction
2. Synthetic Methods of Au NPs
3. Functionalization of Au NPs
3.1. Functionalization via Inorganic Moieties from p-Block
3.1.1. Clusters of Fullerene (C60)
3.1.2. Carborane Clusters
3.1.3. Clusters of POSS
3.1.4. Silica
3.1.5. Carbon Nanotubes
3.1.6. Polyoxometalate Compounds (POM)
3.2. Functionalization via Organometallic Complexes
3.2.1. Ferroncenyl Complexes
3.2.2. Au NPs/Organometallics
3.3. Functionalization of d-block Element Coordinating Complexes
3.3.1. Prussian Blue Derivatives
3.3.2. Metal Complexes of Polypyridyls
3.4. Functionalizated Coordinated Complexes on Au NPs for Numerous Applications
3.5. Functionalization via Shiff Base Coordinated and Carboxylates Linked Complexes
3.6. Functionalization via Bio-Inorganic Complexes
3.7. Functionalization via Crown Ether Devices
3.8. Functionalization via f-block Elements Coordination Complexes
4. Modification of Au NPs via Small Molecules for Biochemical Analysis
4.1. Surface Conjugation of Au NPs by Click-Based Chemistry
4.2. Modification of Au NPs via Co-Ordination Based Recognition
4.2.1. Modification via Amino Acids
4.2.2. Modification via Sulfhydryl Compounds
4.3. Modification via Ligand Exchange for Biochemical Analysis
5. Interaction of Biomolecules with Au NPs for Catalysis
5.1. Interaction of DNA
5.2. Interaction of Proteins
6. Catalysis via Biomolecules Anchored Au NPs
6.1. Catalysis via the Au NPs Itself
6.2. Catalysis via Anchoring Ligands
6.3. Catalysis via Functionalization of Asymmetric Oligonucleotides
7. Surface Chemistry of Au NPs for Various Applications
7.1. Au NP for Health Applications
7.1.1. Surface Chemistry of Au NPs Enabled Prophylaxis
7.1.2. Au NPs Modified Proteins/Peptides as Vaccines
7.1.3. Au NPs Modified Carbohydrates as Vaccines
7.1.4. Au NPs Modified DNA as Vaccines
7.1.5. The Au NPs Surface Enables Diagnosis
Metal Ions Diagnosis
The Surface Functionalized Proteins-Au NPs with Probes

Recognition of Bacteria, Viruses and Cells via Au NPs
7.1.6. The Surface Chemistry of Au NPs Enables Treatment
Functionalized Au NPs as Antibiotics
Functionalized Au NPs for Cancer Therapy
7.2. Other Applications
7.2.1. Gold Nanoparticles as a Catalyst
7.2.2. Gold Nanoparticles as A Photocatalyst
Au NPs for Water Splitting
Role of Au NPs in Redox/Chemical synthesis
7.2.3. Use of Au NPs in Sensing
Sensors Based on Change in LSPR Absorption of Au NPs
Sensing of Proteins
Sensing of Oligonucleotides for Inhirtence Tracing
Biosensors Bassed on Au NP SPR Scattering Approach
8. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wiesner, M.; Bottero, J.-Y. Environmental Nanotechnology; McGraw-Hill Professional Publishing New York: New York, NY, USA, 2007. [Google Scholar]
- Di Guglielmo, C.; López, D.R.; De Lapuente, J.; Mallafre, J.M.L.; Suàrez, M.B. Embryotoxicity of cobalt ferrite and gold nanoparticles: A first in vitro approach. Reprod. Toxicol. 2010, 30, 271–276. [Google Scholar]
- Eck, D.; Helm, C.A.; Wagner, N.J.; Vaynberg, K.A. Plasmon resonance measurements of the adsorption and adsorption kinetics of a biopolymer onto gold nanocolloids. Langmuir 2001, 17, 957–960. [Google Scholar]
- Edwards, P.P.; Thomas, J.M. Gold in a metallic divided state—from faraday to present-day nanoscience. Angew. Chem. Int. Ed. 2007, 46, 5480–5486. [Google Scholar]
- Kang, B.; Mackey, M.A.; El-Sayed, M.A. Nuclear targeting of gold nanoparticles in cancer cells induces DNA damage, causing cytokinesis arrest and apoptosis. J. Am. Chem. Soc. 2010, 132, 1517–1519. [Google Scholar]
- Shang, C.; Liu, Z.-P. Origin and activity of gold nanoparticles as aerobic oxidation catalysts in aqueous solution. J. Am. Chem. Soc. 2011, 133, 9938–9947. [Google Scholar]
- Abadeer, N.S.; Murphy, C.J. Recent progress in cancer thermal therapy using gold nanoparticles. J. Phys. Chem. C 2016, 120, 4691–4716. [Google Scholar]
- Saleh, T.A. Spectroscopy: Between Modeling, Simulation and Practical Investigation. Spectr. Anal. Rev. 2014, 2, 1–2. [Google Scholar]
- Saleh, T.A. Sensing of chlorpheniramine in pharmaceutical applications by sequential injector coupled with potentiometer. J. Pharm. Anal. 2011, 1, 246–250. [Google Scholar]
- Kelly, K.L.; Coronado, E.; Zhao, L.L.; Schatz, G.C. The optical properties of metal nanoparticles: The influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar]
- Takami, A.; Kurita, H.; Koda, S. Laser-induced size reduction of noble metal particles. J. Phys. Chem. B 1999, 103, 1226–1232. [Google Scholar]
- Chen, D. Design, synthesis and properties of highly functional nanostructured photocatalysts. Recent Pat. Nanotechnol. 2008, 2, 183–189. [Google Scholar]
- Balamurugan, B.; Maruyama, T. Evidence of an enhanced interband absorption in Au nanoparticles: Size-dependent electronic structure and optical properties. Appl. Phys. Lett. 2005, 87, 143105. [Google Scholar]
- Chen, X.; Zheng, Z.; Ke, X.; Jaatinen, E.; Xie, T.; Wang, D.; Guo, C.; Zhao, J.; Zhu, H. Supported silver nanoparticles as photocatalysts under ultraviolet and visible light irradiation. Green Chem. 2010, 12, 414–419. [Google Scholar]
- Litter, M.I. Heterogeneous photocatalysis: Transition metal ions in photocatalytic systems. Appl. Catal. B Environ. 1999, 23, 89–114. [Google Scholar]
- Link, S.; El-Sayed, M. Steady state and time resolved optical properties of metallic nanoparticles the surface plasmon absorption as an analytical tool to inverstigate particle properties. Int. Rev. Phys. Chem. 2000, 19, 409. [Google Scholar]
- Furube, A.; Du, L.; Hara, K.; Katoh, R.; Tachiya, M. Ultrafast plasmon-induced electron transfer from gold nanodots into TiO2 nanoparticles. J. Am. Chem. Soc. 2007, 129, 14852–14853. [Google Scholar]
- Grirrane, A.; Corma, A.; García, H. Gold-catalyzed synthesis of aromatic azo compounds from anilines and nitroaromatics. Science 2008, 322, 1661–1664. [Google Scholar]
- Khumaeni, A.; Budi, W.S.; Sutanto, H. Synthesis and characterization of high-purity gold nanoparticles by laser ablation method using low-energy Nd: YAG laser 1064 nm. J. Phys. Conf. Ser. 2017, 909, 012037. [Google Scholar]
- Eskandari-Nojedehi, M.; Jafarizadeh-Malmiri, H.; Rahbar-Shahrouzi, J. Hydrothermal green synthesis of gold nanoparticles using mushroom (Agaricus bisporus) extract: Physico-chemical characteristics and antifungal activity studies. Green Process. Synth. 2018, 7, 38–47. [Google Scholar]
- Gutierrez-Wing, C.; Esparza, R.; Vargas-Hernandez, C.; Garcia, M.F.; Jose-Yacaman, M. Microwave-assisted synthesis of gold nanoparticles self-assembled into self-supported superstructures. Nanoscale 2012, 4, 2281–2287. [Google Scholar]
- Sakai, T.; Enomoto, H.; Sakai, H.; Abe, M. Hydrogen-assisted fabrication of spherical gold nanoparticles through sonochemical reduction of tetrachloride gold (III) ions in water. Ultrason. Sonochem. 2014, 21, 946–950. [Google Scholar]
- Hussain, R.K.; Abdulghani, A.J. Synthesis of gold nanoparticles via chemical reduction of Au (III) ions by isatin in aqueous solutions: Ligand concentrations and pH effects. Baghdad Sci. J. 2014, 11, 1201–1216. [Google Scholar]
- Sadeghi, B.; Mohammadzadeh, M.; Babakhani, B. Green synthesis of gold nanoparticles using Stevia rebaudiana leaf extracts: Characterization and their stability. J. Photochem. Photobiol. B Biol. 2015, 148, 101–106. [Google Scholar]
- Zhu, L.; Zhang, C.; Guo, C.; Wang, X.; Sun, P.; Zhou, D.; Chen, W.; Xue, G. New insight into intermediate precursors of Brust–Schiffrin gold nanoparticles synthesis. J. Phys. Chem. C 2013, 117, 11399–11404. [Google Scholar]
- Hoo, X.-F.; Razak, K.A.; Ridhuan, N.S.; Nor, N.M.; Zakaria, N.D. Synthesis of tunable size gold nanoparticles using seeding growth method and its application in glucose sensor. AIP Conf. Proc. 2017, 1877, 030001. [Google Scholar]
- Ruivo, A.; Ventura, M.; da Silva, M.G.; Laia, C. Synthesis of gold nanoparticles in sol–gel glass porogens containing [bmim][BF 4] ionic liquid. J. Sol-Gel Sci. Technol. 2013, 68, 234–244. [Google Scholar]
- Masanori Sakamoto, M.F.; Majima, T. Light as a construction tool of metal nanoparticles: Synthesis and mechanism. J. Photochem. Photobiol. C 2009, 10, 33–56. [Google Scholar]
- Mian Rong Lee, H.K.L.; Yijie Yang, C.S.L.; Koh, C.L.L.; Lee, Y.H.; Phang, I.Y.; Ling, X.Y. Direct Metal Writing and Precise Positioning of Gold Nanoparticles within Microfluidic Channels for SERS Sensing of Gaseous Analytes. Acs Appl. Mater. Interfaces 2017, 9, 45. [Google Scholar]
- Izquierdo-Lorenzo, I.; Jradi, S.; Adam, P.-M. Direct laser writing of random Au nanoparticle three-dimensional structures for highly reproducible micro-SERS measurements. R. Soc. Chem. 2014, 4, 4128–4133. [Google Scholar]
- Ritacco, T.; Pagliusi, P.; Giocondo, M. Insight into diffusive and convective processes affecting gold nanoparticles microclustering by multiphoton photoreduction. Colloids Surf. A Phys. Eng. Asp. 2021, 610, 125927. [Google Scholar]
- Chen, Y.-S.; Hung, Y.-C.; Liau, I.; Huang, G.S. Assessment of the in vivo toxicity of gold nanoparticles. Nanoscale Res. Lett. 2009, 4, 858. [Google Scholar]
- Colvin, V.L. The potential environmental impact of engineered nanomaterials. Nat. Biotechnol. 2003, 21, 1166–1170. [Google Scholar]
- Shedbalkar, U.; Singh, R.; Wadhwani, S.; Gaidhani, S.; Chopade, B. Microbial synthesis of gold nanoparticles: Current status and future prospects. Adv. Colloid Interface Sci. 2014, 209, 40–48. [Google Scholar]
- Thakkar, K.N.; Mhatre, S.S.; Parikh, R.Y. Biological synthesis of metallic nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 257–262. [Google Scholar]
- Dass, A.; Guo, R.; Tracy, J.B.; Balasubramanian, R.; Douglas, A.D.; Murray, R.W. Gold nanoparticles with perfluorothiolate ligands. Langmuir 2008, 24, 310–315. [Google Scholar]
- Kroto, H.; Heath, J.O.; Brien, S.C.; Curl, R.F.; Smalley, R.E. C60 Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar]
- Nakamura, E.; Isobe, H. Functionalized fullerenes in water. The first 10 years of their chemistry, biology, and nanoscience. Acc. Chem. Res. 2003, 36, 807–815. [Google Scholar]
- Brust, M.; Kiely, C.J.; Bethell, D.; Schiffrin, D.J. C60 mediated aggregation of gold nanoparticles. J. Am. Chem. Soc. 1998, 120, 12367–12368. [Google Scholar]
- Fujihara, H.; Nakai, H. Fullerenethiolate-functionalized gold nanoparticles: A new class of surface-confined metal-c60 nanocomposites. Langmuir 2001, 17, 6393–6395. [Google Scholar]
- Sudeep, P.; Ipe, B.I.; Thomas, K.G.; George, M.; Barazzouk, S.; Hotchandani, S.; Kamat, P.V. Fullerene-functionalized gold nanoparticles. A self-assembled photoactive antenna-metal nanocore assembly. Nano Lett. 2002, 2, 29–35. [Google Scholar]
- Shon, Y.-S.; Choo, H. [60]Fullerene-linked gold nanoparticles: Synthesis and layer-by-layer growth on a solid surface. Chem. Commun. 2002, 2560–2561. [Google Scholar] [CrossRef]
- Liu, J.; Alvarez, J.; Ong, W.; Kaifer, A.E. Network aggregates formed by C60 and gold nanoparticles capped with γ-cyclodextrin hosts. Nano Lett. 2001, 1, 57–60. [Google Scholar]
- Hasobe, T.; Imahori, H.; Kamat, P.V.; Fukuzumi, S. Quaternary self-organization of porphyrin and fullerene units by clusterization with gold nanoparticles on SnO2 electrodes for organic solar cells. J. Am. Chem. Soc. 2003, 125, 14962–14963. [Google Scholar]
- Islam, M.T.; Molugu, S.K.; Cooke, P.H.; Noveron, J.C. Fullerene stabilized gold nanoparticles. New J. Chem. 2015, 39, 5923–5926. [Google Scholar]
- Frare, M.; Pilot, R.; De Filippo, C.; Weber, V.; Signorini, R.; Maggini, M.; Bozio, R. Fullerene functionalized gold nanoparticles for optical limiting of continuous wave lasers. Appl. Phys. B 2019, 125, 1–12. [Google Scholar]
- Plesek, J. Potential applications of the boron cluster compounds. Chem. Rev. 1992, 92, 269–278. [Google Scholar]
- Hawthorne, M.F.; Maderna, A. Applications of Radiolabeled Boron Clusters to the Diagnosis and Treatment of Cancer. Chem. Rev. 1999, 99, 3421–3434. [Google Scholar]
- Barbera, G.; Vaca, A.; Teixidor, F.; Sillanpää, R.; Kivekäs, R.; Vinas, C. Designed synthesis of new ortho-carborane derivatives: From mono-to polysubstituted frameworks. Inorg. Chem. 2008, 47, 7309–7316. [Google Scholar]
- Dash, B.P.; Satapathy, R.; Maguire, J.A.; Hosmane, N.S. Synthesis of a new class of carborane-containing star-shaped molecules via silicon tetrachloride promoted cyclotrimerization reactions. Org. Lett. 2008, 10, 2247–2250. [Google Scholar]
- Smith, H., Jr.; Obenland, C.; Papetti, S. A New Series of Organoboranes. IX. The Preparation and Some Reactions of Sulfur-Carborane Derivatives. Inorg. Chem. 1966, 5, 1013–1015. [Google Scholar]
- Plešek, J.; Heřmánek, S. Syntheses and properties of substituted icosahedral carborane thiols. Collect. Czechoslov. Chem. Commun. 1981, 46, 687–692. [Google Scholar]
- Baše, T.; Bastl, Z.; Plzák, Z.; Grygar, T.; Plešek, J.; Carr, M.J.; Malina, V.; Šubrt, J.; Bohácek, J.; Večerníková, E. Carboranethiol-modified gold surfaces. A study and comparison of modified cluster and flat surfaces. Langmuir 2005, 21, 7776–7785. [Google Scholar]
- Baše, T.S.; Bastl, Z.; Šlouf, M.; Klementová, M.; Šubrt, J.; Vetushka, A.; Ledinský, M.; Fejfar, A.; Machácek, J.; Carr, M.J. Gold micrometer crystals modified with carboranethiol derivatives. J. Phys. Chem. C 2008, 112, 14446–14455. [Google Scholar]
- Grzelczak, M.P.; Danks, S.P.; Klipp, R.C.; Belic, D.; Zaulet, A.; Kunstmann-Olsen, C.; Bradley, D.F.; Tsukuda, T.; Viñas, C.; Teixidor, F. Ion transport across biological membranes by carborane-capped gold nanoparticles. Acs Nano 2017, 11, 12492–12499. [Google Scholar]
- Wang, J.; Chen, L.; Ye, J.; Li, Z.; Jiang, H.; Yan, H.; Stogniy, M.Y.; Sivaev, I.B.; Bregadze, V.I.; Wang, X. Carborane derivative conjugated with gold nanoclusters for targeted cancer cell imaging. Biomacromolecules 2017, 18, 1466–1472. [Google Scholar]
- Bogireddy, N.; Pal, U.; Gomez, L.M.; Agarwal, V. Size controlled green synthesis of gold nanoparticles using Coffea arabica seed extract and their catalytic performance in 4-nitrophenol reduction. Rsc Adv. 2018, 8, 24819–24826. [Google Scholar]
- Schmid, G.; Pugin, R.; Malm, J.O.; Bovin, J.O. Silsesquioxanes as ligands for gold clusters. Eur. J. Inorg. Chem. 1998, 1998, 813–817. [Google Scholar]
- Carroll, J.B.; Frankamp, B.L.; Rotello, V.M. Self-assembly of gold nanoparticles through tandem hydrogen bonding and polyoligosilsequioxane (POSS)–POSS recognition processes. Chem. Commun. 2002, 1892–1893. [Google Scholar]
- Carroll, J.B.; Frankamp, B.L.; Srivastava, S.; Rotello, V.M. Electrostatic self-assembly of structured gold nanoparticle/polyhedral oligomeric silsesquioxane (POSS) nanocomposites. J. Mater. Chem. 2004, 14, 690–694. [Google Scholar]
- Zhang, Y.; Hao, J.; Xu, X.; Chen, X.; Wang, J. Protein Corona-Triggered Catalytic Inhibition of Insufficient POSS Polymer-Caged Gold Nanoparticles for Sensitive Colorimetric Detection of Metallothioneins. Anal. Chem. 2019, 92, 2080–2087. [Google Scholar]
- Xia, S.; Yang, Y.; Zhu, W.; Lü, C. Quaternized polyhedral oligomeric silsesquioxanes stabilized Pd nanoparticles as efficient nanocatalysts for reduction reaction. Colloids Surf. A Physicochem. Eng. Asp. 2020, 585, 124110. [Google Scholar]
- Hanske, C.; Sanz-Ortiz, M.N.; Liz-Marzán, L.M. Silica-Coated Plasmonic Metal Nanoparticles in Action. Adv. Mater. 2018, 30, 1707003. [Google Scholar]
- Chapman, B.S.; Wu, W.-C.; Li, Q.; Holten-Andersen, N.; Tracy, J.B. Heteroaggregation approach for depositing magnetite nanoparticles onto Silica-overcoated gold nanorods. Chem. Mater. 2017, 29, 10362–10368. [Google Scholar]
- Liz-Marzán, L.M.; Giersig, M.; Mulvaney, P. Synthesis of nanosized gold-silica core-shell particles. Langmuir 1996, 12, 4329–4335. [Google Scholar]
- Han, Y.; Jiang, J.; Lee, S.S.; Ying, J.Y. Reverse microemulsion-mediated synthesis of silica-coated gold and silver nanoparticles. Langmuir 2008, 24, 5842–5848. [Google Scholar]
- Casavola, M.; Buonsanti, R.; Caputo, G.; Cozzoli, P.D. Colloidal strategies for preparing oxide-based hybrid nanocrystals. Eur. J. Inorg. Chem. 2008, 2008, 837–854. [Google Scholar]
- Rodríguez-Fernández, J.; Pastoriza-Santos, I.; Perez-Juste, J.; García de Abajo, F.J.; Liz-Marzan, L.M. The effect of silica coating on the optical response of sub-micrometer gold spheres. J. Phys. Chem. C 2007, 111, 13361–13366. [Google Scholar]
- Ruach-Nir, I.; Bendikov, T.A.; Doron-Mor, I.; Barkay, Z.; Vaskevich, A.; Rubinstein, I. Silica-stabilized gold island films for transmission localized surface plasmon sensing. J. Am. Chem. Soc. 2007, 129, 84–92. [Google Scholar]
- Liu, G.; Ji, H.; Yang, X.; Wang, Y. Synthesis of a Au/silica/polymer trilayer composite and the corresponding hollow polymer microsphere with a movable Au core. Langmuir 2008, 24, 1019–1025. [Google Scholar]
- Tovmachenko, O.G.; Graf, C.; van den Heuvel, D.J.; van Blaaderen, A.; Gerritsen, H.C. Fluorescence enhancement by metal-core/silica-shell nanoparticles. Adv. Mater. 2006, 18, 91–95. [Google Scholar]
- Kamata, K.; Lu, Y.; Xia, Y. Synthesis and characterization of monodispersed core-shell spherical colloids with movable cores. J. Am. Chem. Soc. 2003, 125, 2384–2385. [Google Scholar]
- Iijima, S. Synthesis of carbon nanotubes. Nature 1991, 354, 56–58. [Google Scholar]
- Andrews, R.; Jacques, D.; Qian, D.; Rantell, T. Multiwall carbon nanotubes: Synthesis and application. Acc. Chem. Res. 2002, 35, 1008–1017. [Google Scholar]
- Koo, W.-T.; Kim, Y.; Savagatrup, S.; Yoon, B.; Jeon, I.; Choi, S.-J.; Kim, I.-D.; Swager, T.M. Porous Ion Exchange Polymer Matrix for Ultrasmall Au Nanoparticle-Decorated Carbon Nanotube Chemiresistors. Chem. Mater. 2019, 31, 5413–5420. [Google Scholar]
- Deshmukh, S.P.; Dhodamani, A.G.; Patil, S.M.; Mullani, S.B.; More, K.V.; Delekar, S.D. Interfacially Interactive Ternary Silver-Supported Polyaniline/Multiwalled Carbon Nanotube Nanocomposites for Catalytic and Antibacterial Activity. Acs Omega 2019, 5, 219–227. [Google Scholar]
- LeeáTan, K. Growth of Pd, Pt, Ag and Au nanoparticles on carbon nanotubes. J. Mater. Chem. 2001, 11, 2378–2381. [Google Scholar]
- Han, L.; Wu, W.; Kirk, F.L.; Luo, J.; Maye, M.M.; Kariuki, N.N.; Lin, Y.; Wang, C.; Zhong, C.-J. A direct route toward assembly of nanoparticle-carbon nanotube composite materials. Langmuir 2004, 20, 6019–6025. [Google Scholar]
- Sainsbury, T.; Ikuno, T.; Okawa, D.; Pacile, D.; Frechet, J.M.; Zettl, A. Self-assembly of gold nanoparticles at the surface of amine-and thiol-functionalized boron nitride nanotubes. J. Phys. Chem. C 2007, 111, 12992–12999. [Google Scholar]
- Jiang, K.; Eitan, A.; Schadler, L.S.; Ajayan, P.M.; Siegel, R.W.; Grobert, N.; Mayne, M.; Reyes-Reyes, M.; Terrones, H.; Terrones, M. Selective attachment of gold nanoparticles to nitrogen-doped carbon nanotubes. Nano Lett. 2003, 3, 275–277. [Google Scholar]
- Correa-Duarte, M.A.; Liz-Marzán, L.M. Carbon nanotubes as templates for one-dimensional nanoparticle assemblies. J. Mater. Chem. 2006, 16, 22–25. [Google Scholar]
- Fu, Q.; Lu, C.; Liu, J. Selective coating of single wall carbon nanotubes with thin SiO2 layer. Nano Lett. 2002, 2, 329–332. [Google Scholar]
- Bottini, M.; Magrini, A.; Rosato, N.; Bergamaschi, A.; Mustelin, T. Dispersion of pristine single-walled carbon nanotubes in water by a thiolated organosilane: Application in supramolecular nanoassemblies. J. Phys. Chem. B 2006, 110, 13685–13688. [Google Scholar]
- Ou, Y.-Y.; Huang, M.H. High-density assembly of gold nanoparticles on multiwalled carbon nanotubes using 1-pyrenemethylamine as interlinker. J. Phys. Chem. B 2006, 110, 2031–2036. [Google Scholar]
- Hill, C.L. Introduction: Polyoxometalates multicomponent molecular vehicles to probe fundamental issues and practical problems. Chem. Rev. 1998, 98, 1–2. [Google Scholar]
- Wang, Y.; Neyman, A.; Arkhangelsky, E.; Gitis, V.; Meshi, L.; Weinstock, I.A. Self-assembly and structure of directly imaged inorganic-anion monolayers on a gold nanoparticle. J. Am. Chem. Soc. 2009, 131, 17412–17422. [Google Scholar]
- Wang, Y.; Zeiri, O.; Sharet, S.; Weinstock, I.A. Role of the alkali-metal cation size in the self-assembly of polyoxometalate-monolayer shells on gold nanoparticles. Inorg. Chem. 2012, 51, 7436–7438. [Google Scholar]
- Sutter, S.; Trepka, B.; Siroky, S.; Hagedorn, K.; Theiß, S.; Baum, P.; Polarz, S. Light-Triggered Boost of Activity of Catalytic Bola-Type Surfactants by a Plasmonic Metal–Support Interaction Effect. Acs Appl. Mater. Interfaces 2019, 11, 15936–15944. [Google Scholar]
- Solarska, R.; Bienkowski, K.; Zoladek, S.; Majcher, A.; Stefaniuk, T.; Kulesza, P.J.; Augustynski, J. Enhanced water splitting at thin film tungsten trioxide photoanodes bearing plasmonic gold–polyoxometalate particles. Angew. Chem. Int. Ed. 2014, 53, 14196–14200. [Google Scholar]
- Dolbecq, A.; Dumas, E.; Mayer, C.R.; Mialane, P. Hybrid organic-inorganic polyoxometalate compounds: From structural diversity to applications. Chem. Rev. 2010, 110, 6009–6048. [Google Scholar]
- Mayer, C.R.; Neveu, S.; Cabuil, V. A nanoscale hybrid system based on gold nanoparticles and heteropolyanions. Angew. Chem. Int. Ed. 2002, 41, 501–503. [Google Scholar]
- Hegde, S.; Joshi, S.; Mukherjee, T.; Kapoor, S. Formation of gold nanoparticles via a thiol functionalized polyoxometalate. Mater. Sci. Eng. C 2013, 33, 2332–2337. [Google Scholar]
- Martín, S.; Takashima, Y.; Lin, C.-G.; Song, Y.-F.; Miras, H.N.; Cronin, L. Integrated synthesis of gold nanoparticles coated with polyoxometalate clusters. Inorg. Chem. 2019, 58, 4110–4116. [Google Scholar]
- Tomane, S.; López-Maya, E.; Boujday, S.; Humblot, V.; Marrot, J.; Rabasso, N.; Castells-Gil, J.; Sicard, C.; Dolbecq, A.; Mialane, P. Correction: One-pot synthesis of a new generation of hybrid bisphosphonate polyoxometalate gold nanoparticles as antibiofilm agents. Nanoscale Adv. 2019, 1, 4173. [Google Scholar]
- Wilton-Ely, J.D. The surface functionalisation of gold nanoparticles with metal complexes. Dalton Trans. 2008, 25–29. [Google Scholar] [CrossRef]
- Stiles, R.L.; Balasubramanian, R.; Feldberg, S.W.; Murray, R.W. Anion-induced adsorption of ferrocenated nanoparticles. J. Am. Chem. Soc. 2008, 130, 1856–1865. [Google Scholar]
- Wolfe, R.L.; Balasubramanian, R.; Tracy, J.B.; Murray, R.W. Fully ferrocenated hexanethiolate monolayer-protected gold clusters. Langmuir 2007, 23, 2247–2254. [Google Scholar]
- Chen, S.; Murray, R.W. Arenethiolate monolayer-protected gold clusters. Langmuir 1999, 15, 682–689. [Google Scholar]
- Li, D.; Zhang, Y.; Jiang, J.; Li, J. Electroactive gold nanoparticles protected by 4-ferrocene thiophenol monolayer. J. Colloid Interface Sci. 2003, 264, 109–113. [Google Scholar]
- Li, D.; Zhang, Y.; Li, J. Electrochemical study of 4-ferrocene thiophenol monolayers assembled on gold nanoparticles. Microelectron. Eng. 2003, 66, 91–94. [Google Scholar]
- Ingram, R.S.; Hostetler, M.J.; Murray, R.W. Poly-hetero-ω-functionalized alkanethiolate-stabilized gold cluster compounds. J. Am. Chem. Soc. 1997, 119, 9175–9178. [Google Scholar]
- Green, S.J.; Stokes, J.J.; Hostetler, M.J.; Pietron, J.; Murray, R.W. Three-dimensional monolayers: Nanometer-sized electrodes of alkanethiolate-stabilized gold cluster molecules. J. Phys. Chem. B 1997, 101, 2663–2668. [Google Scholar]
- Ornelas, C.; Méry, D.; Cloutet, E.; Aranzaes, J.R.; Astruc, D. Cross olefin metathesis for the selective functionalization, ferrocenylation, and solubilization in water of olefin-terminated dendrimers, polymers, and gold nanoparticles and for a divergent dendrimer construction. J. Am. Chem. Soc. 2008, 130, 1495–1506. [Google Scholar]
- Labande, A.; Astruc, D. Colloids as redox sensors: Recognition of H2PO4− and HSO4− by amidoferrocenylalkylthiol–gold nanoparticles. Chem. Commun. 2000, 124, 1007–1008. [Google Scholar]
- Wang, Y.; Salmon, L.; Ruiz, J.; Astruc, D. Metallodendrimers in three oxidation states with electronically interacting metals and stabilization of size-selected gold nanoparticles. Nat. Commun. 2014, 5, 1–12. [Google Scholar]
- Labande, A.; Ruiz, J.; Astruc, D. Supramolecular gold nanoparticles for the redox recognition of oxoanions: Syntheses, titrations, stereoelectronic effects, and selectivity. J. Am. Chem. Soc. 2002, 124, 1782–1789. [Google Scholar]
- Horikoshi, T.; Itoh, M.; Kurihara, M.; Kubo, K.; Nishihara, H. Synthesis, redox behavior and electrodeposition of biferrocene-modified gold clusters. J. Electroanal. Chem. 1999, 473, 113–116. [Google Scholar]
- Yamada, M.; Tadera, T.; Kubo, K.; Nishihara, H. Electrochemical deposition of biferrocene derivative-attached gold nanoparticles and the morphology of the formed film. J. Phys. Chem. B 2003, 107, 3703–3711. [Google Scholar]
- Dong, T.-Y.; Shih, H.-W.; Chang, L.-S. Synthesis and redox behavior of biferrocenyl-functionalized ruthenium (II) terpyridine gold clusters. Langmuir 2004, 20, 9340–9347. [Google Scholar]
- Daniel, M.-C.; Ruiz, J.; Nlate, S.; Palumbo, J.; Blais, J.-C.; Astruc, D. Gold nanoparticles containing redox-active supramolecular dendrons that recognize H2PO4−. Chem. Commun. 2001, 2000–2001. [Google Scholar]
- Daniel, M.-C.; Ruiz, J.; Nlate, S.; Blais, J.-C.; Astruc, D. Nanoscopic assemblies between supramolecular redox active metallodendrons and gold nanoparticles: Synthesis, characterization, and selective recognition of H2PO4−, HSO4−, and adenosine-5′-triphosphate (ATP2-) anions. J. Am. Chem. Soc. 2003, 125, 2617–2628. [Google Scholar]
- Astruc, D.; Daniel, M.-C.; Ruiz, J. Dendrimers and gold nanoparticles as exo-receptors sensing biologically important anions. Chem. Commun. 2004, 2637–2649. [Google Scholar]
- Liu, F.; Liu, X.; Astruc, D.; Gu, H. Dendronized triazolyl-containing ferrocenyl polymers as stabilizers of gold nanoparticles for recyclable two-phase reduction of 4-nitrophenol. J. Colloid Interface Sci. 2019, 533, 161–170. [Google Scholar]
- Opuchlik, L.J.; Pawłowska, J.; Sęk, S.; Bilewicz, R. Ferrocenylated gold nanoparticles self-assemble at carbon surfaces to form stable films. J. Electroanal. Chem. 2018, 825, 22–29. [Google Scholar]
- Vitale, F.; Vitaliano, R.; Battocchio, C.; Fratoddi, I.; Piscopiello, E.; Tapfer, L.; Russo, M.V. Synthesis and characterization of gold nanoparticles stabilized by palladium (II) phosphine thiol. J. Organomet. Chem. 2008, 693, 1043–1048. [Google Scholar]
- Bartz, M.; Küther, J.; Seshadri, R.; Tremel, W. Colloid-Bound Catalysts for Ring-Opening Metathesis Polymerization: A Combination of Homogenous and Heterogeneous Properties. Angew. Chem. Int. Ed. 1998, 37, 2466–2468. [Google Scholar]
- Wang, S.; Sim, W.-S. Au nanoparticles encapsulated in Ru carbonyl carboxylate shells. Langmuir 2006, 22, 7861–7866. [Google Scholar]
- Belser, T.; Stöhr, M.; Pfaltz, A. Immobilization of rhodium complexes at thiolate monolayers on gold surfaces: Catalytic and structural studies. J. Am. Chem. Soc. 2005, 127, 8720–8731. [Google Scholar]
- Kumar, S.S.; Joseph, J.; Phani, K.L. Novel method for deposition of Gold-Prussian blue nanocomposite films induced by electrochemically formed gold nanoparticles: Characterization and application to electrocatalysis. Chem. Mater. 2007, 19, 4722–4730. [Google Scholar]
- Crespilho, F.N.; Zucolotto, V.; Brett, C.M.; Oliveira, O.N.; Nart, F.C. Enhanced charge transport and incorporation of redox mediators in layer-by-layer films containing PAMAM-encapsulated gold nanoparticles. J. Phys. Chem. B 2006, 110, 17478–17483. [Google Scholar]
- Qiu, J.-D.; Peng, H.-Z.; Liang, R.-P.; Li, J.; Xia, X.-H. Synthesis, characterization, and immobilization of Prussian blue-modified Au nanoparticles: Application to electrocatalytic reduction of H2O2. Langmuir 2007, 23, 2133–2137. [Google Scholar]
- Dumur, F.; Dumas, E.; Mayer, C.R. Functionalization of Gold Nanoparticles by Inorganic Entities. Nanomaterials 2020, 10, 548. [Google Scholar]
- Toma, S.H.; Bonacin, J.A.; Araki, K.; Toma, H.E. Controlled Stabilization and Flocculation of Gold Nanoparticles by Means of 2-Pyrazin-2-ylethanethiol and Pentacyanidoferrate (II) Complexes. Eur. J. Inorg. Chem. 2007, 2007, 3356–3364. [Google Scholar]
- Nunes, F.S.; Bonifácio, L.D.S.; Araki, K.; Toma, H.E. Interaction of 2-and 4-mercaptopyridine with pentacyanoferrates and gold nanoparticles. Inorg. Chem. 2006, 45, 94–101. [Google Scholar]
- Cheng, P.P.H.; Silvester, D.; Wang, G.; Kalyuzhny, G.; Douglas, A.; Murray, R.W. Dynamic and static quenching of fluorescence by 1–4 nm diameter gold monolayer-protected clusters. J. Phys. Chem. B 2006, 110, 4637–4644. [Google Scholar]
- Brewer, S.H.; Glomm, W.R.; Johnson, M.C.; Knag, M.K.; Franzen, S. Probing BSA binding to citrate-coated gold nanoparticles and surfaces. Langmuir 2005, 21, 9303–9307. [Google Scholar]
- Xu, X.-H.N.; Huang, S.; Brownlow, W.; Salaita, K.; Jeffers, R.B. Size and temperature dependence of surface plasmon absorption of gold nanoparticles induced by tris (2, 2′-bipyridine) ruthenium (II). J. Phys. Chem. B 2004, 108, 15543–15551. [Google Scholar]
- Pramod, P.; Sudeep, P.; Thomas, K.G.; Kamat, P.V. Photochemistry of ruthenium trisbipyridine functionalized on gold nanoparticles. J. Phys. Chem. B 2006, 110, 20737–20741. [Google Scholar]
- Jebb, M.; Sudeep, P.; Pramod, P.; Thomas, K.G.; Kamat, P.V. Ruthenium (II) trisbipyridine functionalized gold nanorods. Morphological changes and excited-state interactions. J. Phys. Chem. B 2007, 111, 6839–6844. [Google Scholar]
- Sun, X.; Du, Y.; Dong, S.; Wang, E. Method for effective immobilization of Ru (bpy) 32+ on an electrode surface for solid-state electrochemiluminescene detection. Anal. Chem. 2005, 77, 8166–8169. [Google Scholar]
- Liu, J.; Lu, Y. Stimuli-responsive disassembly of nanoparticle aggregates for light-up colorimetric sensing. J. Am. Chem. Soc. 2005, 127, 12677–12683. [Google Scholar]
- Kuwahara, Y.; Akiyama, T.; Yamada, S. Construction of gold nanoparticle-ruthenium (II) tris (2, 2′-bipyridine) self-assembled multistructures and their photocurrent responses. Thin Solid Film. 2001, 393, 273–277. [Google Scholar]
- Akiyama, T.; Inoue, K.; Kuwahara, Y.; Terasaki, N.; Niidome, Y.; Yamada, S. Particle-size effects on the photocurrent efficiency of nanostructured assemblies consisting of gold nanoparticles and a ruthenium complex–viologen linked thiol. J. Electroanal. Chem. 2003, 550, 303–307. [Google Scholar]
- Akiyama, T.; Inoue, K.; Kuwahara, Y.; Niidome, Y.; Terasaki, N.; Nitahara, S.; Yamada, S. Facile fabrication of morphology-controlled gold nanoparticle architectures by electrolyte-induced agglomeration and their photoelectrochemical applications. Langmuir 2005, 21, 793–796. [Google Scholar]
- Lahav, M.; Heleg-Shabtai, V.; Wasserman, J.; Katz, E.; Willner, I.; Dürr, H.; Hu, Y.-Z.; Bossmann, S.H. Photoelectrochemistry with Integrated Photosensitizer-Electron Acceptor and Au-Nanoparticle Arrays. J. Am. Chem. Soc. 2000, 122, 11480–11487. [Google Scholar]
- Shipway, A.N.; Lahav, M.; Willner, I. Nanostructured gold colloid electrodes. Adv. Mater. 2000, 12, 993–998. [Google Scholar]
- Chen, S.; Pei, R.; Zhao, T.; Dyer, D.J. Gold Nanoparticle Assemblies by Metal Ion-Pyridine Complexation and Their Rectified Quantized Charging in Aqueous Solutions. J. Phys. Chem. B 2002, 106, 1903–1908. [Google Scholar]
- Soller, T.; Ringler, M.; Wunderlich, M.; Klar, T.; Feldmann, J.; Josel, H.-P.; Markert, Y.; Nichtl, A.; Kürzinger, K. Radiative and nonradiative rates of phosphors attached to gold nanoparticles. Nano Lett. 2007, 7, 1941–1946. [Google Scholar]
- Slim, M.; Durisic, N.; Grutter, P.; Sleiman, H.F. DNA–Protein Noncovalent Cross-Linking: Ruthenium Dipyridophenazine Biotin Complex for the Assembly of Proteins and Gold Nanoparticles on DNA Templates. ChemBioChem 2007, 8, 804–812. [Google Scholar]
- Shultz, D.A.; Kumar, R.K.; Bin-Salamon, S.; Kirk, M.L. Valence tautomerization and exchange coupling in a cobalt–nitronylnitroxide–semiquinone complex. Polyhedron 2005, 24, 2876–2879. [Google Scholar]
- Barmin, R.A.; Rudakovskaya, P.G.; Gusliakova, O.I.; Sindeeva, O.A.; Prikhozhdenko, E.S.; Maksimova, E.A.; Obukhova, E.N.; Chernyshev, V.S.; Khlebtsov, B.N.; Solovev, A.A.; et al. Air-Filled Bubbles Stabilized by Gold Nanoparticle/Photodynamic Dye Hybrid Structures for Theranostics. Nanomaterials 2021, 11, 415. [Google Scholar]
- Mayer, C.R.; Dumas, E.; Miomandre, F.; Méallet-Renault, R.; Warmont, F.; Vigneron, J.; Pansu, R.; Etcheberry, A.; Sécheresse, F. Polypyridyl ruthenium complexes as coating agent for the formation of gold and silver nanocomposites in different media. Preliminary luminescence and electrochemical studies. New J. Chem. 2006, 30, 1628–1637. [Google Scholar]
- Pérez León, C.; Kador, L.; Peng, B.; Thelakkat, M. Influence of the solvent on the surface-enhanced Raman spectra of ruthenium (II) bipyridyl complexes. J. Phys. Chem. B 2005, 109, 5783–5789. [Google Scholar]
- Huang, W.; Tanaka, H.; Ogawa, T. Effects of Metal-Ion Complexation for the Self-Assembled Nanocomposite Films Composed of Gold Nanoparticles and 3, 8-Bis (terthiophenyl) phenanthroline-Based Dithiols Bridging 1 μm Gap Gold Electrodes: Morphology, Temperature Dependent Electronic Conduction, and Photoresponse. J. Phys. Chem. C 2008, 112, 11513–11526. [Google Scholar]
- Ono, F.; Kanemasa, S.; Tanaka, J. Reusable nano-sized chiral bisoxazoline catalysts. Tetrahedron Lett. 2005, 46, 7623–7626. [Google Scholar]
- Hone, D.C.; Walker, P.I.; Evans-Gowing, R.; FitzGerald, S.; Beeby, A.; Chambrier, I.; Cook, M.J.; Russell, D.A. Generation of cytotoxic singlet oxygen via phthalocyanine-stabilized gold nanoparticles: A potential delivery vehicle for photodynamic therapy. Langmuir 2002, 18, 2985–2987. [Google Scholar]
- Beer, P.D.; Cormode, D.P.; Davis, J.J. Zinc metalloporphyrin-functionalised nanoparticle anion sensors. Chem. Commun. 2004, 414–415. [Google Scholar]
- Ozawa, H.; Kawao, M.; Tanaka, H.; Ogawa, T. Synthesis of dendron-protected porphyrin wires and preparation of a one-dimensional assembly of gold nanoparticles chemically linked to the π-conjugated wires. Langmuir 2007, 23, 6365–6371. [Google Scholar]
- Aubin-Tam, M.-E.; Hamad-Schifferli, K. Gold nanoparticle-cytochrome C complexes: The effect of nanoparticle ligand charge on protein structure. Langmuir 2005, 21, 12080–12084. [Google Scholar]
- Sheet, D.; Bera, A.; Jana, R.D.; Paine, T.K. Oxidizing Ability of a Dioxygen-Activating Nonheme Iron (II)-Benzilate Complex Immobilized on Gold Nanoparticles. Inorg. Chem. 2019, 58, 4828–4841. [Google Scholar]
- Huang, C.C.; Yang, Z.; Lee, K.H.; Chang, H.T. Synthesis of highly fluorescent gold nanoparticles for sensing mercury (II). Angew. Chem. 2007, 119, 6948–6952. [Google Scholar]
- Huang, C.-C.; Chang, H.-T. Parameters for selective colorimetric sensing of mercury (II) in aqueous solutions using mercaptopropionic acid-modified gold nanoparticles. Chem. Commun. 2007, 1215–1217. [Google Scholar]
- Zamborini, F.P.; Hicks, J.F.; Murray, R.W. Quantized double layer charging of nanoparticle films assembled using carboxylate/(Cu2+ or Zn2+)/carboxylate bridges. J. Am. Chem. Soc. 2000, 122, 4514–4515. [Google Scholar]
- Zamborini, F.P.; Leopold, M.C.; Hicks, J.F.; Kulesza, P.J.; Malik, M.A.; Murray, R.W. Electron hopping conductivity and vapor sensing properties of flexible network polymer films of metal nanoparticles. J. Am. Chem. Soc. 2002, 124, 8958–8964. [Google Scholar]
- Hicks, J.F.; Zamborini, F.P.; Murray, R.W. Dynamics of electron transfers between electrodes and monolayers of nanoparticles. J. Phys. Chem. B 2002, 106, 7751–7757. [Google Scholar]
- Sheibley, D.; Tognarelli, D.; Szymanik, R.; Leopold, M.C. Ultra-fast formation and characterization of stable nanoparticle film assemblies. J. Mater. Chem. 2005, 15, 491–498. [Google Scholar]
- Xu, R.; Wang, D.; Zhang, J.; Li, Y. Shape-dependent catalytic activity of silver nanoparticles for the oxidation of styrene. Chem. Asian J. 2006, 1, 888–893. [Google Scholar]
- Wanunu, M.; Popovitz-Biro, R.; Cohen, H.; Vaskevich, A.; Rubinstein, I. Coordination-based gold nanoparticle layers. J. Am. Chem. Soc. 2005, 127, 9207–9215. [Google Scholar]
- Mayer, C.R.; Cucchiaro, G.; Jullien, J.; Dumur, F.; Marrot, J.; Dumas, E.; Sécheresse, F. Functionalization of gold nanoparticles by iron (III) complexes derived from Schiff base ligands. Eur. J. Inorg. Chem. 2008, 2008, 3614–3623. [Google Scholar]
- Guo, C.; Boullanger, P.; Jiang, L.; Liu, T. Highly sensitive gold nanoparticles biosensor chips modified with a self-assembled bilayer for detection of Con A. Biosens. Bioelectron. 2007, 22, 1830–1834. [Google Scholar]
- Türkekul, K.; Üzer, A.; Can, Z.; Erçağ, E.; Apak, R. Colorimetric Sensing of the Insensitive Energetic Material 3-Nitro-1, 2, 4-triazol-5-one (NTO) Using l-Cysteine Stabilized Gold Nanoparticles and Copper (II). Anal. Lett. 2019, 52, 2809–2821. [Google Scholar]
- Abad, J.M.; Mertens, S.F.; Pita, M.; Fernández, V.M.; Schiffrin, D.J. Functionalization of thioctic acid-capped gold nanoparticles for specific immobilization of histidine-tagged proteins. J. Am. Chem. Soc. 2005, 127, 5689–5694. [Google Scholar]
- Reynolds, A.J.; Haines, A.H.; Russell, D.A. Gold glyconanoparticles for mimics and measurement of metal ion-mediated carbohydrate-carbohydrate interactions. Langmuir 2006, 22, 1156–1163. [Google Scholar]
- Si, S.; Kotal, A.; Mandal, T.K. One-dimensional assembly of peptide-functionalized gold nanoparticles: An approach toward mercury ion sensing. J. Phys. Chem. C 2007, 111, 1248–1255. [Google Scholar]
- Lee, J.S.; Han, M.S.; Mirkin, C.A. Colorimetric detection of mercuric ion (Hg2+) in aqueous media using DNA-functionalized gold nanoparticles. Angew. Chem. Int. Ed. 2007, 46, 4093–4096. [Google Scholar]
- Liu, J.; Lu, Y. A colorimetric lead biosensor using DNAzyme-directed assembly of gold nanoparticles. J. Am. Chem. Soc. 2003, 125, 6642–6643. [Google Scholar]
- Liu, J.; Lu, Y. Accelerated color change of gold nanoparticles assembled by DNAzymes for simple and fast colorimetric Pb2+ detection. J. Am. Chem. Soc. 2004, 126, 12298–12305. [Google Scholar]
- Liu, J.; Lu, Y. Optimization of a Pb2+-directed gold nanoparticle/DNAzyme assembly and its application as a colorimetric biosensor for Pb2+. Chem. Mater. 2004, 16, 3231–3238. [Google Scholar]
- Liu, J.; Lu, Y. Colorimetric Cu2+ detection with a ligation DNAzyme and nanoparticles. Chem. Commun. 2007, 4872–4874. [Google Scholar] [CrossRef]
- Marubayashi, K.; Takizawa, S.; Kawakusu, T.; Arai, T.; Sasai, H. Monolayer-Protected Au Cluster (MPC)-Supported Ti− BINOLate Complex. Org. Lett. 2003, 5, 4409–4412. [Google Scholar]
- Rogers, N.J.; Claire, S.; Harris, R.M.; Farabi, S.; Zikeli, G.; Styles, I.B.; Hodges, N.J.; Pikramenou, Z. High coating of Ru (II) complexes on gold nanoparticles for single particle luminescence imaging in cells. Chem. Commun. 2014, 50, 617–619. [Google Scholar]
- Thangavel, N.; Jayakumar, I.; Ravichandran, M.; Ganesan, V.V.; Nair, B.U. Photocrosslinking of collagen using Ru (II)-polypyridyl complex functionalized gold nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 215, 196–202. [Google Scholar]
- Cox, J.A.; Kittredge, K.W.; Ca, D.V. Measurement platforms fabricated by layer-by-layer assembly of crown ether functionalized gold nanoclusters. J. Solid State Electrochem. 2004, 8, 722–726. [Google Scholar]
- Lin, S.Y.; Wu, S.H.; Chen, C.h. A simple strategy for prompt visual sensing by gold nanoparticles: General applications of interparticle hydrogen bonds. Angew. Chem. 2006, 118, 5070–5073. [Google Scholar]
- Wang, C.; Yu, C. Detection of chemical pollutants in water using gold nanoparticles as sensors: A review. Rev. Anal. Chem. 2013, 32, 1–14. [Google Scholar]
- Yamauchi, A.; Hayashita, T.; Nishizawa, S.; Watanabe, M.; Teramae, N. Benzo-15-crown-5 fluoroionophore/γ-cyclodextrin complex with remarkably high potassium ion sensitivity and selectivity in water. J. Am. Chem. Soc. 1999, 121, 2319–2320. [Google Scholar]
- Xia, W.-S.; Schmehl, R.H.; Li, C.-J. A highly selective fluorescent chemosensor for K+ from a bis-15-crown-5 derivative. J. Am. Chem. Soc. 1999, 121, 5599–5600. [Google Scholar]
- Siddique, S.; Chow, J.C.L. Gold Nanoparticles for Drug Delivery and Cancer Therapy. Appl. Sci. 2020, 10, 3824. [Google Scholar]
- Lin, S.-Y.; Liu, S.-W.; Lin, C.-M.; Chen, C.-h. Recognition of potassium ion in water by 15-crown-5 functionalized gold nanoparticles. Anal. Chem. 2002, 74, 330–335. [Google Scholar]
- Pompano, R.R.; Wortley, P.G.; Moatz, L.M.; Tognarelli, D.; Kittredge, K.W.; Leopold, M.C. Crown ether-metal “sandwiches” as linking mechanisms in assembled nanoparticle films. Thin Solid Film. 2006, 510, 311–319. [Google Scholar]
- Lin, S.-Y.; Chen, C.-h.; Lin, M.-C.; Hsu, H.-F. A cooperative effect of bifunctionalized nanoparticles on recognition: Sensing alkali ions by crown and carboxylate moieties in aqueous media. Anal. Chem. 2005, 77, 4821–4828. [Google Scholar]
- Gao, J.; Fu, J.; Lin, C.; Lin, J.; Han, Y.; Yu, X.; Pan, C. Formation and photoluminescence of silver nanoparticles stabilized by a two-armed polymer with a crown ether core. Langmuir 2004, 20, 9775–9779. [Google Scholar]
- Kuang, H.; Chen, W.; Yan, W.; Xu, L.; Zhu, Y.; Liu, L.; Chu, H.; Peng, C.; Wang, L.; Kotov, N.A. Crown ether assembly of gold nanoparticles: Melamine sensor. Biosens. Bioelectron. 2011, 26, 2032–2037. [Google Scholar]
- Sung, S.; Holmes, H.; Wainwright, L.; Toscani, A.; Stasiuk, G.J.; White, A.J.; Bell, J.D.; Wilton-Ely, J.D. Multimetallic complexes and functionalized gold nanoparticles based on a combination of d-and f-elements. Inorg. Chem. 2014, 53, 1989–2005. [Google Scholar]
- Truman, L.K.; Bradberry, S.J.; Comby, S.; Kotova, O.; Gunnlaugsson, T. Surface-Modified Gold Nanoparticles Possessing Two-Channel Responsive EuIII/TbIII Cyclen Complexes as Luminescent Logic Gate Mimics. ChemPhysChem 2017, 18, 1746–1751. [Google Scholar]
- Lewis, D.J.; Day, T.M.; MacPherson, J.V.; Pikramenou, Z. Luminescent nanobeads: Attachment of surface reactive Eu (III) complexes to gold nanoparticles. Chem. Commun. 2006, 1433–1435. [Google Scholar] [CrossRef]
- Massue, J.; Quinn, S.J.; Gunnlaugsson, T. Lanthanide luminescent displacement assays: The sensing of phosphate anions using Eu (III)-Cyclen-conjugated gold nanoparticles in aqueous solution. J. Am. Chem. Soc. 2008, 130, 6900–6901. [Google Scholar]
- Ipe, B.I.; Yoosaf, K.; Thomas, K.G. Functionalized gold nanoparticles as phosphorescent nanomaterials and sensors. J. Am. Chem. Soc. 2006, 128, 1907–1913. [Google Scholar]
- Alric, C.; Taleb, J.; Le Duc, G.; Mandon, C.; Billotey, C.; Le Meur-Herland, A.; Brochard, T.; Vocanson, F.; Janier, M.; Perriat, P. Gadolinium chelate coated gold nanoparticles as contrast agents for both X-ray computed tomography and magnetic resonance imaging. J. Am. Chem. Soc. 2008, 130, 5908–5915. [Google Scholar]
- Debouttière, P.J.; Roux, S.; Vocanson, F.; Billotey, C.; Beuf, O.; Favre-Réguillon, A.; Lin, Y.; Pellet-Rostaing, S.; Lamartine, R.; Perriat, P. Design of gold nanoparticles for magnetic resonance imaging. Adv. Funct. Mater. 2006, 16, 2330–2339. [Google Scholar]
- Moriggi, L.; Cannizzo, C.; Dumas, E.; Mayer, C.R.; Ulianov, A.; Helm, L. Gold nanoparticles functionalized with gadolinium chelates as high-relaxivity MRI contrast agents. J. Am. Chem. Soc. 2009, 131, 10828–10829. [Google Scholar]
- Liu, J.; Xiong, Z.; Zhang, J.; Peng, C.; Klajnert-Maculewicz, B.; Shen, M.; Shi, X. Zwitterionic gadolinium (III)-complexed dendrimer-entrapped gold nanoparticles for enhanced computed tomography/magnetic resonance imaging of lung cancer metastasis. Acs Appl. Mater. Interfaces 2019, 11, 15212–15221. [Google Scholar]
- Martins, J.; Ferreira, P.M.; Geraldes, C.F.; Helm, L.; Prata, M.I.; Rodrigues, S.P.; Rodrigues, T.B.; Calle, D.; López-Larrubia, P.; Cerdan, S. Gold nanoparticles functionalised with fast water exchanging Gd3+ chelates: Linker effects on the relaxivity. Dalton Trans. 2015, 44, 4016–4031. [Google Scholar]
- Haiss, W.; Thanh, N.T.; Aveyard, J.; Fernig, D.G. Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar]
- Perrault, S.D.; Chan, W.C. Synthesis and surface modification of highly monodispersed, spherical gold nanoparticles of 50–200 nm. J. Am. Chem. Soc. 2009, 131, 17042–17043. [Google Scholar]
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.; Rotello, V.M. Gold nanoparticles in chemical and biological sensing. Chem. Rev. 2012, 112, 2739–2779. [Google Scholar]
- Li, K.; Wang, K.; Qin, W.; Deng, S.; Li, D.; Shi, J.; Huang, Q.; Fan, C. DNA-directed assembly of gold nanohalo for quantitative plasmonic imaging of single-particle catalysis. J. Am. Chem. Soc. 2015, 137, 4292–4295. [Google Scholar]
- Young, K.L.; Ross, M.B.; Blaber, M.G.; Rycenga, M.; Jones, M.R.; Zhang, C.; Senesi, A.J.; Lee, B.; Schatz, G.C.; Mirkin, C.A. Using DNA to design plasmonic metamaterials with tunable optical properties. Adv. Mater. 2014, 26, 653–659. [Google Scholar]
- Xu, X.; Han, M.S.; Mirkin, C.A. A gold-nanoparticle-based real-time colorimetric screening method for endonuclease activity and inhibition. Angew. Chem. Int. Ed. 2007, 46, 3468–3470. [Google Scholar]
- Zhao, Q.; Huang, H.; Zhang, L.; Wang, L.; Zeng, Y.; Xia, X.; Liu, F.; Chen, Y. Strategy to fabricate naked-eye readout ultrasensitive plasmonic nanosensor based on enzyme mimetic gold nanoclusters. Anal. Chem. 2016, 88, 1412–1418. [Google Scholar]
- Cui, H.-F.; Xu, T.-B.; Sun, Y.-L.; Zhou, A.-W.; Cui, Y.-H.; Liu, W.; Luong, J.H. Hairpin DNA as a biobarcode modified on gold nanoparticles for electrochemical DNA detection. Anal. Chem. 2015, 87, 1358–1365. [Google Scholar]
- Colombo, M.; Mazzucchelli, S.; Collico, V.; Avvakumova, S.; Pandolfi, L.; Corsi, F.; Porta, F.; Prosperi, D. Protein-Assisted One-Pot Synthesis and Biofunctionalization of Spherical Gold Nanoparticles for Selective Targeting of Cancer Cells. Angew. Chem. Int. Ed. 2012, 51, 9272–9275. [Google Scholar]
- Sun, J.; Xianyu, Y.; Jiang, X. Point-of-care biochemical assays using gold nanoparticle-implemented microfluidics. Chem. Soc. Rev. 2014, 43, 6239–6253. [Google Scholar]
- Chen, Y.; Xianyu, Y.; Jiang, X. Surface Modification of Gold Nanoparticles with Small Molecules for Biochemical Analysis. Acc. Chem. Res. 2017, 50, 310–319. [Google Scholar]
- Liu, D.; Wang, Z.; Jiang, X. Gold nanoparticles for the colorimetric and fluorescent detection of ions and small organic molecules. Nanoscale 2011, 3, 1421–1433. [Google Scholar]
- Li, N.; Binder, W.H. Click-chemistry for nanoparticle-modification. J. Mater. Chem. 2011, 21, 16717–16734. [Google Scholar]
- Xie, R.; Hong, S.; Feng, L.; Rong, J.; Chen, X. Cell-selective metabolic glycan labeling based on ligand-targeted liposomes. J. Am. Chem. Soc. 2012, 134, 9914–9917. [Google Scholar]
- Colombo, M.; Sommaruga, S.; Mazzucchelli, S.; Polito, L.; Verderio, P.; Galeffi, P.; Corsi, F.; Tortora, P.; Prosperi, D. Site-specific conjugation of ScFvs antibodies to nanoparticles by bioorthogonal strain-promoted alkyne–nitrone cycloaddition. Angew. Chem. 2012, 124, 511–514. [Google Scholar]
- Lin, L.; Tian, X.; Hong, S.; Dai, P.; You, Q.; Wang, R.; Feng, L.; Xie, C.; Tian, Z.Q.; Chen, X. A bioorthogonal Raman reporter strategy for SERS detection of glycans on live cells. Angew. Chem. Int. Ed. 2013, 52, 7266–7271. [Google Scholar]
- Ramil, C.P.; Lin, Q. Bioorthogonal chemistry: Strategies and recent developments. Chem. Commun. 2013, 49, 11007–11022. [Google Scholar]
- Alonso, F.; Moglie, Y.; Radivoy, G. Copper nanoparticles in click chemistry. Acc. Chem. Res. 2015, 48, 2516–2528. [Google Scholar]
- Wang, Q.; Chan, T.R.; Hilgraf, R.; Fokin, V.V.; Sharpless, K.B.; Finn, M. Bioconjugation by copper (I)-catalyzed azide-alkyne [3+ 2] cycloaddition. J. Am. Chem. Soc. 2003, 125, 3192–3193. [Google Scholar]
- Zhou, Y.; Wang, S.; Zhang, K.; Jiang, X. Visual detection of copper (II) by azide-and alkyne-functionalized gold nanoparticles using click chemistry. Angew. Chem. Int. Ed. 2008, 47, 7454–7456. [Google Scholar]
- Zhu, K.; Zhang, Y.; He, S.; Chen, W.; Shen, J.; Wang, Z.; Jiang, X. Quantification of proteins by functionalized gold nanoparticles using click chemistry. Anal. Chem. 2012, 84, 4267–4270. [Google Scholar]
- Qu, W.; Liu, Y.; Liu, D.; Wang, Z.; Jiang, X. Copper-Mediated Amplification Allows Readout of Immunoassays by the Naked Eye. Angew. Chem. Int. Ed. 2011, 50, 3442–3445. [Google Scholar]
- Xianyu, Y.; Wang, Z.; Jiang, X. A plasmonic nanosensor for immunoassay via enzyme-triggered click chemistry. Acs Nano 2014, 8, 12741–12747. [Google Scholar]
- Xianyu, Y.; Wang, Z.; Sun, J.; Wang, X.; Jiang, X. Colorimetric logic gates through molecular recognition and plasmonic nanoparticles. Small 2014, 10, 4833–4838. [Google Scholar]
- Zhang, L.; Feng, Q.; Wang, J.; Sun, J.; Shi, X.; Jiang, X. Microfluidic synthesis of rigid nanovesicles for hydrophilic reagents delivery. Angew. Chem. 2015, 127, 4024–4028. [Google Scholar]
- Xianyu, Y.; Xie, Y.; Wang, N.; Wang, Z.; Jiang, X. A dispersion-dominated chromogenic strategy for colorimetric sensing of glutathione at the nanomolar level using gold nanoparticles. Small 2015, 11, 5510–5514. [Google Scholar]
- Guo, Y.; Zhang, Y.; Shao, H.; Wang, Z.; Wang, X.; Jiang, X. Label-free colorimetric detection of cadmium ions in rice samples using gold nanoparticles. Anal. Chem. 2014, 86, 8530–8534. [Google Scholar]
- Liu, D.; Chen, W.; Sun, K.; Deng, K.; Zhang, W.; Wang, Z.; Jiang, X. Resettable, multi-readout logic gates based on controllably reversible aggregation of gold nanoparticles. Angew. Chem. 2011, 123, 4189–4193. [Google Scholar]
- Chen, W.; Cao, F.; Zheng, W.; Tian, Y.; Xianyu, Y.; Xu, P.; Zhang, W.; Wang, Z.; Deng, K.; Jiang, X. Detection of the nanomolar level of total Cr [(III) and (VI)] by functionalized gold nanoparticles and a smartphone with the assistance of theoretical calculation models. Nanoscale 2015, 7, 2042–2049. [Google Scholar]
- Zheng, W.; Li, H.; Chen, W.; Ji, J.; Jiang, X. Recyclable colorimetric detection of trivalent cations in aqueous media using zwitterionic gold nanoparticles. Anal. Chem. 2016, 88, 4140–4146. [Google Scholar]
- Liu, D.; Qu, W.; Chen, W.; Zhang, W.; Wang, Z.; Jiang, X. Highly sensitive, colorimetric detection of mercury (II) in aqueous media by quaternary ammonium group-capped gold nanoparticles at room temperature. Anal. Chem. 2010, 82, 9606–9610. [Google Scholar]
- Liu, D.; Wang, S.; Swierczewska, M.; Huang, X.; Bhirde, A.A.; Sun, J.; Wang, Z.; Yang, M.; Jiang, X.; Chen, X. Highly robust, recyclable displacement assay for mercuric ions in aqueous solutions and living cells. Acs Nano 2012, 6, 10999–11008. [Google Scholar]
- Liu, D.; Chen, W.; Tian, Y.; He, S.; Zheng, W.; Sun, J.; Wang, Z.; Jiang, X. A highly sensitive gold-nanoparticle-based assay for acetylcholinesterase in cerebrospinal fluid of transgenic mice with Alzheimer’s disease. Adv. Healthc. Mater. 2012, 1, 90–95. [Google Scholar]
- Liu, D.; Chen, W.; Wei, J.; Li, X.; Wang, Z.; Jiang, X. A highly sensitive, dual-readout assay based on gold nanoparticles for organophosphorus and carbamate pesticides. Anal. Chem. 2012, 84, 4185–4191. [Google Scholar]
- Watson, J.D.; Crick, F.H. Molecular structure of nucleic acids: A structure for deoxyribose nucleic acid. Nature 1953, 171, 737–738. [Google Scholar]
- Alivisatos, A.P.; Johnsson, K.P.; Peng, X.; Wilson, T.E.; Loweth, C.J.; Bruchez, M.P.; Schultz, P.G. Organization of’nanocrystal molecules’ using DNA. Nature 1996, 382, 609–611. [Google Scholar]
- Pinto, Y.Y.; Le, J.D.; Seeman, N.C.; Musier-Forsyth, K.; Taton, T.A.; Kiehl, R.A. Sequence-encoded self-assembly of multiple-nanocomponent arrays by 2D DNA scaffolding. Nano Lett. 2005, 5, 2399–2402. [Google Scholar]
- Zheng, J.; Constantinou, P.E.; Micheel, C.; Alivisatos, A.P.; Kiehl, R.A.; Seeman, N.C. Two-dimensional nanoparticle arrays show the organizational power of robust DNA motifs. Nano Lett. 2006, 6, 1502–1504. [Google Scholar]
- Sharma, J.; Chhabra, R.; Liu, Y.; Ke, Y.; Yan, H. DNA-templated self-assembly of two-dimensional and periodical gold nanoparticle arrays. Angew. Chem. 2006, 118, 744–749. [Google Scholar]
- Lee, J.H.; Wernette, D.P.; Yigit, M.V.; Liu, J.; Wang, Z.; Lu, Y. Site-specific control of distances between gold nanoparticles using phosphorothioate anchors on DNA and a short bifunctional molecular fastener. Angew. Chem. 2007, 119, 9164–9168. [Google Scholar]
- Ludueña, R.F.; Roach, M.C.; Trcka, P.P.; Weintraub, S. N, N-Bis (α-iodoacetyl)-2, 2′-dithiobis (ethylamine), a reversible crosslinking reagent for protein sulfhydryl groups. Anal. Biochem. 1981, 117, 76–80. [Google Scholar]
- Ozbay, E. Plasmonics: Merging photonics and electronics at nanoscale dimensions. Science 2006, 311, 189–193. [Google Scholar]
- Liu, Z.; Searson, P.C. Single nanoporous gold nanowire sensors. J. Phys. Chem. B 2006, 110, 4318–4322. [Google Scholar]
- Chirea, M.; Freitas, A.; Vasile, B.S.; Ghitulica, C.; Pereira, C.M.; Silva, F. Gold nanowire networks: Synthesis, characterization, and catalytic activity. Langmuir 2011, 27, 3906–3913. [Google Scholar]
- Bai, X.; Gao, Y.; Liu, H.-G.; Zheng, L. Synthesis of amphiphilic ionic liquids terminated gold nanorods and their superior catalytic activity for the reduction of nitro compounds. J. Phys. Chem. C 2009, 113, 17730–17736. [Google Scholar]
- Richter, J. Metallization of DNA. Phys. E Low Dimens. Syst. Nanostruct. 2003, 16, 157–173. [Google Scholar]
- Ongaro, A.; Griffin, F.; Beecher, P.; Nagle, L.; Iacopino, D.; Quinn, A.; Redmond, G.; Fitzmaurice, D. DNA-templated assembly of conducting gold nanowires between gold electrodes on a silicon oxide substrate. Chem. Mater. 2005, 17, 1959–1964. [Google Scholar]
- Kim, H.J.; Roh, Y.; Hong, B. Selective formation of a latticed nanostructure with the precise alignment of DNA-templated gold nanowires. Langmuir 2010, 26, 18315–18319. [Google Scholar]
- Harnack, O.; Ford, W.E.; Yasuda, A.; Wessels, J.M. Tris (hydroxymethyl) phosphine-capped gold particles templated by DNA as nanowire precursors. Nano Lett. 2002, 2, 919–923. [Google Scholar]
- Yonezawa, T.; Onoue, S.-Y.; Kimizuka, N. Metal coating of DNA molecules by cationic, metastable gold nanoparticles. Chem. Lett. 2002, 31, 1172–1173. [Google Scholar]
- Patolsky, F.; Weizmann, Y.; Lioubashevski, O.; Willner, I. Au-nanoparticle nanowires based on DNA and polylysine templates. Angew. Chem. Int. Ed. 2002, 41, 2323–2327. [Google Scholar]
- Haruta, M. Catalysis of gold nanoparticles deposited on metal oxides. Cattech 2002, 6, 102–115. [Google Scholar]
- Fu, T.J.; Seeman, N.C. DNA double-crossover molecules. Biochemistry 1993, 32, 3211–3220. [Google Scholar]
- Ding, B.; Sha, R.; Seeman, N.C. Pseudohexagonal 2D DNA crystals from double crossover cohesion. J. Am. Chem. Soc. 2004, 126, 10230–10231. [Google Scholar]
- Le, J.D.; Pinto, Y.; Seeman, N.C.; Musier-Forsyth, K.; Taton, T.A.; Kiehl, R.A. DNA-templated self-assembly of metallic nanocomponent arrays on a surface. Nano Lett. 2004, 4, 2343–2347. [Google Scholar]
- Mastroianni, A.J.; Claridge, S.A.; Alivisatos, A.P. Pyramidal and chiral groupings of gold nanocrystals assembled using DNA scaffolds. J. Am. Chem. Soc. 2009, 131, 8455–8459. [Google Scholar]
- Chen, J.; Seeman, N.C. Synthesis from DNA of a molecule with the connectivity of a cube. Nature 1991, 350, 631–633. [Google Scholar]
- Erben, C.M.; Goodman, R.P.; Turberfield, A.J. A self-assembled DNA bipyramid. J. Am. Chem. Soc. 2007, 129, 6992–6993. [Google Scholar]
- Zhang, Y.; Seeman, N.C. Construction of a DNA-truncated octahedron. J. Am. Chem. Soc. 1994, 116, 1661–1669. [Google Scholar]
- Riordan, T.H. A Brief Overview of DNA Origami and Its Applications; Duke University: Durham, NC, USA.
- Andersen, E.S.; Dong, M.; Nielsen, M.M.; Jahn, K.; Subramani, R.; Mamdouh, W.; Golas, M.M.; Sander, B.; Stark, H.; Oliveira, C.L. Self-assembly of a nanoscale DNA box with a controllable lid. Nature 2009, 459, 73–76. [Google Scholar]
- Kuzuya, A.; Komiyama, M. Design and construction of a box-shaped 3D-DNA origami. Chem. Commun. 2009, 4182–4184. [Google Scholar]
- Kuzyk, A.; Schreiber, R.; Fan, Z.; Pardatscher, G.; Roller, E.-M.; Högele, A.; Simmel, F.C.; Govorov, A.O.; Liedl, T. DNA-based self-assembly of chiral plasmonic nanostructures with tailored optical response. Nature 2012, 483, 311–314. [Google Scholar]
- Yang, C.; Manocchi, A.K.; Lee, B.; Yi, H. Viral templated palladium nanocatalysts for dichromate reduction. Appl. Catal. B Environ. 2010, 93, 282–291. [Google Scholar]
- Nam, Y.S.; Magyar, A.P.; Lee, D.; Kim, J.-W.; Yun, D.S.; Park, H.; Pollom, T.S.; Weitz, D.A.; Belcher, A.M. Biologically templated photocatalytic nanostructures for sustained light-driven water oxidation. Nat. Nanotechnol. 2010, 5, 340–344. [Google Scholar]
- Scheibel, T.; Parthasarathy, R.; Sawicki, G.; Lin, X.-M.; Jaeger, H.; Lindquist, S.L. Conducting nanowires built by controlled self-assembly of amyloid fibers and selective metal deposition. Proc. Natl. Acad. Sci. USA 2003, 100, 4527–4532. [Google Scholar]
- Glover, J.R.; Kowal, A.S.; Schirmer, E.C.; Patino, M.M.; Liu, J.-J.; Lindquist, S. Self-seeded fibers formed by Sup35, the protein determinant of [PSI+], a heritable prion-like factor of S. cerevisiae. Cell 1997, 89, 811–819. [Google Scholar]
- Steinmetz, N.F.; Evans, D.J. Utilisation of plant viruses in bionanotechnology. Org. Biomol. Chem. 2007, 5, 2891–2902. [Google Scholar]
- Lin, T.; Chen, Z.; Usha, R.; Stauffacher, C.V.; Dai, J.-B.; Schmidt, T.; Johnson, J.E. The refined crystal structure of cowpea mosaic virus at 2.8 Å resolution. Virology 1999, 265, 20–34. [Google Scholar]
- Steinmetz, N.F.; Lomonossoff, G.P.; Evans, D.J. Cowpea mosaic virus for material fabrication: Addressable carboxylate groups on a programmable nanoscaffold. Langmuir 2006, 22, 3488–3490. [Google Scholar]
- Steinmetz, N.F.; Lomonossoff, G.P.; Evans, D.J. Decoration of cowpea mosaic virus with multiple, redox-active, organometallic complexes. Small 2006, 2, 530–533. [Google Scholar]
- Blum, A.S.; Soto, C.M.; Wilson, C.D.; Cole, J.D.; Kim, M.; Gnade, B.; Chatterji, A.; Ochoa, W.F.; Lin, T.; Johnson, J.E. Cowpea mosaic virus as a scaffold for 3-D patterning of gold nanoparticles. Nano Lett. 2004, 4, 867–870. [Google Scholar]
- Zafeiratos, S.; Piccinin, S.; Teschner, D. Alloys in catalysis: Phase separation and surface segregation phenomena in response to the reactive environment. Catal. Sci. Technol. 2012, 2, 1787–1801. [Google Scholar]
- Evans, D.J. The bionanoscience of plant viruses: Templates and synthons for new materials. J. Mater. Chem. 2008, 18, 3746–3754. [Google Scholar]
- Shenton, W.; Douglas, T.; Young, M.; Stubbs, G.; Mann, S. Inorganic–organic nanotube composites from template mineralization of tobacco mosaic virus. Adv. Mater. 1999, 11, 253–256. [Google Scholar]
- Knez, M.; Kadri, A.; Wege, C.; Gösele, U.; Jeske, H.; Nielsch, K. Atomic layer deposition on biological macromolecules: Metal oxide coating of tobacco mosaic virus and ferritin. Nano Lett. 2006, 6, 1172–1177. [Google Scholar]
- Dujardin, E.; Peet, C.; Stubbs, G.; Culver, J.N.; Mann, S. Organization of metallic nanoparticles using tobacco mosaic virus templates. Nano Lett. 2003, 3, 413–417. [Google Scholar]
- Lee, S.-Y.; Royston, E.; Culver, J.N.; Harris, M.T. Improved metal cluster deposition on a genetically engineered tobacco mosaic virus template. Nanotechnology 2005, 16, S435. [Google Scholar]
- Lim, J.-S.; Kim, S.-M.; Lee, S.-Y.; Stach, E.A.; Culver, J.N.; Harris, M.T. Formation of Au/Pd alloy nanoparticles on TMV. J. Nanomater. 2010, 2010, 620505. [Google Scholar]
- Khan, A.A.; Fox, E.K.; Górzny, M.Ł.; Nikulina, E.; Brougham, D.F.; Wege, C.; Bittner, A.M. pH control of the electrostatic binding of gold and iron oxide nanoparticles to tobacco mosaic virus. Langmuir 2013, 29, 2094–2098. [Google Scholar]
- Guli, M.; Lambert, E.M.; Li, M.; Mann, S. Template-Directed Synthesis of Nanoplasmonic Arrays by Intracrystalline Metalization of Cross-Linked Lysozyme Crystals. Angew. Chem. 2010, 122, 530–533. [Google Scholar]
- Wei, H.; Wang, Z.; Zhang, J.; House, S.; Gao, Y.-G.; Yang, L.; Robinson, H.; Tan, L.H.; Xing, H.; Hou, C. Time-dependent, protein-directed growth of gold nanoparticles within a single crystal of lysozyme. Nat. Nanotechnol. 2011, 6, 93. [Google Scholar]
- Wei, H.; Lu, Y. Catalysis of gold nanoparticles within lysozyme single crystals. Chem. Asian J. 2012, 7, 680–683. [Google Scholar]
- McMillan, R.A.; Paavola, C.D.; Howard, J.; Chan, S.L.; Zaluzec, N.J.; Trent, J.D. Ordered nanoparticle arrays formed on engineered chaperonin protein templates. Nat. Mater. 2002, 1, 247–252. [Google Scholar]
- Trent, J.D.; Kagawa, H.K.; Yaoi, T.; Olle, E.; Zaluzec, N.J. Chaperonin filaments: The archaeal cytoskeleton? Proc. Natl. Acad. Sci. USA 1997, 94, 5383–5388. [Google Scholar]
- Ellis, M.J.; Knapp, S.; Koeck, P.J.; Fakoor-Biniaz, Z.; Ladenstein, R.; Hebert, H. Two-Dimensional Crystallization of the Chaperonin TF55 from the Hyperthermophilic ArchaeonSulfolobus solfataricus. J. Struct. Biol. 1998, 123, 30–36. [Google Scholar]
- Bond, G.; Louis, C.; Thompson, D. Catalysis by Gold Imperial; College Press: Joplin, MO, USA, 2006. [Google Scholar]
- Hashmi, A.S.K.; Hutchings, G.J. Gold catalysis. Angew. Chem. Int. Ed. 2006, 45, 7896–7936. [Google Scholar]
- Della Pina, C.; Falletta, E.; Rossi, M. Update on selective oxidation using gold. Chem. Soc. Rev. 2012, 41, 350–369. [Google Scholar]
- Mills, G.; Gordon, M.S.; Metiu, H. Oxygen adsorption on Au clusters and a rough Au (111) surface: The role of surface flatness, electron confinement, excess electrons, and band gap. J. Chem. Phys. 2003, 118, 4198–4205. [Google Scholar]
- Bond, G.C.; Thompson, D.T. Catalysis by gold. Catal. Rev. 1999, 41, 319–388. [Google Scholar]
- Wales, D.J. Structure, dynamics, and thermodynamics of clusters: Tales from topographic potential surfaces. Science 1996, 271, 925–929. [Google Scholar]
- Boyen, H.-G.; Kästle, G.; Weigl, F.; Koslowski, B.; Dietrich, C.; Ziemann, P.; Spatz, J.P.; Riethmüller, S.; Hartmann, C.; Möller, M. Oxidation-resistant gold-55 clusters. Science 2002, 297, 1533–1536. [Google Scholar]
- Bond, G.C.; Thompson, D.T. Gold-catalysed oxidation of carbon monoxide. Gold Bull. 2000, 33, 41–50. [Google Scholar]
- Benkovic, S.J.; Hammes-Schiffer, S. A perspective on enzyme catalysis. Science 2003, 301, 1196–1202. [Google Scholar]
- Comotti, M.; Della Pina, C.; Matarrese, R.; Rossi, M. The catalytic activity of “naked” gold particles. Angew. Chem. Int. Ed. 2004, 43, 5812–5815. [Google Scholar]
- Boronat, M.; Corma, A. Oxygen activation on gold nanoparticles: Separating the influence of particle size, particle shape and support interaction. Dalton Trans. 2010, 39, 8538–8546. [Google Scholar]
- Kotov, N.A. Inorganic nanoparticles as protein mimics. Science 2010, 330, 188–189. [Google Scholar]
- Witt, S.; Wohlfahrt, G.; Schomburg, D.; Hecht, H.-J.; Kalisz, H.M. Conserved arginine-516 of Penicillium amagasakiense glucose oxidase is essential for the efficient binding of β-D-glucose. Biochem. J. 2000, 347, 553–559. [Google Scholar]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar]
- Zheng, X.; Liu, Q.; Jing, C.; Li, Y.; Li, D.; Luo, W.; Wen, Y.; He, Y.; Huang, Q.; Long, Y.T. Catalytic gold nanoparticles for nanoplasmonic detection of DNA hybridization. Angew. Chem. 2011, 123, 12200–12204. [Google Scholar]
- Newman, J.D.; Turner, A.P. Home blood glucose biosensors: A commercial perspective. Biosens. Bioelectron. 2005, 20, 2435–2453. [Google Scholar]
- Beltrame, P.; Comotti, M.; Della Pina, C.; Rossi, M. Aerobic oxidation of glucose: II. Catalysis by colloidal gold. Appl. Catal. A Gen. 2006, 297, 1–7. [Google Scholar]
- Biella, S.; Prati, L.; Rossi, M. Selective oxidation of D-glucose on gold catalyst. J. Catal. 2002, 206, 242–247. [Google Scholar]
- Luo, W.; Zhu, C.; Su, S.; Li, D.; He, Y.; Huang, Q.; Fan, C. Self-catalyzed, self-limiting growth of glucose oxidase-mimicking gold nanoparticles. Acs Nano 2010, 4, 7451–7458. [Google Scholar]
- Cedervall, T.; Lynch, I.; Lindman, S.; Berggård, T.; Thulin, E.; Nilsson, H.; Dawson, K.A.; Linse, S. Understanding the nanoparticle–protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2050–2055. [Google Scholar]
- Lacerda, S.H.D.P.; Park, J.J.; Meuse, C.; Pristinski, D.; Becker, M.L.; Karim, A.; Douglas, J.F. Interaction of gold nanoparticles with common human blood proteins. Acs Nano 2010, 4, 365–379. [Google Scholar]
- Röcker, C.; Pötzl, M.; Zhang, F.; Parak, W.J.; Nienhaus, G.U. A quantitative fluorescence study of protein monolayer formation on colloidal nanoparticles. Nat. Nanotechnol. 2009, 4, 577–580. [Google Scholar]
- Babitzke, P.; Gollnick, P. Posttranscription Initiation Control of Tryptophan Metabolism in Bacillus subtilis by thetrp RNA-Binding Attenuation Protein (TRAP), anti-TRAP, and RNA Structure. J. Bacteriol. 2001, 183, 5795–5802. [Google Scholar]
- Chen, X.-p.; Antson, A.A.; Yang, M.; Li, P.; Baumann, C.; Dodson, E.J.; Dodson, G.G.; Gollnick, P. Regulatory features of the trp operon and the crystal structure of the trp RNA-binding attenuation protein from Bacillus stearothermophilus. J. Mol. Biol. 1999, 289, 1003–1016. [Google Scholar]
- Antson, A.A.; Otridge, J.; Brzozowski, A.M.; Dodson, E.J.; Dodson, G.G.; Wilson, K.S.; Smith, T.M.; Yang, M.; Kurecki, T.; Gollnick, P. The structure of trp RNA-binding attenuation protein. Nature 1995, 374, 693–700. [Google Scholar]
- Heddle, J.G.; Okajima, T.; Scott, D.J.; Akashi, S.; Park, S.-Y.; Tame, J.R. Dynamic allostery in the ring protein TRAP. J. Mol. Biol. 2007, 371, 154–167. [Google Scholar]
- Heddle, J.G.; Yokoyama, T.; Yamashita, I.; Park, S.-Y.; Tame, J.R. Rounding up: Engineering 12-membered rings from the cyclic 11-mer TRAP. Structure 2006, 14, 925–933. [Google Scholar]
- Heddle, J.G.; Fujiwara, I.; Yamadaki, H.; Yoshii, S.; Nishio, K.; Addy, C.; Yamashita, I.; Tame, J.R. Using the Ring-Shaped Protein TRAP to Capture and Confine Gold Nanodots on a Surface. Small 2007, 3, 1950–1956. [Google Scholar]
- Malay, A.D.; Heddle, J.G.; Tomita, S.; Iwasaki, K.; Miyazaki, N.; Sumitomo, K.; Yanagi, H.; Yamashita, I.; Uraoka, Y. Gold nanoparticle-induced formation of artificial protein capsids. Nano Lett. 2012, 12, 2056–2059. [Google Scholar]
- Wittstock, A.; Zielasek, V.; Biener, J.; Friend, C.; Bäumer, M. Nanoporous gold catalysts for selective gas-phase oxidative coupling of methanol at low temperature. Science 2010, 327, 319–322. [Google Scholar]
- Häkkinen, H. The gold–sulfur interface at the nanoscale. Nat. Chem. 2012, 4, 443. [Google Scholar]
- Zhang, D.; Neumann, O.; Wang, H.; Yuwono, V.M.; Barhoumi, A.; Perham, M.; Hartgerink, J.D.; Wittung-Stafshede, P.; Halas, N.J. Gold nanoparticles can induce the formation of protein-based aggregates at physiological pH. Nano Lett. 2009, 9, 666–671. [Google Scholar]
- Aubin-Tam, M.-E.; Hamad-Schifferli, K. Structure and function of nanoparticle–protein conjugates. Biomed. Mater. 2008, 3, 034001. [Google Scholar]
- Treuel, L.; Malissek, M.; Gebauer, J.S.; Zellner, R. The influence of surface composition of nanoparticles on their interactions with serum albumin. ChemPhysChem 2010, 11, 3093–3099. [Google Scholar]
- Lynch, I.; Dawson, K.A. Protein-nanoparticle interactions. Nano Today 2008, 3, 40–47. [Google Scholar]
- Mancin, F.; Prins, L.J.; Scrimin, P. Catalysis on gold-nanoparticle-passivating monolayers. Curr. Opin. Colloid Interface Sci. 2013, 18, 61–69. [Google Scholar]
- Pengo, P.; Polizzi, S.; Pasquato, L.; Scrimin, P. Carboxylate-imidazole cooperativity in dipeptide-functionalized gold nanoparticles with esterase-like activity. J. Am. Chem. Soc. 2005, 127, 1616–1617. [Google Scholar]
- Mikami, Y.; Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Catalytic activity of unsupported gold nanoparticles. Catal. Sci. Technol. 2013, 3, 58–69. [Google Scholar]
- Manea, F.; Houillon, F.B.; Pasquato, L.; Scrimin, P. Nanozymes: Gold-nanoparticle-based transphosphorylation catalysts. Angew. Chem. 2004, 116, 6291–6295. [Google Scholar]
- Pingoud, A.; Fuxreiter, M.; Pingoud, V.; Wende, W. Type II restriction endonucleases: Structure and mechanism. Cell. Mol. Life Sci. 2005, 62, 685. [Google Scholar]
- Pommier, Y.; Leo, E.; Zhang, H.; Marchand, C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 2010, 17, 421–433. [Google Scholar]
- Zaupa, G.; Mora, C.; Bonomi, R.; Prins, L.J.; Scrimin, P. Catalytic self-assembled monolayers on Au nanoparticles: The source of catalysis of a transphosphorylation reaction. Chem. Eur. J. 2011, 17, 4879–4889. [Google Scholar]
- Northrop, D.B. Follow the protons: A low-barrier hydrogen bond unifies the mechanisms of the aspartic proteases. Acc. Chem. Res. 2001, 34, 790–797. [Google Scholar]
- Hsu, M.-H.; Josephrajan, T.; Yeh, C.-S.; Shieh, D.-B.; Su, W.-C.; Hwu, J.R. Novel arylhydrazone-conjugated gold nanoparticles with DNA-cleaving ability: The first DNA-nicking nanomaterial. Bioconjugate Chem. 2007, 18, 1709–1712. [Google Scholar]
- Hwu, J.R.; Lin, C.C.; Chuang, S.H.; King, K.Y.; Su, T.-R.; Tsay, S.-C. Aminyl and iminyl radicals from arylhydrazones in the photo-induced DNA cleavage. Bioorganic Med. Chem. 2004, 12, 2509–2515. [Google Scholar]
- Bonomi, R.; Selvestrel, F.; Lombardo, V.; Sissi, C.; Polizzi, S.; Mancin, F.; Tonellato, U.; Scrimin, P. Phosphate diester and DNA hydrolysis by a multivalent, nanoparticle-based catalyst. J. Am. Chem. Soc. 2008, 130, 15744–15745. [Google Scholar]
- Fillon, Y.; Verma, A.; Ghosh, P.; Ernenwein, D.; Rotello, V.M.; Chmielewski, J. Peptide ligation catalyzed by functionalized gold nanoparticles. J. Am. Chem. Soc. 2007, 129, 6676–6677. [Google Scholar]
- Dawson, P.E.; Muir, T.W.; Clark-Lewis, I.; Kent, S. Synthesis of proteins by native chemical ligation. Science 1994, 266, 776–779. [Google Scholar]
- Yarus, M. Getting past the RNA world: The initial Darwinian ancestor. Cold Spring Harb. Perspect. Biol. 2011, 3, a003590. [Google Scholar]
- Silverman, S.K. Deoxyribozymes: DNA catalysts for bioorganic chemistry. Org. Biomol. Chem. 2004, 2, 2701–2706. [Google Scholar]
- Fiammengo, R.; Jäschke, A. Nucleic acid enzymes. Curr. Opin. Biotechnol. 2005, 16, 614–621. [Google Scholar]
- Lu, Y.; Liu, J. Functional DNA nanotechnology: Emerging applications of DNAzymes and aptamers. Curr. Opin. Biotechnol. 2006, 17, 580–588. [Google Scholar]
- Zhao, W.; Lam, J.C.; Chiuman, W.; Brook, M.A.; Li, Y. Enzymatic cleavage of nucleic acids on gold nanoparticles: A generic platform for facile colorimetric biosensors. Small 2008, 4, 810–816. [Google Scholar]
- Santoro, S.W.; Joyce, G.F. A general purpose RNA-cleaving DNA enzyme. Proc. Natl. Acad. Sci. USA 1997, 94, 4262–4266. [Google Scholar]
- Li, J.; Lu, Y. A highly sensitive and selective catalytic DNA biosensor for lead ions. J. Am. Chem. Soc. 2000, 122, 10466–10467. [Google Scholar]
- Brennan, J.L.; Hatzakis, N.S.; Tshikhudo, T.R.; Dirvianskyte, N.; Razumas, V.; Patkar, S.; Vind, J.; Svendsen, A.; Nolte, R.J.; Rowan, A.E. Bionanoconjugation via click chemistry: The creation of functional hybrids of lipases and gold nanoparticles. Bioconjugate Chem. 2006, 17, 1373–1375. [Google Scholar]
- Prati, L.; Spontoni, P.; Gaiassi, A. From renewable to fine chemicals through selective oxidation: The case of glycerol. Top. Catal. 2009, 52, 288. [Google Scholar]
- Villa, A.; Wang, D.; Su, D.S.; Prati, L. Gold sols as catalysts for glycerol oxidation: The role of stabilizer. ChemCatChem 2009, 1, 510–514. [Google Scholar]
- Wang, Z.; Lévy, R.; Fernig, D.G.; Brust, M. The peptide route to multifunctional gold nanoparticles. Bioconjugate Chem. 2005, 16, 497–500. [Google Scholar]
- Xu, X.; Wang, Y.; Huo, F.; Mirkin, C.A. Asymmetric Functionalization of Gold Nanoparticles with Oligonucleotides. J. Am. Chem. Soc. 2006, 128, 9286–9287. [Google Scholar]
- Lazarides, A.A.; Schatz, G.C. DNA-linked metal nanosphere materials: Structural basis for the optical properties. J. Phys. Chem. B 2000, 104, 460–467. [Google Scholar]
- Dykman, L.; Khlebtsov, N. Immunological properties of gold nanoparticles. Chem Sci 2017, 8, 1719–1735. [Google Scholar]
- Chen, Y.-S.; Hung, Y.-C.; Lin, W.-H.; Huang, G.S. Assessment of gold nanoparticles as a size-dependent vaccine carrier for enhancing the antibody response against synthetic foot-and-mouth disease virus peptide. Nanotechnology 2010, 21, 195101. [Google Scholar]
- Connor, E.E.; Mwamuka, J.; Gole, A.; Murphy, C.J.; Wyatt, M.D. Gold nanoparticles are taken up by human cells but do not cause acute cytotoxicity. Small 2005, 1, 325–327. [Google Scholar]
- Pelliccia, M.; Andreozzi, P.; Paulose, J.; D’Alicarnasso, M.; Cagno, V.; Donalisio, M.; Civra, A.; Broeckel, R.M.; Haese, N.; Silva, P.J. Additives for vaccine storage to improve thermal stability of adenoviruses from hours to months. Nat. Commun. 2016, 7, 1–7. [Google Scholar]
- Zhang, J.; Mou, L.; Jiang, X. Surface chemistry of gold nanoparticles for healthrelated applications. R. Soc. Chem. 2020, 11, 923–936. [Google Scholar]
- Gole, A.; Murphy, C.J. Azide-derivatized gold nanorods: Functional materials for “click” chemistry. Langmuir 2008, 24, 266–272. [Google Scholar]
- Wilson, C.G.; Sisco, P.N.; Gadala-Maria, F.A.; Murphy, C.J.; Goldsmith, E.C. Polyelectrolyte-coated gold nanorods and their interactions with type I collagen. Biomaterials 2009, 30, 5639–5648. [Google Scholar]
- Niikura, K.; Matsunaga, T.; Suzuki, T.; Kobayashi, S.; Yamaguchi, H.; Orba, Y.; Kawaguchi, A.; Hasegawa, H.; Kajino, K.; Ninomiya, T. Gold nanoparticles as a vaccine platform: Influence of size and shape on immunological responses in vitro and in vivo. Acs Nano 2013, 7, 3926–3938. [Google Scholar]
- Yang, J.A.; Johnson, B.J.; Wu, S.; Woods, W.S.; George, J.M.; Murphy, C.J. Study of wild-type α-synuclein binding and orientation on gold nanoparticles. Langmuir 2013, 29, 4603–4615. [Google Scholar]
- Torelli, M.D.; Putans, R.A.; Tan, Y.; Lohse, S.E.; Murphy, C.J.; Hamers, R.J. Quantitative determination of ligand densities on nanomaterials by X-ray photoelectron spectroscopy. Acs Appl. Mater. Interfaces 2015, 7, 1720–1725. [Google Scholar]
- Huang, R.; Carney, R.P.; Ikuma, K.; Stellacci, F.; Lau, B.L. Effects of surface compositional and structural heterogeneity on nanoparticle–protein interactions: Different protein configurations. Acs Nano 2014, 8, 5402–5412. [Google Scholar]
- Hong, R.; Fischer, N.O.; Verma, A.; Goodman, C.M.; Emrick, T.; Rotello, V.M. Control of protein structure and function through surface recognition by tailored nanoparticle scaffolds. J. Am. Chem. Soc. 2004, 126, 739–743. [Google Scholar]
- Kumar, R.; Ray, P.C.; Datta, D.; Bansal, G.P.; Angov, E.; Kumar, N. Nanovaccines for malaria using Plasmodium falciparum antigen Pfs25 attached gold nanoparticles. Vaccine 2015, 33, 5064–5071. [Google Scholar]
- Chen, H.; Zhang, J.; Gao, Y.; Liu, S.; Koh, K.; Zhu, X.; Yin, Y. Sensitive cell apoptosis assay based on caspase-3 activity detection with graphene oxide-assisted electrochemical signal amplification. Biosens. Bioelectron. 2015, 68, 777–782. [Google Scholar]
- Fischer, M. Surface plasmon resonance: A general introduction. Methods Mol. Biol. 2010, 627, 1–14. [Google Scholar]
- Stone, J.W.; Thornburg, N.J.; Blum, D.L.; Kuhn, S.J.; Wright, D.W.; Crowe, J.E., Jr. Gold nanorod vaccine for respiratory syncytial virus. Nanotechnology 2013, 24, 295102. [Google Scholar]
- Almeida, J.P.M.; Lin, A.Y.; Figueroa, E.R.; Foster, A.E.; Drezek, R.A. In vivo gold nanoparticle delivery of peptide vaccine induces anti-tumor immune response in prophylactic and therapeutic tumor models. Small 2015, 11, 1453–1459. [Google Scholar]
- Chen, H.; Zhang, J.; Liu, X.; Gao, Y.; Ye, Z.; Li, G. Colorimetric copper (II) ion sensor based on the conformational change of peptide immobilized onto the surface of gold nanoparticles. Anal. Methods 2014, 6, 2580–2585. [Google Scholar]
- Gao, Y.; Zou, F.; Wu, B.; Wang, X.; Zhang, J.; Koh, K.; Chen, H. CB [7]-mediated signal amplification approach for sensitive surface plasmon resonance spectroscopy. Biosens. Bioelectron. 2016, 81, 207–213. [Google Scholar]
- Tao, W.; Ziemer, K.S.; Gill, H.S. Gold nanoparticle–M2e conjugate coformulated with CpG induces protective immunity against influenza A virus. Nanomedicine 2014, 9, 237–251. [Google Scholar]
- Brinas, R.P.; Sundgren, A.; Sahoo, P.; Morey, S.; Rittenhouse-Olson, K.; Wilding, G.E.; Deng, W.; Barchi, J.J., Jr. Design and synthesis of multifunctional gold nanoparticles bearing tumor-associated glycopeptide antigens as potential cancer vaccines. Bioconjugate Chem. 2012, 23, 1513–1523. [Google Scholar]
- Yu, X.; Feizpour, A.; Ramirez, N.-G.P.; Wu, L.; Akiyama, H.; Xu, F.; Gummuluru, S.; Reinhard, B.M. Glycosphingolipid-functionalized nanoparticles recapitulate CD169-dependent HIV-1 uptake and trafficking in dendritic cells. Nat. Commun. 2014, 5, 1–12. [Google Scholar]
- Doores, K.J.; Bonomelli, C.; Harvey, D.J.; Vasiljevic, S.; Dwek, R.A.; Burton, D.R.; Crispin, M.; Scanlan, C.N. Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens. Proc. Natl. Acad. Sci. USA 2010, 107, 13800–13805. [Google Scholar]
- Marradi, M.; Di Gianvincenzo, P.; Enríquez-Navas, P.M.; Martínez-Ávila, O.M.; Chiodo, F.; Yuste, E.; Angulo, J.; Penadés, S. Gold nanoparticles coated with oligomannosides of HIV-1 glycoprotein gp120 mimic the carbohydrate epitope of antibody 2G12. J. Mol. Biol. 2011, 410, 798–810. [Google Scholar]
- Parry, A.L.; Clemson, N.A.; Ellis, J.; Bernhard, S.S.; Davis, B.G.; Cameron, N.R. ‘Multicopy multivalent’glycopolymer-stabilized gold nanoparticles as potential synthetic cancer vaccines. J. Am. Chem. Soc. 2013, 135, 9362–9365. [Google Scholar]
- Tian, Y.; Wang, H.; Liu, Y.; Mao, L.; Chen, W.; Zhu, Z.; Liu, W.; Zheng, W.; Zhao, Y.; Kong, D. A peptide-based nanofibrous hydrogel as a promising DNA nanovector for optimizing the efficacy of HIV vaccine. Nano Lett. 2014, 14, 1439–1445. [Google Scholar]
- Xu, L.; Liu, Y.; Chen, Z.; Li, W.; Liu, Y.; Wang, L.; Liu, Y.; Wu, X.; Ji, Y.; Zhao, Y. Surface-engineered gold nanorods: Promising DNA vaccine adjuvant for HIV-1 treatment. Nano Lett. 2012, 12, 2003–2012. [Google Scholar]
- Zhou, X.; Zhang, X.; Yu, X.; Zha, X.; Fu, Q.; Liu, B.; Wang, X.; Chen, Y.; Chen, Y.; Shan, Y. The effect of conjugation to gold nanoparticles on the ability of low molecular weight chitosan to transfer DNA vaccine. Biomaterials 2008, 29, 111–117. [Google Scholar]
- Zhou, W.; Gao, X.; Liu, D.; Chen, X. Gold nanoparticles for in vitro diagnostics. Chem. Rev. 2015, 115, 10575–10636. [Google Scholar]
- Guo, Y.; Wang, Z.; Qu, W.; Shao, H.; Jiang, X. Colorimetric detection of mercury, lead and copper ions simultaneously using protein-functionalized gold nanoparticles. Biosens. Bioelectron. 2011, 26, 4064–4069. [Google Scholar]
- Miranda, O.R.; Li, X.; Garcia-Gonzalez, L.; Zhu, Z.-J.; Yan, B.; Bunz, U.H.; Rotello, V.M. Colorimetric bacteria sensing using a supramolecular enzyme–nanoparticle biosensor. J. Am. Chem. Soc. 2011, 133, 9650–9653. [Google Scholar]
- Lien, C.-W.; Tseng, Y.-T.; Huang, C.-C.; Chang, H.-T. Logic control of enzyme-like gold nanoparticles for selective detection of lead and mercury ions. Anal. Chem. 2014, 86, 2065–2072. [Google Scholar]
- Kim, Y.; Johnson, R.C.; Hupp, J.T. Gold nanoparticle-based sensing of “spectroscopically silent” heavy metal ions. Nano Lett. 2001, 1, 165–167. [Google Scholar]
- Xu, X.; Daniel, W.L.; Wei, W.; Mirkin, C.A. Colorimetric Cu2+ detection using DNA-modified gold-nanoparticle aggregates as probes and click chemistry. Small 2010, 6, 623–626. [Google Scholar]
- Wang, C.-I.; Huang, C.-C.; Lin, Y.-W.; Chen, W.-T.; Chang, H.-T. Catalytic gold nanoparticles for fluorescent detection of mercury (II) and lead (II) ions. Anal. Chim. Acta 2012, 745, 124–130. [Google Scholar]
- Lin, Y.-W.; Huang, C.-C.; Chang, H.-T. Gold nanoparticle probes for the detection of mercury, lead and copper ions. Analyst 2011, 136, 863–871. [Google Scholar]
- Lien, C.-W.; Chen, Y.-C.; Chang, H.-T.; Huang, C.-C. Logical regulation of the enzyme-like activity of gold nanoparticles by using heavy metal ions. Nanoscale 2013, 5, 8227–8234. [Google Scholar]
- Tsai, C.-S.; Yu, T.-B.; Chen, C.-T. Gold nanoparticle-based competitive colorimetric assay for detection of protein–protein interactions. Chem. Commun. 2005, 4273–4275. [Google Scholar]
- Pavlov, V.; Xiao, Y.; Shlyahovsky, B.; Willner, I. Aptamer-functionalized Au nanoparticles for the amplified optical detection of thrombin. J. Am. Chem. Soc. 2004, 126, 11768–11769. [Google Scholar]
- Wang, X.; Ramstrom, O.; Yan, M. Quantitative analysis of multivalent ligand presentation on gold glyconanoparticles and the impact on lectin binding. Anal. Chem. 2010, 82, 9082–9089. [Google Scholar]
- Xianyu, Y.; Chen, Y.; Jiang, X. Horseradish peroxidase-mediated, iodide-catalyzed cascade reaction for plasmonic immunoassays. Anal. Chem. 2015, 87, 10688–10692. [Google Scholar]
- Zhang, J.; Zheng, W.; Jiang, X. Ag+-Gated Surface Chemistry of Gold Nanoparticles and Colorimetric Detection of Acetylcholinesterase. Small 2018, 14, 1801680. [Google Scholar]
- Wang, C.-I.; Chen, W.-T.; Chang, H.-T. Enzyme mimics of Au/Ag nanoparticles for fluorescent detection of acetylcholine. Anal. Chem. 2012, 84, 9706–9712. [Google Scholar]
- Yang, M.; Zhang, W.; Zheng, W.; Cao, F.; Jiang, X. Inkjet-printed barcodes for a rapid and multiplexed paper-based assay compatible with mobile devices. Lab Chip 2017, 17, 3874–3882. [Google Scholar]
- Chen, Y.; Sun, J.; Xianyu, Y.; Yin, B.; Niu, Y.; Wang, S.; Cao, F.; Zhang, X.; Wang, Y.; Jiang, X. A dual-readout chemiluminescent-gold lateral flow test for multiplex and ultrasensitive detection of disease biomarkers in real samples. Nanoscale 2016, 8, 15205–15212. [Google Scholar]
- Deng, J.; Yang, M.; Wu, J.; Zhang, W.; Jiang, X. A self-contained chemiluminescent lateral flow assay for point-of-care testing. Anal. Chem. 2018, 90, 9132–9137. [Google Scholar]
- Yang, M.; Zhang, W.; Yang, J.; Hu, B.; Cao, F.; Zheng, W.; Chen, Y.; Jiang, X. Skiving stacked sheets of paper into test paper for rapid and multiplexed assay. Sci. Adv. 2017, 3, eaao4862. [Google Scholar]
- Chen, J.; Jackson, A.A.; Rotello, V.M.; Nugen, S.R. Colorimetric Detection of Escherichia coli Based on the Enzyme-Induced Metallization of Gold Nanorods. Small 2016, 12, 2469–2475. [Google Scholar]
- Wei, J.; Zheng, L.; Lv, X.; Bi, Y.; Chen, W.; Zhang, W.; Shi, Y.; Zhao, L.; Sun, X.; Wang, F. Analysis of influenza virus receptor specificity using glycan-functionalized gold nanoparticles. Acs Nano 2014, 8, 4600–4607. [Google Scholar]
- Yang, X.; Dang, Y.; Lou, J.; Shao, H.; Jiang, X. D-alanyl-D-alanine-modified gold nanoparticles form a broad-spectrum sensor for bacteria. Theranostics 2018, 8, 1449. [Google Scholar]
- Miller, K.P.; Wang, L.; Benicewicz, B.C.; Decho, A.W. Inorganic nanoparticles engineered to attack bacteria. Chem. Soc. Rev. 2015, 44, 7787–7807. [Google Scholar]
- Gupta, A.; Mumtaz, S.; Li, C.-H.; Hussain, I.; Rotello, V.M. Combatting antibiotic-resistant bacteria using nanomaterials. Chem. Soc. Rev. 2019, 48, 415–427. [Google Scholar]
- Cui, Y.; Zhao, Y.; Tian, Y.; Zhang, W.; Lü, X.; Jiang, X. The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials 2012, 33, 2327–2333. [Google Scholar]
- Li, X.; Robinson, S.M.; Gupta, A.; Saha, K.; Jiang, Z.; Moyano, D.F.; Sahar, A.; Riley, M.A.; Rotello, V.M. Functional gold nanoparticles as potent antimicrobial agents against multi-drug-resistant bacteria. Acs Nano 2014, 8, 10682–10686. [Google Scholar]
- Huo, S.; Jiang, Y.; Gupta, A.; Jiang, Z.; Landis, R.F.; Hou, S.; Liang, X.-J.; Rotello, V.M. Fully zwitterionic nanoparticle antimicrobial agents through tuning of core size and ligand structure. Acs Nano 2016, 10, 8732–8737. [Google Scholar]
- Gupta, A.; Saleh, N.M.; Das, R.; Landis, R.F.; Bigdeli, A.; Motamedchaboki, K.; Campos, A.R.; Pomeroy, K.; Mahmoudi, M.; Rotello, V.M. Synergistic antimicrobial therapy using nanoparticles and antibiotics for the treatment of multidrug-resistant bacterial infection. Nano Futures 2017, 1, 015004. [Google Scholar]
- Zhao, Y.; Chen, Z.; Chen, Y.; Xu, J.; Li, J.; Jiang, X. Synergy of non-antibiotic drugs and pyrimidinethiol on gold nanoparticles against superbugs. J. Am. Chem. Soc. 2013, 135, 12940–12943. [Google Scholar]
- Wei, S.-C.; Chang, L.; Huang, C.-C.; Chang, H.-T. Dual-functional gold nanoparticles with antimicrobial and proangiogenic activities improve the healing of multidrug-resistant bacteria-infected wounds in diabetic mice. Biomater. Sci. 2019, 7, 4482–4490. [Google Scholar]
- Feng, Y.; Chen, W.; Jia, Y.; Tian, Y.; Zhao, Y.; Long, F.; Rui, Y.; Jiang, X. N-Heterocyclic molecule-capped gold nanoparticles as effective antibiotics against multi-drug resistant bacteria. Nanoscale 2016, 8, 13223–13227. [Google Scholar]
- Yang, X.; Yang, J.; Wang, L.; Ran, B.; Jia, Y.; Zhang, L.; Yang, G.; Shao, H.; Jiang, X. Pharmaceutical intermediate-modified gold nanoparticles: Against multidrug-resistant bacteria and wound-healing application via an electrospun scaffold. Acs Nano 2017, 11, 5737–5745. [Google Scholar]
- Ju, P.; Hu, J.; Li, F.; Cao, Y.; Li, L.; Shi, D.; Hao, Y.; Zhang, M.; He, J.; Ni, P. A biodegradable polyphosphoester-functionalized poly (disulfide) nanocarrier for reduction-triggered intracellular drug delivery. J. Mater. Chem. B 2018, 6, 7263–7273. [Google Scholar]
- Yang, X.; Wei, Q.; Shao, H.; Jiang, X. Multivalent Aminosaccharide-Based Gold Nanoparticles as Narrow-Spectrum Antibiotics in Vivo. Acs Appl. Mater. Interfaces 2019, 11, 7725–7730. [Google Scholar]
- Zhao, X.; Jia, Y.; Li, J.; Dong, R.; Zhang, J.; Ma, C.; Wang, H.; Rui, Y.; Jiang, X. Indole derivative-capped gold nanoparticles as an effective bactericide in vivo. Acs Appl. Mater. Interfaces 2018, 10, 29398–29406. [Google Scholar]
- Li, Y.; Tian, Y.; Zheng, W.; Feng, Y.; Huang, R.; Shao, J.; Tang, R.; Wang, P.; Jia, Y.; Zhang, J. Composites of bacterial cellulose and small molecule-decorated gold nanoparticles for treating Gram-Negative bacteria-infected wounds. Small 2017, 13, 1700130. [Google Scholar]
- Zheng, W.; Jia, Y.; Chen, W.; Wang, G.; Guo, X.; Jiang, X. Universal coating from electrostatic self-assembly to prevent multidrug-resistant bacterial colonization on medical devices and solid surfaces. Acs Appl. Mater. Interfaces 2017, 9, 21181–21189. [Google Scholar]
- Xie, Y.; Liu, Y.; Yang, J.; Liu, Y.; Hu, F.; Zhu, K.; Jiang, X. Gold Nanoclusters for Targeting Methicillin-Resistant Staphylococcus aureus In Vivo. Angew. Chem. Int. Ed. 2018, 57, 3958–3962. [Google Scholar]
- Chen, W.Y.; Chang, H.Y.; Lu, J.K.; Huang, Y.C.; Harroun, S.G.; Tseng, Y.T.; Li, Y.J.; Huang, C.C.; Chang, H.T. Self-assembly of antimicrobial peptides on gold nanodots: Against multidrug-resistant bacteria and wound-healing application. Adv. Funct. Mater. 2015, 25, 7189–7199. [Google Scholar]
- Hu, B.; Kong, F.; Gao, X.; Jiang, L.; Li, X.; Gao, W.; Xu, K.; Tang, B. Avoiding Thiol Compound Interference: A Nanoplatform Based on High-Fidelity Au–Se Bonds for Biological Applications. Angew. Chem. 2018, 130, 5404–5407. [Google Scholar]
- Smith, C.A.; Narouz, M.R.; Lummis, P.A.; Singh, I.; Nazemi, A.; Li, C.-H.; Crudden, C.M. N-Heterocyclic carbenes in materials chemistry. Chem. Rev. 2019, 119, 4986–5056. [Google Scholar]
- Narouz, M.R.; Osten, K.M.; Unsworth, P.J.; Man, R.W.; Salorinne, K.; Takano, S.; Tomihara, R.; Kaappa, S.; Malola, S.; Dinh, C.-T. N-heterocyclic carbene-functionalized magic-number gold nanoclusters. Nat. Chem. 2019, 11, 419–425. [Google Scholar]
- Narouz, M.R.; Takano, S.; Lummis, P.A.; Levchenko, T.I.; Nazemi, A.; Kaappa, S.; Malola, S.; Yousefalizadeh, G.; Calhoun, L.A.; Stamplecoskie, K.G. Robust, highly luminescent Au13 superatoms protected by N-heterocyclic carbenes. J. Am. Chem. Soc. 2019, 141, 14997–15002. [Google Scholar]
- Salorinne, K.; Man, R.W.; Li, C.H.; Taki, M.; Nambo, M.; Crudden, C.M. Water-Soluble N-Heterocyclic Carbene-Protected Gold Nanoparticles: Size-Controlled Synthesis, Stability, and Optical Properties. Angew. Chem. Int. Ed. 2017, 56, 6198–6202. [Google Scholar]
- Dreaden, E.C.; Alkilany, A.M.; Huang, X.; Murphy, C.J.; El-Sayed, M.A. The golden age: Gold nanoparticles for biomedicine. Chem. Soc. Rev. 2012, 41, 2740–2779. [Google Scholar]
- Giljohann, D.A.; Seferos, D.S.; Daniel, W.L.; Massich, M.D.; Patel, P.C.; Mirkin, C.A. Gold nanoparticles for biology and medicine. Angew. Chem. Int. Ed. 2010, 49, 3280–3294. [Google Scholar]
- Hainfeld, J.F.; Slatkin, D.N.; Smilowitz, H.M. The use of gold nanoparticles to enhance radiotherapy in mice. Phys. Med. Biol. 2004, 49, N309. [Google Scholar]
- Her, S.; Jaffray, D.A.; Allen, C. Gold nanoparticles for applications in cancer radiotherapy: Mechanisms and recent advancements. Adv. Drug Deliv. Rev. 2017, 109, 84–101. [Google Scholar]
- Dreaden, E.C.; Austin, L.A.; Mackey, M.A.; El-Sayed, M.A. Size matters: Gold nanoparticles in targeted cancer drug delivery. Ther. Deliv. 2012, 3, 457–478. [Google Scholar]
- Mosquera, J.S.; Henriksen-Lacey, M.; García, I.; Martínez-Calvo, M.; Rodríguez, J.S.; Mascareñas, J.L.; Liz-Marzán, L.M. Cellular uptake of gold nanoparticles triggered by host–guest interactions. J. Am. Chem. Soc. 2018, 140, 4469–4472. [Google Scholar]
- Mizuhara, T.; Saha, K.; Moyano, D.F.; Kim, C.S.; Yan, B.; Kim, Y.K.; Rotello, V.M. Acylsulfonamide-functionalized Zwitterionic gold nanoparticles for enhanced cellular uptake at tumor pH. Angew. Chem. 2015, 127, 6667–6670. [Google Scholar]
- Zhang, X.-D.; Wu, D.; Shen, X.; Chen, J.; Sun, Y.-M.; Liu, P.-X.; Liang, X.-J. Size-dependent radiosensitization of PEG-coated gold nanoparticles for cancer radiation therapy. Biomaterials 2012, 33, 6408–6419. [Google Scholar]
- Zhang, X.D.; Chen, J.; Luo, Z.; Wu, D.; Shen, X.; Song, S.S.; Sun, Y.M.; Liu, P.X.; Zhao, J.; Huo, S. Enhanced Tumor Accumulation of Sub-2 nm Gold Nanoclusters for Cancer Radiation Therapy. Adv. Healthc. Mater. 2014, 3, 133–141. [Google Scholar]
- Hirsch, L.R.; Stafford, R.J.; Bankson, J.A.; Sershen, S.R.; Rivera, B.; Price, R.; Hazle, J.D.; Halas, N.J.; West, J.L. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc. Natl. Acad. Sci. USA 2003, 100, 13549–13554. [Google Scholar]
- Loo, C.; Lowery, A.; Halas, N.; West, J.; Drezek, R. Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett. 2005, 5, 709–711. [Google Scholar]
- Alkilany, A.M.; Thompson, L.B.; Boulos, S.P.; Sisco, P.N.; Murphy, C.J. Gold nanorods: Their potential for photothermal therapeutics and drug delivery, tempered by the complexity of their biological interactions. Adv. Drug Deliv. Rev. 2012, 64, 190–199. [Google Scholar]
- Huang, X.; El-Sayed, I.H.; Qian, W.; El-Sayed, M.A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [Google Scholar]
- Aioub, M.; Panikkanvalappil, S.R.; El-Sayed, M.A. Platinum-coated gold nanorods: Efficient reactive oxygen scavengers that prevent oxidative damage toward healthy, untreated cells during plasmonic photothermal therapy. Acs Nano 2017, 11, 579–586. [Google Scholar]
- Volsi, A.L.; de Aberasturi, D.J.; Henriksen-Lacey, M.; Giammona, G.; Licciardi, M.; Liz-Marzán, L.M. Inulin coated plasmonic gold nanoparticles as a tumor-selective tool for cancer therapy. J. Mater. Chem. B 2016, 4, 1150–1155. [Google Scholar]
- Shen, S.; Tang, H.; Zhang, X.; Ren, J.; Pang, Z.; Wang, D.; Gao, H.; Qian, Y.; Jiang, X.; Yang, W. Targeting mesoporous silica-encapsulated gold nanorods for chemo-photothermal therapy with near-infrared radiation. Biomaterials 2013, 34, 3150–3158. [Google Scholar]
- Zhang, Z.; Wang, L.; Wang, J.; Jiang, X.; Li, X.; Hu, Z.; Ji, Y.; Wu, X.; Chen, C. Mesoporous silica-coated gold nanorods as a light-mediated multifunctional theranostic platform for cancer treatment. Adv. Mater. 2012, 24, 1418–1423. [Google Scholar]
- Mallat, T.; Baiker, A. Oxidation of alcohols with molecular oxygen on solid catalysts. Chem. Rev. 2004, 104, 3037–3058. [Google Scholar]
- Corma, A.; Garcia, H. Supported gold nanoparticles as catalysts for organic reactions. Chem. Soc. Rev. 2008, 37, 2096–2126. [Google Scholar]
- Haruta, M.; Yamada, N.; Kobayashi, T.; Iijima, S. Gold catalysts prepared by coprecipitation for low-temperature oxidation of hydrogen and of carbon monoxide. J. Catal. 1989, 115, 301–309. [Google Scholar]
- Enache, D.I.; Edwards, J.K.; Landon, P.; Solsona-Espriu, B.; Carley, A.F.; Herzing, A.A.; Watanabe, M.; Kiely, C.J.; Knight, D.W.; Hutchings, G.J. Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/TiO2 catalysts. Science 2006, 311, 362–365. [Google Scholar]
- Hayashi, T.; Inagaki, T.; Itayama, N.; Baba, H. Selective oxidation of alcohol over supported gold catalysts: Methyl glycolate formation from ethylene glycol and methanol. Catal. Today 2006, 117, 210–213. [Google Scholar]
- Corma, A.; Domine, M.E. Gold supported on a mesoporous CeO2 matrix as an efficient catalyst in the selective aerobic oxidation of aldehydes in the liquid phase. Chem. Commun. 2005, 4042–4044. [Google Scholar] [CrossRef]
- Klitgaard, S.K.; Egeblad, K.; Mentzel, U.V.; Popov, A.G.; Jensen, T.; Taarning, E.; Nielsen, I.S.; Christensen, C.H. Oxidations of amines with molecular oxygen using bifunctional gold–titania catalysts. Green Chem. 2008, 10, 419–423. [Google Scholar]
- Haruta, M. When gold is not noble: Catalysis by nanoparticles. Chem. Rec. 2003, 3, 75–87. [Google Scholar]
- Boronat, M.; Corma, A. Origin of the different activity and selectivity toward hydrogenation of single metal Au and Pt on TiO2 and bimetallic Au− Pt/TiO2 catalysts. Langmuir 2010, 26, 16607–16614. [Google Scholar]
- Zhang, Z.; Shao, C.; Zou, P.; Zhang, P.; Zhang, M.; Mu, J.; Guo, Z.; Li, X.; Wang, C.; Liu, Y. In situ assembly of well-dispersed gold nanoparticles on electrospun silica nanotubes for catalytic reduction of 4-nitrophenol. Chem. Commun. 2011, 47, 3906–3908. [Google Scholar]
- Milone, C.; Ingoglia, R.; Tropeano, M.; Neri, G.; Galvagno, S. First example of selective hydrogenation of unconstrained α, β-unsaturated ketone to α, β-unsaturated alcohol by molecular hydrogen. Chem. Commun. 2003, 868–869. [Google Scholar]
- Ide, M.S.; Hao, B.; Neurock, M.; Davis, R.J. Mechanistic insights on the hydrogenation of α, β-unsaturated ketones and aldehydes to unsaturated alcohols over metal catalysts. Acs Catal. 2012, 2, 671–683. [Google Scholar]
- Rashid, M.H.; Mandal, T.K. Templateless synthesis of polygonal gold nanoparticles: An unsupported and reusable catalyst with superior activity. Adv. Funct. Mater. 2008, 18, 2261–2271. [Google Scholar]
- Panigrahi, S.; Basu, S.; Praharaj, S.; Pande, S.; Jana, S.; Pal, A.; Ghosh, S.K.; Pal, T. Synthesis and size-selective catalysis by supported gold nanoparticles: Study on heterogeneous and homogeneous catalytic process. J. Phys. Chem. C 2007, 111, 4596–4605. [Google Scholar]
- Kuroda, K.; Ishida, T.; Haruta, M. Reduction of 4-nitrophenol to 4-aminophenol over Au nanoparticles deposited on PMMA. J. Mol. Catal. A Chem. 2009, 298, 7–11. [Google Scholar]
- Abay, A.K.; Chen, X.; Kuo, D.-H. Highly efficient noble metal free copper nickel oxysulfide nanoparticles for catalytic reduction of 4-nitrophenol, methyl blue, and rhodamine-B organic pollutants. New J. Chem. 2017, 41, 5628–5638. [Google Scholar]
- Wu, G.; Liu, X.; Zhou, P.; Wang, L.; Hegazy, M.; Huang, X.; Huang, Y. A facile approach for the reduction of 4-nitrophenol and degradation of congo red using gold nanoparticles or laccase decorated hybrid inorganic nanoparticles/polymer-biomacromolecules vesicles. Mater. Sci. Eng. C 2019, 94, 524–553. [Google Scholar]
- Das, S.K.; Dickinson, C.; Lafir, F.; Brougham, D.F.; Marsili, E. Synthesis, characterization and catalytic activity of gold nanoparticles biosynthesized with Rhizopus oryzae protein extract. R. Soc. Chem. 2012, 14, 1322–1334. [Google Scholar]
- Punnoose, M.S.; Bijimol, D.; Abraham, T.; Plathanam, N.J.; Mathew, B. Synthesis, Characterization and Catalytic Activity of Gold Nanoparticles synthesized using a Green Route. AIP Conf. Proc. 2019, 2269, 030003. [Google Scholar]
- Kumar, I.; Mondal, M.; Meyappan, V.; Sakthivel, N. Green one-pot synthesis of gold nanoparticles using Sansevieria roxburghiana leaf extract for the catalytic degradation of toxic organic pollutants. Mater. Res. Bull. 2019, 117, 18–27. [Google Scholar]
- Kobayashi, H.; Teranishi, M.; Negishi, R.; Naya, S.-I.; Tada, H. Reaction mechanism of the multiple-electron oxygen reduction reaction on the surfaces of gold and platinum nanoparticles loaded on titanium (IV) oxide. J. Phys. Chem. Lett. 2016, 7, 5002–5007. [Google Scholar]
- Tanaka, A.; Teramura, K.; Hosokawa, S.; Kominami, H.; Tanaka, T. Visible light-induced water splitting in an aqueous suspension of a plasmonic Au/TiO 2 photocatalyst with metal co-catalysts. Chem. Sci. 2017, 8, 2574–2580. [Google Scholar]
- Buehler, N.; Meier, K.; Reber, J.F. Photochemical hydrogen production with cadmium sulfide suspensions. J. Phys. Chem. 1984, 88, 3261–3268. [Google Scholar]
- Tada, H.; Mitsui, T.; Kiyonaga, T.; Akita, T.; Tanaka, K. All-solid-state Z-scheme in CdS–Au–TiO 2 three-component nanojunction system. Nat. Mater. 2006, 5, 782–786. [Google Scholar]
- Naya, S.-i.; Kume, T.; Akashi, R.; Fujishima, M.; Tada, H. Red-light-driven water splitting by Au (core)–CdS (shell) half-cut nanoegg with heteroepitaxial junction. J. Am. Chem. Soc. 2018, 140, 1251–1254. [Google Scholar]
- Tada, H. Overall water splitting and hydrogen peroxide synthesis by gold nanoparticle-based plasmonic photocatalysts. R. Soc. Chem. 2019, 1, 4238–4245. [Google Scholar]
- Reineck, P.; Lee, G.P.; Brick, D.; Karg, M.; Mulvaney, P.; Bach, U. A solid-state plasmonic solar cell via metal nanoparticle self-assembly. Adv. Mater 2012, 24, 4750–4755. [Google Scholar]
- Meissner, D.; Memming, R.; Bahnemann, K.D. Fundamental problems of water splitting at cadmium sulfide. Phys. Lett 1986, 127, 419. [Google Scholar]
- Lang, X.; Chen, X.; Zhao, J. Heterogeneous visible light photocatalysis for selective organic transformations. Chem. Soc. Rev. 2014, 43, 473–486. [Google Scholar]
- Naya, S.I.; Niwa, T.; Kume, T.; Tada, H. Visible-Light-Induced Electron Transport from Small to Large Nanoparticles in Bimodal Gold Nanoparticle-Loaded Titanium (IV) Oxide. Angew. Chem. 2014, 126, 7433–7437. [Google Scholar]
- Li, X.; Chen, C.; Zhao, J. Mechanism of photodecomposition of H2O2 on TiO2 surfaces under visible light irradiation. Langmuir 2001, 17, 4118–4122. [Google Scholar]
- Minero, C.; Mariella, G.; Maurino, V.; Pelizzetti, E. Photocatalytic transformation of organic compounds in the presence of inorganic anions. 1. Hydroxyl-mediated and direct electron-transfer reactions of phenol on a titanium dioxide-fluoride system. Langmuir 2000, 16, 2632–2641. [Google Scholar]
- Park, H.; Choi, W. Effects of TiO2 surface fluorination on photocatalytic reactions and photoelectrochemical behaviors. J. Phys. Chem. B 2004, 108, 4086–4093. [Google Scholar]
- Maurino, V.; Minero, C.; Mariella, G.; Pelizzetti, E. Sustained production of H2O2 on irradiated TiO2–fluoride systems. Chem. Commun. 2005, 2627–2629. [Google Scholar]
- Hirakawa, H.; Shiota, S.; Shiraishi, Y.; Sakamoto, H.; Ichikawa, S.; Hirai, T. Au nanoparticles supported on BiVO4: Effective inorganic photocatalysts for H2O2 production from water and O2 under visible light. Acs Catal. 2016, 6, 4976–4982. [Google Scholar]
- Shiraishi, Y.; Kanazawa, S.; Sugano, Y.; Tsukamoto, D.; Sakamoto, H.; Ichikawa, S.; Hirai, T. Highly selective production of hydrogen peroxide on graphitic carbon nitride (g-C3N4) photocatalyst activated by visible light. Acs Catal. 2014, 4, 774–780. [Google Scholar]
- Shiraishi, Y.; Kanazawa, S.; Kofuji, Y.; Sakamoto, H.; Ichikawa, S.; Tanaka, S.; Hirai, T. Sunlight-driven hydrogen peroxide production from water and molecular oxygen by metal-free photocatalysts. Angew. Chem. Int. Ed. 2014, 53, 13454–13459. [Google Scholar]
- Teranishi, M.; Naya, S.-I.; Tada, H. In situ liquid phase synthesis of hydrogen peroxide from molecular oxygen using gold nanoparticle-loaded titanium (IV) dioxide photocatalyst. J. Am. Chem. Soc. 2010, 132, 7850–7851. [Google Scholar]
- Schasfoort, R.; Tudos, A. Handbook of Surface Plasmon Resonance; RSC Publishing: Cambridge, UK, 2008. [Google Scholar]
- Link, S.; El-Sayed, M.A. Shape and size dependence of radiative, non-radiative and photothermal properties of gold nanocrystals. Int. Rev. Phys. Chem. 2000, 19, 409–453. [Google Scholar]
- Underwood, S.; Mulvaney, P. Effect of the solution refractive index on the color of gold colloids. Langmuir 1994, 10, 3427–3430. [Google Scholar]
- Anker, J.N.; Hall, W.P.; Lyandres, O.; Shah, N.C.; Zhao, J.; Van Duyne, R.P. Biosensing with plasmonic nanosensors. Nanosci. Technol. A Collect. Rev. Nat. J. 2010, 308–319. [Google Scholar]
- Fan, M.; Thompson, M.; Andrade, M.L.; Brolo, A.G. Silver nanoparticles on a plastic platform for localized surface plasmon resonance biosensing. Anal. Chem. 2010, 82, 6350–6352. [Google Scholar]
- Kealley, C.S.; Arnold, M.D.; Porkovich, A.; Cortie, M.B. Sensors based on monochromatic interrogation of a localised surface plasmon resonance. Sens. Actuators B Chem. 2010, 148, 34–40. [Google Scholar]
- Watanabe, S.; Sonobe, M.; Arai, M.; Tazume, Y.; Matsuo, T.; Nakamura, T.; Yoshida, K. Enhanced optical sensing of anions with amide-functionalized gold nanoparticles. Chem. Commun. 2002, 2866–2867. [Google Scholar] [CrossRef]
- Fahnestock, K.J.; Manesse, M.; McIlwee, H.A.; Schauer, C.L.; Boukherroub, R.; Szunerits, S. Selective detection of hexachromium ions by localized surface plasmon resonance measurements using gold nanoparticles/chitosan composite interfaces. Analyst 2009, 134, 881–886. [Google Scholar]
- Kalyuzhny, G.; Vaskevich, A.; Schneeweiss, M.A.; Rubinstein, I. Transmission surface-plasmon resonance (T-SPR) measurements for monitoring adsorption on ultrathin gold island films. Chem. A Eur. J. 2002, 8, 3849–3857. [Google Scholar]
- Tokareva, I.; Minko, S.; Fendler, J.H.; Hutter, E. Nanosensors based on responsive polymer brushes and gold nanoparticle enhanced transmission surface plasmon resonance spectroscopy. J. Am. Chem. Soc. 2004, 126, 15950–15951. [Google Scholar]
- Englebienne, P.; Van Hoonacker, A.; Verhas, M. High-throughput screening using the surface plasmon resonance effect of colloidal gold nanoparticles. Analyst 2001, 126, 1645–1651. [Google Scholar]
- Prabhakar, A.; Mukherji, S. A novel C-shaped, gold nanoparticle coated, embedded polymer waveguide for localized surface plasmon resonance based detection. Lab Chip 2010, 10, 3422–3425. [Google Scholar]
- Matsui, J.; Akamatsu, K.; Hara, N.; Miyoshi, D.; Nawafune, H.; Tamaki, K.; Sugimoto, N. SPR sensor chip for detection of small molecules using molecularly imprinted polymer with embedded gold nanoparticles. Anal. Chem. 2005, 77, 4282–4285. [Google Scholar]
- Briglin, S.M.; Gao, T.; Lewis, N.S. Detection of organic mercaptan vapors using thin films of alkylamine-passivated gold nanocrystals. Langmuir 2004, 20, 299–305. [Google Scholar]
- Frasconi, M.; Tortolini, C.; Botre, F.; Mazzei, F. Multifunctional Au nanoparticle dendrimer-based surface plasmon resonance biosensor and its application for improved insulin detection. Anal. Chem. 2010, 82, 7335–7342. [Google Scholar]
- Fu, E.; Ramsey, S.A.; Yager, P. Dependence of the signal amplification potential of colloidal gold nanoparticles on resonance wavelength in surface plasmon resonance-based detection. Anal. Chim. Acta 2007, 599, 118–123. [Google Scholar]
- Hutter, E.; Pileni, M.-P. Detection of DNA hybridization by gold nanoparticle enhanced transmission surface plasmon resonance spectroscopy. J. Phys. Chem. B 2003, 107, 6497–6499. [Google Scholar]
- Hutter, E.; Fendler, J.; Roy, D. Surface plasmon resonance studies of gold and silver nanoparticles linked to gold and silver substrates by 2-aminoethanethiol and 1, 6-hexanedithiol. J. Phys. Chem. B 2001, 105, 11159–11168. [Google Scholar]
- Lyon, L.A.; Musick, M.D.; Natan, M.J. Colloidal Au-enhanced surface plasmon resonance immunosensing. Anal. Chem. 1998, 70, 5177–5183. [Google Scholar]
- Pieper-Fürst, U.; Stöcklein, W.F.; Warsinke, A. Gold nanoparticle-enhanced surface plasmon resonance measurement with a highly sensitive quantification for human tissue inhibitor of metalloproteinases-2. Anal. Chim. Acta 2005, 550, 69–76. [Google Scholar]
- Mitchell, J.S.; Lowe, T.E. Ultrasensitive detection of testosterone using conjugate linker technology in a nanoparticle-enhanced surface plasmon resonance biosensor. Biosens. Bioelectron. 2009, 24, 2177–2183. [Google Scholar]
- Chang, Y.-F.; Chen, R.-C.; Lee, Y.-J.; Chao, S.-C.; Su, L.-C.; Li, Y.-C.; Chou, C. Localized surface plasmon coupled fluorescence fiber-optic biosensor for alpha-fetoprotein detection in human serum. Biosens. Bioelectron. 2009, 24, 1610–1614. [Google Scholar]
- Chang, Y.-F.; Wang, S.-F.; Huang, J.C.; Su, L.-C.; Yao, L.; Li, Y.-C.; Wu, S.-C.; Chen, Y.-M.A.; Hsieh, J.-P.; Chou, C. Detection of swine-origin influenza A (H1N1) viruses using a localized surface plasmon coupled fluorescence fiber-optic biosensor. Biosens. Bioelectron. 2010, 26, 1068–1073. [Google Scholar]
- Fang, S.; Lee, H.J.; Wark, A.W.; Corn, R.M. Attomole microarray detection of microRNAs by nanoparticle-amplified SPR imaging measurements of surface polyadenylation reactions. J. Am. Chem. Soc. 2006, 128, 14044–14046. [Google Scholar]
- He, L.; Musick, M.D.; Nicewarner, S.R.; Salinas, F.G.; Benkovic, S.J.; Natan, M.J.; Keating, C.D. Colloidal Au-enhanced surface plasmon resonance for ultrasensitive detection of DNA hybridization. J. Am. Chem. Soc. 2000, 122, 9071–9077. [Google Scholar]
- Yao, X.; Li, X.; Toledo, F.; Zurita-Lopez, C.; Gutova, M.; Momand, J.; Zhou, F. Subattomole oligonucleotide and p53 cDNA determinations via a high-resolution SPR combined with oligonucleotide-capped gold nanoparticle signal amplification. Anal. Biochem. 2006, 354, 220–228. [Google Scholar]
- Bailey, R.C.; Nam, J.-M.; Mirkin, C.A.; Hupp, J.T. Real-time multicolor DNA detection with chemoresponsive diffraction gratings and nanoparticle probes. J. Am. Chem. Soc. 2003, 125, 13541–13547. [Google Scholar]
- Du, B.A.; Li, Z.P.; Liu, C.H. One-step homogeneous detection of DNA hybridization with gold nanoparticle probes by using a linear light-scattering technique. Angew. Chem. Int. Ed. 2006, 45, 8022–8025. [Google Scholar]
- Jans, H.; Liu, X.; Austin, L.; Maes, G.; Huo, Q. Dynamic light scattering as a powerful tool for gold nanoparticle bioconjugation and biomolecular binding studies. Anal. Chem. 2009, 81, 9425–9432. [Google Scholar]
- Wang, X.; Xu, Y.; Xu, X.; Hu, K.; Xiang, M.; Li, L.; Liu, F.; Li, N. Direct determination of urinary lysozyme using surface plasmon resonance light-scattering of gold nanoparticles. Talanta 2010, 82, 693–697. [Google Scholar]
- Xie, C.; Xu, F.; Huang, X.; Dong, C.; Ren, J. Single gold nanoparticles counter: An ultrasensitive detection platform for one-step homogeneous immunoassays and DNA hybridization assays. J. Am. Chem. Soc. 2009, 131, 12763–12770. [Google Scholar]
- Wu, C.; Xiong, C.; Wang, L.; Lan, C.; Ling, L. Sensitive and selective localized surface plasmon resonance light-scattering sensor for Ag+ with unmodified gold nanoparticles. Analyst 2010, 135, 2682–2687. [Google Scholar]
- El-Sayed, I.H.; Huang, X.; El-Sayed, M.A. Surface plasmon resonance scattering and absorption of anti-EGFR antibody conjugated gold nanoparticles in cancer diagnostics: Applications in oral cancer. Nano Lett. 2005, 5, 829–834. [Google Scholar]

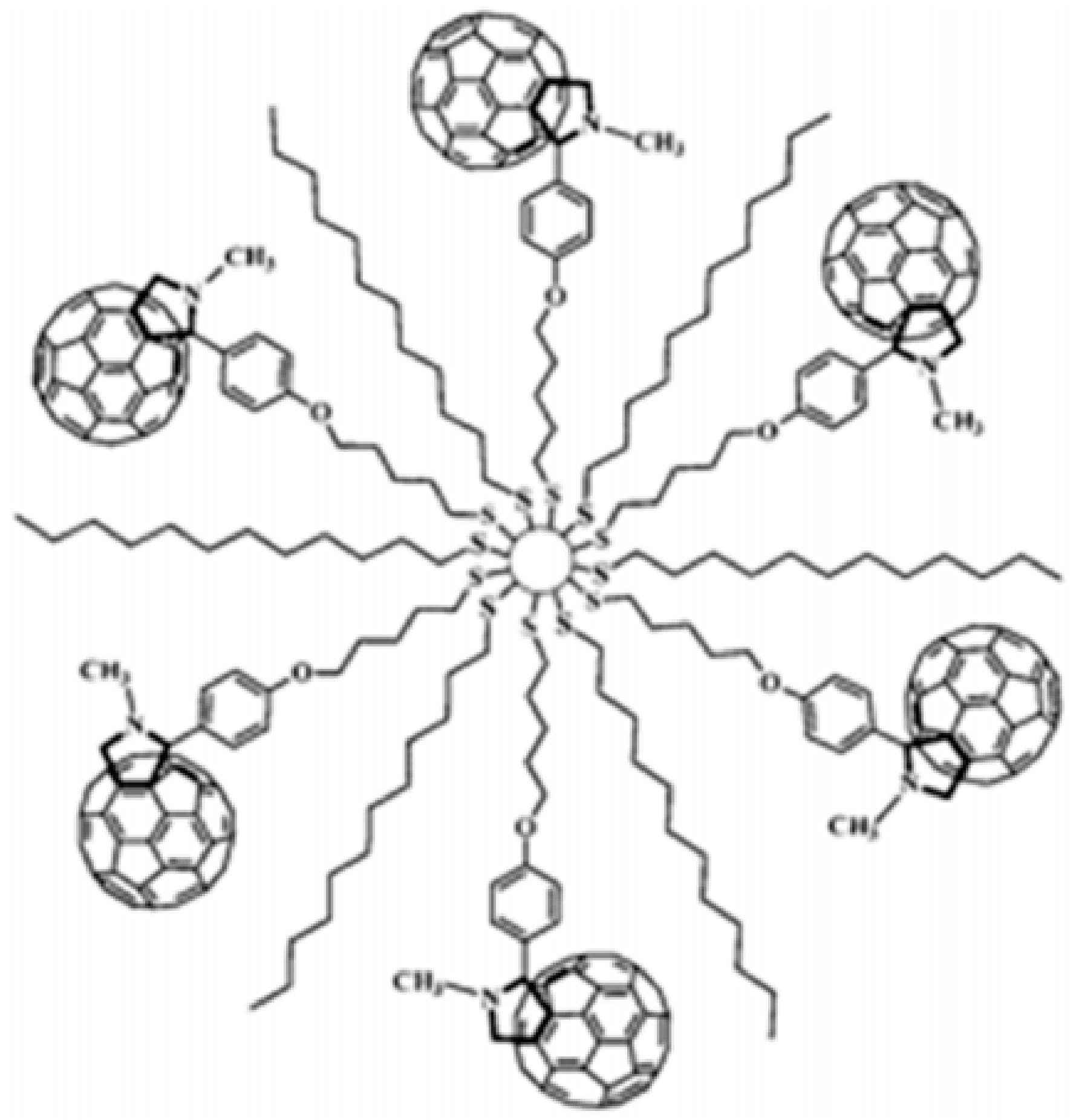


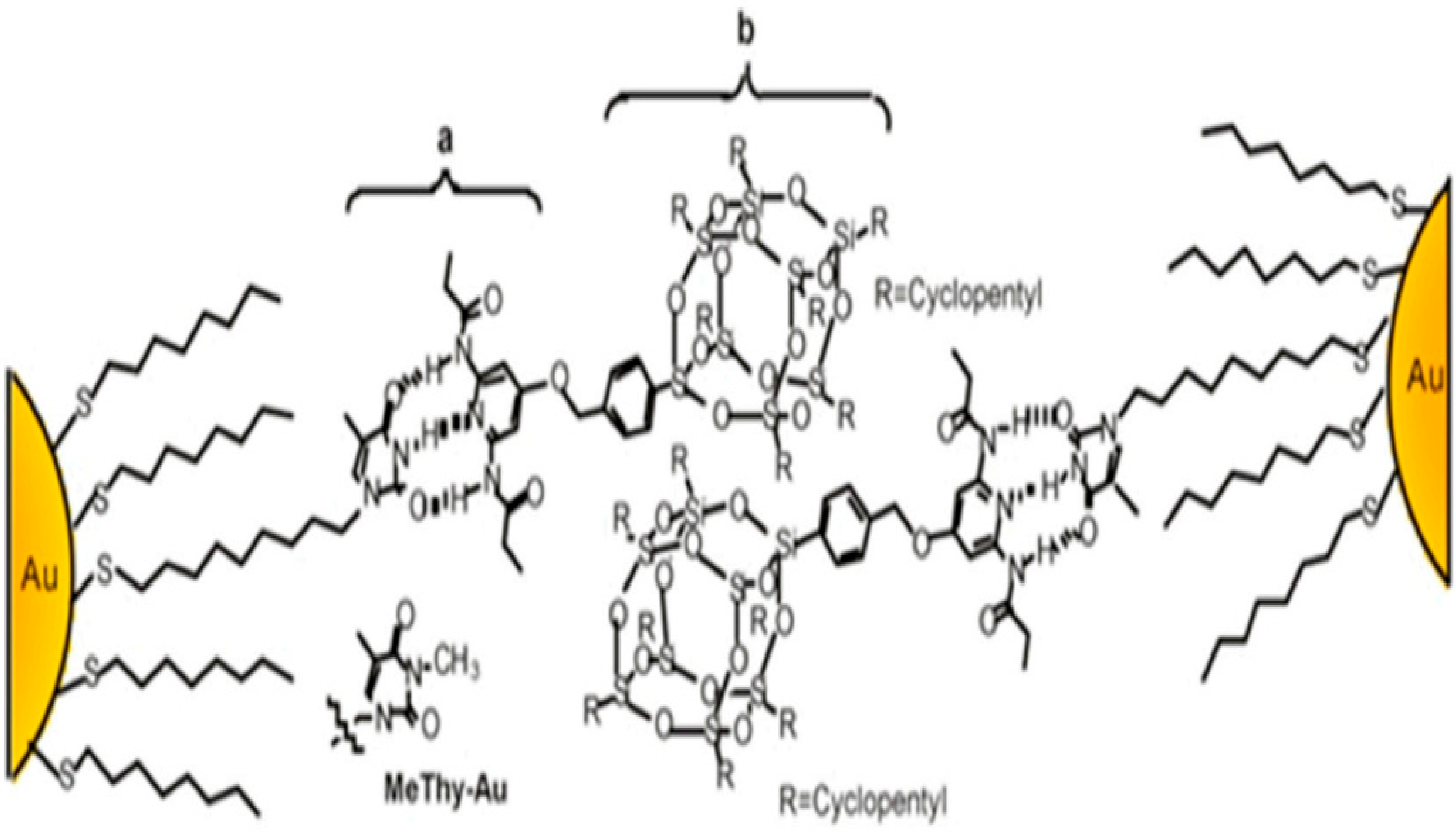











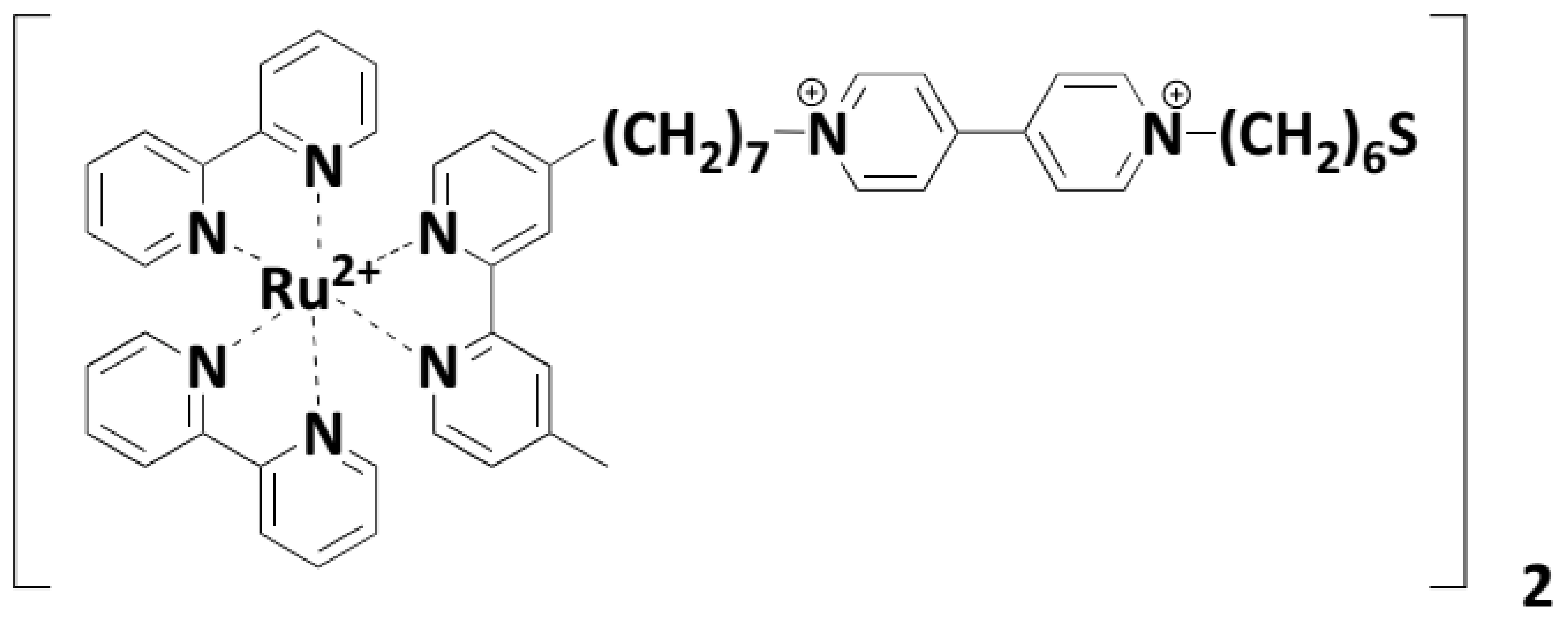







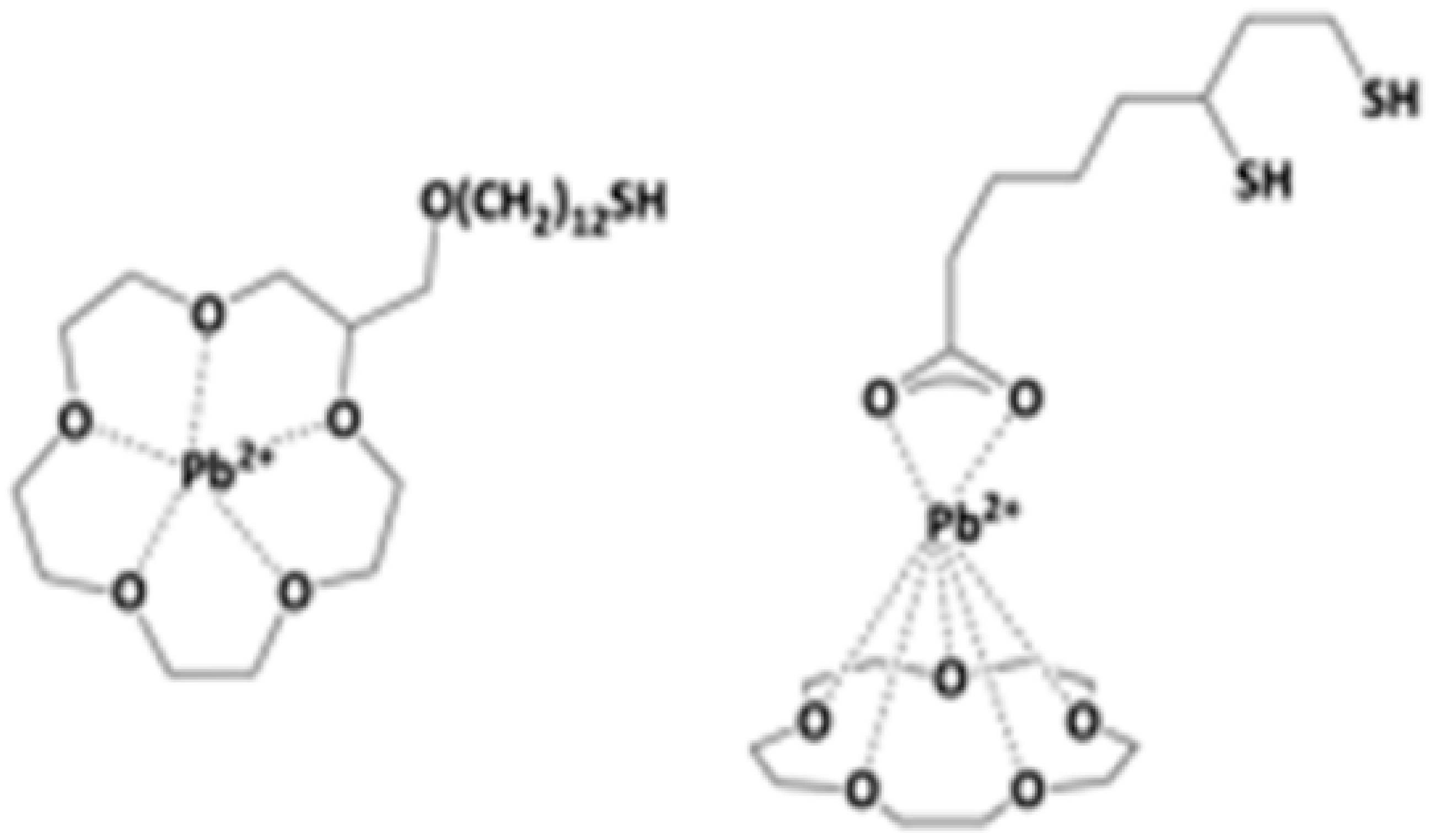



































Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaseen, M.; Humayun, M.; Khan, A.; Usman, M.; Ullah, H.; Tahir, A.A.; Ullah, H. Preparation, Functionalization, Modification, and Applications of Nanostructured Gold: A Critical Review. Energies 2021, 14, 1278. https://doi.org/10.3390/en14051278
Yaseen M, Humayun M, Khan A, Usman M, Ullah H, Tahir AA, Ullah H. Preparation, Functionalization, Modification, and Applications of Nanostructured Gold: A Critical Review. Energies. 2021; 14(5):1278. https://doi.org/10.3390/en14051278
Chicago/Turabian StyleYaseen, Muhammad, Muhammad Humayun, Abbas Khan, Muhammad Usman, Habib Ullah, Asif Ali Tahir, and Habib Ullah. 2021. "Preparation, Functionalization, Modification, and Applications of Nanostructured Gold: A Critical Review" Energies 14, no. 5: 1278. https://doi.org/10.3390/en14051278
APA StyleYaseen, M., Humayun, M., Khan, A., Usman, M., Ullah, H., Tahir, A. A., & Ullah, H. (2021). Preparation, Functionalization, Modification, and Applications of Nanostructured Gold: A Critical Review. Energies, 14(5), 1278. https://doi.org/10.3390/en14051278










