Nonedible Vegetable Oil-Based Polyols in Anticorrosive and Antimicrobial Polyurethane Coatings
Abstract
:1. Introduction
2. Diisocyanates and Polyols for Polyurethanes
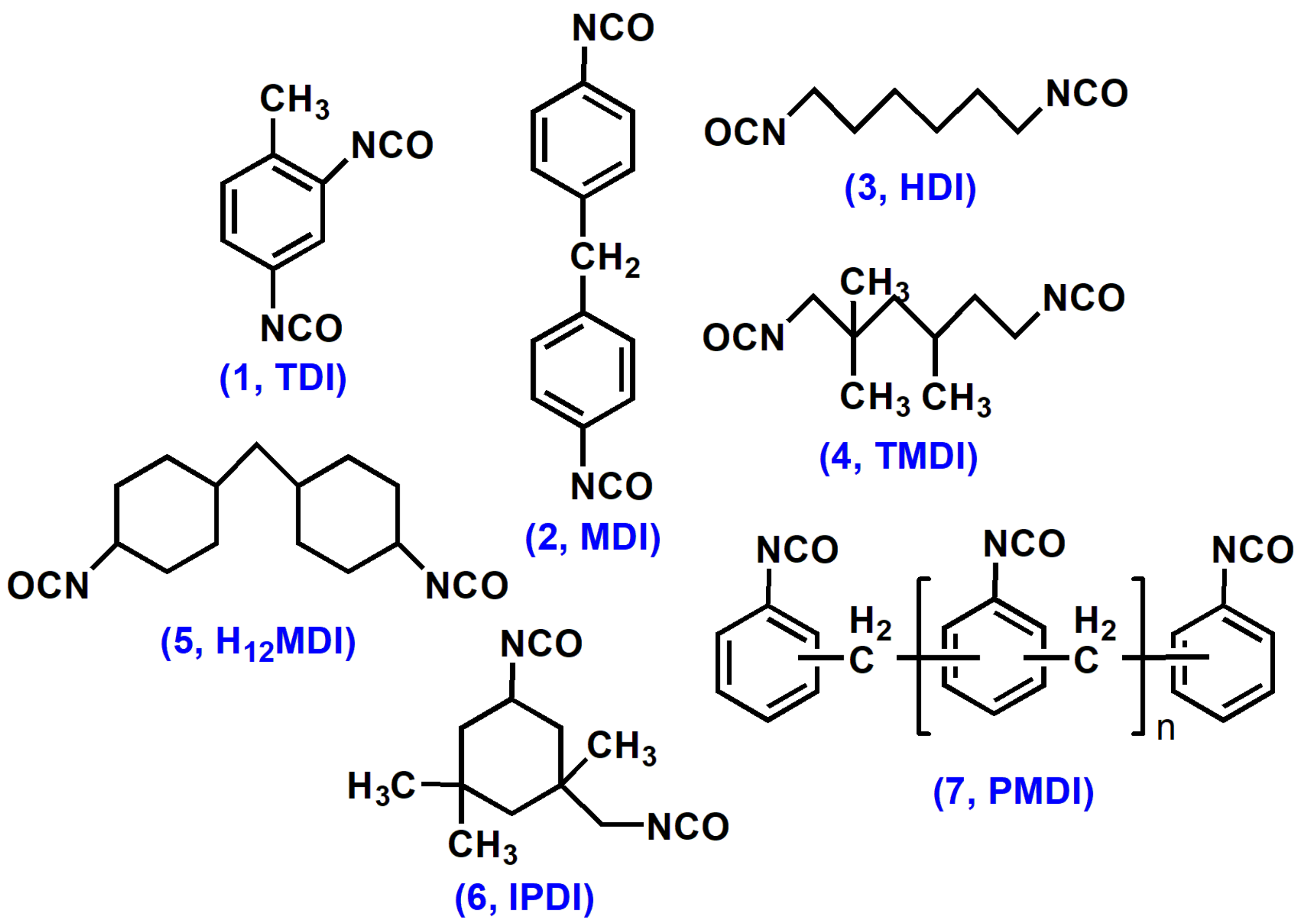
2.1. Diisocyanates for Polyurethanes
2.2. Conventional Polyols for Polyurethanes
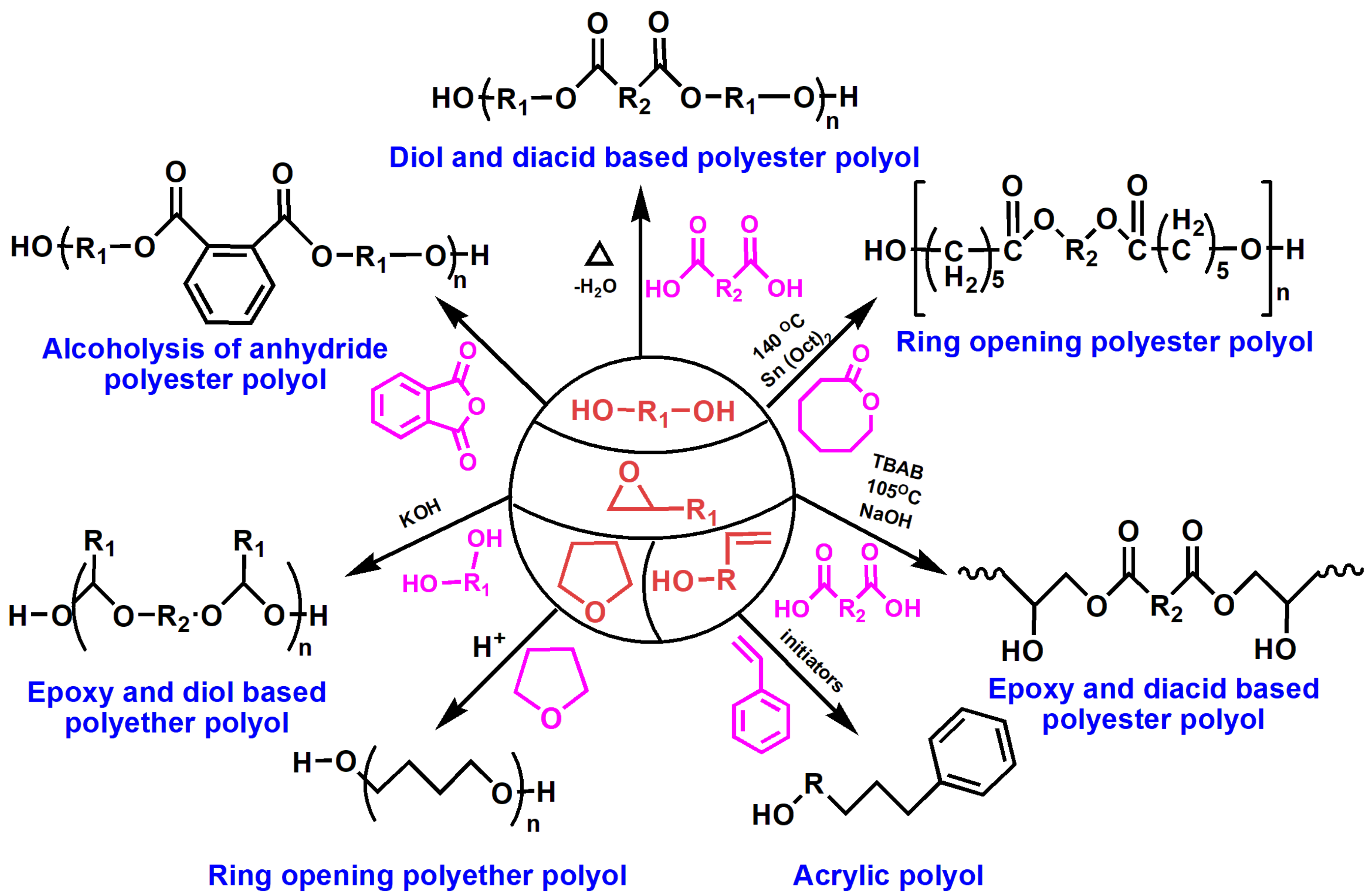
2.3. Polyols from Renewable Resources for Polyurethanes
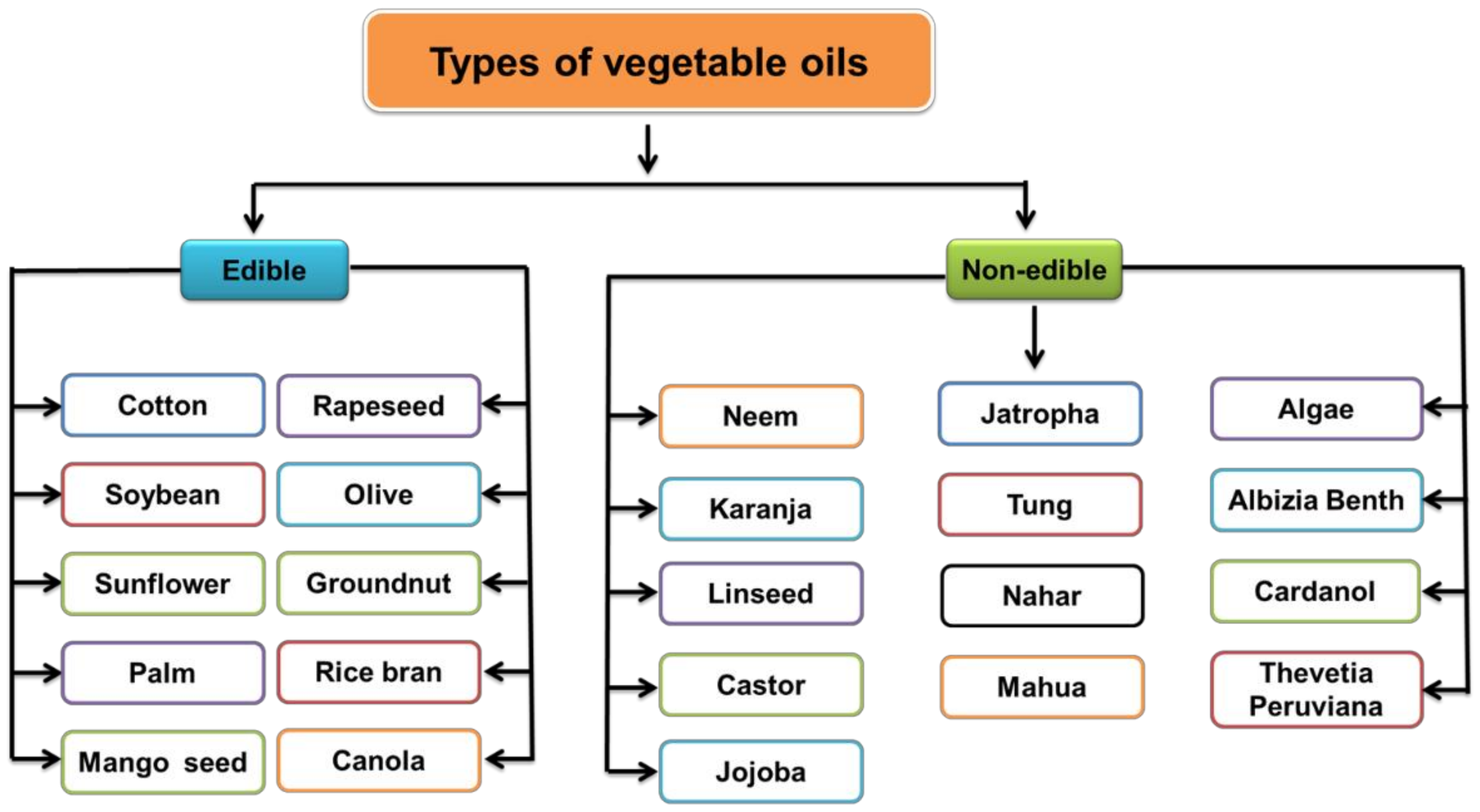
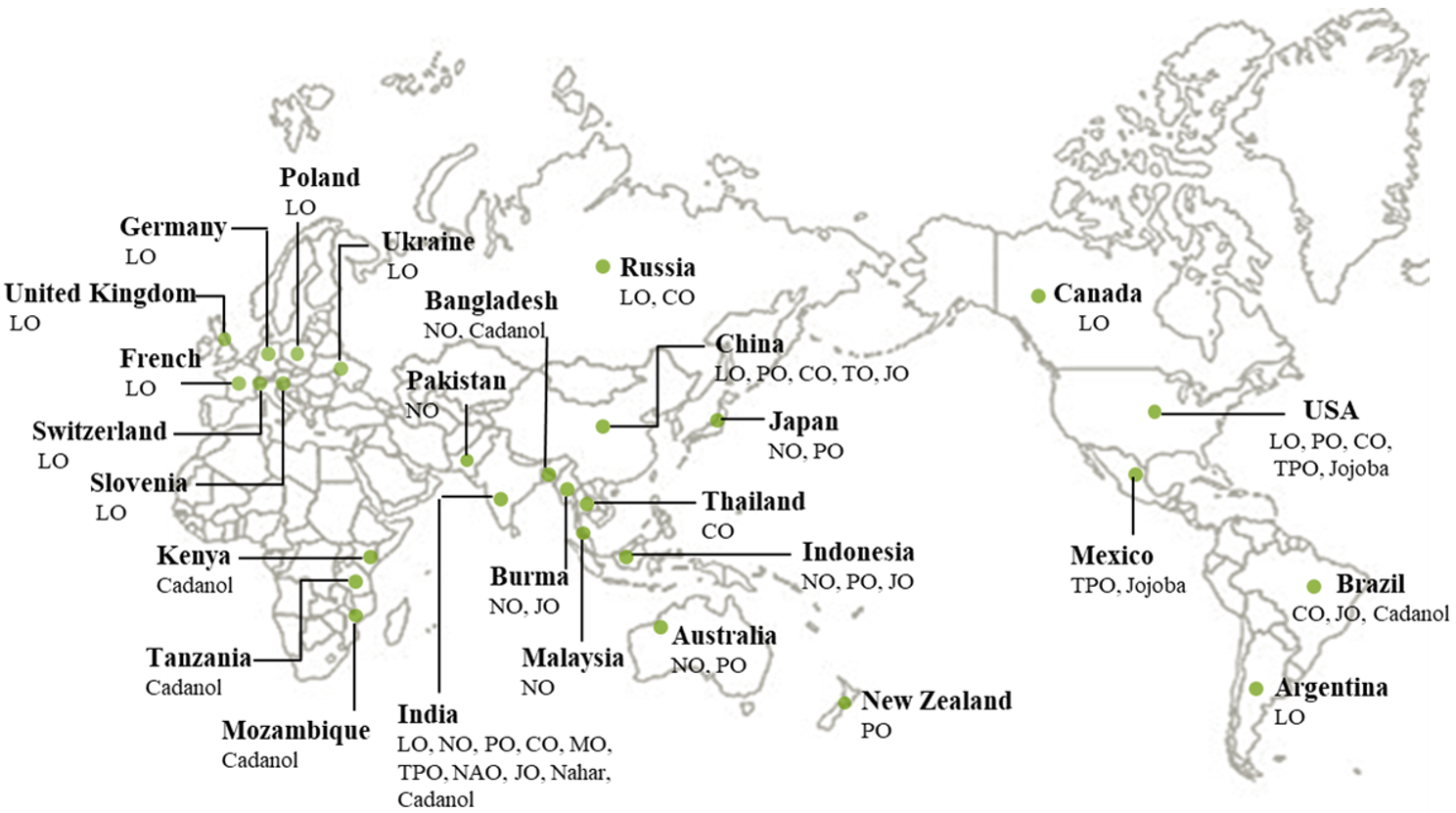
3. Nonedible Vegetable Oils for Polyols
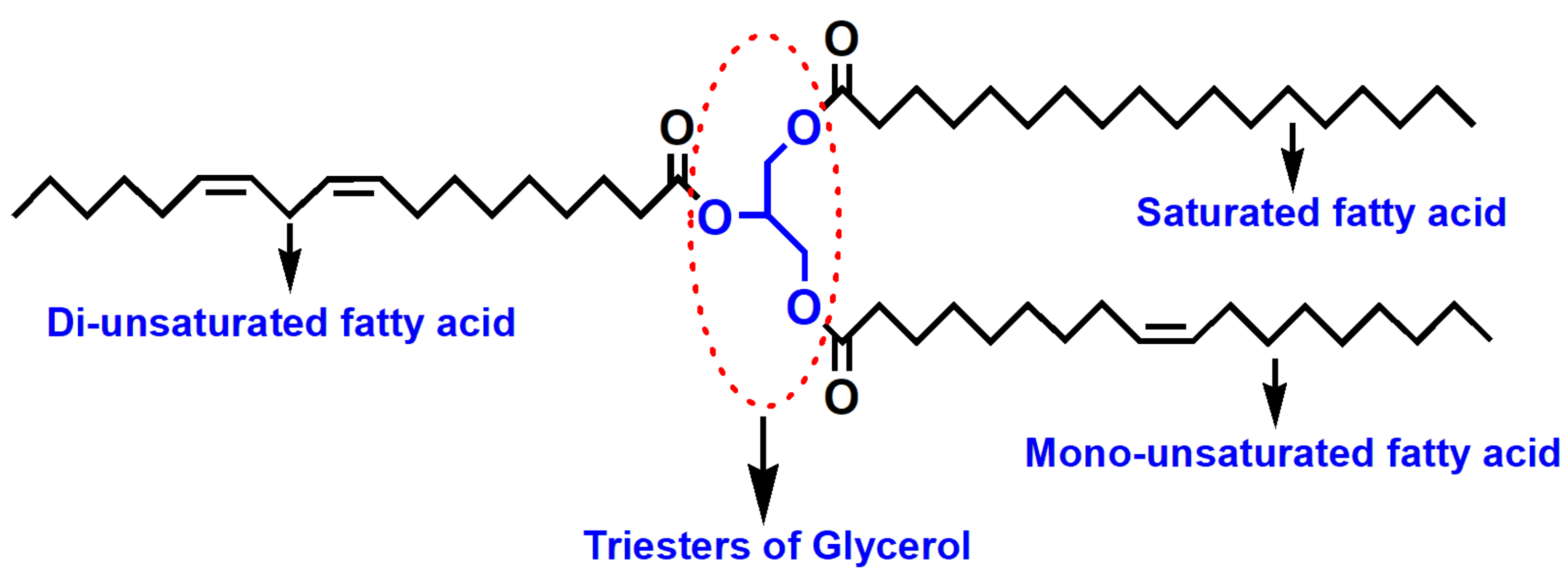
3.1. Nonedible Vegetable Oils
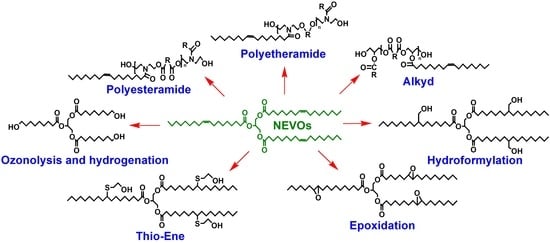
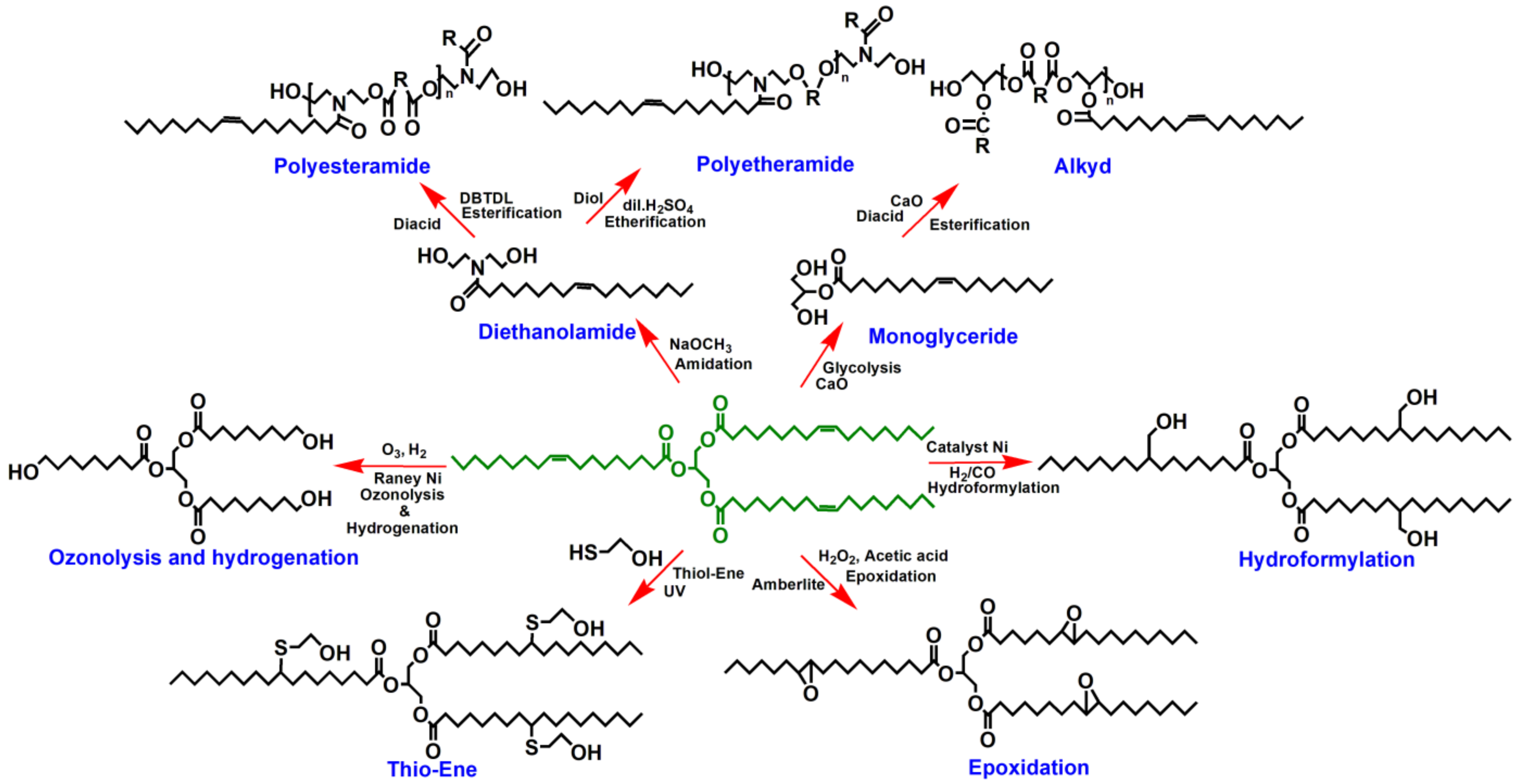
3.2. Chemical Transformation of Nonedible Vegetable Oils to Polyols
4. Nonedible Vegetable Oil-Based Polyurethane Coatings
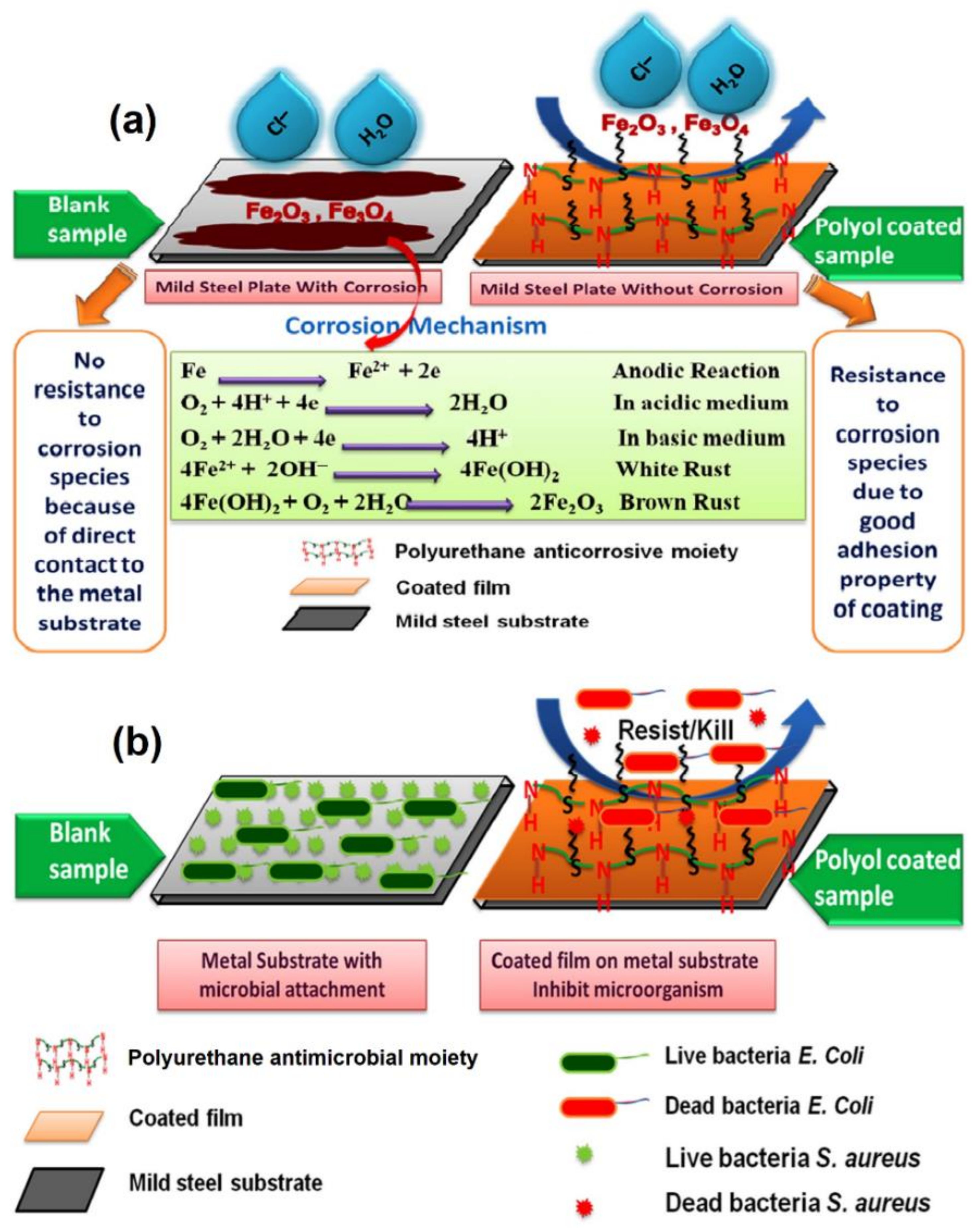
4.1. Nonedible Vegetable Oil-Based Anticorrosive Polyurethane Coatings
4.1.1. Anticorrosive Polyurethane Coatings: Adhesion
4.1.2. Anticorrosive Polyurethane Coatings: Crosslinking
4.1.3. Anticorrosive Polyurethane Coatings: Organic-Inorganic Hybrids
4.1.4. Anticorrosive Polyurethane Coatings with Additives: Nanocomposites
4.1.5. Anticorrosive Polyurethane Coatings with Additives: Microcapsules
4.2. Nonedible Vegetable Oil-Based Antimicrobial Polyurethane Coatings
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABO | Albizia benth seed oil |
| Ag | silver |
| AO | algae oil |
| CO | castor oil |
| EDTA | ethylenediaminetetraacetic acid |
| GOx | graphene oxide |
| HAP | hydroxyapatite |
| JO | jatropha oil |
| KO | karanja oil |
| LO | linseed oil |
| MO | mahua oil |
| NAO | Nahar seed oil |
| NEVO | nonedible vegetable oil |
| NO | neem seed oil |
| NP | nanoparticle |
| PEsA | polyesteramide |
| PEtA | polyetheramide |
| PO | pongamia glabra seed oils |
| PU | polyurethane |
| PUFA | poly(urethane fatty amide) |
| TDI | toluene diisocyanate |
| TEOS | tetraethoxyorthosilicate |
| TO | tung oil |
| TPO | thevetia peruviana seed oil |
| VO | vegetable oil |
References
- Petrović, Z.S. Polyurethanes from vegetable oils. Polym. Rev. 2008, 48, 109–155. [Google Scholar] [CrossRef]
- Szycher, M. Szycher’s Handbook of Polyurethanes, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar] [CrossRef]
- Rokicki, G.; Parzuchowski, P.G.; Mazurek, M. Non-isocyanate polyurethanes: Synthesis, properties, and applications. Polym. Adv. Technol. 2015, 26, 707–761. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Raju, K.V.S.N. Structural engineering of polyurethane coatings for high performance applications. Prog. Polym. Sci. 2007, 32, 352–418. [Google Scholar] [CrossRef]
- Noreen, A.; Zia, K.M.; Zuber, M.; Tabasum, S.; Zahoor, A.F. Bio-based polyurethane: An efficient and environment friendly coating systems: A review. Prog. Org. Coat. 2016, 91, 25–32. [Google Scholar] [CrossRef]
- Gite, V.; Rajput, S.D.; Yemul, O.S. Advances in polyurethane coating technologies. Chem. Wkly. 2012, 58, 201–208. [Google Scholar]
- Datta, J.; Kasprzyk, P. Thermoplastic polyurethanes derived from petrochemical or renewable resources: A comprehensive review. Polym. Eng. Sci. 2018, 58, E14–E35. [Google Scholar] [CrossRef] [Green Version]
- Hojabri, L.; Kong, X.; Narine, S.S. Fatty acid-derived diisocyanate and biobased polyurethane produced from vegetable oil: Synthesis, polymerization, and characterization. Biomacromolecules 2009, 10, 884–891. [Google Scholar] [CrossRef] [PubMed]
- Hojabri, L.; Kong, X.; Narine, S.S. Novel long chain unsaturated diisocyanate from fatty acid: Synthesis, characterization, and application in bio-based polyurethane. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 3302–3310. [Google Scholar] [CrossRef]
- Sunder, A.; Türk, H.; Haag, R.; Frey, H. Copolymers of glycidol and glycidyl ethers: Design of branched polyether polyols by combination of latent cyclic AB2 and ABR monomers. Macromolecules 2000, 33, 7682–7692. [Google Scholar] [CrossRef]
- Herrmann, A.; Kaufmann, L.; Dey, P.; Haag, R.; Schedler, U. Bioorthogonal in situ hydrogels based on polyether polyols for new biosensor materials with high sensitivity. ACS Appl. Mater. Interfaces 2018, 10, 11382–11390. [Google Scholar] [CrossRef]
- Sunder, A.; Mülhaupt, R.; Haag, R.; Frey, H. Hyperbranched polyether polyols: A modular approach to complex polymer architectures. Adv. Mater. 2000, 12, 235–239. [Google Scholar] [CrossRef]
- Gallezot, P. Conversion of biomass to selected chemical products. Chem. Soc. Rev. 2012, 41, 1538–1558. [Google Scholar] [CrossRef]
- Khanderay, J.C.; Gite, V.V. Fully biobased polyester polyols derived from renewable resources toward preparation of polyurethane and their application for coatings. J. Appl. Polym. Sci. 2019, 136, 47558. [Google Scholar] [CrossRef]
- Gandini, A. The irruption of polymers from renewable resources on the scene of macromolecular science and technology. Green Chem. 2011, 13, 1061–1083. [Google Scholar] [CrossRef]
- Meier, M.A.R.; Metzger, J.O.; Schubert, U.S. Plant oil renewable resources as green alternatives in polymer science. Chem. Soc. Rev. 2007, 36, 1788–1802. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Kulkarni, R.D.; Patil, C.K.; Gite, V.V. Utilization of renewable bio-based resources, viz. sorbitol, diol, and diacid, in the preparation of two pack PU anticorrosive coatings. RSC Adv. 2016, 6, 9843–9850. [Google Scholar] [CrossRef]
- Donnelly, M.J.; Stanford, J.L.; Still, R.H. The conversion of polysaccharides into polyurethanes: A review. Carbohydr. Polym. 1991, 14, 221–240. [Google Scholar] [CrossRef]
- Chowdhury, R.A.; Clarkson, C.M.; Shrestha, S.; El Awad Azrak, S.M.; Mavlan, M.; Youngblood, J.P. High-performance waterborne polyurethane coating based on a blocked isocyanate with cellulose nanocrystals (CNC) as the polyol. ACS Appl. Polym. Mater. 2019, 2, 385–393. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, D.; Zhou, M.; Xia, Y.; Chen, X.; Oliver, S.; Shi, S.; Lei, L. Bio-based omniphobic polyurethane coating providing anti-smudge and anti-corrosion protection. Prog. Org. Coat. 2020, 148, 105844. [Google Scholar] [CrossRef]
- Adekunle, K.F. A review of vegetable oil-based polymers: Synthesis and applications. Open J. Polym. Chem. 2015, 5, 34–40. [Google Scholar] [CrossRef] [Green Version]
- Banković-Ilić, I.B.; Stamenković, O.S.; Veljković, V.B. Biodiesel production from non-edible plant oils. Renew. Sustain. Energy Rev. 2012, 16, 3621–3647. [Google Scholar] [CrossRef]
- Gaikwad, M.S.; Gite, V.V.; Mahulikar, P.P.; Hundiwale, D.G.; Yemul, O.S. Eco-friendly polyurethane coatings from cottonseed and karanja oil. Prog. Org. Coat. 2015, 86, 164–172. [Google Scholar] [CrossRef]
- Kumar, A.; Sharma, S. Potential non-edible oil resources as biodiesel feedstock: An Indian perspective. Renew. Sustain. Energy Rev. 2011, 15, 1791–1800. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Das, L.M.; Babu, M.K.G.; Naik, S.N. Biodiesel development from high acid value polanga seed oil and performance evaluation in a CI engine. Fuel 2007, 86, 448–454. [Google Scholar] [CrossRef]
- Patil, C.K.; Jirimali, H.D.; Paradeshi, J.S.; Chaudhari, B.L.; Alagi, P.K.; Hong, S.C.; Gite, V.V. Synthesis of biobased polyols using algae oil for multifunctional polyurethane coatings. Green Mater. 2018, 6, 165–177. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Development of linseed oil-TiO2 green nanocomposites as antimicrobial coatings. J. Mater. Chem. B 2015, 3, 4458–4471. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.B.; Tatiya, P.D.; Hedaoo, R.K.; Kulkarni, R.D.; Gite, V.V. Polyurethane prepared from neem oil polyesteramides for self-healing anticorrosive coatings. Ind. Eng. Chem. Res. 2013, 52, 10189–10197. [Google Scholar] [CrossRef]
- Karmee, S.K.; Mahesh, P.; Ravi, R.; Chadha, A. Kinetic study of the base-catalyzed transesterification of monoglycerides from Pongamia oil. J. Am. Oil Chem. Soc. 2004, 81, 425–430. [Google Scholar] [CrossRef]
- Bora, M.M.; Deka, R.; Ahmed, N.; Kakati, D.K. Karanja (Millettia pinnata (L.) Panigrahi) seed oil as a renewable raw material for the synthesis of alkyd resin. Ind. Crop. Prod. 2014, 61, 106–114. [Google Scholar] [CrossRef]
- Das, G.; Karak, N. Epoxidized Mesua ferrea L. seed oil-based reactive diluent for BPA epoxy resin and their green nanocomposites. Prog. Org. Coat. 2009, 66, 59–64. [Google Scholar] [CrossRef]
- Thakur, S.; Karak, N. Castor oil-based hyperbranched polyurethanes as advanced surface coating materials. Prog. Org. Coat. 2013, 76, 157–164. [Google Scholar] [CrossRef]
- Akintayo, C.O.; Akintayo, E.T.; Azeez, M.A. Albizia benth oil maleinised polyesteramides: Synthesis, structure, and characteristics. ISRN Polym. Sci. 2012, 2012, 708520. [Google Scholar] [CrossRef] [Green Version]
- Mosiewicki, M.A.; Casado, U.; Marcovich, N.E.; Aranguren, M.I. Polyurethanes from tung oil: Polymer characterization and composites. Polym. Eng. Sci. 2009, 49, 685–692. [Google Scholar] [CrossRef]
- Pawar, M.S.; Kadam, A.S.; Yemul, O.S. Development of polyetheramide based corrosion protective polyurethane coating from mahua oil. Prog. Org. Coat. 2015, 89, 143–149. [Google Scholar] [CrossRef]
- Bora, M.M.; Gogoi, P.; Deka, D.C.; Kakati, D.K. Synthesis and characterization of yellow oleander (Thevetia peruviana) seed oil-based alkyd resin. Ind. Crop. Prod. 2014, 52, 721–728. [Google Scholar] [CrossRef]
- Ashraful, A.M.; Masjuki, H.H.; Kalam, M.A.; Rizwanul Fattah, I.M.; Imtenan, S.; Shahir, S.A.; Mobarak, H.M. Production and comparison of fuel properties, engine performance, and emission characteristics of biodiesel from various non-edible vegetable oils: A review. Energy Convers. Manag. 2014, 80, 202–228. [Google Scholar] [CrossRef]
- Ahamad, S.; Naqvi, F.; Verma, K.L.; Yadav, S. Studies on a newly developed linseed oil-based alumina-filled polyesteramide anticorrosive coating. J. Appl. Polym. Sci. 1999, 72, 1679–1687. [Google Scholar] [CrossRef]
- Dutta, N.; Karak, N.; Dolui, S.K. Synthesis and characterization of polyester resins based on nahar seed oil. Prog. Org. Coat. 2004, 49, 146–152. [Google Scholar] [CrossRef]
- Saravari, O.; Praditvatanakit, S. Preparation and properties of urethane alkyd based on a castor oil/jatropha oil mixture. Prog. Org. Coat. 2013, 76, 698–704. [Google Scholar] [CrossRef]
- Hlaing, N.N.; Oo, M.M. Manufacture of alkyd resin from castor oil. World Acad. Sci. Eng. Technol. 2008, 48, 155–161. [Google Scholar]
- Samadzadeh, M.; Boura, S.H.; Peikari, M.; Ashrafi, A.; Kasiriha, M. Tung oil: An autonomous repairing agent for self-healing epoxy coatings. Prog. Org. Coat. 2011, 70, 383–387. [Google Scholar] [CrossRef]
- Shaker, N.O.; Alian, N.A.; El-Sawy, M.M. Preparation, characterization and evaluation of jojoba seed oil-modified alkyd resins. Chem. Sin. 2012, 3, 1157–1162. [Google Scholar]
- Bhatia, V.K.; Chaudhry, A.; Sivasankaran, G.A.; Bisht, R.P.S.; Kashyap, M. Modification of jojoba oil for lubricant formulations. J. Am. Oil Chem. Soc. 1990, 67, 1–7. [Google Scholar] [CrossRef]
- Khanderay, J.C.; Gite, V.V. Vegetable oil-based polyurethane coatings: Recent developments in India. Green Mater. 2017, 5, 109–122. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M. Microwave assisted synthesis of urethane modified polyesteramide coatings from jatropha seed oil. J. Polym. Environ. 2011, 19, 784–792. [Google Scholar] [CrossRef]
- Aplin, P.J.; Eliseo, T. Ingestion of castor oil plant seeds. Med. J. Aust. 1997, 167, 260–261. [Google Scholar] [CrossRef]
- Shaw, D.; Pearn, J. Oleander poisoning. Med. J. Aust. 1979, 2, 267–269. [Google Scholar] [CrossRef]
- Azam, M.M.; Waris, A.; Nahar, N.M. Prospects and potential of fatty acid methyl esters of some non-traditional seed oils for use as biodiesel in India. Biomass Bioenergy 2005, 29, 293–302. [Google Scholar] [CrossRef]
- Patil, C.K.; Jirimali, H.D.; Paradeshi, J.S.; Chaudhari, B.L.; Alagi, P.K.; Mahulikar, P.P.; Hong, S.C.; Gite, V.V. Chemical transformation of renewable algae oil to polyetheramide polyols for polyurethane coatings. Prog. Org. Coat. 2021, 151, 106084. [Google Scholar] [CrossRef]
- Naik, S.N.; Goud, V.V.; Rout, P.K.; Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renew. Sustain. Energy Rev. 2010, 14, 578–597. [Google Scholar] [CrossRef]
- Klein, J.; Zikeli, S.; Claupein, W.; Gruber, S. Linseed (Linum usitatissimum) as an oil crop in organic farming: Abiotic impacts on seed ingredients and yield. Org. Agric. 2017, 7, 1–19. [Google Scholar] [CrossRef]
- Čeh, B.; Štraus, S.; Hladnik, A.; Kušar, A. Impact of linseed variety, location and production year on seed yield, oil content and its composition. Agronomy 2020, 10, 1770. [Google Scholar] [CrossRef]
- El-Hamidi, M.; Zaher, F.A. Production of vegetable oils in the world and in Egypt: An overview. Bull. Natl. Res. Cent. 2018, 42, 19. [Google Scholar] [CrossRef]
- Zafar, F.; Sharmin, E.; Ashraf, S.M.; Ahmad, S. Studies on poly(styrene-co-maleic anhydride)-modified polyesteramide-based anticorrosive coatings synthesized from a sustainable resource. J. Appl. Polym. Sci. 2004, 92, 2538–2544. [Google Scholar] [CrossRef]
- El-Wahab, H.A.; El-Fattah, M.A.; Ghazy, M.B.M. Synthesis and characterization of new modified anti-corrosive polyesteramide resins incorporated pyromellitimide ring for surface coating. Prog. Org. Coat. 2011, 72, 353–359. [Google Scholar] [CrossRef]
- Zafar, F.; Ashraf, S.M.; Ahmad, S. Studies on zinc-containing linseed oil based polyesteramide. React. Funct. Polym. 2007, 67, 928–935. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M. Development of ambient cured polyesteramide coatings from linseed oil: A sustainable resource. J. Polym. Environ. 2011, 19, 391–397. [Google Scholar] [CrossRef]
- Pathan, S.; Ahmad, S. Synergistic effects of linseed oil based waterborne alkyd and 3-isocynatopropyl triethoxysilane: Highly transparent, mechanically robust, thermally stable, hydrophobic, anticorrosive coatings. ACS Sustain. Chem. Eng. 2016, 4, 3062–3075. [Google Scholar] [CrossRef]
- İşeri-Çağlar, D.; Baştürk, E.; Oktay, B.; Kahraman, M.V. Preparation and evaluation of linseed oil based alkyd paints. Prog. Org. Coat. 2014, 77, 81–86. [Google Scholar] [CrossRef]
- Sharmin, E.; Rahman, O.u.; Zafar, F.; Akram, D.; Alam, M.; Ahmad, S. Linseed oil polyol/ZnO bionanocomposite towards mechanically robust, thermally stable, hydrophobic coatings: A novel synergistic approach utilising a sustainable resource. RSC Adv. 2015, 5, 47928–47944. [Google Scholar] [CrossRef]
- Alam, M.; Ray, A.R.; Ahmad, S. Synthesis and characterization of poly(esteramide-urethane) from linseed oil as anticorrosive coatings. J. Appl. Polym. Sci. 2009, 114, 3268–3273. [Google Scholar] [CrossRef]
- Alam, M.; Ashraf, S.M.; Ahmad, S. Pyridine-poly(urethane ester amide) coatings from linseed oil. J. Polym. Res. 2008, 15, 343–350. [Google Scholar] [CrossRef]
- Alam, M.; Ashraf, S.M.; Ray, A.R.; Ahmad, S. Development of anticorrosive poly(ether-urethane) amide coatings from linseed oil: A sustainable resource. J. Polym. Environ. 2010, 18, 208–215. [Google Scholar] [CrossRef]
- Yadav, S.; Zafar, F.; Hasnat, A.; Ahmad, S. Poly(urethane fatty amide) resin from linseed oil-A renewable resource. Prog. Org. Coat. 2009, 64, 27–32. [Google Scholar] [CrossRef]
- Bagle, A.V.; Jadhav, R.S.; Gite, V.V.; Hundiwale, D.G.; Mahulikar, P.P. Controlled release study of phenol formaldehyde microcapsules containing neem oil as an insecticide. Int. J. Polym. Mater. 2013, 62, 421–425. [Google Scholar] [CrossRef]
- Biswas, K.; Chattopadhyay, I.; Banerjee, R.K.; Bandyopadhyay, U. Biological activities and medicinal properties of neem (Azadirachta indica). Curr. Sci. 2002, 82, 1336–1345. [Google Scholar]
- Barnwal, B.K.; Sharma, M.P. Prospects of biodiesel production from vegetable oils in India. Renew. Sustain. Energy Rev. 2005, 9, 363–378. [Google Scholar] [CrossRef]
- Nayan, N.K.; Kumar, S.; Singh, R.K. Production of the liquid fuel by thermal pyrolysis of neem seed. Fuel 2013, 103, 437–443. [Google Scholar] [CrossRef]
- Vismaya; Sapna Eipeson, W.; Manjunatha, J.R.; Srinivas, P.; Sindhu Kanya, T.C. Extraction and recovery of karanjin: A value addition to karanja (Pongamia pinnata) seed oil. Ind. Crop. Prod. 2010, 32, 118–122. [Google Scholar] [CrossRef]
- Khayoon, M.S.; Olutoye, M.A.; Hameed, B.H. Utilization of crude karanj (Pongamia pinnata) oil as a potential feedstock for the synthesis of fatty acid methyl esters. Bioresour. Technol. 2012, 111, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Goud, V.V.; Pradhan, N.C.; Patwardhan, A.V. Epoxidation of karanja (Pongamia glabra) oil by H2O2. J. Am. Oil Chem. Soc. 2006, 83, 635–640. [Google Scholar] [CrossRef]
- Das, G.; Karak, N. Mesua ferrea L. seed oil-based epoxy resins. J. Appl. Polym. Sci. 2010, 118, 128–134. [Google Scholar] [CrossRef]
- Mahapatra, S.S.; Karak, N. Synthesis and characterization of polyesteramide resins from Nahar seed oil for surface coating applications. Prog. Org. Coat. 2004, 51, 103–108. [Google Scholar] [CrossRef]
- Dutta, S.; Karak, N. Synthesis, characterization of poly(urethane amide) resins from Nahar seed oil for surface coating applications. Prog. Org. Coat. 2005, 53, 147–152. [Google Scholar] [CrossRef]
- Kalita, H.; Karak, N. Bio-based hyperbranched polyurethane/Fe3O4 nanocomposites as shape memory materials. Polym. Adv. Technol. 2013, 24, 819–823. [Google Scholar] [CrossRef]
- Dutta, S.; Karak, N.; Baruah, S. Jute-fiber-reinforced polyurethane green composites based on Mesua ferrea L. seed oil. J. Appl. Polym. Sci. 2010, 115, 843–850. [Google Scholar] [CrossRef]
- Dutta, S.; Karak, N.; Jana, T. Evaluation of Mesua ferrea L. seed oil modified polyurethane paints. Prog. Org. Coat. 2009, 65, 131–135. [Google Scholar] [CrossRef]
- Yadava, D.; Vasudev, S.; Singh, N.; Mohapatra, T.; Prabhu, K. Breeding major oil crops: Present status and future research needs. In Technological Innovations in Major World Oil Crops; Gupta, S.K., Ed.; Springer Science+Business Media, LLC: Berlin/Heidelberg, Germany, 2013; Volume 1, pp. 17–51. [Google Scholar] [CrossRef]
- Oyman, Z.O.; Ming, W.; Linde, R.v.d. Oxidation of drying oils containing non-conjugated and conjugated double bonds catalyzed by a cobalt catalyst. Prog. Org. Coat. 2005, 54, 198–204. [Google Scholar] [CrossRef]
- Biermann, U.; Butte, W.; Holtgrefe, R.; Feder, W.; Metzger, J.O. Esters of calendula oil and tung oil as reactive diluents for alkyd resins. Eur. J. Lipid Sci. Technol. 2010, 112, 103–109. [Google Scholar] [CrossRef]
- Ribeiro da Silva, V.; Mosiewicki, M.A.; Yoshida, M.I.; Coelho da Silva, M.; Stefani, P.M.; Marcovich, N.E. Polyurethane foams based on modified tung oil and reinforced with rice husk ash I: Synthesis and physical chemical characterization. Polym. Test. 2013, 32, 438–445. [Google Scholar] [CrossRef]
- Deka, D.C.; Basumatary, S. High quality biodiesel from yellow oleander (Thevetia peruviana) seed oil. Biomass Bioenergy 2011, 35, 1797–1803. [Google Scholar] [CrossRef]
- Brooks, W.H. Jojoba—A north american desert shrub; its ecology, possible commercialization, and potential as an introduction into other arid regions. J. Arid Environ. 1978, 1, 227–236. [Google Scholar] [CrossRef]
- Voirin, C.; Caillol, S.; Sadavarte, N.V.; Tawade, B.V.; Boutevin, B.; Wadgaonkar, P.P. Functionalization of cardanol: Towards biobased polymers and additives. Polym. Chem. 2014, 5, 3142–3162. [Google Scholar] [CrossRef]
- Ma, H.-X.; Li, J.-J.; Qiu, J.-J.; Liu, Y.; Liu, C.-M. Renewable cardanol-based star-shaped prepolymer containing a phosphazene core as a potential biobased green fire-retardant coating. ACS Sustain. Chem. Eng. 2017, 5, 350–359. [Google Scholar] [CrossRef]
- Kozubek, A.; Zarnowski, R.; Stasiuk, M.; Gubernator, J. Natural amphiphilic phenols as bioactive compounds. Cell. Mol. Biol. Lett. 2001, 6, 351–355. [Google Scholar]
- Kanehashi, S.; Yokoyama, K.; Masuda, R.; Kidesaki, T.; Nagai, K.; Miyakoshi, T. Preparation and characterization of cardanol-based epoxy resin for coating at room temperature curing. J. Appl. Polym. Sci. 2013, 130, 2468–2478. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, X.; Zhu, J.; Liu, X.; Wang, Z.; Yan, J. UV-curable coatings from multiarmed cardanol-based acrylate oligomers. ACS Sustain. Chem. Eng. 2015, 3, 1313–1320. [Google Scholar] [CrossRef]
- Bo, C.; Hu, L.; Jia, P.; Liang, B.; Zhou, J.; Zhou, Y. Structure and thermal properties of phosphorus-containing polyol synthesized from cardanol. RSC Adv. 2015, 5, 106651–106660. [Google Scholar] [CrossRef]
- Kathalewar, M.; Sabnis, A. Preparation of novel CNSL-based urethane polyol via nonisocyanate route: Curing with melamine-formaldehyde resin and structure–property relationship. J. Appl. Polym. Sci. 2015, 132, 41391. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, J.; Chen, S.; Zhou, Y. Synthesis and fire properties of rigid polyurethane foams made from a polyol derived from melamine and cardanol. Polym. Degrad. Stabil. 2014, 110, 27–34. [Google Scholar] [CrossRef]
- Patel, C.J.; Mannari, V. Air-drying bio-based polyurethane dispersion from cardanol: Synthesis and characterization of coatings. Prog. Org. Coat. 2014, 77, 997–1006. [Google Scholar] [CrossRef]
- Suresh, K.I. Rigid polyurethane foams from cardanol: Synthesis, structural characterization, and evaluation of polyol and foam properties. ACS Sustain. Chem. Eng. 2013, 1, 232–242. [Google Scholar] [CrossRef]
- Kavalek, M.; Havrland, B.; Ivanova, T.; Hutla, P.; Skopec, P. Utilization of Jatropha Curcas L. seed cake for production of solid biofuels. In Proceedings of the 12th International Scientific Conference Engineering for Rural Development, Jelgava, Latvia, 23–24 May 2013; pp. 536–540. [Google Scholar]
- Patil, C.K.; Rajput, S.D.; Marathe, R.J.; Kulkarni, R.D.; Phadnis, H.; Sohn, D.; Mahulikar, P.P.; Gite, V.V. Synthesis of bio-based polyurethane coatings from vegetable oil and dicarboxylic acids. Prog. Org. Coat. 2017, 106, 87–95. [Google Scholar] [CrossRef]
- Pfister, D.P.; Xia, Y.; Larock, R.C. Recent advances in vegetable oil-based polyurethanes. ChemSusChem 2011, 4, 703–717. [Google Scholar] [CrossRef]
- Shirke, A.G.; Dholakiya, B.Z.; Kuperkar, K. Enhancement of physico-chemical and anti-corrosive properties of tung oil based polyurethane coating via modification using anhydrides and inorganic acid. Surf. Interfaces 2019, 15, 180–190. [Google Scholar] [CrossRef]
- Alagi, P.; Ghorpade, R.; Jang, J.H.; Patil, C.; Jirimali, H.; Gite, V.; Hong, S.C. Functional soybean oil-based polyols as sustainable feedstocks for polyurethane coatings. Ind. Crop. Prod. 2018, 113, 249–258. [Google Scholar] [CrossRef]
- Alagi, P.; Ghorpade, R.; Jang, J.H.; Patil, C.; Jirimali, H.; Gite, V.; Hong, S.C. Controlled hydroxyl functionality of soybean oil-based polyols for polyurethane coatings with improved anticorrosion properties. Macromol. Res. 2018, 26, 696–703. [Google Scholar] [CrossRef]
- Alagi, P.; Choi, Y.J.; Hong, S.C. Preparation of vegetable oil-based polyols with controlled hydroxyl functionalities for thermoplastic polyurethane. Eur. Polym. J. 2016, 78, 46–60. [Google Scholar] [CrossRef]
- Alagi, P.; Choi, Y.J.; Seog, J.; Hong, S.C. Efficient and quantitative chemical transformation of vegetable oils to polyols through a thiol-ene reaction for thermoplastic polyurethanes. Ind. Crop. Prod. 2016, 87, 78–88. [Google Scholar] [CrossRef]
- Patil, C.K.; Jirimali, H.D.; Mahajan, M.S.; Paradeshi, J.S.; Chaudhari, B.L.; Gite, V.V. Functional anti-corrosive and anti-bacterial surface coatings based on mercaptosuccinic and thiodipropionic acids and algae oil as renewable feedstock. React. Funct. Polym. 2019, 139, 142–152. [Google Scholar] [CrossRef]
- Patil, C.K.; Jirimali, H.D.; Paradeshi, J.S.; Chaudhari, B.L.; Gite, V.V. Functional antimicrobial and anticorrosive polyurethane composite coatings from algae oil and silver doped egg shell hydroxyapatite for sustainable development. Prog. Org. Coat. 2019, 128, 127–136. [Google Scholar] [CrossRef]
- Ahmad, S.; Ashraf, S.M.; Sharmin, E.; Zafar, F.; Hasnat, A. Studies on ambient cured polyurethane modified epoxy coatings synthesized from a sustainable resource. Prog. Cryst. Growth Charact. Mater. 2002, 45, 83–88. [Google Scholar] [CrossRef]
- Sharmin, E.; Ashraf, S.M.; Ahmad, S. Synthesis, characterization, antibacterial and corrosion protective properties of epoxies, epoxy-polyols and epoxy-polyurethane coatings from linseed and Pongamia glabra seed oils. Int. J. Biol. Macromol. 2007, 40, 407–422. [Google Scholar] [CrossRef] [PubMed]
- Akram, D.; Sharmin, E.; Ahmad, S. Development and characterization of boron incorporated linseed oil polyurethanes. J. Appl. Polym. Sci. 2010, 116, 499–508. [Google Scholar] [CrossRef]
- Sharmin, E.; Zafar, F.; Akram, D.; Ahmad, S. Plant oil polyol nanocomposite for antibacterial polyurethane coating. Prog. Org. Coat. 2013, 76, 541–547. [Google Scholar] [CrossRef]
- Pouladi, J.; Mirabedini, S.M.; Eivaz Mohammadloo, H.; Rad, N.G. Synthesis of novel plant oil-based isocyanate-free urethane coatings and study of their anti-corrosion properties. Eur. Polym. J. 2021, 153, 110502. [Google Scholar] [CrossRef]
- Ahmad, S.; Ashraf, S.M.; Hasnat, A.; Yadav, S.; Jamal, A. Studies on urethane-modified alumina-filled polyesteramide anticorrosive coatings cured at ambient temperature. J. Appl. Polym. Sci. 2001, 82, 1855–1865. [Google Scholar] [CrossRef]
- Paraskar, P.M.; Prabhudesai, M.S.; Kulkarni, R.D. Synthesis and characterizations of air-cured polyurethane coatings from vegetable oils and itaconic acid. React. Funct. Polym. 2020, 156, 104734. [Google Scholar] [CrossRef]
- Shaik, M.R.; Alam, M.; Alandis, N.M. Development of sustainable resource based poly(urethane-etheramide)/Fe2O3 nanocomposite as anticorrosive coating materials. J. Polym. Eng. 2015, 35, 905–916. [Google Scholar] [CrossRef]
- Riaz, U.; Ashraf, S.M.; Ahmad, S. Comparative study of polyaniline and poly(1-naphthylamine) dispersed oil polyurethane coatings. Anti-Corros. Methods Mater. 2008, 55, 308–316. [Google Scholar] [CrossRef]
- Ahmad, S.; Zafar, F.; Sharmin, E.; Garg, N.; Kashif, M. Synthesis and characterization of corrosion protective polyurethanefattyamide/silica hybrid coating material. Prog. Org. Coat. 2012, 73, 112–117. [Google Scholar] [CrossRef]
- Yücedag, F.; Atalay-Oral, C.; Erkal, S.; Sirkecioglu, A.; Karasartova, D.; Sahin, F.; Tantekin-Ersolmaz, S.B.; Güner, F.S. Antibacterial oil-based polyurethane films for wound dressing applications. J. Appl. Polym. Sci. 2010, 115, 1347–1357. [Google Scholar] [CrossRef]
- Naik, R.B.; Ratna, D.; Singh, S.K. Synthesis and characterization of novel hyperbranched alkyd and isocyanate trimer based high solid polyurethane coatings. Prog. Org. Coat. 2014, 77, 369–379. [Google Scholar] [CrossRef]
- Marathe, R.; Tatiya, P.; Chaudhari, A.; Lee, J.; Mahulikar, P.; Sohn, D.; Gite, V. Neem acetylated polyester polyol-Renewable source based smart PU coatings containing quinoline (corrosion inhibitor) encapsulated polyurea microcapsules for enhance anticorrosive property. Ind. Crop. Prod. 2015, 77, 239–250. [Google Scholar] [CrossRef]
- Chaudhari, A.; Gite, V.; Rajput, S.; Mahulikar, P.; Kulkarni, R. Development of eco-friendly polyurethane coatings based on neem oil polyetheramide. Ind. Crop. Prod. 2013, 50, 550–556. [Google Scholar] [CrossRef]
- Chaudhari, A.B.; Anand, A.; Rajput, S.D.; Kulkarni, R.D.; Gite, V.V. Synthesis, characterization and application of Azadirachta indica juss (neem oil) fatty amides (AIJFA) based polyurethanes coatings: A renewable novel approach. Prog. Org. Coat. 2013, 76, 1779–1785. [Google Scholar] [CrossRef]
- Chaudhari, A.; Kuwar, A.; Mahulikar, P.; Hundiwale, D.; Kulkarni, R.; Gite, V. Development of anticorrosive two pack polyurethane coatings based on modified fatty amide of Azadirachta indica Juss oil cured at room temperature—A sustainable resource. RSC Adv. 2014, 4, 17866–17872. [Google Scholar] [CrossRef]
- Chaudhari, A.; Kulkarni, R.; Mahulikar, P.; Sohn, D.; Gite, V. Development of PU coatings from neem oil based alkyds prepared by the monoglyceride route. J. Am. Oil Chem. Soc. 2015, 92, 733–741. [Google Scholar] [CrossRef] [Green Version]
- Ahmadi, Y.; Ahmad, S. Surface-active antimicrobial and anticorrosive oleo-polyurethane/graphene oxide nanocomposite coatings: Synergistic effects of in-situ polymerization and π-π interaction. Prog. Org. Coat. 2019, 127, 168–180. [Google Scholar] [CrossRef]
- Ranade, S.; Neelakantan, L. Exploring in situ integration of pongamia oil to improve barrier properties of polyurethane coatings. J. Appl. Polym. Sci. 2020, 137, 49553. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M. Development of poly(urethane esteramide) coatings from Pongamia glabra oil as anticorrosive applications. Int. J. Polym. Anal. Charact. 2015, 20, 330–343. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M.; Ahmad, N.; Naushad, M. Synthesis and characterization of poly(urethane-ether azomethine) fatty amide based corrosion resistant coatings from Pongamia glabra oil: An eco-friendly approach. J. Chem. 2016, 2016, 5623126. [Google Scholar] [CrossRef] [Green Version]
- Zafar, F.; Kashif, M.; Sharmin, E.; Ahmad, S. Studies on boron containing poly(urethane fattyamide). Macromol. Symp. 2010, 290, 79–84. [Google Scholar] [CrossRef]
- Siyanbola, T.O.; Neelambaram, P.; Mohanty, S.; Somisetti, V.; Basak, P.; Narayan, R.; Kothapalli, R.V.S.N. The effects of carbonized Eucalyptus globulus leaves on castor seed oil based urethane coating system. Prog. Org. Coat. 2019, 131, 42–48. [Google Scholar] [CrossRef]
- Bhosale, N.; Shaik, A.; Mandal, S.K. Synthesis and characterization of castor oil based hybrid polymers and their polyurethane–urea/silica coatings. RSC Adv. 2015, 5, 103625–103635. [Google Scholar] [CrossRef]
- Gite, V.V.; Kulkarni, R.D.; Hundiwale, D.G.; Kapadi, U.R. Synthesis and characterisation of polyurethane coatings based on trimer of isophorone diisocyanate (IPDI) and monoglycerides of oils. Surf. Coat. Int. B Coat. Trans. 2006, 89, 117–122. [Google Scholar] [CrossRef]
- Shaik, A.; Narayan, R.; Raju, K.V.S.N. Synthesis and properties of siloxane-crosslinked polyurethane-urea/silica hybrid films from castor oil. J. Coat. Technol. Res. 2014, 11, 397–407. [Google Scholar] [CrossRef]
- Bakhshi, H.; Yeganeh, H.; Yari, A.; Nezhad, S.K. Castor oil-based polyurethane coatings containing benzyl triethanol ammonium chloride: Synthesis, characterization, and biological properties. J. Mater. Sci. 2014, 49, 5365–5377. [Google Scholar] [CrossRef]
- Siyanbola, T.O.; Enishetty, R.; Kumar, R.; James, O.O.; Olasehinde, G.I.; Kaki, S.; Narayan, R.; Raju, K.V.S.N. Specific crosslinking effects of poly(epichlorohydrin)-triol on urethane polymer matrix of castor seed oil-based coatings. J. Coat. Technol. Res. 2021, 18, 129–141. [Google Scholar] [CrossRef]
- Morsi, S.M.M.; Mohamed, H.A.; Abdel Ghany, N.A. Development of advanced-functional polyurethane/red iron oxide composites as protective one coating systems for steel. Prog. Org. Coat. 2019, 136, 105236. [Google Scholar] [CrossRef]
- Akram, D.; Sharmin, E.; Ahmad, S. Synthesis, characterization and corrosion protective properties of boron-modified polyurethane from natural polyol. Prog. Org. Coat. 2008, 63, 25–32. [Google Scholar] [CrossRef]
- Nardeli, J.V.; Fugivara, C.S.; Taryba, M.; Montemor, M.F.; Benedetti, A.V. Biobased self-healing polyurethane coating with Zn micro-flakes for corrosion protection of AA7475. Chem. Eng. J. 2021, 404, 126478. [Google Scholar] [CrossRef]
- Nardeli, J.V.; Fugivara, C.S.; Taryba, M.; Montemor, M.F.; Ribeiro, S.J.L.; Benedetti, A.V. Novel healing coatings based on natural-derived polyurethane modified with tannins for corrosion protection of AA2024-T3. Corros. Sci. 2020, 162, 108213. [Google Scholar] [CrossRef]
- Nardeli, J.V.; Fugivara, C.S.; Pinto, E.R.P.; Polito, W.L.; Messaddeq, Y.; Ribeiro, S.J.L.; Benedetti, A.V. Preparation of polyurethane monolithic resins and modification with a condensed tannin-yielding self-healing property. Polymers 2019, 11, 1890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uscátegui, Y.L.; Arévalo, F.R.; Díaz, L.E.; Cobo, M.I.; Valero, M.F. Microbial degradation, cytotoxicity and antibacterial activity of polyurethanes based on modified castor oil and polycaprolactone. J. Biomater. Sci. Polym. Ed. 2016, 27, 1860–1879. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Badran, B.M.; Rabie, A.M.; Morsi, S.M.M. Synthesis and characterization of aqueous (polyurethane/aromatic polyamide sulfone) copolymer dispersions from castor oil. Prog. Org. Coat. 2014, 77, 965–974. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Morsi, S.M.M.; Badran, B.M.; Rabie, A.M. Eco-friendly protective coatings based on poly(urethane sulfone amide) dispersions for carbon steel. J. Coat. Technol. Res. 2017, 14, 437–446. [Google Scholar] [CrossRef]
- Shaik, M.R.; Alam, M.; Alandis, N.M. Development of castor oil based poly(urethane-esteramide)/TiO2 nanocomposites as anticorrosive and antimicrobial coatings. J. Nanomater. 2015, 2015, 745217. [Google Scholar] [CrossRef] [Green Version]
- Akram, D.; Hakami, O.; Sharmin, E.; Ahmad, S. Castor and linseed oil polyurethane/TEOS hybrids as protective coatings: A synergistic approach utilising plant oil polyols, a sustainable resource. Prog. Org. Coat. 2017, 108, 1–14. [Google Scholar] [CrossRef]
- Pathak, R.; Kathalewar, M.; Wazarkar, K.; Sabnis, A. Non-isocyanate polyurethane (NIPU) from tris-2-hydroxy ethyl isocyanurate modified fatty acid for coating applications. Prog. Org. Coat. 2015, 89, 160–169. [Google Scholar] [CrossRef]
- Akintayo, C.O.; Akintayo, E.T.; Ziegler, T. Studies on newly developed urethane modified polyetheramide coatings from Albizia benth oil. Prog. Org. Coat. 2011, 71, 89–97. [Google Scholar] [CrossRef]
- Huang, Y.; Pang, L.; Wang, H.; Zhong, R.; Zeng, Z.; Yang, J. Synthesis and properties of UV-curable tung oil based resins via modification of Diels–Alder reaction, nonisocyanate polyurethane and acrylates. Prog. Org. Coat. 2013, 76, 654–661. [Google Scholar] [CrossRef]
- Raychura, A.J.; Dholakiya, B.Z.; Patel, K.I.; Jauhari, S. Development of non-traditional vegetable-oil-based two-pack polyurethane for wood-finished coating: An alternative approach. ChemistrySelect 2018, 3, 10837–10842. [Google Scholar] [CrossRef]
- Raychura, A.J.; Jauhari, S.; Dholakiya, B.Z. Development of wood protective polyurethane coatings from mahua oil-based polyetheramide polyol: A renewable approach. Soft Mater. 2018, 16, 209–219. [Google Scholar] [CrossRef]
- Raychura, A.J.; Jauhari, S.; Patel, K.I.; Dholakiya, B.Z. A renewable approach toward the development of mahua oil-based wood protective polyurethane coatings: Synthesis and performance evaluation. J. Appl. Polym. Sci. 2018, 135, 46722. [Google Scholar] [CrossRef]
- Siyanbola, T.O.; Sasidhar, K.; Anjaneyulu, B.; Kumar, K.P.; Rao, B.V.S.K.; Narayan, R.; Olaofe, O.; Akintayo, E.T.; Raju, K.V.S.N. Anti-microbial and anti-corrosive poly(ester amide urethane) siloxane modified ZnO hybrid coatings from Thevetia peruviana seed oil. J. Mater. Sci. 2013, 48, 8215–8227. [Google Scholar] [CrossRef]
- Siyanbola, T.O.; Sasidhar, K.; Rao, B.V.S.K.; Narayan, R.; Olaofe, O.; Akintayo, E.T.; Raju, K.V.S.N. Development of functional polyurethane–ZnO hybrid nanocomposite coatings from Thevetia peruviana seed oil. J. Am. Oil Chem. Soc. 2015, 92, 267–275. [Google Scholar] [CrossRef]
- Athawale, V.; Shetty, N. Synthesis and characterisation of low-cost cardanol polyurethanes. Pigment. Resin Technol. 2010, 39, 9–14. [Google Scholar] [CrossRef]
- Balgude, D.; Sabnis, A.; Ghosh, S.K. Synthesis and characterization of cardanol based aqueous 2K polyurethane coatings. Eur. Polym. J. 2016, 85, 620–634. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M. Microwave-assisted preparation of urethane-modified polyetheramide coatings from Jatropha seed oil. High Perform. Polym. 2012, 24, 538–545. [Google Scholar] [CrossRef]
- Alam, M.; Alandis, N.M.; Ahmad, N.; Alam, M.A.; Sharmin, E. Jatropha seed oil derived poly(esteramide-urethane)/fumed silica nanocomposite coatings for corrosion protection. Open Chem. 2019, 17, 206–219. [Google Scholar] [CrossRef]
- Sørensen, P.A.; Kiil, S.; Dam-Johansen, K.; Weinell, C.E. Anticorrosive coatings: A review. J. Coat. Technol. Res. 2009, 6, 135–176. [Google Scholar] [CrossRef]
- Alam, M.; Ray, A.R.; Ashraf, S.M.; Ahmad, S. Synthesis, characterization and performance of amine modified linseed oil fatty amide coatings. J. Am. Oil Chem. Soc. 2009, 86, 573–580. [Google Scholar] [CrossRef]
- Somisetti, V.; Narayan, R.; Kothapalli, R.V.S.N. Multifunctional polyurethane coatings derived from phosphated cardanol and undecylenic acid based polyols. Prog. Org. Coat. 2019, 134, 91–102. [Google Scholar] [CrossRef]
- Voit, B. Hyperbranched polymers-All problems solved after 15 years of research? J. Polym. Sci. Part A Polym. Chem. 2005, 43, 2679–2699. [Google Scholar] [CrossRef] [Green Version]
- Bechi, D.M.; Luca, M.A.d.; Martinelli, M.; Mitidieri, S. Organic–inorganic coatings based on epoxidized castor oil with APTES/TIP and TEOS/TIP. Prog. Org. Coat. 2013, 76, 736–742. [Google Scholar] [CrossRef]
- Akram, D.; Sharmin, E.; Ahmad, S. Synthesis and characterization of boron incorporated polyester polyol from linseed oil: A sustainable material. Macromol. Symp. 2009, 277, 130–137. [Google Scholar] [CrossRef]
- Ong, H.R.; Ramli, R.; Khan, M.M.R.; Yunus, R.M. The influence of CuO nanoparticle on non-edible rubber seed oil based alkyd resin preparation and its antimicrobial activity. Prog. Org. Coat. 2016, 101, 245–252. [Google Scholar] [CrossRef] [Green Version]
- Esser-Kahn, A.P.; Odom, S.A.; Sottos, N.R.; White, S.R.; Moore, J.S. Triggered release from polymer capsules. Macromolecules 2011, 44, 5539–5553. [Google Scholar] [CrossRef]
- Cho, S.H.; White, S.R.; Braun, P.V. Self-healing polymer coatings. Adv. Mater. 2009, 21, 645–649. [Google Scholar] [CrossRef]
- Zhu, D.Y.; Rong, M.Z.; Zhang, M.Q. Self-healing polymeric materials based on microencapsulated healing agents: From design to preparation. Prog. Polym. Sci. 2015, 49–50, 175–220. [Google Scholar] [CrossRef]
- Suryanarayana, C.; Rao, K.C.; Kumar, D. Preparation and characterization of microcapsules containing linseed oil and its use in self-healing coatings. Prog. Org. Coat. 2008, 63, 72–78. [Google Scholar] [CrossRef]
- Samadzadeh, M.; Boura, S.H.; Peikari, M.; Kasiriha, S.M.; Ashrafi, A. A review on self-healing coatings based on micro/nanocapsules. Prog. Org. Coat. 2010, 68, 159–164. [Google Scholar] [CrossRef]
- Tatiya, P.D.; Hedaoo, R.K.; Mahulikar, P.P.; Gite, V.V. Novel polyurea microcapsules using dendritic functional monomer: Synthesis, characterization, and its use in self-healing and anticorrosive polyurethane coatings. Ind. Eng. Chem. Res. 2013, 52, 1562–1570. [Google Scholar] [CrossRef]
- Vasilev, K.; Cook, J.; Griesser, H.J. Antibacterial surfaces for biomedical devices. Expert Rev. Med. Devices 2009, 6, 553–567. [Google Scholar] [CrossRef] [PubMed]
- Tiller, J.C. Antimicrobial Surfaces. In Bioactive Surfaces. Advances in Polymer Science; Börner, H.G., Lutz, J.-F., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 240. [Google Scholar] [CrossRef]
- Yari, A.; Yeganeh, H.; Bakhshi, H.; Gharibi, R. Preparation and characterization of novel antibacterial castor oil-based polyurethane membranes for wound dressing application. J. Biomed. Mater. Res. A 2014, 102, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Mandal, B.; Majumdar, S.G.; Maity, C.R. Chemical and nutritional evaluation of Pongamia glabra oil and Acacia auriculaeformis oil. J. Am. Oil Chem. Soc. 1984, 61, 1447–1449. [Google Scholar] [CrossRef]
- Sharmin, E.; Akram, D.; Zafar, F.; Ashraf, S.M.; Ahmad, S. Plant oil polyol based poly(ester urethane) metallohybrid coatings. Prog. Org. Coat. 2012, 73, 118–122. [Google Scholar] [CrossRef]
- Zafar, F.; Ashraf, S.M.; Ahmad, S. In situ development of Zn/Cd-incorporated poly(esteramide-urethane) from sustainable resource. J. Appl. Polym. Sci. 2008, 110, 584–593. [Google Scholar] [CrossRef]
- Thakur, S.; Barua, S.; Karak, N. Self-healable castor oil based tough smart hyperbranched polyurethane nanocomposite with antimicrobial attributes. RSC Adv. 2015, 5, 2167–2176. [Google Scholar] [CrossRef]
- Chamberlain, N.R.; Mehrtens, B.G.; Xiong, Z.; Kapral, F.A.; Boardman, J.L.; Rearick, J.I. Correlation of carotenoid production, decreased membrane fluidity, and resistance to oleic acid killing in Staphylococcus aureus 18Z. Infect. Immun. 1991, 59, 4332–4337. Available online: http://iai.asm.org/content/59/12/4332.abstract (accessed on 1 February 2021). [CrossRef] [PubMed] [Green Version]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver nanoparticles: Green synthesis and their antimicrobial activities. Adv. Colloid Interface Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Kvítek, L.; Panáček, A.; Soukupová, J.; Kolář, M.; Večeřová, R.; Prucek, R.; Holecová, M.; Zbořil, R. Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles (NPs). J. Phys. Chem. C 2008, 112, 5825–5834. [Google Scholar] [CrossRef]
- Fu, H.; Wang, Y.; Li, X.; Chen, W. Synthesis of vegetable oil-based waterborne polyurethane/silver-halloysite antibacterial nanocomposites. Compos. Sci. Technol. 2016, 126, 86–93. [Google Scholar] [CrossRef]
- Liang, H.; Liu, L.; Lu, J.; Chen, M.; Zhang, C. Castor oil-based cationic waterborne polyurethane dispersions: Storage stability, thermo-physical properties and antibacterial properties. Ind. Crop. Prod. 2018, 117, 169–178. [Google Scholar] [CrossRef]

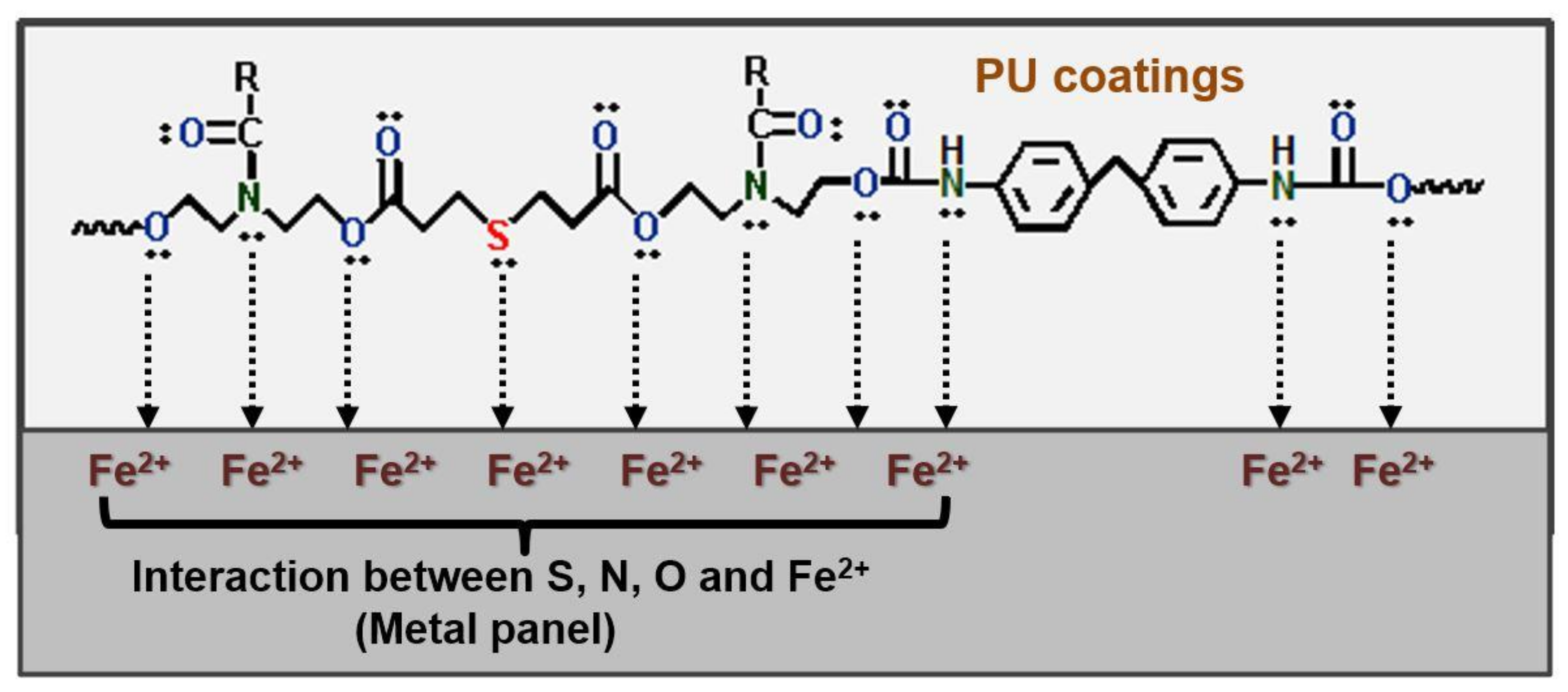
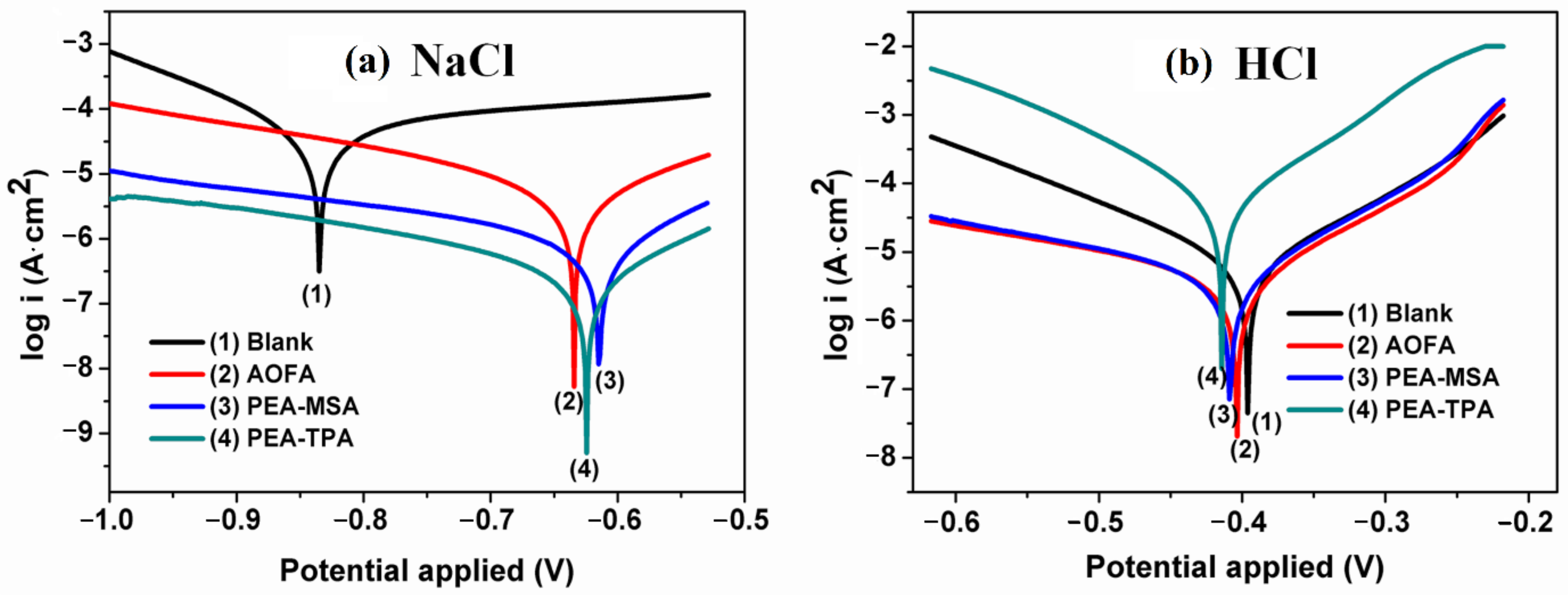

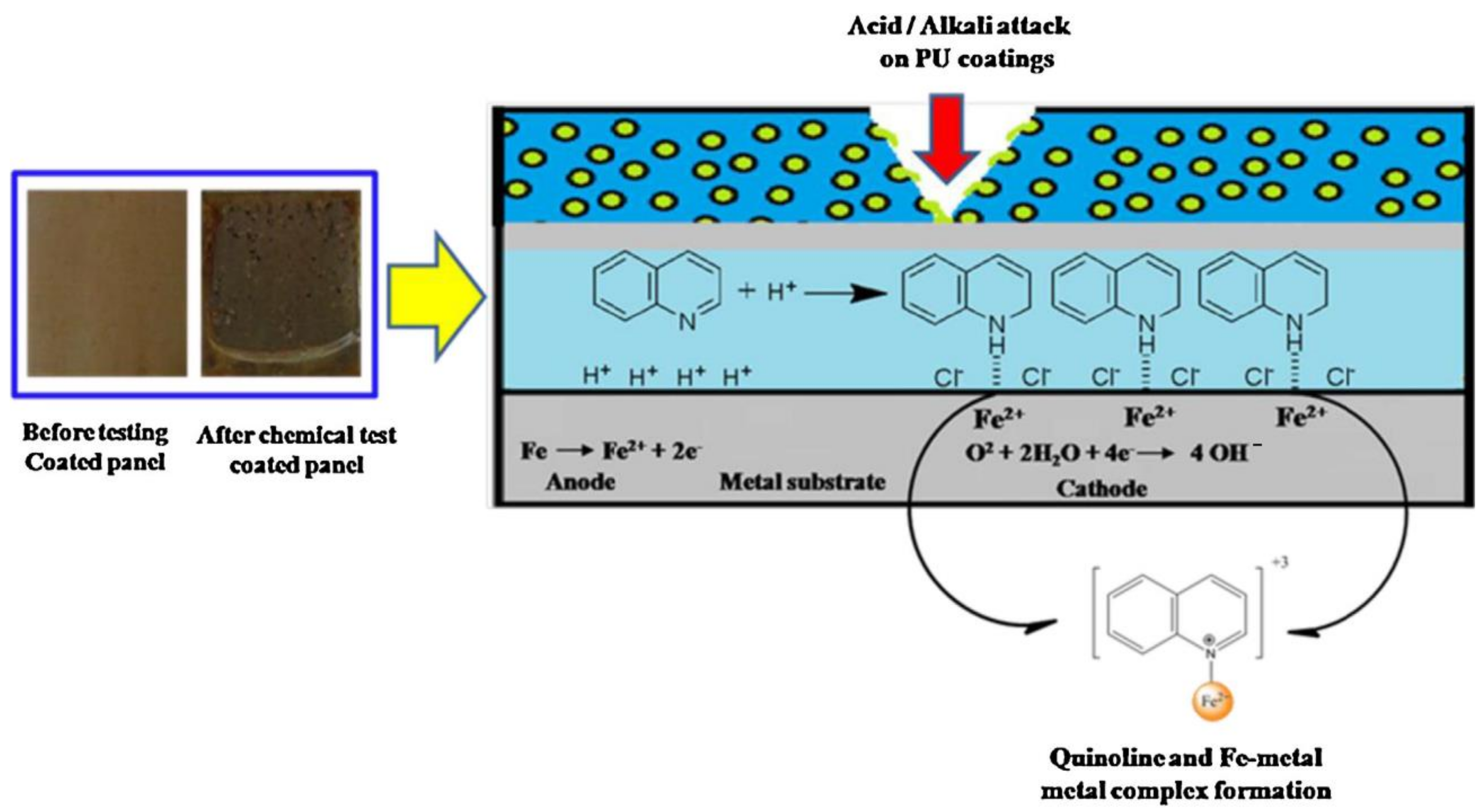

| NEVOs | Type of Fatty Acids | Oleic | Linoleic | Linol-Enic | Palmitic | Stearic | Ricino-Leic | α-Eleos-Tearic Acid | cis-11,14-Eicosa-Dienoic Acid | Arac-Hidic | Ligno-Ceric | Behenic | Unknown | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Carbon | 18 | 18 | 18 | 16 | 18 | 18 | 18 | 20 | 20 | 24 | 22 | - | ||
| No. of C=C Bonds | 1 | 2 | 3 | 0 | 0 | 1 | 3 | 2 | 0 | 0 | 0 | - | ||
| AO | 77.6 | 2.2 | 1.4 | 9.2 | 3.2 | - | - | 1.5 | - | - | - | 3.7 | [26] | |
| LO | 18.5 | 17.3 | 53.2 | 6.6 | 4.4 | - | - | - | - | - | - | - | [27] | |
| NO | 50.0 | 5.2 | - | 11.9 | 30.0 | - | - | - | 2.9 | - | - | - | [28] | |
| PO (KO) | 44.5~71.3 | 10.8~18.3 | - | 3.7~7.9 | 2.4~8.9 | - | - | - | 2.2~4.7 | 1.1~3.5 | 4.2~5.3 | - | [29,30] | |
| NAO | 52.3 | 22.3 | - | 15.9 | 9.5 | - | - | - | - | - | - | - | [31] | |
| CO | - | - | - | - | - | 92.0~95.0 | - | - | - | - | - | 5.0~8.0 | [32] | |
| ABO | 20.7 | 47.9 | 5.8 | 14.1 | 11.2 | [33] | ||||||||
| TO | 3.5~12.7 | 8.0~10.0 | - | - | - | - | 77.0~82.0 | - | - | - | - | [34] | ||
| MO | 35.8 | 16.6 | - | 22.3 | 22.5 | - | - | - | - | - | - | 2.8 | [35] | |
| TPO | 43.7 | 19.9 | - | 23.3 | 10.7 | - | - | - | 2.4 | - | - | - | [36] | |
| Jojoba | 43.5~66 | 25.2~34.4 | [37] | |||||||||||
| JO | 34.3~44.7 | 31.4~43.2 | 13.6~15.1 | 7.1~7.4 | [37] | |||||||||
| NEVOs | Specific Gravity (g/cm3) (25 °C) | Refractive Index | Saponi-Fication Value (mg KOH/g) | Iodine Value (g of I2/100 g) | Acid Value (mg KOH/g) | Hydroxyl Value (mg KOH/g) | References |
|---|---|---|---|---|---|---|---|
| AO | N.A. a | N.A. a | 193 | 85 | 0.2 | - | [26] |
| LO | 0.896 | 1.478 | 160 | 181 | 8.3 | - | [38] |
| NO | 0.920 | 1.503 | 186 | 65 | 1.0 | - | [28] |
| PO (KO) | 0.938 | 1.467 | 182 | 89.9 | 11.5 | - | [30] |
| NAO | 0.890 | 1.473 | 260 | 89.3 | 14.3 | - | [30,31,39] |
| CO | 0.913 | 1.474 | 185 | 82 | 0.6 | 165 | [40,41] |
| ABO | 0.915 | 1.473 | 185 | 104.5 | 8.1 | 0.3 | [33] |
| TO | 0.915 | 1.517 | 187 | 167 | 2.6 | - | [34,42] |
| MO | 0.904 | 1.477 | 187 | 62.4 | 0.4 | [35] | |
| TPO | 0.912 | 1.464 | 121 | 71.4 | 0.66 | - | [30] |
| Jojoba | 0.866 | 1.464 | 100 | 95 | 2.0 | - | [43,44] |
| Cardanol | 0.941~0.924 | 1693~1686 | 47~58 | 215~235 | 12.1~15.4 | 170~250 | [45] |
| JO | 0.903 | 1.475 | 190 | 97 | 2.1 | - | [40,46] |
| NEVOs | Synthetic Intermediate/Pathway | Functional Ingredients | Functionality | References |
|---|---|---|---|---|
| AO | PEsA | Mercaptosuccinic acid, thiodipropionic acid | Antibacterial, anticorrosive | [103] |
| PEsA, alkyd | Phthalic anhydride, itaconic, maleic, dimer acids | Antibacterial, anticorrosive | [26] | |
| PEsA | Ricinoleic acid, silver (Ag)-hydroxyapatite (HAP) nanoparticles (NPs) | Antibacterial, anticorrosive | [104] | |
| PEtA | Fatty amide | Antibacterial, anticorrosive | [50] | |
| LO | Epoxidation, hydroxylation | LO, TDI | Anticorrosive | [105] |
| Epoxidation, hydroxylation | Long hydrocarbon chains of fatty triester residues | Antibacterial | [106] | |
| Epoxidation, hydroxylation, polyesterification | Boric acid | Anticorrosive | [107] | |
| Epoxidation, esterification | Copper acetate | Antibacterial | [108] | |
| Epoxidation, carbonation | Hydroxyl groups of nonisocyate PU | Anticorrosive | [109] | |
| PEsA | Fatty amide, pyridine | Anticorrosive | [63] | |
| PEsA | Fatty amide, EDTA | Anticorrosive | [62] | |
| PEsA | Fatty amide, aluminium hydroxide | Anticorrosive | [110] | |
| PEsA | Fatty amide | Anticorrosive | [111] | |
| PEtA | Fatty amide, resorcinol | Anticorrosive | [64] | |
| PEtA | Fatty amide, Fe2O3 NPs | Anticorrosive | [112] | |
| Fatty amide | Fatty amide, poly(1-naphthylamine) | Anticorrosive | [113] | |
| Diethanolamine | Fatty amide | Anticorrosive | [65] | |
| Diethanolamide | Fatty amide, tetraethoxyorthosilicate (TEOS) | Anticorrosive | [114] | |
| Polyesterification | - | Antibacterial | [115] | |
| Alkyd | Hyperbranching structure | Anticorrosive | [116] | |
| NO | Epoxidation | Quinoline microcapsule | Anticorrosive | [117] |
| PEtA | Fatty amide, TiO2 NP | Anticorrosive | [118] | |
| Fatty amide | Diethanolamine | Anticorrosive | [119] | |
| Fatty amide | Fatty amide, piperazine | Anticorrosive | [120] | |
| Alkyd | Anhydride, itaconic, and dimer fatty acids | Anticorrosive | [121] | |
| PO (KO) | Epoxidation, hydroxylation | Bioflavonoids-karanjin and pongamol | Antibacterial | [106] |
| Epoxidation, acrylation | Acrylic acid, graphene oxide (GOx) | Antibacterial, anticorrosive | [122] | |
| Monoglyceride, polyol mixture | - | Anticorrosive | [123] | |
| PEsA | Fatty amide, ethylene glycol tetraacetic acid | Anticorrosive | [124] | |
| PEsA | Fatty amide | Anticorrosive | [111] | |
| PEtA | Fatty amide, ethane 1,2-di(azomethine) Bisphenol | Anticorrosive | [125] | |
| PEsA, boronation | Fatty amide, boric acid | Anticorrosive | [126] | |
| NAO | Polyol mixture | Fatty amide | Anticorrosive | [75] |
| CO | - | COOH functionalized carbon nanorods | Anticorrosive | [127] |
| Acrylation | Alkoxy silane | Anticorrosive | [128] | |
| Monoglyceride | - | Anticorrosive | [32] | |
| Monoglyceride | Trimer of isocyanate | Anticorrosive | [129] | |
| Polyol mixture | 3-Amino propyl trimethoxy silane, 3-glycidoxy propyl trimethoxy silane | Antibacterial | [130] | |
| Polyol mixture | Benzyl triethanol ammonium chloride | Antibacterial | [131] | |
| Polyol mixture | Poly(epichlorohydrin)-triol, PMDI | Antibacterial, anticorrosive | [132] | |
| Polyol mixture | Red iron oxide pigment | anticorrosive | [133] | |
| Esterification | Boric acid, phthalic anhydride | Anticorrosive | [134] | |
| Transesterification | Zinc flakes | Anticorrosive | [135] | |
| Transesterification | Tannin | Anticorrosive | [136,137] | |
| Transesterification | - | Anticorrosive | [40] | |
| Transesterification, polyol mixture | Pentaerythritol, polycaprolactone, | Antibacterial | [138] | |
| Transesterification, copolymerization | Bis(4-aminophenyl)sulfone | Anticorrosive | [139,140] | |
| PEsA | Fatty amide, TiO2 | Anticorrosive, antibacterial | [141] | |
| PEsA | Fatty amide | Anticorrosive | [111] | |
| Hydrolysis, condensation | TEOS | Anticorrosive, antibacterial | [142] | |
| Epoxidation | Tris-2-hydroxy ethyl isocyanurate, hydroxyl groups | Anticorrosive | [143] | |
| ABO | PEtA | Fatty amide | Anticorrosive | [144] |
| TO | Hydroxylation, alcoholysis | Anhydrides, boric acid | Anticorrosive | [98] |
| Diels-Alder | Glycidyl methacrylate | Anticorrosive | [145] | |
| MO | Fatty amide | Fatty amide, diethanolamine | Anticorrosive, Antibacterial | [146] |
| PEtA | Fatty amide | Anticorrosive | [35] | |
| PEtA | Fatty amide, diglycidyl ether of bisphenol-A | Antibacterial | [147] | |
| PEtA | Fatty amide, Bisphenol-C | Antibacterial | [148] | |
| TPO | PEsA, glycerides | Fatty amide, ZnO NPs | Anticorrosive | [149,150] |
| Cardanol | Epoxidation, hydroxylation | - | Anticorrosive | [151] |
| Epoxidation, hydroxylation | - | Anticorrosive | [152] | |
| Cardanol-epoxy condensation | Autoxidative catalyst drier | Anticorrosive | [93] | |
| JO | PEtA | Fatty amide, hydroquinone | Anticorrosive | [153] |
| PEsA | Fatty amide, itaconic acid | Anticorrosive | [46] | |
| PEsA | Fatty amide, fumed silica NPs | Anticorrosive | [154] |
| Ag-HAP Content | Ecorr (mV) | Icorr (µA cm−2) | Rp (kΩ) | Corrosion Rate (mm/Year) | Inhibition Efficiency (%) |
|---|---|---|---|---|---|
| Blank | −842.90 | 9.38130 | 1.7244 | 0.10901 | − |
| 0% | −600.450 | 0.0056990 | 57.0990 | 0.00662 | 99.39 |
| 0.5% | −563.080 | 0.0053659 | 38.3980 | 0.00623 | 99.94 |
| 1% | −459.450 | 0.0032487 | 81.4370 | 0.00377 | 99.96 |
| 2% | −591.100 | 0.0027605 | 85.4140 | 0.00320 | 99.97 |
| 4% | −517.430 | 0.0018696 | 128.700 | 0.00217 | 99.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patil, C.K.; Jung, D.W.; Jirimali, H.D.; Baik, J.H.; Gite, V.V.; Hong, S.C. Nonedible Vegetable Oil-Based Polyols in Anticorrosive and Antimicrobial Polyurethane Coatings. Polymers 2021, 13, 3149. https://doi.org/10.3390/polym13183149
Patil CK, Jung DW, Jirimali HD, Baik JH, Gite VV, Hong SC. Nonedible Vegetable Oil-Based Polyols in Anticorrosive and Antimicrobial Polyurethane Coatings. Polymers. 2021; 13(18):3149. https://doi.org/10.3390/polym13183149
Chicago/Turabian StylePatil, Chandrashekhar K., Dong Wook Jung, Harishchandra D. Jirimali, Joon Hyun Baik, Vikas V. Gite, and Sung Chul Hong. 2021. "Nonedible Vegetable Oil-Based Polyols in Anticorrosive and Antimicrobial Polyurethane Coatings" Polymers 13, no. 18: 3149. https://doi.org/10.3390/polym13183149