Temporospatial Nestedness in Consciousness: An Updated Perspective on the Temporospatial Theory of Consciousness
Abstract
:1. Time and Space in the Brain
2. Four Mechanisms in the TTC
3. Dimensions of Consciousness
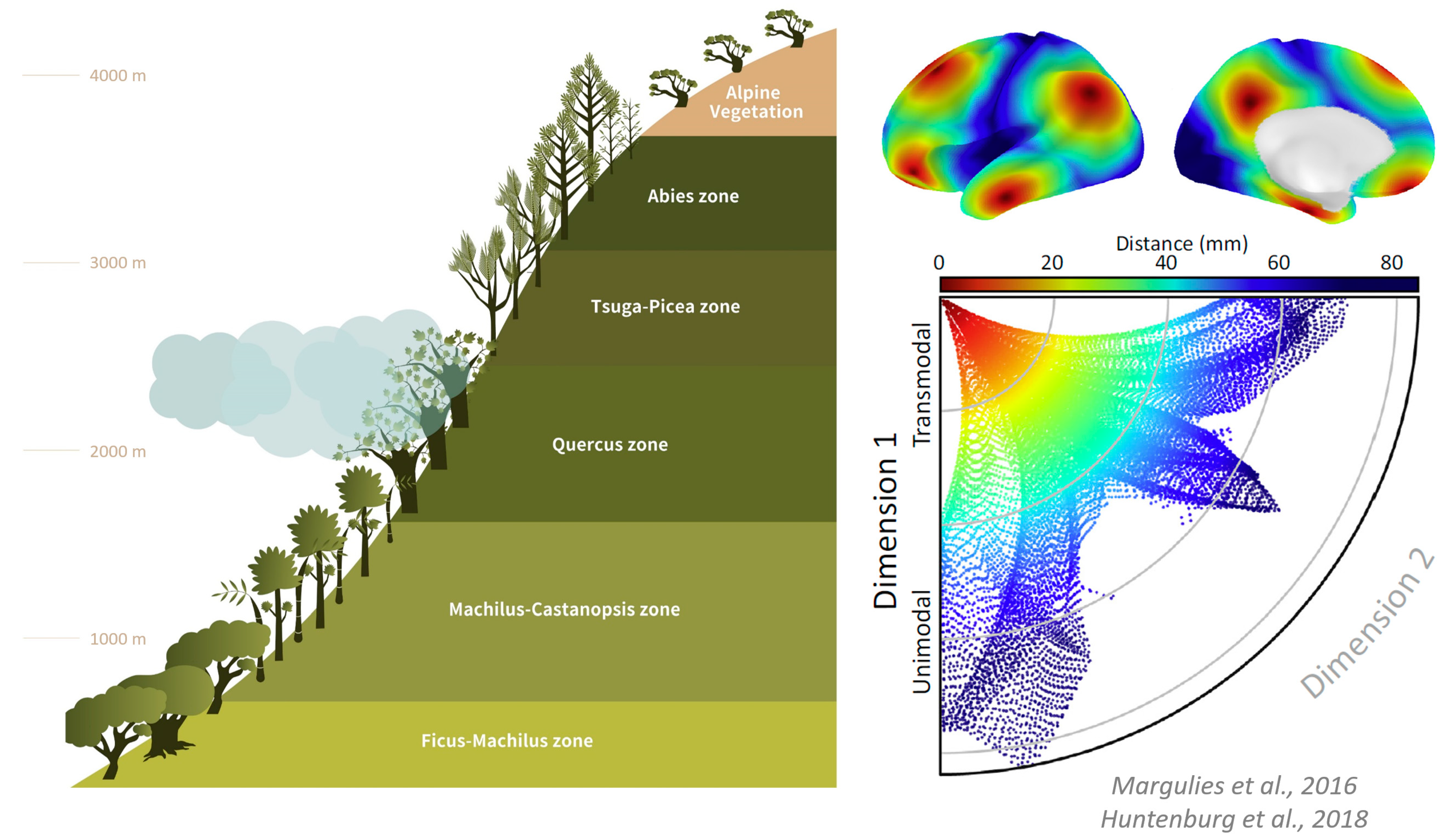
4. Functional Geometry—Spatial Organization Rules
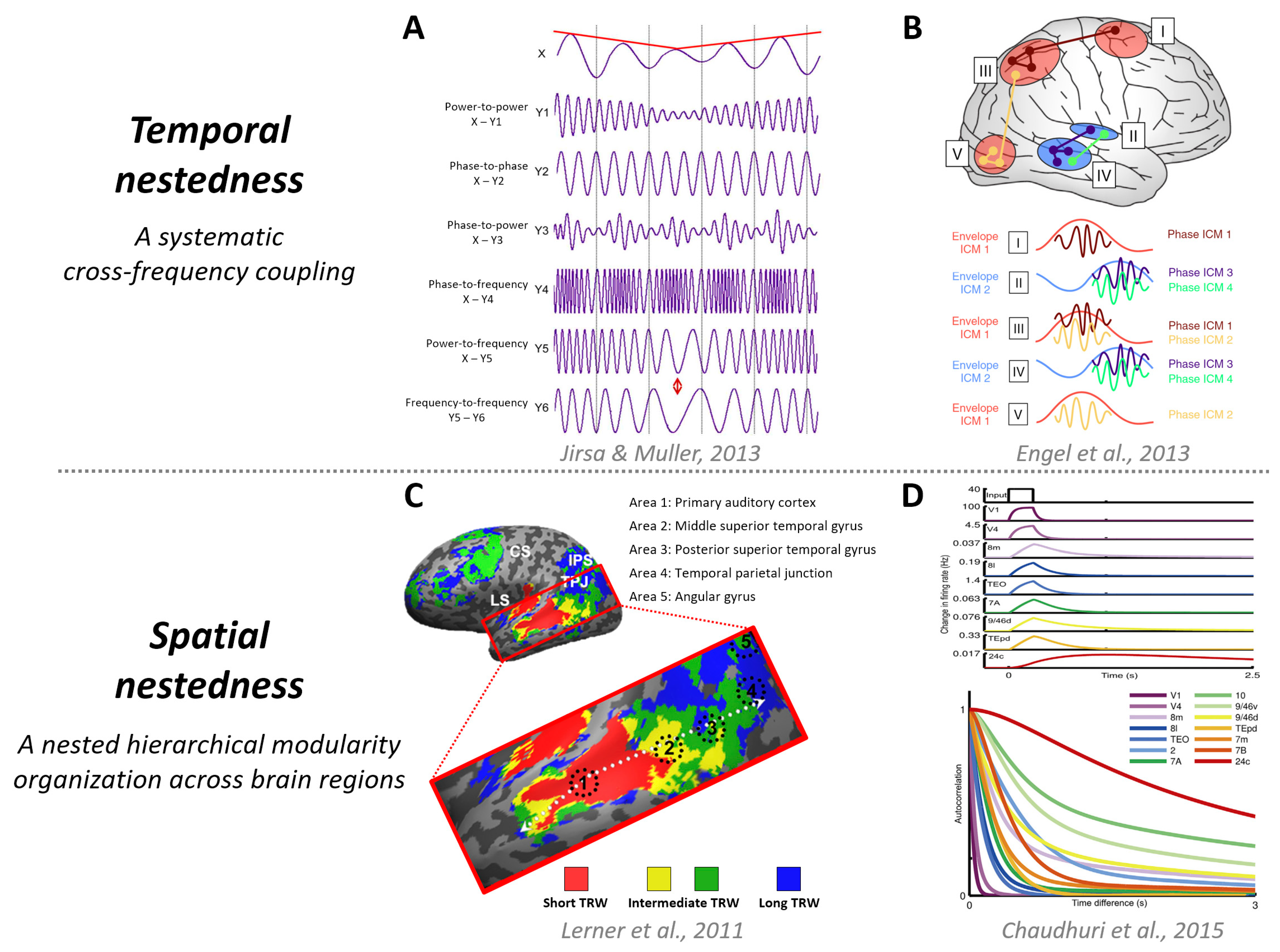
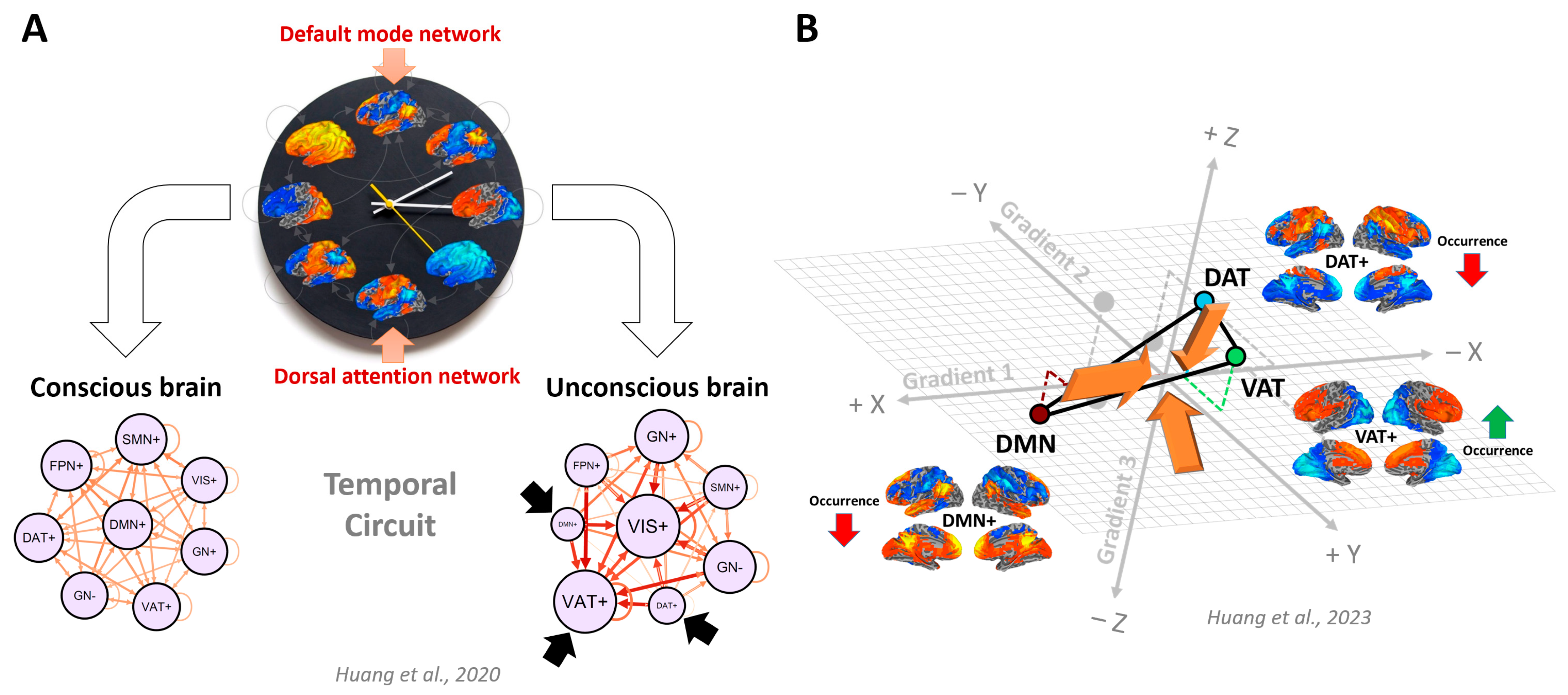
5. Temporal Circuit—Regularities in Time
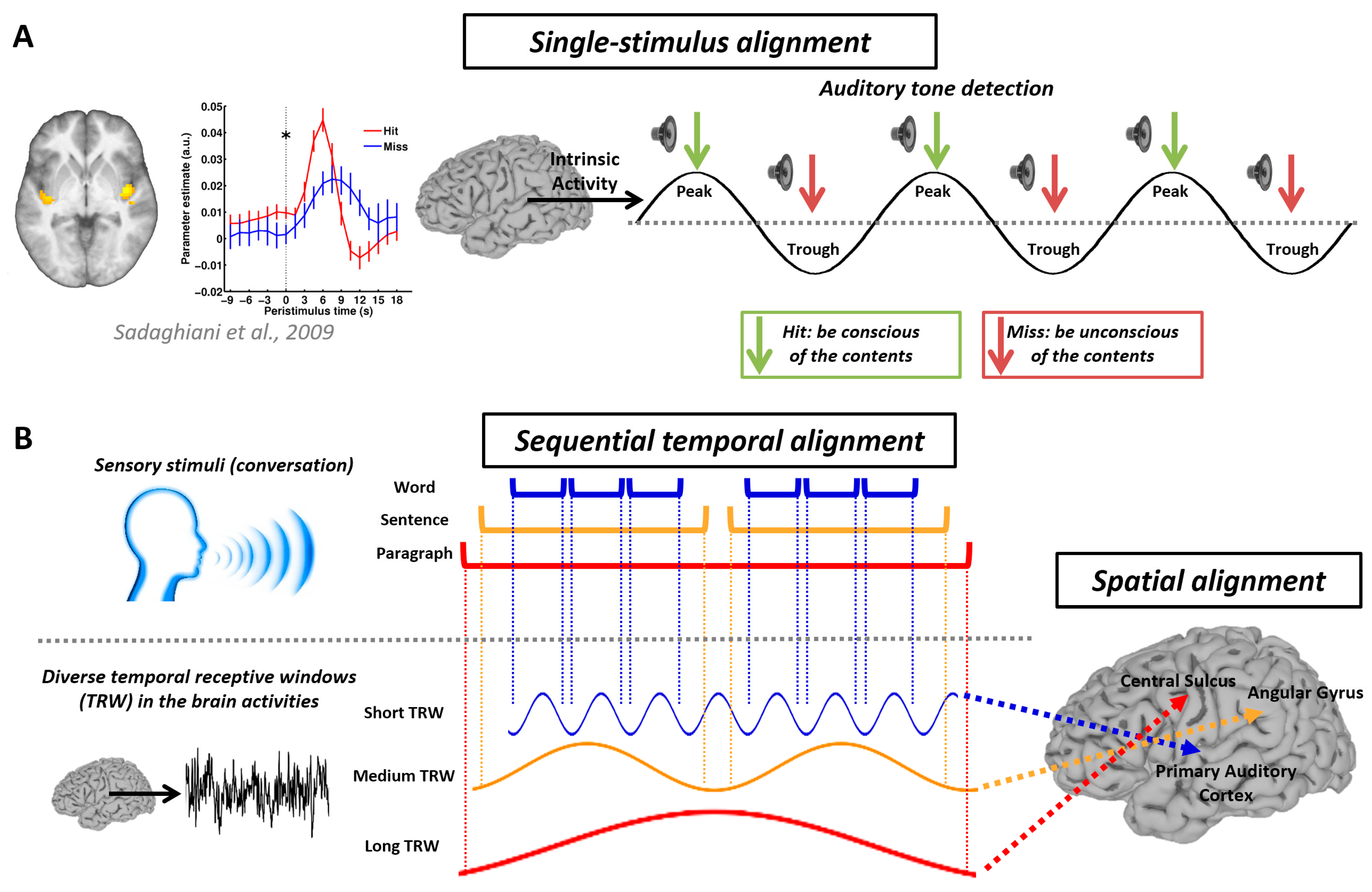
6. Temporal Circuit Nested within Functional Geometry—Temporospatial Nestedness
7. Concluding Remarks
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Aru, J.; Suzuki, M.; Larkum, M.E. Cellular Mechanisms of Conscious Processing. Trends Cogn. Sci. 2020, 24, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Alkire, M.T.; Hudetz, A.G.; Tononi, G. Consciousness and Anesthesia. Science 2008, 322, 876–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baars, B.J. The Conscious Access Hypothesis: Origins and Recent Evidence. Trends Cogn. Sci. 2002, 6, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Seth, A.K.; Bayne, T. Theories of Consciousness. Nat. Rev. Neurosci. 2022, 23, 439–452. [Google Scholar] [CrossRef]
- Bayne, T.; Seth, A.K.; Massimini, M. From Complexity to Consciousness. Trends Neurosci. 2020, 43, 546–547. [Google Scholar] [CrossRef]
- Koch, C.; Massimini, M.; Boly, M.; Tononi, G. Neural Correlates of Consciousness: Progress and Problems. Nat. Rev. Neurosci. 2016, 17, 307–321. [Google Scholar] [CrossRef]
- Rees, G.; Kreiman, G.; Koch, C. Neural Correlates of Consciousness in Humans. Nat. Rev. Neurosci. 2002, 3, 261–270. [Google Scholar] [CrossRef]
- Tononi, G.; Boly, M.; Massimini, M.; Koch, C. Integrated Information Theory: From Consciousness to Its Physical Substrate. Nat. Rev. Neurosci. 2016, 17, 450–461. [Google Scholar] [CrossRef]
- Tononi, G.; Koch, C. The Neural Correlates of Consciousness. Ann. N. Y. Acad. Sci. 2008, 1124, 239–261. [Google Scholar] [CrossRef]
- Tsuchiya, N.; Wilke, M.; Frässle, S.; Lamme, V.A.F. No-Report Paradigms: Extracting the True Neural Correlates of Consciousness. Trends Cogn. Sci. 2015, 19, 757–770. [Google Scholar] [CrossRef]
- Mashour, G.A.; Hudetz, A.G. Neural Correlates of Unconsciousness in Large-Scale Brain Networks. Trends Neurosci. 2018, 41, 150–160. [Google Scholar] [CrossRef]
- Brown, R.; Lau, H.; LeDoux, J.E. Understanding the Higher-Order Approach to Consciousness. Trends Cogn. Sci. 2019, 23, 754–768. [Google Scholar] [CrossRef]
- Lau, H.; Rosenthal, D. Empirical Support for Higher-Order Theories of Conscious Awareness. Trends Cogn. Sci. 2011, 15, 365–373. [Google Scholar] [CrossRef]
- Lamme, V.A.F. Towards a True Neural Stance on Consciousness. Trends Cogn. Sci. 2006, 10, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Dehaene, S.; Changeux, J.-P. Experimental and Theoretical Approaches to Conscious Processing. Neuron 2011, 70, 200–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Northoff, G.; Wainio-Theberge, S.; Evers, K. Is Temporo-Spatial Dynamics the “Common Currency” of Brain and Mind? In Quest of “Spatiotemporal Neuroscience”. Phys. Life Rev. 2020, 33, 34–54. [Google Scholar] [CrossRef]
- Fingelkurts, A.A.; Fingelkurts, A.A.; Neves, C.F.H. Natural World Physical, Brain Operational, and Mind Phenomenal Space–Time. Phys. Life Rev. 2010, 7, 195–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Northoff, G.; Huang, Z. How Do the Brain’s Time and Space Mediate Consciousness and Its Different Dimensions? Temporo-Spatial Theory of Consciousness (TTC). Neurosci. Biobehav. Rev. 2017, 80, 630–645. [Google Scholar] [CrossRef]
- Northoff, G.; Zilio, F. Temporo-Spatial Theory of Consciousness (TTC)—Bridging the Gap of Neuronal Activity and Phenomenal States. Behav. Brain Res. 2022, 424, 113788. [Google Scholar] [CrossRef]
- Northoff, G.; Zilio, F. From Shorter to Longer Timescales: Converging Integrated Information Theory (IIT) with the Temporo-Spatial Theory of Consciousness (TTC). Entropy 2022, 24, 270. [Google Scholar] [CrossRef]
- Hubel, D.H.; Wiesel, T.N. Receptive Fields, Binocular Interaction and Functional Architecture in the Cat’s Visual Cortex. J. Physiol. 1962, 160, 106–154. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.A.; Dumoulin, S.O.; Harvey, B.M.; Ress, D. Measurement of Population Receptive Fields in Human Early Visual Cortex Using Back-Projection Tomography. J. Vis. 2014, 14, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, P.; Markan, C.M. An Adaptable Neuromorphic Model of Orientation Selectivity Based on Floating Gate Dynamics. Front. Neurosci. 2014, 8, 54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasson, U.; Yang, E.; Vallines, I.; Heeger, D.J.; Rubin, N. A Hierarchy of Temporal Receptive Windows in Human Cortex. J. Neurosci. 2008, 28, 2539–2550. [Google Scholar] [CrossRef] [Green Version]
- Hasson, U.; Chen, J.; Honey, C.J. Hierarchical Process Memory: Memory as an Integral Component of Information Processing. Trends Cogn. Sci. 2015, 19, 304–313. [Google Scholar] [CrossRef] [Green Version]
- Lerner, Y.; Honey, C.J.; Silbert, L.J.; Hasson, U. Topographic Mapping of a Hierarchy of Temporal Receptive Windows Using a Narrated Story. J. Neurosci. 2011, 31, 2906–2915. [Google Scholar] [CrossRef] [Green Version]
- Honey, C.J.; Thesen, T.; Donner, T.H.; Silbert, L.J.; Carlson, C.E.; Devinsky, O.; Doyle, W.K.; Rubin, N.; Heeger, D.J.; Hasson, U. Slow Cortical Dynamics and the Accumulation of Information over Long Timescales. Neuron 2012, 76, 423–434. [Google Scholar] [CrossRef] [Green Version]
- Jirsa, V.; Müller, V. Cross-Frequency Coupling in Real and Virtual Brain Networks. Front. Comput. Neurosci. 2013, 7, 78. [Google Scholar] [CrossRef] [Green Version]
- Weaver, K.E.; Wander, J.D.; Ko, A.L.; Casimo, K.; Grabowski, T.J.; Ojemann, J.G.; Darvas, F. Directional Patterns of Cross Frequency Phase and Amplitude Coupling within the Resting State Mimic Patterns of FMRI Functional Connectivity. Neuroimage 2016, 128, 238–251. [Google Scholar] [CrossRef]
- Wang, L.; Saalmann, Y.B.; Pinsk, M.A.; Arcaro, M.J.; Kastner, S. Electrophysiological Low-Frequency Coherence and Cross-Frequency Coupling Contribute to BOLD Connectivity. Neuron 2012, 76, 1010–1020. [Google Scholar] [CrossRef] [Green Version]
- Hyafil, A.; Giraud, A.-L.; Fontolan, L.; Gutkin, B. Neural Cross-Frequency Coupling: Connecting Architectures, Mechanisms, and Functions. Trends Neurosci. 2015, 38, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Menara, T.; Bassett, D.S.; Pasqualetti, F. Phase-Amplitude Coupling in Neuronal Oscillator Networks. Phys. Rev. Res. 2021, 3, 023218. [Google Scholar] [CrossRef]
- Akao, A.; Shirasaka, S.; Jimbo, Y.; Ermentrout, B.; Kotani, K. Theta-Gamma Cross-Frequency Coupling Enables Covariance between Distant Brain Regions. arXiv 2019, arXiv:1903.12155. [Google Scholar] [CrossRef]
- Sotero, R.C. Topology, Cross-Frequency, and Same-Frequency Band Interactions Shape the Generation of Phase-Amplitude Coupling in a Neural Mass Model of a Cortical Column. PLoS Comput. Biol. 2016, 12, e1005180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, B.J.; Zempel, J.M.; Snyder, A.Z.; Raichle, M.E. The Temporal Structures and Functional Significance of Scale-Free Brain Activity. Neuron 2010, 66, 353–369. [Google Scholar] [CrossRef] [Green Version]
- Siebenhühner, F.; Wang, S.H.; Arnulfo, G.; Lampinen, A.; Nobili, L.; Palva, J.M.; Palva, S. Genuine Cross-Frequency Coupling Networks in Human Resting-State Electrophysiological Recordings. PLoS Biol. 2020, 18, e3000685. [Google Scholar] [CrossRef]
- Tort, A.B.L.; Kramer, M.A.; Thorn, C.; Gibson, D.J.; Kubota, Y.; Graybiel, A.M.; Kopell, N.J. Dynamic Cross-Frequency Couplings of Local Field Potential Oscillations in Rat Striatum and Hippocampus during Performance of a T-Maze Task. Proc. Natl. Acad. Sci. USA 2008, 105, 20517–20522. [Google Scholar] [CrossRef] [PubMed]
- Engel, A.K.; Gerloff, C.; Hilgetag, C.C.; Nolte, G. Intrinsic Coupling Modes: Multiscale Interactions in Ongoing Brain Activity. Neuron 2013, 80, 867–886. [Google Scholar] [CrossRef] [Green Version]
- Chaudhuri, R.; Knoblauch, K.; Gariel, M.-A.; Kennedy, H.; Wang, X.-J. A Large-Scale Circuit Mechanism for Hierarchical Dynamical Processing in the Primate Cortex. Neuron 2015, 88, 419–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chialvo, D.R. Emergent Complex Neural Dynamics. Nat. Phys. 2010, 6, 744–750. [Google Scholar] [CrossRef] [Green Version]
- Farbood, M.M.; Heeger, D.J.; Marcus, G.; Hasson, U.; Lerner, Y. The Neural Processing of Hierarchical Structure in Music and Speech at Different Timescales. Front. Neurosci. 2015, 9, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, R.; van den Brink, R.L.; Pfeffer, T.; Voytek, B. Neuronal Timescales Are Functionally Dynamic and Shaped by Cortical Microarchitecture. Elife 2020, 9, e61277. [Google Scholar] [CrossRef]
- Golesorkhi, M.; Gomez-Pilar, J.; Zilio, F.; Berberian, N.; Wolff, A.; Yagoub, M.C.E.; Northoff, G. The Brain and Its Time: Intrinsic Neural Timescales Are Key for Input Processing. Commun. Biol. 2021, 4, 970. [Google Scholar] [CrossRef] [PubMed]
- Kiebel, S.J.; Daunizeau, J.; Friston, K.J. A Hierarchy of Time-Scales and the Brain. PLoS Comput. Biol. 2008, 4, e1000209. [Google Scholar] [CrossRef] [PubMed]
- Klar, P.; Çatal, Y.; Langner, R.; Huang, Z.; Northoff, G. Scale-free Dynamics of Core-periphery Topography. Hum. Brain Mapp. 2023, 44, 1997–2017. [Google Scholar] [CrossRef]
- Manea, A.M.; Zilverstand, A.; Ugurbil, K.; Heilbronner, S.R.; Zimmermann, J. Intrinsic Timescales as an Organizational Principle of Neural Processing across the Whole Rhesus Macaque Brain. Elife 2022, 11, e75540. [Google Scholar] [CrossRef]
- Murray, J.D.; Bernacchia, A.; Freedman, D.J.; Romo, R.; Wallis, J.D.; Cai, X.; Padoa-Schioppa, C.; Pasternak, T.; Seo, H.; Lee, D.; et al. A Hierarchy of Intrinsic Timescales across Primate Cortex. Nat. Neurosci. 2014, 17, 1661–1663. [Google Scholar] [CrossRef] [Green Version]
- Nanda, A.; Johnson, G.W.; Mu, Y.; Ahrens, M.B.; Chang, C.; Englot, D.J.; Breakspear, M.; Rubinov, M. Time-Resolved Correlation of Distributed Brain Activity Tracks E-I Balance and Accounts for Diverse Scale-Free Phenomena. Cell. Rep. 2023, 42, 112254. [Google Scholar] [CrossRef] [PubMed]
- Raut, R.V.; Snyder, A.Z.; Raichle, M.E. Hierarchical Dynamics as a Macroscopic Organizing Principle of the Human Brain. Proc. Natl. Acad. Sci. USA 2020, 117, 20890–20897. [Google Scholar] [CrossRef]
- Runyan, C.A.; Piasini, E.; Panzeri, S.; Harvey, C.D. Distinct Timescales of Population Coding across Cortex. Nature 2017, 548, 92–96. [Google Scholar] [CrossRef] [Green Version]
- Varela, F.; Lachaux, J.-P.; Rodriguez, E.; Martinerie, J. The Brainweb: Phase Synchronization and Large-Scale Integration. Nat. Rev. Neurosci. 2001, 2, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J. Macroscopic Gradients of Synaptic Excitation and Inhibition in the Neocortex. Nat. Rev. Neurosci. 2020, 21, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.; Berberian, N.; Golesorkhi, M.; Gomez-Pilar, J.; Zilio, F.; Northoff, G. Intrinsic Neural Timescales: Temporal Integration and Segregation. Trends Cogn. Sci. 2022, 26, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Yeshurun, Y.; Nguyen, M.; Hasson, U. Amplification of Local Changes along the Timescale Processing Hierarchy. Proc. Natl. Acad. Sci. USA 2017, 114, 9475–9480. [Google Scholar] [CrossRef] [PubMed]
- Zilio, F.; Gomez-Pilar, J.; Cao, S.; Zhang, J.; Zang, D.; Qi, Z.; Tan, J.; Hiromi, T.; Wu, X.; Fogel, S.; et al. Are Intrinsic Neural Timescales Related to Sensory Processing? Evidence from Abnormal Behavioral States. Neuroimage 2021, 226, 117579. [Google Scholar] [CrossRef] [PubMed]
- Margulies, D.S.; Ghosh, S.S.; Goulas, A.; Falkiewicz, M.; Huntenburg, J.M.; Langs, G.; Bezgin, G.; Eickhoff, S.B.; Castellanos, F.X.; Petrides, M.; et al. Situating the Default-Mode Network along a Principal Gradient of Macroscale Cortical Organization. Proc. Natl. Acad. Sci. USA 2016, 113, 12574–12579. [Google Scholar] [CrossRef]
- Burt, J.B.; Demirtaş, M.; Eckner, W.J.; Navejar, N.M.; Ji, J.L.; Martin, W.J.; Bernacchia, A.; Anticevic, A.; Murray, J.D. Hierarchy of Transcriptomic Specialization across Human Cortex Captured by Structural Neuroimaging Topography. Nat. Neurosci. 2018, 21, 1251–1259. [Google Scholar] [CrossRef]
- Deco, G.; Vidaurre, D.; Kringelbach, M.L. Revisiting the Global Workspace Orchestrating the Hierarchical Organization of the Human Brain. Nat. Hum. Behav. 2021, 5, 497–511. [Google Scholar] [CrossRef]
- Demirtaş, M.; Burt, J.B.; Helmer, M.; Ji, J.L.; Adkinson, B.D.; Glasser, M.F.; Essen, D.C.V.; Sotiropoulos, S.N.; Anticevic, A.; Murray, J.D. Hierarchical Heterogeneity across Human Cortex Shapes Large-Scale Neural Dynamics. Neuron 2019, 101, 1181–1194. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Kong, R.; Kong, X.; Liégeois, R.; Orban, C.; Deco, G.; van den Heuvel, M.P.; Yeo, B.T.T. Inversion of a Large-Scale Circuit Model Reveals a Cortical Hierarchy in the Dynamic Resting Human Brain. Sci. Adv. 2019, 5, eaat7854. [Google Scholar] [CrossRef] [Green Version]
- Mukamel, E.A.; Pirondini, E.; Babadi, B.; Wong, K.F.K.; Pierce, E.T.; Harrell, P.G.; Walsh, J.L.; Salazar-Gomez, A.F.; Cash, S.S.; Eskandar, E.N.; et al. A Transition in Brain State during Propofol-Induced Unconsciousness. J. Neurosci. 2014, 34, 839–845. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Li, X.; Hagihira, S.; Sleigh, J.W. Cross-Frequency Coupling during Isoflurane Anaesthesia as Revealed by Electroencephalographic Harmonic Wavelet Bicoherence. Bja Br. J. Anaesth. 2013, 110, 409–419. [Google Scholar] [CrossRef] [Green Version]
- Chamadia, S.; Pedemonte, J.C.; Hahm, E.Y.; Mekonnen, J.; Ibala, R.; Gitlin, J.; Ethridge, B.R.; Qu, J.; Vazquez, R.; Rhee, J.; et al. Delta Oscillations Phase Limit Neural Activity during Sevoflurane Anesthesia. Commun. Biol. 2019, 2, 415. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wen, X.; Ou, G.; Li, S.; Chen, Y.; Zhang, J.; Liang, Z. Propofol Modulates Neural Dynamics of Thalamo-Cortical System Associated with Anesthetic Levels in Rats. Cogn. Neurodyn. 2022, 1–19. [Google Scholar] [CrossRef]
- Huang, Z.; Tarnal, V.; Vlisides, P.E.; Janke, E.L.; McKinney, A.M.; Picton, P.; Mashour, G.A.; Hudetz, A.G. Asymmetric Neural Dynamics Characterize Loss and Recovery of Consciousness. Neuroimage 2021, 236, 118042. [Google Scholar] [CrossRef]
- Huang, Z.; Liu, X.; Mashour, G.A.; Hudetz, A.G. Timescales of Intrinsic BOLD Signal Dynamics and Functional Connectivity in Pharmacologic and Neuropathologic States of Unconsciousness. J. Neurosci. 2018, 38, 2304–2317. [Google Scholar] [CrossRef] [Green Version]
- Signorelli, C.M.; Uhrig, L.; Kringelbach, M.; Jarraya, B.; Deco, G. Hierarchical Disruption in the Cortex of Anesthetized Monkeys as a New Signature of Consciousness Loss. Neuroimage 2021, 227, 117618. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Mashour, G.A.; Hudetz, A.G. Functional Geometry of the Cortex Encodes Dimensions of Consciousness. Nat. Commun. 2023, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Podvalny, E.; Flounders, M.W.; King, L.E.; Holroyd, T.; He, B.J. A Dual Role of Prestimulus Spontaneous Neural Activity in Visual Object Recognition. Nat. Commun. 2019, 10, 3910. [Google Scholar] [CrossRef] [Green Version]
- Ergenoglu, T.; Demiralp, T.; Bayraktaroglu, Z.; Ergen, M.; Beydagi, H.; Uresin, Y. Alpha Rhythm of the EEG Modulates Visual Detection Performance in Humans. Cogn. Brain Res. 2004, 20, 376–383. [Google Scholar] [CrossRef]
- Boly, M.; Balteau, E.; Schnakers, C.; Degueldre, C.; Moonen, G.; Luxen, A.; Phillips, C.; Peigneux, P.; Maquet, P.; Laureys, S. Baseline Brain Activity Fluctuations Predict Somatosensory Perception in Humans. Proc. Natl. Acad. Sci. USA 2007, 104, 12187–12192. [Google Scholar] [CrossRef]
- Sadaghiani, S.; Hesselmann, G.; Kleinschmidt, A. Distributed and Antagonistic Contributions of Ongoing Activity Fluctuations to Auditory Stimulus Detection. J. Neurosci. 2009, 29, 13410–13417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al, E.; Iliopoulos, F.; Forschack, N.; Nierhaus, T.; Grund, M.; Motyka, P.; Gaebler, M.; Nikulin, V.V.; Villringer, A. Heart–Brain Interactions Shape Somatosensory Perception and Evoked Potentials. Proc. Natl. Acad. Sci. USA 2020, 117, 10575–10584. [Google Scholar] [CrossRef] [PubMed]
- Sadaghiani, S.; Scheeringa, R.; Lehongre, K.; Morillon, B.; Giraud, A.-L.; Kleinschmidt, A. Intrinsic Connectivity Networks, Alpha Oscillations, and Tonic Alertness: A Simultaneous Electroencephalography/Functional Magnetic Resonance Imaging Study. J. Neurosci. 2010, 30, 10243–10250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hesselmann, G.; Kell, C.A.; Kleinschmidt, A. Ongoing Activity Fluctuations in HMT+ Bias the Perception of Coherent Visual Motion. J. Neurosci. 2008, 28, 14481–14485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadaghiani, S.; Poline, J.-B.; Kleinschmidt, A.; D’Esposito, M. Ongoing Dynamics in Large-Scale Functional Connectivity Predict Perception. Proc. Natl. Acad. Sci. USA 2015, 112, 8463–8468. [Google Scholar] [CrossRef]
- Benwell, C.S.Y.; Tagliabue, C.F.; Veniero, D.; Cecere, R.; Savazzi, S.; Thut, G. Prestimulus EEG Power Predicts Conscious Awareness But Not Objective Visual Performance. Eneuro 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Ploner, M.; Lee, M.C.; Wiech, K.; Bingel, U.; Tracey, I. Prestimulus Functional Connectivity Determines Pain Perception in Humans. Proc. Natl. Acad. Sci. USA 2010, 107, 355–360. [Google Scholar] [CrossRef]
- Van Dijk, H.; Schoffelen, J.-M.; Oostenveld, R.; Jensen, O. Prestimulus Oscillatory Activity in the Alpha Band Predicts Visual Discrimination Ability. J. Neurosci. 2008, 28, 1816–1823. [Google Scholar] [CrossRef] [Green Version]
- Romei, V.; Brodbeck, V.; Michel, C.; Amedi, A.; Pascual-Leone, A.; Thut, G. Spontaneous Fluctuations in Posterior α-Band EEG Activity Reflect Variability in Excitability of Human Visual Areas. Cereb. Cortex 2008, 18, 2010–2018. [Google Scholar] [CrossRef]
- Iemi, L.; Chaumon, M.; Crouzet, S.M.; Busch, N.A. Spontaneous Neural Oscillations Bias Perception by Modulating Baseline Excitability. J. Neurosci. 2017, 37, 807–819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Vugt, B.; Dagnino, B.; Vartak, D.; Safaai, H.; Panzeri, S.; Dehaene, S.; Roelfsema, P.R. The Threshold for Conscious Report: Signal Loss and Response Bias in Visual and Frontal Cortex. Science 2018, 360, 537–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Northoff, G.; Klar, P.; Bein, M.; Safron, A. As without, so within: How the Brain’s Temporo-Spatial Alignment to the Environment Shapes Consciousness. Interface Focus 2023, 13, 20220076. [Google Scholar] [CrossRef]
- Safron, A. An Integrated World Modeling Theory (IWMT) of Consciousness: Combining Integrated Information and Global Neuronal Workspace Theories With the Free Energy Principle and Active Inference Framework; Toward Solving the Hard Problem and Characterizing Agentic Causation. Front. Artif. Intell. 2020, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- Safron, A. Integrated World Modeling Theory Expanded: Implications for the Future of Consciousness. Front. Comput. Neurosci. 2022, 16, 642397. [Google Scholar] [CrossRef] [PubMed]
- Casali, A.G.; Gosseries, O.; Rosanova, M.; Boly, M.; Sarasso, S.; Casali, K.R.; Casarotto, S.; Bruno, M.-A.; Laureys, S.; Tononi, G.; et al. A Theoretically Based Index of Consciousness Independent of Sensory Processing and Behavior. Sci. Transl. Med. 2013, 5, 198ra105. [Google Scholar] [CrossRef]
- Fingelkurts, A.A.; Fingelkurts, A.A. Present Moment, Past, and Future: Mental Kaleidoscope. Front. Psychol. 2014, 5, 395. [Google Scholar] [CrossRef] [Green Version]
- Siclari, F.; Baird, B.; Perogamvros, L.; Bernardi, G.; LaRocque, J.J.; Riedner, B.; Boly, M.; Postle, B.R.; Tononi, G. The Neural Correlates of Dreaming. Nat. Neurosci. 2017, 20, 872–878. [Google Scholar] [CrossRef] [Green Version]
- Cul, A.D.; Dehaene, S.; Reyes, P.; Bravo, E.; Slachevsky, A. Causal Role of Prefrontal Cortex in the Threshold for Access to Consciousness. Brain 2009, 132, 2531–2540. [Google Scholar] [CrossRef] [Green Version]
- Block, N. Two Neural Correlates of Consciousness. Trends Cogn. Sci. 2005, 9, 46–52. [Google Scholar] [CrossRef] [Green Version]
- Mashour, G.A.; Roelfsema, P.; Changeux, J.-P.; Dehaene, S. Conscious Processing and the Global Neuronal Workspace Hypothesis. Neuron 2020, 105, 776–798. [Google Scholar] [CrossRef] [PubMed]
- Moutard, C.; Dehaene, S.; Malach, R. Spontaneous Fluctuations and Non-Linear Ignitions: Two Dynamic Faces of Cortical Recurrent Loops. Neuron 2015, 88, 194–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dehaene, S.; Charles, L.; King, J.-R.; Marti, S. Toward a Computational Theory of Conscious Processing. Curr. Opin. Neurobiol. 2014, 25, 76–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dehaene, S.; Kerszberg, M.; Changeux, J.-P. A Neuronal Model of a Global Workspace in Effortful Cognitive Tasks. Proc. Natl. Acad. Sci. USA 1998, 95, 14529–14534. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.N.; Purdon, P.L.; Dort, C.J.V. General Anesthesia and Altered States of Arousal: A Systems Neuroscience Analysis. Annu. Rev. Neurosci. 2011, 34, 601–628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laureys, S. The Neural Correlate of (Un)Awareness: Lessons from the Vegetative State. Trends Cogn. Sci. 2005, 9, 556–559. [Google Scholar] [CrossRef]
- Fingelkurts, A.A.; Fingelkurts, A.A.; Bagnato, S.; Boccagni, C.; Galardi, G. Do We Need a Theory-Based Assessment of Consciousness in the Field of Disorders of Consciousness? Front. Hum. Neurosci. 2014, 8, 402. [Google Scholar] [CrossRef] [Green Version]
- Monti, M.M. Cognition in the Vegetative State. Annu. Rev. Clin. Psychol. 2012, 8, 431–454. [Google Scholar] [CrossRef] [Green Version]
- Schiff, N.D. Cognitive Motor Dissociation Following Severe Brain Injuries. Jama Neurol. 2015, 72, 1413–1415. [Google Scholar] [CrossRef]
- Owen, A.M.; Coleman, M.R.; Boly, M.; Davis, M.H.; Laureys, S.; Pickard, J.D. Detecting Awareness in the Vegetative State. Science 2006, 313, 1402. [Google Scholar] [CrossRef] [Green Version]
- Timmermann, C.; Bauer, P.R.; Gosseries, O.; Vanhaudenhuyse, A.; Vollenweider, F.; Laureys, S.; Singer, T.; Group, M. and L.E. (MLE) E.R.; Antonova, E.; Lutz, A. A Neurophenomenological Approach to Non-Ordinary States of Consciousness: Hypnosis, Meditation, and Psychedelics. Trends Cogn. Sci. 2023, 27, 139–159. [Google Scholar] [CrossRef]
- Bayne, T.; Hohwy, J.; Owen, A.M. Are There Levels of Consciousness? Trends Cogn. Sci. 2016, 20, 405–413. [Google Scholar] [CrossRef] [Green Version]
- Bayne, T.; Carter, O. Dimensions of Consciousness and the Psychedelic State. Neurosci. Conscious. 2018, 2018, niy008. [Google Scholar] [CrossRef] [Green Version]
- Fortier-Davy, M.; Millière, R. The Multi-Dimensional Approach to Drug-Induced States a Commentary on Bayne and Carter’s “Dimensions of Consciousness and the Psychedelic State”. Neurosci. Conscious. 2020, 2020, niaa004. [Google Scholar] [CrossRef] [PubMed]
- Birch, J.; Schnell, A.K.; Clayton, N.S. Dimensions of Animal Consciousness. Trends Cogn. Sci. 2020, 24, 789–801. [Google Scholar] [CrossRef]
- Walter, J. Consciousness as a Multidimensional Phenomenon: Implications for the Assessment of Disorders of Consciousness. Neurosci. Conscious. 2021, 2021, niab047. [Google Scholar] [CrossRef]
- Carlén, M. What Constitutes the Prefrontal Cortex? Science 2017, 358, 478–482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.-J.; Friston, K. Structural and Functional Brain Networks: From Connections to Cognition. Science 2013, 342, 1238411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernhardt, B.C.; Smallwood, J.; Keilholz, S.; Margulies, D.S. Gradients in Brain Organization. Neuroimage 2022, 251, 118987. [Google Scholar] [CrossRef] [PubMed]
- Huntenburg, J.M.; Bazin, P.-L.; Margulies, D.S. Large-Scale Gradients in Human Cortical Organization. Trends Cogn. Sci. 2018, 22, 21–31. [Google Scholar] [CrossRef] [Green Version]
- Keller, A.S.; Sydnor, V.J.; Pines, A.; Fair, D.A.; Bassett, D.S.; Satterthwaite, T.D. Hierarchical Functional System Development Supports Executive Function. Trends Cogn. Sci. 2023, 27, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.; Jefferies, E.; Rueschemeyer, S.-A.; Sormaz, M.; Wang, H.; Margulies, D.S.; Smallwood, J. Distant from Input: Evidence of Regions within the Default Mode Network Supporting Perceptually-Decoupled and Conceptually-Guided Cognition. Neuroimage 2018, 171, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, J.; Turnbull, A.; Wang, H.; Ho, N.S.P.; Poerio, G.L.; Karapanagiotidis, T.; Konu, D.; Mckeown, B.; Zhang, M.; Murphy, C.; et al. The Neural Correlates of Ongoing Conscious Thought. Iscience 2021, 24, 102132. [Google Scholar] [CrossRef] [PubMed]
- Cross, N.; Paquola, C.; Pomares, F.B.; Perrault, A.A.; Jegou, A.; Nguyen, A.; Aydin, U.; Bernhardt, B.C.; Grova, C.; Dang-Vu, T.T. Cortical Gradients of Functional Connectivity Are Robust to State-Dependent Changes Following Sleep Deprivation. Neuroimage 2021, 226, 117547. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, J.; Wu, J.; Mashour, G.A.; Hudetz, A.G. Temporal Circuit of Macroscale Dynamic Brain Activity Supports Human Consciousness. Sci. Adv. 2020, 6, eaaz0087. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Tarnal, V.; Vlisides, P.E.; Janke, E.L.; McKinney, A.M.; Picton, P.; Mashour, G.A.; Hudetz, A.G. Anterior Insula Regulates Brain Network Transitions That Gate Conscious Access. Cell Rep. 2021, 35, 109081. [Google Scholar] [CrossRef]
- Menon, V. Large-Scale Brain Networks and Psychopathology: A Unifying Triple Network Model. Trends Cogn. Sci. 2011, 15, 483–506. [Google Scholar] [CrossRef]
- Uddin, L.Q. Salience Processing and Insular Cortical Function and Dysfunction. Nat. Rev. Neurosci. 2015, 16, 55–61. [Google Scholar] [CrossRef]
- Klar, P.; Çatal, Y.; Langner, R.; Huang, Z.; Northoff, G. Scale-Free Dynamics in the Core-Periphery Topography and Task Alignment Decline from Conscious to Unconscious States. Commun. Biol. 2023, 6, 499. [Google Scholar] [CrossRef]
- Vossel, S.; Geng, J.J.; Fink, G.R. Dorsal and Ventral Attention Systems. Neurosci. 2014, 20, 150–159. [Google Scholar] [CrossRef]
- Corbetta, M.; Shulman, G.L. Control of Goal-Directed and Stimulus-Driven Attention in the Brain. Nat. Rev. Neurosci. 2002, 3, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, J.; Bernhardt, B.C.; Leech, R.; Bzdok, D.; Jefferies, E.; Margulies, D.S. The Default Mode Network in Cognition: A Topographical Perspective. Nat. Rev. Neurosci. 2021, 22, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Raichle, M.E.; MacLeod, A.M.; Snyder, A.Z.; Powers, W.J.; Gusnard, D.A.; Shulman, G.L. A Default Mode of Brain Function. Proc. Natl. Acad. Sci. USA 2001, 98, 676–682. [Google Scholar] [CrossRef]
- Demertzi, A.; Kucyi, A.; Ponce-Alvarez, A.; Keliris, G.A.; Whitfield-Gabrieli, S.; Deco, G. Functional Network Antagonism and Consciousness. Netw. Neurosci. 2022, 6, 998–1009. [Google Scholar] [CrossRef]
- Schooler, J.W.; Smallwood, J.; Christoff, K.; Handy, T.C.; Reichle, E.D.; Sayette, M.A. Meta-Awareness, Perceptual Decoupling and the Wandering Mind. Trends Cogn. Sci. 2011, 15, 319–326. [Google Scholar] [CrossRef]
- Raichle, M.E. The Brain’s Default Mode Network. Annu. Rev. Neurosci. 2013, 38, 433–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fornito, A.; Harrison, B.J.; Zalesky, A.; Simons, J.S. Competitive and Cooperative Dynamics of Large-Scale Brain Functional Networks Supporting Recollection. Proc. Natl. Acad. Sci. USA 2012, 109, 12788–12793. [Google Scholar] [CrossRef]
- Critchley, H.D.; Wiens, S.; Rotshtein, P.; Öhman, A.; Dolan, R.J. Neural Systems Supporting Interoceptive Awareness. Nat. Neurosci. 2004, 7, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Faivre, N.; Salomon, R.; Blanke, O. Visual Consciousness and Bodily Self-Consciousness. Curr. Opin. Neurol. 2015, 28, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Fingelkurts, A.A.; Fingelkurts, A.A.; Kallio-Tamminen, T. Selfhood Triumvirate: From Phenomenology to Brain Activity and Back Again. Conscious. Cogn. 2020, 86, 103031. [Google Scholar] [CrossRef]
- Northoff, G. Immanuel Kant’s Mind and the Brain’s Resting State. Trends Cogn. Sci. 2012, 16, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Sainburg, L.E.; Kuang, S.; Han, F.; Williams, J.W.; Liu, Y.; Zhang, N.; Zhang, X.; Leopold, D.A.; Liu, X. Brain Activity Fluctuations Propagate as Waves Traversing the Cortical Hierarchy. Cereb. Cortex 2021, 31, bhab064. [Google Scholar] [CrossRef]
- Raut, R.V.; Snyder, A.Z.; Mitra, A.; Yellin, D.; Fujii, N.; Malach, R.; Raichle, M.E. Global Waves Synchronize the Brain’s Functional Systems with Fluctuating Arousal. Sci. Adv. 2021, 7, eabf2709. [Google Scholar] [CrossRef]
- Yousefi, B.; Keilholz, S. Propagating Patterns of Intrinsic Activity along Macroscale Gradients Coordinate Functional Connections across the Whole Brain. Neuroimage 2021, 231, 117827. [Google Scholar] [CrossRef] [PubMed]
- Bolt, T.; Nomi, J.S.; Bzdok, D.; Salas, J.A.; Chang, C.; Yeo, B.T.T.; Uddin, L.Q.; Keilholz, S.D. A Parsimonious Description of Global Functional Brain Organization in Three Spatiotemporal Patterns. Nat. Neurosci. 2022, 25, 1093–1103. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Z. Temporospatial Nestedness in Consciousness: An Updated Perspective on the Temporospatial Theory of Consciousness. Entropy 2023, 25, 1074. https://doi.org/10.3390/e25071074
Huang Z. Temporospatial Nestedness in Consciousness: An Updated Perspective on the Temporospatial Theory of Consciousness. Entropy. 2023; 25(7):1074. https://doi.org/10.3390/e25071074
Chicago/Turabian StyleHuang, Zirui. 2023. "Temporospatial Nestedness in Consciousness: An Updated Perspective on the Temporospatial Theory of Consciousness" Entropy 25, no. 7: 1074. https://doi.org/10.3390/e25071074




