A Review and Evaluation of the Data Supporting Internal Use of Helichrysum italicum
Abstract
:1. Introduction
2. Methodology
3. Traditional Uses and Scientific Data
4. Human Clinical Observations
5. Registered Clinical Trials
5.1. H. italicum Herb
5.2. Individual Bioactive Substances
5.2.1. Phenolic Acids
5.2.3. Other Compounds
6. Recent Advances in H. italicum Studies
7. Critical Perspective on Safety and Efficacy
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Author, Year, Country | Study Type | Condition | Spec. “Italicum” Specie, Plant Parts, Compos. | Formulation, Dosage | No. of Participants, Sex | Age (y) | Duration | Main Outcome/ Results | Safety, Adverse Events |
|---|---|---|---|---|---|---|---|---|---|
| NA (Aboca S.p.A.), 2005, Italy [56] | Randomized, controlled, parallel | Post-surgery pain | Yes, flowering tops, single component | Granules for oral suspension, 2.5 g | 45, m/f | 18–65, >65 | NA | NA | NA |
| Galeone, 2012, Italy [57] | Randomized, controlled | Bacterial and non-bacterial prostatitis | No, NA, comb. with other herbs | Suppository (2 g) | 60, m | 20–50 | 28 d, follow-up after 60 and 120 d | Improvement of urinary symptoms, no difference in microbiological results | Minor side effects |
| Varney, 2013, USA [59] | Pilot, randomized, controlled, double-blind | Mental exhaustion, moderate burnout | Yes, essential oil, mixture with two other EOs | Plastic inhaler, 2 drops of H. italicum EO per inhaler | 14, m/f | 25–45, 45–65, >65 | 3 weeks | Reduction in perceived level of mental fatigue/burnout | Not reported |
| Cohen, 2013, Israel [60] | Randomized, controlled, single-blind, parallel, multi-center | Cough associated with upper respiratory tract infections | Yes, NA, comb. with other herbs and honey | Syrup, 20 mL in three doses per day | 150, m/f | 2–5 | 4 d | Reduced cough frequency, severity, better sleep quality | Safe, well tolerated |
| Canciani, 2014, Italy [61] | Randomized, double-blind, multicenter, placebo-controlled | Cough associated with upper respiratory tract infections, persisting > 7 d | Yes, NA, comb. with other herbs and honey | Syrup, 4 doses per day (5 mL each) | 102, m/f | 3–6 | 8 d | The significant decrease of cough, especially evident at night | Adverse events unrelated to the treatment |
| Di Vico, 2019, Italy [58] | Pilot, randomized, controlled | Chronic prostatitis, chronic pelvic pain syndrome | No, NA, comb. with other herbs | Suppository (2 g), 1 supp./day | 30, m | 23–49 | 1 month | Subjective pain relief, decrease in urinary symptoms | None reported |
References
- Faqi, A.S.; Yan, J.S. Chapter 30—Nonclinical safety assessment of botanical products. In A Comprehensive Guide to Toxicology in Nonclinical Drug Development, 2nd ed.; Faqi, A.S., Ed.; Academic Press: Boston, MA, USA, 2017; pp. 813–823. ISBN 978–0-12-803620-4. [Google Scholar]
- Carmona, F.; Soares Pereira, A.M. Herbal medicines: Old and new concepts, truths and misunderstandings. Rev. Bras. Farmacogn. 2013, 23, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Rivera, D.; Obon, C.; Inocencio, C.; Heinrich, M.; Verde, A.; Fajardo, J.; Llorach, R. The ethnobotanical study of local mediterranean food plants as medicinal resources in southern Spain. J. Physiol. Pharmacol. 2005, 56, 97–114. [Google Scholar]
- Akaberi, M.; Sahebkar, A.; Azizi, N.; Emami, S.A. Everlasting flowers: Phytochemistry and pharmacology of the genus Helichrysum. Ind. Crop. Prod. 2019, 138, 111471. [Google Scholar] [CrossRef]
- Perrini, R.; Alba, V.; Ruta, C.; Morone-Fortunato, I.; Blanco, A.; Montemurro, C. An evaluation of a new approach to the regeneration of Helichrysum italicum (Roth) G. Don, and the molecular characterization of the variation among sets of differently derived regenerants. Cell. Mol. Biol. Lett. 2009, 14, 377–394. [Google Scholar] [CrossRef]
- Galbany-Casals, M.; Blanco-Moreno, J.M.; Garcia-Jacas, N.; Breitwieser, I.; Smissen, R.D. Genetic variation in mediterranean Helichrysum italicum (asteraceae; gnaphalieae): Do disjunct populations of subsp. microphyllum have a common origin? Plant Biol. 2011, 13, 678–687. [Google Scholar] [CrossRef]
- Antunes Viegas, D.; Palmeira-de-Oliveira, A.; Salgueiro, L.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, R. Helichrysum italicum: From traditional use to scientific data. J. Ethnopharmacol. 2014, 151, 54–65. [Google Scholar] [CrossRef]
- Les, F.; Venditti, A.; Cásedas, G.; Frezza, C.; Guiso, M.; Sciubba, F.; Serafini, M.; Bianco, A.; Valero, M.S.; López, V. Everlasting Flower (Helichrysum stoechas moench) as a potential source of bioactive molecules with antiproliferative, antioxidant, antidiabetic and neuroprotective properties. Ind. Crop. Prod. 2017, 108, 295–302. [Google Scholar] [CrossRef]
- Pljevljakušić, D.; Bigović, D.; Janković, T.; Jelačić, S.; Šavikin, K. Sandy everlasting (Helichrysum arenarium (l.) moench): Botanical, chemical and biological properties. Front. Plant. Sci. 2018, 9, 1123. [Google Scholar] [CrossRef] [Green Version]
- Ninčević, T.; Grdiša, M.; Šatović, Z.; Jug-Dujaković, M. Helichrysum italicum (Roth) G. Don: Taxonomy, biological activity, biochemical and genetic diversity. Ind. Crop. Prod. 2019, 138, 111487. [Google Scholar] [CrossRef]
- Blažević, N.; Petričić, J.; Stanić, G.; Maleš, Ž. Variations in yields and composition of immortelle (Helichrysum italicum, Roth Guss.) essential oil from different locations and vegetation periods along adriatic coast. Acta Pharm. 1995, 45, 517–522. [Google Scholar]
- Benítez, G.; González-Tejero, M.R.; Molero-Mesa, J. Pharmaceutical ethnobotany in the western part of granada province (southern Spain): Ethnopharmacological synthesis. J. Ethnopharmacol. 2010, 129, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appendino, G.; Ottino, M.; Marquez, N.; Bianchi, F.; Giana, A.; Ballero, M.; Sterner, O.; Fiebich, B.L.; Munoz, E. Arzanol, an anti-inflammatory and anti-HIV-1 Phloroglucinol alpha-pyrone from Helichrysum italicum Ssp. microphyllum. J. Nat. Prod. 2007, 70, 608–612. [Google Scholar] [CrossRef]
- Maksimovic, S.; Tadic, V.; Skala, D.; Zizovic, I. Separation of phytochemicals from Helichrysum italicum: An analysis of different isolation techniques and biological activity of prepared extracts. Phytochemistry 2017, 138, 9–28. [Google Scholar] [CrossRef]
- Facino, R.M.; Carini, M.; Mariani, M.; Cipriani, C. Anti erythematous and photoprotective activities in guinea pigs and in man of topically applied flavonoids from Helichrysum italicum G. Don. Acta Ther. 1988, 14, 323–346. [Google Scholar]
- Facino, R.M.; Carini, M.; Franzoi, L.; Pirola, O.; Bosisio, E. Phytochemical characterization and radical scavenger activity of flavonoids from Helichrysum italicum G. Don (compositae). Pharmacol. Res. 1990, 22, 709–721. [Google Scholar] [CrossRef]
- Pietta, P.; Mauri, P.; Gardana, C.; Facino, R.M.; Carini, M. High-performance liquid chromatographic determination of flavonoid glucosides from Helichrysum italicum. J. Chromatogr. A 1991, 537, 449–452. [Google Scholar] [CrossRef]
- Pietta, P.; Mauri, P.; Facino, R.M.; Carini, M. Analysis of flavonoids by MECC with ultraviolet diode array detection. J. Pharm. Biomed. Anal. 1992, 10, 1041–1045. [Google Scholar] [CrossRef]
- Zapesochnaya, G.G.; Dzyadevich, T.V.; Karasartov, B.S. Phenolic compounds of Helichrysum italicum. Chem. Nat. Compd. 1990, 26, 342–343. [Google Scholar] [CrossRef]
- Zapesochnaya, G.G.; Kurkin, V.A.; Kudryavtseva, T.V.; Karasartov, B.S.; Cholponbaev, K.S.; Tyukavkina, N.A.; Ruchkin, V.E. Dicaffeolyquinic acids from Helichrysum italicum and achillea cartilaginea. Chem. Nat. Compd. 1992, 28, 40–44. [Google Scholar] [CrossRef]
- Nostro, A.; Bisignano, G.; Angela Cannatelli, M.; Crisafi, G.; Paola Germanò, M.; Alonzo, V. Effects of Helichrysum italicum extract on growth and enzymatic activity of staphylococcus aureus. Int. J. Antimicrob. Agents 2001, 17, 517–520. [Google Scholar] [CrossRef]
- Nostro, A.; Cannatelli, M.A.; Musolino, A.D.; Procopio, F.; Alonzo, V. Helichrysum italicum extract interferes with the production of enterotoxins by staphylococcus aureus. Lett. Appl. Microbiol. 2002, 35, 181–184. [Google Scholar] [CrossRef] [Green Version]
- Nostro, A.; Cannatelli, M.A.; Marino, A.; Picerno, I.; Pizzimenti, F.C.; Scoglio, M.E.; Spataro, P. Evaluation of antiherpesvirus-1 and genotoxic activities of Helichrysum italicum extract. New Microbiol. 2003, 26, 125–128. [Google Scholar] [PubMed]
- Nostro, A.; Cannatelli, M.A.; Crisafi, G.; Musolino, A.D.; Procopio, F.; Alonzo, V. Modifications of hydrophobicity, In Vitro adherence and cellular aggregation of streptococcus mutans by Helichrysum italicum extract. Lett. Appl. Microbiol. 2004, 38, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Sala, A.; Recio, M.C.; Giner, R.M.; Máñez, S.; Ríos, J.L. New acetophenone glucosides isolated from extracts of Helichrysum italicum with antiinflammatory activity. J. Nat. Prod. 2001, 64, 1360–1362. [Google Scholar] [CrossRef]
- Sala, A.; Recio, M.; Giner, R.M.; Máñez, S.; Tournier, H.; Schinella, G.; Ríos, J.-L. Anti-inflammatory and antioxidant properties of Helichrysum italicum. J. Pharm. Pharmacol. 2002, 54, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Sala, A.; Recio, M.C.; Schinella, G.R.; Máñez, S.; Giner, R.M.; Ríos, J.-L. A new dual inhibitor of arachidonate metabolism isolated from Helichrysum italicum. Eur. J. Pharmacol. 2003, 460, 219–226. [Google Scholar] [CrossRef]
- Sala, A.; Recio, M.C.; Schinella, G.R.; Máñez, S.; Giner, R.M.; Cerdá-Nicolás, M.; Ríos, J.-L. Assessment of the anti-inflammatory activity and free radical scavenger activity of tiliroside. Eur. J. Pharmacol. 2003, 461, 53–61. [Google Scholar] [CrossRef]
- Schinella, G.R.; Tournier, H.A.; Prieto, J.M.; de Buschiazzo, P.M.; Ríos, J.L. Antioxidant activity of anti-inflammatory plant extracts. Life Sci. 2002, 70, 1023–1033. [Google Scholar] [CrossRef]
- Schinella, G.R.; Tournier, H.A.; Máñez, S.; de Buschiazzo, P.M.; del Carmen Recio, M.; Ríos, J.L. Tiliroside and gnaphaliin inhibit human low density lipoprotein oxidation. Fitoterapia 2007, 78, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wollenweber, E.; Christ, M.; Dunstan, R.H.; Roitman, J.N.; Stevens, J.F. Exudate Flavonoids in some gnaphalieae and inuleae (asteraceae). Z. Nat. C 2005, 60, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.; Deiana, M.; Atzeri, A.; Corona, G.; Incani, A.; Melis, M.P.; Appendino, G.; Dessì, M.A. Evaluation of the antioxidant and cytotoxic activity of arzanol, a prenylated α-pyrone–phloroglucinol etherodimer from Helichrysum italicum subsp.microphyllum. Chem. Biol. Interact. 2007, 165, 117–126. [Google Scholar] [CrossRef]
- Rosa, A.; Pollastro, F.; Atzeri, A.; Appendino, G.; Melis, M.P.; Deiana, M.; Incani, A.; Loru, D.; Dessì, M.A. Protective role of arzanol against lipid peroxidation in biological systems. Chem. Phys. Lipids 2011, 164, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Koeberle, A.; Dehm, F.; Pollastro, F.; Appendino, G.; Northoff, H.; Rossi, A.; Sautebin, L.; Werz, O. Arzanol, a prenylated heterodimeric phloroglucinyl pyrone, inhibits eicosanoid biosynthesis and exhibits anti-inflammatory efficacy In Vivo. Biochem. Pharmacol. 2011, 81, 259–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Abrosca, B.; Buommino, E.; D’Angelo, G.; Coretti, L.; Scognamiglio, M.; Severino, V.; Pacifico, S.; Donnarumma, G.; Fiorentino, A. Spectroscopic identification and anti-biofilm properties of polar metabolites from the medicinal plant Helichrysum italicum against pseudomonas aeruginosa. Bioorg. Med. Chem. 2013, 21, 7038–7046. [Google Scholar] [CrossRef]
- Taglialatela-Scafati, O.; Pollastro, F.; Chianese, G.; Minassi, A.; Gibbons, S.; Arunotayanun, W.; Mabebie, B.; Ballero, M.; Appendino, G. Antimicrobial phenolics and unusual glycerides from Helichrysum italicum subsp. microphyllum. J. Nat. Prod. 2013, 76, 346–353. [Google Scholar] [CrossRef]
- de la Garza, A.L.; Etxeberria, U.; Lostao, M.P.; San Román, B.; Barrenetxe, J.; Martínez, J.A.; Milagro, F.I. Helichrysum and grapefruit extracts inhibit carbohydrate digestion and absorption, improving postprandial glucose levels and hyperinsulinemia in rats. J. Agric. Food Chem. 2013, 61, 12012–12019. [Google Scholar] [CrossRef] [PubMed]
- Rigano, D.; Formisano, C.; Senatore, F.; Piacente, S.; Pagano, E.; Capasso, R.; Borrelli, F.; Izzo, A.A. Intestinal antispasmodic effects of Helichrysum italicum (Roth) Don ssp. italicum and chemical identification of the active ingredients. J Ethnopharmacol. 2013, 150, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Rigano, D.; Formisano, C.; Pagano, E.; Senatore, F.; Piacente, S.; Masullo, M.; Capasso, R.; Izzo, A.A.; Borrelli, F. A new acetophenone derivative from flowers of Helichrysum italicum (Roth) Don ssp. italicum. Fitoterapia 2014, 99, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Mari, A.; Napolitano, A.; Masullo, M.; Pizza, C.; Piacente, S. Identification and quantitative determination of the polar constituents in Helichrysum italicum flowers and derived food supplements. J Pharm. Biomed. Anal. 2014, 96, 249–255. [Google Scholar] [CrossRef]
- Pereira, C.G.; Barreira, L.; Bijttebier, S.; Pieters, L.; Neves, V.; Rodrigues, M.; Rivas, R.; Varela, J.; Custodio, L. Chemical profiling of infusions and decoctions of Helichrysum italicum Subsp. picardii by UHPLC-PDA-MS and In Vitro biological activities comparatively with green tea (Camellia sinensis) and rooibos tisane (Aspalathus linearis). J. Pharm. Biomed. Anal. 2017, 145, 593–603. [Google Scholar] [CrossRef]
- Kothavade, P.S.; Nagmoti, D.M.; Bulani, V.D.; Juvekar, A.R. Arzanol, a potent MPGES-1 inhibitor: Novel anti-inflammatory agent. Sci. World J. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Espíndola, K.M.M.; Ferreira, R.G.; Narvaez, L.E.M.; Silva Rosario, A.C.R.; da Silva, A.H.M.; Silva, A.G.B.; Vieira, A.P.O.; Monteiro, M.C. Chemical and pharmacological aspects of caffeic acid and its activity in hepatocarcinoma. Front. Oncol. 2019, 9, 541. [Google Scholar] [CrossRef] [Green Version]
- Miao, M.; Xiang, L. Chapter Three—Pharmacological action and potential targets of chlorogenic acid. In Advances in Pharmacology; Du, G., Ed.; Pharmacological Advances in Natural Product Drug Discovery; Academic Press: Boston, MA, USA, 2020; Volume 87, pp. 71–88. [Google Scholar]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of naringenin: A review of clinical trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mlala, S.; Oyedeji, A.O.; Gondwe, M.; Oyedeji, O.O. Ursolic acid and its derivatives as bioactive agents. Molecules 2019, 24, 2751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santini, L. Rassegna clinico-statistica sulle proprietà terapeutiche dell élicrisio. Minerva Med. 1952, 43, 714–719. [Google Scholar] [PubMed]
- Benigni, R.; Capra, C.; Cattorini, P. Piante Medicinali: Chimica, Farmacologia e Terapia; Inverni & Della Beffa: Milano, Italy, 1962. [Google Scholar]
- Vannini, C. Atti II Seminario Internazionale Piante Medicinali e Medicina Tradizionale; Maremagnum: Città di Castello, Italy, 1981. [Google Scholar]
- Campanini, E. Helichrysum angustifolium: Esperienze cliniche sulla psoriasi. Acta Phytother. 1995, 1, 8–10. [Google Scholar]
- Appendino, G.; Taglialatela-Scafati, O.; Minassi, A.; Pollastro, F.; Ballero, M.; Maxia, A.; Sanna, C. Helichrysum italicum: The sleeping giant of mediterranean herbal medicine. Herb. J. Am. Bot. Counc. 2015, 105, 34–45. [Google Scholar]
- Voinchet, V.; Giraud-Robert, A.-M. Utilisation de l’huile essentielle d’hélichryse italienne et de l’huile végétale de rose musquée après intervention de chirurgie plastique réparatrice et esthétique. Phytothérapie 2007, 5, 67–72. [Google Scholar] [CrossRef]
- Granger, C.; Brown, A.; Aladren, S.; Narda, M. Night cream containing melatonin, carnosine and Helichrysum italicum extract helps reduce skin reactivity and signs of photodamage: Ex Vivo and clinical studies. Dermatol. Ther. 2020, 10, 1315–1329. [Google Scholar] [CrossRef] [PubMed]
- Randomised Parallel Study against Placebo for the Determination of Efficacy of Liophilised Salvia Officinalis and Helichrysum italicum in Pain Treatment from Post-Surgery Pain in 45 Male or Female Patients. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2005-000958-71/IT/ (accessed on 30 December 2020).
- Galeone, G.; Spadavecchia, R.; Balducci, M.T.; Pagliarulo, V. The role of proxelan in the treatment of chronic prostatitis. Results of a randomized trial. Minerva Urol. Nefrol. 2012, 64, 135–141. [Google Scholar] [PubMed]
- Di Vico, T.; Durante, J.; Polito, C.; Tognarelli, A.; Canale, D.; Caglieresi, C.; Morelli, G.; Bartoletti, R. Pumpkin seeds, centella asiatica, boswellia, Helichrysum, acetate vitamin e, melaleuca alternifolia and hyaluronic acid phytocomplex monotherapy effects in patients with chronic pelvic pain syndrome. Minerva Urol. Nefrol. 2020, 72, 236–242. [Google Scholar] [CrossRef]
- Varney, E.; Buckle, J. Effect of inhaled essential oils on mental exhaustion and moderate burnout: A small pilot study. J. Altern. Complement. Med. 2013, 19, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Cohen, H.A.; Blau, H.; Gur, S.; Moshe, H.; Ran, B. Randomized, single-blinded study to evaluate the efficacy of grintuss and mucolit pediatric syrups for cough due to upper respiratory tract infection. Pediatric Pulmonol. 2015, 50, S53–S83. [Google Scholar] [CrossRef]
- Canciani, M.; Murgia, V.; Caimmi, D.; Anapurapu, S.; Licari, A.; Marseglia, G.L. Efficacy of Grintuss® pediatric syrup in treating cough in children: A randomized, multicenter, double blind, placebo-controlled clinical trial. Ital. J. Pediatr. 2014, 40, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calapai, G. Randomized, Controlled, Double Blind Clinical Study on the Assessment of Efficacy, Safety and Palatability of “KalobaTuss® Children”, a Syrup for the Treatment of Cough in Pediatric Age (3–6 Years). Available online: https://clinicaltrials.gov/ct2/show/NCT04073251 (accessed on 30 December 2020).
- Sendker, J.; Sheridan, H. Composition and quality control of herbal medicines. In Toxicology of Herbal Products; Pelkonen, O., Duez, P., Vuorela, P.M., Vuorela, H., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 29–65. ISBN 978-3-319-43806-1. [Google Scholar]
- 2-Principles of herbal pharmacology. In Principles and Practice of Phytotherapy, 2nd ed.; Bone, K.; Mills, S. (Eds.) Churchill Livingstone: Saint Louis, MO, USA, 2013; pp. 17–82. ISBN 978-0-443-06992-5. [Google Scholar]
- Karača, S.; Trifković, K.; Bušić, A.; Đorđević, V.; Belščak-Cvitanović, A.; Cebin, A.V.; Bugarski, B.; Komes, D. The functional potential of immortelle (Helichrysum italicum) based edible films reinforced with proteins and hydrogel particles. LWT 2019, 99, 387–395. [Google Scholar] [CrossRef]
- Kramberger, K.; Barlič-Maganja, D.; Bandelj, D.; Baruca Arbeiter, A.; Peeters, K.; Miklavčič Višnjevec, A.; Jenko Pražnikar, Z. HPLC-DAD-ESI-QTOF-MS determination of bioactive compounds and antioxidant activity comparison of the hydroalcoholic and water extracts from two Helichrysum italicum species. Metabolites 2020, 10, 403. [Google Scholar] [CrossRef] [PubMed]
- Records in Cochrane Library on Chlorogenic Acid. Available online: https://www.cochranelibrary.com/advanced-search?q=chlorogenic%20acid&t=1 (accessed on 2 May 2021).
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Records in Cochrane Library on Caffeic Acid. Available online: https://www.cochranelibrary.com/advanced-search?q=caffeic%20acid&t=1 (accessed on 2 May 2021).
- Phase Ⅱ Study of Pinocembrin Injection to Treat Ischemic Stroke—Randomized, Double-Blind, Placebo-Controlled, Multicenter Study. Available online: https://clinicaltrials.gov/ct2/show/NCT02059785 (accessed on 29 April 2021).
- Cao, G.; Ying, P.; Yan, B.; Xue, W.; Li, K.; Shi, A.; Sun, T.; Yan, J.; Hu, X. Pharmacokinetics, safety, and tolerability of single and multiple-doses of pinocembrin injection administered intravenously in healthy subjects. J. Ethnopharmacol. 2015, 168, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Records in Cochrane Library on Quercetin. Available online: https://www.cochranelibrary.com/advanced-search (accessed on 2 May 2021).
- Records in Cochrane Library on Naringenin. Available online: https://www.cochranelibrary.com/advanced-search?q=naringenin&t=1 (accessed on 2 May 2021).
- Rebello, C.J.; Beyl, R.A.; Lertora, J.J.L.; Greenway, F.L.; Ravussin, E.; Ribnicky, D.M.; Poulev, A.; Kennedy, B.J.; Castro, H.F.; Campagna, S.R.; et al. Safety and pharmacokinetics of naringenin: A randomized, controlled, single-ascending-dose clinical trial. Diabetes Obes. Metab. 2020, 22, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Records in Cochrane Library on Luteolin. Available online: https://www.cochranelibrary.com/advanced-search?q=luteolin&t=1 (accessed on 2 May 2021).
- Qian, Z.; Wang, X.; Song, Z.; Zhang, H.; Zhou, S.; Zhao, J.; Wang, H. A phase I trial to evaluate the multiple-dose safety and antitumor activity of ursolic acid liposomes in subjects with advanced solid tumors. BioMed Res. Int. 2015, 2015, e809714. [Google Scholar] [CrossRef] [Green Version]
- Dzamic, A.M.; Mileski, K.S.; Ciric, A.D.; Ristic, M.S.; Sokovic, M.D.; Marin, P.D. Essential oil composition, antioxidant and antimicrobial properties of essential oil and deodorized extracts of Helichrysum italicum (Roth) G. Don. J. Essent. Oil Bear. Plants 2019, 22, 493–503. [Google Scholar] [CrossRef]
- Addis, R.; Cruciani, S.; Santaniello, S.; Bellu, E.; Sarais, G.; Ventura, C.; Maioli, M.; Pintore, G. Fibroblast proliferation and migration in wound healing by phytochemicals: Evidence for a novel synergic outcome. Int. J. Med. Sci. 2020, 17, 1030–1042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pilić, Z.; Martinović, I. Effect of Helichrysum italicum on the corrosion of copper in simulated acid rain solution. Chem. Biochem. Eng. Q. 2019, 33, 449–457. [Google Scholar] [CrossRef]
- Eksi, M.; Sevgi, O.; Akburak, S.; Yurtseven, H.; Esin, İ. Assessment of recycled or locally available materials as green roof substrates. Ecol. Eng. 2020, 156, 105966. [Google Scholar] [CrossRef]
- Kramberger, K.; Pražnikar, Z.J.; Baruca Arbeiter, A.; Petelin, A.; Bandelj, D.; Kenig, S. A comparative study of the antioxidative effects of Helichrysum italicum and Helichrysum arenarium infusions. Antioxidants 2021, 10, 380. [Google Scholar] [CrossRef] [PubMed]
- Baruca Arbeiter, A.; Hladnik, M.; Jakše, J.; Bandelj, D. First set of microsatellite markers for immortelle (Helichrysum italicum (Roth) G. Don): A step towards the selection of the most promising genotypes for cultivation. Ind. Crop. Prod. 2021, 162, 113298. [Google Scholar] [CrossRef]
- Foti, C.; Guida, S.; Antelmi, A.; Romita, P.; Corazza, M. Allergic contact dermatitis caused by Helichrysum italicum contained in an emollient cream. Contact Dermat. 2013, 69, 62–63. [Google Scholar] [CrossRef]
- Appendino, G. Helichrysum italicum: Back to medicine from the tinsel of luxury. Planta Med. 2015, 81, PL_09. [Google Scholar] [CrossRef]
- Biagi, M.; Pecorari, R.; Appendino, G.; Miraldi, E.; Magnano, A.R.; Governa, P.; Cettolin, G.; Giachetti, D. Herbal products in Italy: The thin line between phytotherapy, nutrition and parapharmaceuticals; a normative overview of the fastest growing market in Europe. Pharmaceuticals 2016, 9, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thakkar, S.; Anklam, E.; Xu, A.; Ulberth, F.; Li, J.; Li, B.; Hugas, M.; Sarma, N.; Crerar, S.; Swift, S.; et al. Regulatory landscape of dietary supplements and herbal medicines from a global perspective. Regul. Toxicol. Pharmacol. 2020, 114, 104647. [Google Scholar] [CrossRef] [PubMed]


| Compound Name | Chemical Structure 1 | Extraction Solvent 2 | Extraction Yield (from Starting Plant Material) 3 | Main Bioactivity 4 |
|---|---|---|---|---|
| Caffeic acid | 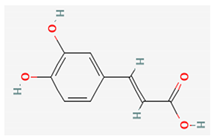 | Methanol | 0.0067% (NS) [38] 0.00042% (flowers) [41] 0.0057–0.015% (aerial parts or flowers) [42] | Antioxidant, anti-inflammatory and anticancer activity [44] |
| Chlorogenic acid |  | Methanol | 0.104% (NS) [38] 0.045% (flowers) [41] 0.52–0.77% (aerial parts or flowers) [42] | Antibacterial [36], antiviral, antioxidant, anti-inflammatory, anticardiovascular, hypoglycemic, and anticancer activity [45] |
| Pinocembrin | 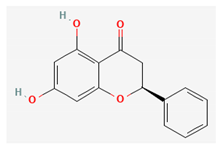 | Acetone [32] Methanol [27] | NA (NS) [32] NA (aerial parts) [27] | Anti-inflammatory [29] |
| Quercetin | 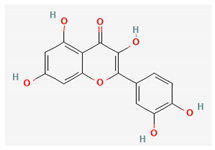 | Methanol | 0.015% (flowers) [41] 0.001–0.0015% (aerial parts or flowers) [42] | Antioxidant, anti-inflammatory, antimicrobial, cardioprotective, gastroprotective and anticancer activity [46] |
| Naringenin | 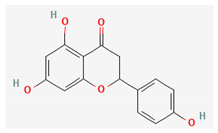 | Methanol | 0.023% (NS) [38] | Antioxidant, antitumor, antiviral, antibacterial, anti-inflammatory, and cardioprotective activity [47] |
| Gnaphaliin |  | Methanol | 0.03% (aerial parts) [31] | Anti-inflammatory [29] |
| Luteolin | 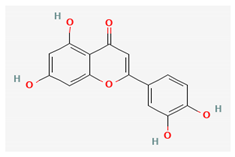 | Ethanol | NA (flowers) [16] | Antiviral [24] |
| Tiliroside |  | Methanol | 0.0063% (aerial parts) [31] 0.0015% (flowers) [41] | Anti-inflammatory and antioxidant [29] |
| Arzanol |  | Acetone | 0.078% (aerial parts) [14] 0.064% (aerial parts and flowers) [33] 0.32% (aerial parts) [37] | Anti-inflammatory [14,35], antiviral [14], antioxidant [33] and antibacterial [37] |
| Ursolic acid |  | Acetone | 0.40% (aerial parts) [37] | Anti-inflammatory, anticancer, antidiabetic, antioxidant and antibacterial effects [48] |
| Author and Date [Reference] | Observation |
|---|---|
| Santini, 1930s [49] | H. italicum decoction administered to patients suffering from bronchitis and asthmatic cough led to improvement of their respiratory condition and unrelated conditions such as psoriasis and arthritis. |
| Santini, 1930–1950 [49] | Two decades of clinical observations led to the conclusion that clinical activity of a decoction and syrup from H. italicum is similar to that of cortisone. Aerosolized decoction of H. italicum showed positive results in the use for allergic rhinitis. |
| Santini, 1950s [49] | Two independent clinical studies on patient with psoriasis confirmed beneficial effects of H. italicum treatment. |
| Benigni, 1950s [50] | A series of clinical studies in various Italian centers substantially confirmed the findings of Santini, showing that “Fraction H” produced using an organic solvent, could, to varying degrees, replace corticosteroids in many of their uses and their adverse side effects were thus avoided. |
| Vannini, 1981 [51] | H. italicum decoction was found to be highly efficacious in treatment of tracheo-bronchitis in a small clinical study in children. |
| Facino, 1988 [16] | Flavonoid fraction was applied to humans 10 min before or after exposure to UVB radiation to evaluate their photoprotective and anti-erythematous activities, respectively. The onset of the erythematous response was completely prevented and a sun protection factor of approximately 5 was provided. |
| Campanini, 1995 [52] | Three weeks of treatment with 5% H. italicum decoction led to improvement of psoriasis in all participants, with relapses observed within two months post-treatment. |
| Voinchet and Giraud-Robert, 2007 [54] | Two drops of H. italicum essential oil, orally twice a day for 10 d, followed by its topical application for 2–3 months in post-operative scars of patients submitted to a plastic surgery of the thorax led to a reduction of local inflammation, edema, bruises, and hematomas. |
| Granger, 2020 [55] | Night cream containing melatonin, carnosine, and H. italicum extract reduced skin damage caused by environmental factors and its nightly use could improve clinical signs of aging with additional skin calming benefits. Hydration and trans-epidermal water loss values were improved within 1 h of use. Wrinkle counts were reduced by up to 18.9%, and brown and UV spot numbers by 5.5% and 13.2%, respectively. Lactic acid-induced stinging was significantly reduced within 7 d of use, with 86.7% of subjects reporting that their skin felt calmer. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kramberger, K.; Kenig, S.; Jenko Pražnikar, Z.; Kočevar Glavač, N.; Barlič-Maganja, D. A Review and Evaluation of the Data Supporting Internal Use of Helichrysum italicum. Plants 2021, 10, 1738. https://doi.org/10.3390/plants10081738
Kramberger K, Kenig S, Jenko Pražnikar Z, Kočevar Glavač N, Barlič-Maganja D. A Review and Evaluation of the Data Supporting Internal Use of Helichrysum italicum. Plants. 2021; 10(8):1738. https://doi.org/10.3390/plants10081738
Chicago/Turabian StyleKramberger, Katja, Saša Kenig, Zala Jenko Pražnikar, Nina Kočevar Glavač, and Darja Barlič-Maganja. 2021. "A Review and Evaluation of the Data Supporting Internal Use of Helichrysum italicum" Plants 10, no. 8: 1738. https://doi.org/10.3390/plants10081738
APA StyleKramberger, K., Kenig, S., Jenko Pražnikar, Z., Kočevar Glavač, N., & Barlič-Maganja, D. (2021). A Review and Evaluation of the Data Supporting Internal Use of Helichrysum italicum. Plants, 10(8), 1738. https://doi.org/10.3390/plants10081738






