Seed Priming with Silicon as a Potential to Increase Salt Stress Tolerance in Lathyrus odoratus
Abstract
:1. Introduction
2. Results
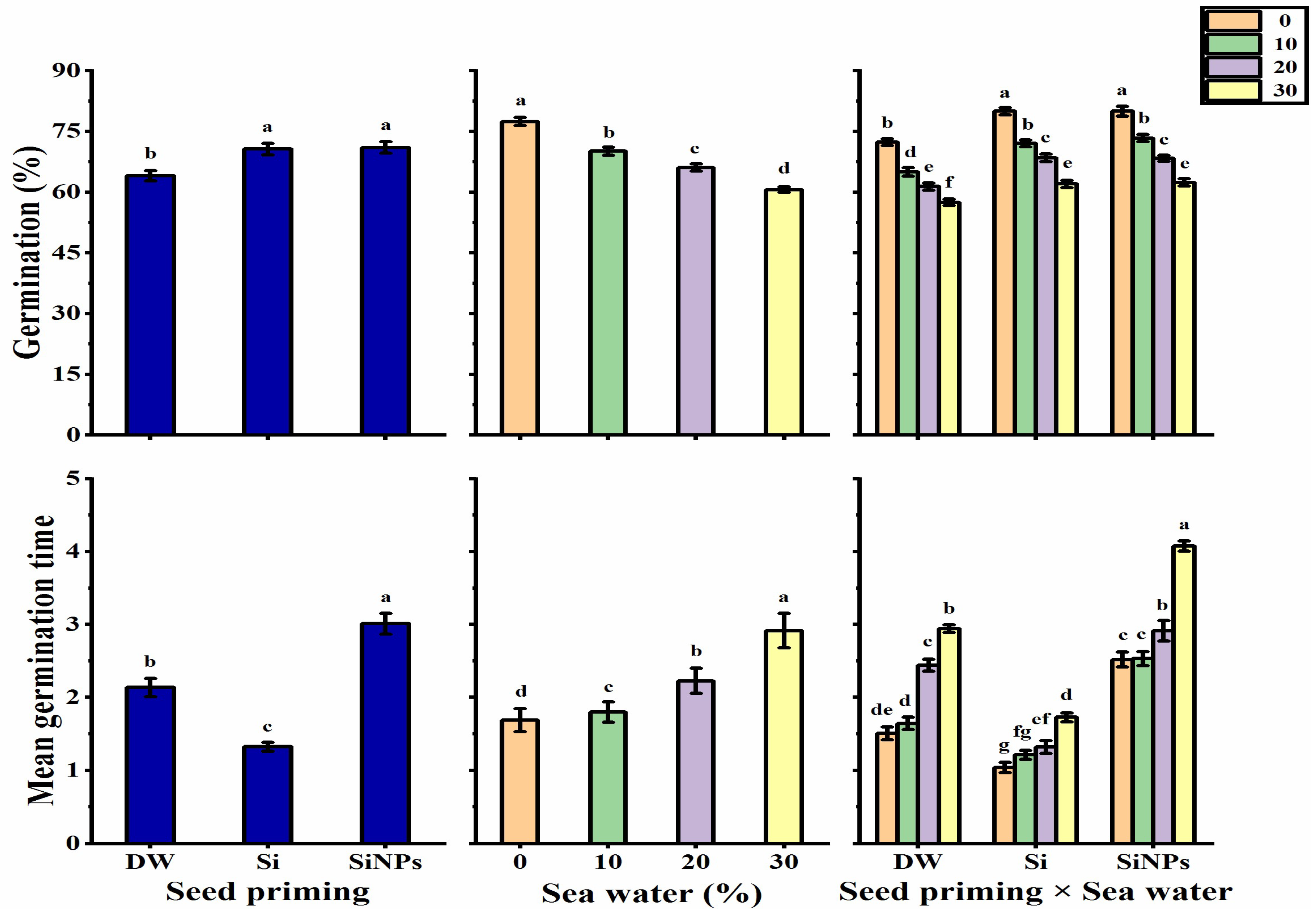
2.1. Germination Characteristics
2.2. Plant Growth
2.3. Leaf Water Status
2.4. Photosynthetic Pigments
2.5. Biochemical Parameters
2.5.1. Total Carbohydrates
2.5.2. Proline Content
2.5.3. Total Phenols and Flavonoids Contents
2.5.4. Oxidative Damage Induced
2.5.5. Antioxidant Enzyme Activities
2.6. Ion Contents
3. Discussion
4. Materials and Methods
4.1. Location and Plant Materials
4.2. Seawater Source and Chemicals
4.3. Experiment Layout
4.4. Germination and Growth Characteristics
4.5. Leaf Water Status
4.6. Photosynthetic Pigments
4.7. Biochemical Parameters
4.7.1. Total Carbohydrates
4.7.2. Proline Content
4.7.3. Phenols and Flavonoids Determination
4.7.4. Lipid Peroxidation Estimation
4.7.5. H2O2 Content
4.7.6. Antioxidant Enzyme Activities
4.7.7. Ion Estimation
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Al-Gretawee, H.; Rayburg, S.; Neave, M. The cooling effect of a medium sized park on an urban environment. Int. J. GEOMATE 2016, 11, 2541–2546. [Google Scholar] [CrossRef]
- Gunawardena, K.R.; Wells, M.J.; Kershaw, T. Utilising green and bluespace to mitigate urban heat island intensity. Sci. Total Environ. 2017, 584–585, 1040–1055. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). Urban Green Space Interventions and Health: A Review of Impacts and Effectiveness; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2017; Available online: http://www.euro.who.int/__data/assets/pdf_file/0010/337690/FULL-REPORT-for-LLP.pdf?ua=1 (accessed on 15 May 2017).
- Zupancic, T.; Westmacott, C.; Bulthuis, M. The Impact of Green Space on Heat and Air Pollution in Urban Communities: A Meta-Narrative Systematic Review; David Suzuki Foundation: Vancouver, Canada, 2015. [Google Scholar]
- Lovasi, G.S.; Quinn, J.W.; Neckerman, K.M.; Perzanowski, M.S.; Rundle, A. Children living in areas with more street trees have lower prevalence of asthma. J. Epidemiol. Community Health 2008, 62, 647–649. [Google Scholar] [CrossRef] [Green Version]
- Hanski, I.; von Hertzen, L.; Fyhrquist, N.; Koskinen, K.; Torppa, K.; Laatikainen, T.; Karisola, P.; Auvinen, P.; Paulin, L.; Mäkelä, M.; et al. Environmental biodiversity, human microbiota, and allergy are interrelated. Proc. Natl. Acad. Sci. USA 2012, 109, 8334–8339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jordan, L.A.; Davitt, D.A.; Morris, R.L.; Neuman, D.S. Foliar damage to ornamental trees sprinkler-irrigated with reuse water. Irrig. Sci. 2001, 21, 17–25. [Google Scholar]
- Wu, L.; Guo, X.; Harivandi, A. Salt tolerance and salt accumulation of landscape plants irrigated by sprinkler and drip irrigation systems. J. Plant Nutr. 2001, 24, 1473–1490. [Google Scholar] [CrossRef]
- Fox, L.J.; Grose, J.N.; Appleton, B.L.; Donohue, S.J. Evaluation of treated effluent as an irrigation source for landscape plants. J. Environ. Hortic. 2005, 23, 174–178. [Google Scholar] [CrossRef]
- Niu, G.; Cabrera, R.I. Growth and physiological responses of landscape plants to saline water irrigation: A review. HortScience 2010, 45, 1605–1609. [Google Scholar] [CrossRef]
- Bradford, K.J. Manipulation of seed water relations via osmotic priming to improve germination under stress conditions. HortScience 1986, 21, 1105–1112. [Google Scholar]
- McDonald, M.B. Seed priming. In Seed Technology and Its Biological Basis; Black, M., Bewley, J.D., Eds.; CRC Press LLC: Boca Raton, FL, USA, 2000; pp. 287–325. [Google Scholar]
- Ghassemi-Golezani, K.; Aliloo, A.A.; Valizadeh, M.; Moghaddam, M. Effects of hydro and osmo-priming on seed germination and field emergence of lentil (Lens culinaris Medik.). Not. Bot. Hort. Agrobot. Cluj 2008, 36, 29–33. [Google Scholar]
- Esmaielpour, B.; Ghassemi-Golezani, K.; Khoei, F.R.; Gregoorian, V.; Toorchi, M. The effect of NaCl priming on cucumber seed-ling growth under salinity stress. J. Food Agric. Environ. 2006, 4, 347–349. [Google Scholar]
- Sivritepe, N.; Sivritepe, H.O.; Eris, A. The effects of NaCl priming on salt tolerance in melon seedlings grown under saline con-ditions. Sci. Hortic. 2003, 97, 229–237. [Google Scholar] [CrossRef]
- Hameed, A.; Afzal, I.; Iqbal, N. Seed priming and salinity induced variations in wheat (Triticum aestivum L.) leaf protein profile. Seed Sci. Technol. 2010, 38, 236–241. [Google Scholar] [CrossRef]
- Hameed, A.; Sheikh, M.A.; Jamil, A.; Basra, S.M.A. Seed priming with sodium silicate enhances seed germination and seedling growth in wheat (Triticum aestivum L.) under water deficit stress induced by polyethylene glycol. Pak. J. Life Soc. Sci. 2013, 11, 19–24. [Google Scholar]
- Bao-Shan, L.; Chun-hui, L.; Li-jun, F.; Shu-chun, Q.; Min, Y. Effect of TMS (nanostructured silicon dioxide) on growth of Changbai larch seedlings. J. For. Res. 2004, 15, 138–140. [Google Scholar] [CrossRef]
- Almeida, D.M.; Oliveira, M.M.; Saibo, N.J. Regulation of Na+ and K+ homeostasis in plants: Towards improved salt stress tolerance in crop plants. Genet. Mol. Biol. 2017, 40, 326–345. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; Gong, H. Beneficial effects of silicon on salt and drought tolerance in plants. Agron. Sustain. Dev. 2014, 34, 455–472. [Google Scholar] [CrossRef] [Green Version]
- Rizwan, M.; Ali, S.; Ibrahim, M.; Farid, M.; Adrees, M.; Bharwana, S.A.; Zia-Ur-Rehman, M.; Qayyum, M.F.; Abbas, F. Mechanisms of silicon-mediated alleviation of drought and salt stress in plants: A review. Environ. Sci. Pollut. Res. 2015, 22, 15416–15431. [Google Scholar] [CrossRef] [PubMed]
- Seal, P.; Das, P.; Biswas, A.K. Versatile potentiality of silicon in mitigation of biotic and abiotic stresses in plants: A review. Am. J. Plant Sci. 2018, 9, 1433–1454. [Google Scholar] [CrossRef] [Green Version]
- Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alamri, S.A.; Alam, P.; Ashraf, M.; Ahmad, P. Potential of exogenously sourced kinetin in protecting Solanum lycopersicum from NaCl-induced oxidative stress through up-regulation of the antioxidant system, ascorbate-glutathione cycle and glyoxalase system. PLoS ONE 2018, 13, e0202175. [Google Scholar] [CrossRef]
- Ahmad, P.; Ahanger, M.A.; Alam, P.; Alyemeni, M.N.; Wijaya, L.; Ali, S.; Ashraf, M. Silicon (Si) Supplementation Alleviates NaCl Toxicity in Mung Bean [Vigna radiata (L.) Wilczek] Through the modifications of physio-biochemical attributes and key antioxidant enzymes. J. Plant Growth Regul. 2019, 38, 70–82. [Google Scholar] [CrossRef]
- Sabaghnia, N.; Janmohammadi, M. Effect of nano-silicon particles application on salinity tolerance in early growth of some lentil genotypes. Ann. Univ. Mariae Curie-Sklodowska Sectio C Biol. 2014, 69, 39–55. [Google Scholar]
- Ashraf, M.; Foolad, M.R. Pre-sowing seed treatment. A shotgun approach to improve germination, plant growth, and crop yield under saline and non-saline conditions. Adv. Agronomy 2005, 88, 223–271. [Google Scholar]
- Eslami, V.; Behdani, M.A.; Ali, S. Effect of salinity on germination and early seedling growth of canola cultivars. Environ. Stress Agric. Sci. 2009, 1, 39–46. [Google Scholar]
- Debez, A.; Chaibi, W.; Bouzid, S. Effect of NaCl and growth regulators on germination of Atriplex halimus L. Cah. Agric. 2001, 10, 135–138. [Google Scholar]
- Zhang, X.H.; Zhou, D.; Cui, J.J.; Ma, H.L.; Lang, D.Y.; Wu, X.L.; Wang, Z.S.; Qiu, H.Y.; Li, M. Effect of silicon on seed germi-nation and the physiological characteristics of Glycyrrhiza uralensis under different levels of salinity. J. Hortic. Sci. Biotechnol. 2015, 90, 439–443. [Google Scholar] [CrossRef]
- Santos, C.V. Regulation of chlorophyll biosynthesis and degradation by salt stress in sunflower leaves. Sci. Hortic. 2004, 103, 93–99. [Google Scholar] [CrossRef]
- Al-Ashkar, I.; Alderfasi, A.; Ben Romdhane, W.; Seleiman, M.F.; El-Said, R.A.; Al-Doss, A. Morphological and genetic diversity within salt tolerance detection in eighteen wheat genotypes. Plants 2020, 9, 287. [Google Scholar] [CrossRef] [Green Version]
- Al-Yasi, H.; Attia, H.; Alamer, K.; Hassan, F.; Ali, E.; Elshazly, S.; Siddique, K.H.M.; Hessini, K. Impact of drought on growth, photosynthesis, osmotic adjustment, and cell wall elasticity in Damask rose. Plant Physiol. Biochem. 2020, 150, 133–139. [Google Scholar] [CrossRef]
- Attia, H.; Al-Yasi, H.; Alamer, K.; Esmat, F.; Hassan, F.; Elshazly, S.; Hessini, K. Induced anti-oxidation efficiency and others by salt stress in Rosa damascena Miller. Sci. Hortic. 2020, 274, 109681. [Google Scholar] [CrossRef]
- Siddiqui, M.H.; Al-Whaibi, M.H. Role of nano-SiO2 in germination of tomato (Lycopersicum esculentum seeds Mill.). Saudi J. Biol. Sci. 2014, 21, 13–17. [Google Scholar] [CrossRef] [Green Version]
- Pesaran, A.A. Moisture Transport in Silica Gel Particle Beds. Ph.D. Thesis, University of California, Los Angeles, CA, USA, 1983. [Google Scholar]
- Zhu, Z.; Wei, G.; Li, J.; Qian, Q.; Yu, J. Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci. 2004, 167, 527–533. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [Green Version]
- Hossain, M.T.; Mori, R.; Soga, K.; Wakabayashi, K.; Kamisaka, S.; Fujii, S.; Yamamoto, R.; Hoson, T. Growth promotion and an increase in cell wall extensibility by silicon in rice and some other poaceae seedlings. J. Plant Res. 2002, 115, 23–27. [Google Scholar] [CrossRef]
- El-Serafy, R.S. Effect of Silicon and Calcium on Productivity and Flower Quality of Carnation. Ph.D. Thesis, Tanta University, Tanta, Eqypt, 2015. [Google Scholar]
- Wang, J.; Naser, N. Improved performance of carbon paste amperometric biosensors through the incorporation of fumed silica. Electroanalytical 1994, 6, 571–575. [Google Scholar] [CrossRef]
- Meng, Y.; Yin, Q.; Yan, Z.; Wang, Y.; Niu, J.; Zhang, J.; Fan, K. Exogenous silicon enhanced salt resistance by maintaining K+/Na+ homeostasis and antioxidant performance in Alfalfa leaves. Front. Plant Sci. 2020, 11, 11. [Google Scholar] [CrossRef]
- Al-Ashkar, I.; Romdhane, W.B.; El-Said, R.A.; Ghazy, A.; Attia, K.; Al-Doss, A. Agro-physiologic responses and stress-related gene expression of four doubled haploid wheat lines under salinity stress conditions. Biology 2021, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Berni, R.; Hausman, J.-F.; Guerriero, G. A review on the beneficial role of silicon against salinity in non-accumulator crops: Tomato as a model. Biomolecules 2020, 10, 1284. [Google Scholar] [CrossRef] [PubMed]
- Romero-Aranda, M.R.; Jurado, O.; Cuartero, J. Silicon alleviates the deleterious salt effect on tomato plant growth by improving plant water status. J. Plant Physiol. 2006, 163, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Yin, L.; Wang, S.; Zhang, M.; Deng, X.; Zhang, S.; Tanaka, K. Enhanced root hydraulic conductance by aquaporin reg-ulation accounts for silicon alleviated salt-induced osmotic stress in Sorghum bicolor L. Environ. Exp. Bot. 2015, 111, 42–51. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Sairam, R.K.; Tyagi, A. Physiology and molecular biology of salinity stress tolerance in plants. Curr. Sci. India 2004, 86, 407–421. [Google Scholar]
- Kishor, P.K.; Sangam, S.; Amrutha, R.N.; Laxmi, P.S.; Naidu, K.R.; Rao, K.R.S.S.; Rao, S.; Reddy, K.J.; Sreenivasulu, N. Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: Its implications in plant growth and abiotic stress tolerance. Curr. Sci. India 2005, 88, 424–438. [Google Scholar]
- Khan, A.; Khan, A.L.; Muneer, S.; Kim, Y.-H.; Al-Rawahi, A.; Al-Harrasi, A. Silicon and salinity: Crosstalk in crop-mediated stress tolerance mechanisms. Front. Plant Sci. 2019, 10, 1429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berni, R.; Luyckx, M.; Xu, X.; Legay, S.; Sergeant, K.; Hausman, J.-F.; Lutts, S.; Cai, G.; Guerriero, G. Reactive oxygen species and heavy metal stress in plants: Impact on the cell wall and secondary metabolism. Environ. Exp. Bot. 2019, 161, 98–106. [Google Scholar] [CrossRef]
- Abdel-Hameed, E.-S.S. Total phenolic contents and free radical scavenging activity of certain Egyptian Ficus species leaf samples. Food Chem. 2009, 114, 1271–1277. [Google Scholar] [CrossRef]
- Di Meo, F.; Lemaur, V.; Cornil, J.; Lazzaroni, R.; Duroux, J.-L.; Olivier, Y.; Trouillas, P. Free radical scavenging by natural polyphenols: Atom versus electron transfer. J. Phys. Chem. A 2013, 117, 2082–2092. [Google Scholar] [CrossRef] [PubMed]
- Li, W.B.; Shi, X.H.; Wang, H.; Zhang, F.S. Effects of silicon on rice leaves resistance to ultraviolet-B. Acta Bot. Sin. 2004, 46, 691–697. [Google Scholar]
- El-Serafy, R.S. Silica Nanoparticles enhances physio-biochemical characters and postharvest quality of Rosa hybrida L. cut flowers. J. Hortic. Res. 2019, 27, 47–54. [Google Scholar] [CrossRef] [Green Version]
- AAl-Ashkar, I.; Alderfasi, A.; El-Hendawy, S.; Al-Suhaibani, N.; El-Kafafi, S.; Seleiman, M.F. Detecting salt tolerance in doubled haploid wheat lines. Agronomy 2019, 9, 211. [Google Scholar] [CrossRef] [Green Version]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef]
- Coskun, D.; Britto, D.T.; Huynh, W.Q.; Kronzucker, H.J. The Role of silicon in higher plants under salinity and drought stress. Front. Plant Sci. 2016, 7, 1072. [Google Scholar] [CrossRef] [Green Version]
- Guo, Q.; Liu, L.; Barkla, B.J. Membrane Lipid Remodeling in Response to Salinity. Int. J. Mol. Sci. 2019, 20, 4264. [Google Scholar] [CrossRef] [Green Version]
- Hamayun, M.; Sohn, E.-Y.; Khan, S.A.; Shinwari, Z.K.; Khan, A.L.; Lee, I.-J. Silicon alleviates the adverse effects of salinity and drought stress on growth and endogenous plant growth hormones of soybean (Glycine max L.). Pak. J. Bot. 2010, 42, 1713–1722. [Google Scholar]
- Himabindu, Y.; Chakradhar, T.; Reddy, M.C.; Kanygin, A.; Redding, K.E.; Chandrasekhar, T. Salt-tolerant genes from halo-phytes are potential key players of salt tolerance in glycophytes. Environ. Exp. Bot. 2016, 124, 39–63. [Google Scholar] [CrossRef] [Green Version]
- Azooz, M.M.; Metwally, A.; Abou-Elhamd, M.F. Jasmonate-induced tolerance of Hassawi okra seedlings to salinity in brackish water. Acta Physiol. Plant. 2015, 37, 77. [Google Scholar] [CrossRef]
- Yan, G.; Fan, X.; Peng, M.; Yin, C.; Xiao, Z.; Liang, Y. Silicon improves rice salinity resistance by alleviating ionic toxicity and osmotic constraint in an organ-specific pattern. Front. Plant Sci. 2020, 11, 260. [Google Scholar] [CrossRef] [PubMed]
- Soleimannejad, Z.; Abdolzadeh, A.; Sadeghipour, H.R. Beneficial effects of silicon application in alleviating salinity stress in halophytic puccinellia distans plants. Silicon 2019, 11, 1001–1010. [Google Scholar] [CrossRef]
- Flam-Shepherd, R.; Huynh, W.Q.; Coskun, D.; Hamam, A.M.; Britto, D.T.; Kronzucker, H.J. Membrane fluxes, bypass flows, and sodium stress in rice: The influence of silicon. J. Exp. Bot. 2018, 69, 1679–1692. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.H.; Roberts, E.H. The quantification of aging and survival in orthodox seeds. Seed Sci. Technol. 1981, 9, 373–409. [Google Scholar]
- Clarke, J.M.; McCaig, T.N. Excised-leaf water retention capability as an indicator of drought resistance of triticum genotypes. Can. J. Plant Sci. 1982, 62, 571–578. [Google Scholar] [CrossRef]
- Barrs, H.D. Determination of water deficits in plant tissues. Water Defic. Plant Growth 1968, 1, 235–368. [Google Scholar]
- El-Serafy, R.S. Phenotypic plasticity, biomass allocation, and biochemical analysis of cordyline seedlings in response to oligo-chitosan foliar spray. J. Soil Sci. Plant Nutr. 2020, 20, 1503–1514. [Google Scholar] [CrossRef]
- Dere, Ş.; Güneş, T.; Sivaci, R. Spectrophotometric determination of chlorophyll-A, B and total carotenoid contents of some algae species using different solvents. Tr. J. Botany 1998, 22, 13–17. [Google Scholar]
- Weinmann, H. Determination of total available carbohydrates in plants. Plant Physiol. 1947, 22, 279–290. [Google Scholar] [CrossRef] [Green Version]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- El-Serafy, R.S.; El-Sheshtawy, A.A. Effect of nitrogen fixing bacteria and moringa leaf extract on fruit yield, estragole content and total phenols of organic fennel. Sci. Hortic. 2020, 265, 109209. [Google Scholar] [CrossRef]
- Boateng, J.; Verghese, M.; Walker, L.T.; Ogutu, S. Effect of processing on antioxidant contents in selected dry beans (Phaseolus spp. L.). LWT 2008, 41, 1541–1547. [Google Scholar] [CrossRef]
- Talukdar, D. Flavonoid-Deficient Mutants in Grass Pea (Lathyrus sativus L.): Genetic control, linkage relationships, and mapping with aconitase and s-nitrosoglutathione reductase isozyme loci. Sci. World J. 2012, 2012, 345983. [Google Scholar] [CrossRef] [Green Version]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Patterson, B.D.; Macrae, E.A.; Ferguson, I.B. Estimation of hydrogen peroxide in plant extracts using titanium (IV). Anal. Biochem. 1984, 139, 487–492. [Google Scholar] [CrossRef]
- Murkherje, S.; Choudhuri, M. Implication of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogenperoxide in Vigna seedlings. Physiol. Plant 1983, 58, 166–170. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen Peroxide is Scavenged by Ascorbate-specific Peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Snyder, G.H. Methods for silicon analysis in plants, soils, and fertilizers. In Studies in Plant Science; Elsevier: Amsterdam, The Netherlands, 2001; Volume 8, pp. 185–196. [Google Scholar]
- Deal, S.B. Flame photometric determination of sodium and potassium in zinc cadmium sulfide phosphors. Anal. Chem. 1954, 26, 598–599. [Google Scholar] [CrossRef]
- Waller, R.A.; Duncan, D.B. A bayes rule for the symmetric multiple comparisons problem. J. Am. Stat. Associ. 1969, 64, 1484–1503. [Google Scholar]









| Component | EC (dS/m) | pH | HCO3 (meq/L) | CL (meq/L) | SO4−2 (meq/L) | Ca+2 (meq/L) | Mg+2 (meq/L) | Na+ (meq/L) | K+ (meq/L) |
|---|---|---|---|---|---|---|---|---|---|
| Concentration | 40.51 | 6.81 | 5.59 | 415 | 74.35 | 42.10 | 14.57 | 435.35 | 1.34 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Serafy, R.S.; El-Sheshtawy, A.-N.A.; Atteya, A.K.G.; Al-Hashimi, A.; Abbasi, A.M.; Al-Ashkar, I. Seed Priming with Silicon as a Potential to Increase Salt Stress Tolerance in Lathyrus odoratus. Plants 2021, 10, 2140. https://doi.org/10.3390/plants10102140
El-Serafy RS, El-Sheshtawy A-NA, Atteya AKG, Al-Hashimi A, Abbasi AM, Al-Ashkar I. Seed Priming with Silicon as a Potential to Increase Salt Stress Tolerance in Lathyrus odoratus. Plants. 2021; 10(10):2140. https://doi.org/10.3390/plants10102140
Chicago/Turabian StyleEl-Serafy, Rasha S., Abdel-Nasser A. El-Sheshtawy, Amira K.G. Atteya, Abdulrahman Al-Hashimi, Arshad Mehmood Abbasi, and Ibrahim Al-Ashkar. 2021. "Seed Priming with Silicon as a Potential to Increase Salt Stress Tolerance in Lathyrus odoratus" Plants 10, no. 10: 2140. https://doi.org/10.3390/plants10102140






