Abstract
A novel bis-iodurated polyether compound, based on an unprecedented tetra-THF backbone, has been isolated as a trace by-product of the oxidation of squalene with the catalytic system RuO2(cat.)/NaIO4. The double erythro configuration of the central portion of the molecule furnishes the first indirect support of the previously postulated pathway operating in the oxidative pentacyclization of the isoprenoid substrate. A bidirectional double oxidative bis-cyclization is invoked to explain the formation of this compound. The isolated substance was successfully subjected to a double rearrangement-ring expansion to give a novel bis-THF-bis-THP compound.
1. Introduction
Some years ago we discovered a novel cascade process catalysed by RuO4 generated in situ by the action of NaIO4 on RuO2, the pre-catalytic species employed to generate RuO4 [1]. This is a unique process by which a poly-THF backbone, made up of adjacently linked THF rings, can be built-up in a single step and in a stereoselective manner starting from polyenes characterized by a repetitive 1,5-diene structural motif [2,3,4,5,6,7,8]. In particular, oxidation of squalene gives rise to penta-THF compound 1 (Scheme 1) containing ten stereogenic centres. Previous studies carried out in our group had suggested that steric and chelation control factors concur to determine the stereochemical outcome of the process [6].
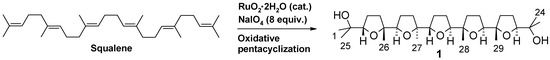
Scheme 1.
Stereoselective synthesis of a pentacyclic poly-THF (1) by RuO4-catalysed oxidative polycyclization of squalene.
Scheme 1.
Stereoselective synthesis of a pentacyclic poly-THF (1) by RuO4-catalysed oxidative polycyclization of squalene.
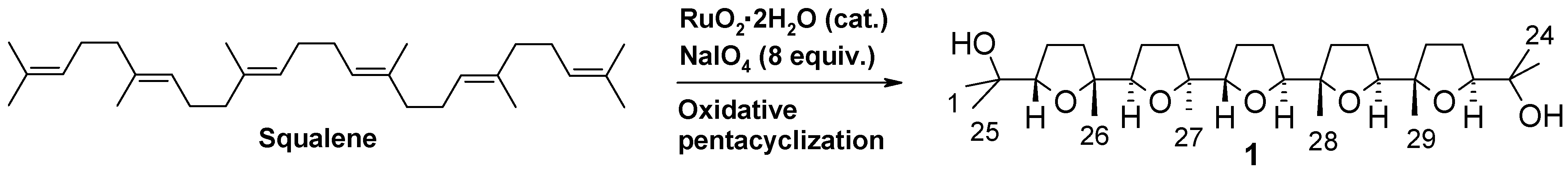
In a more recent investigation, the use of different cyclization conditions led to a different stereochemistry of the process [9]. In particular, four new C30 isomeric heptacyclic polyether substances (compounds 2-5, Scheme 2) were obtained through a unique seven-step cascade process featuring a pentacyclization of squalene followed by a double oxidative spiroketalization at the two bis-THF termini of the first-formed penta-THF intermediates. Careful HPLC isolation of latter substances for X-ray studies [9] allowed also the isolation of tetra-THF 6 (Scheme 2), a very minor side-product of the process, possessing a CS-symmetric structure embodying two terminal cis-threo-trans bis-THF moieties connected by a central bis-iodurated tetracarbonious segment. The determination of the stereostructure of compound 6, the mechanistic implication of its isolation as well as its double rearrangement-ring expansion to a new bis-THP polyether compound, are discussed in the present paper.
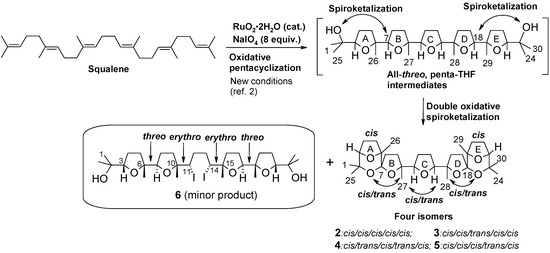
Scheme 2.
Synthesis of polycyclic polyethers by one-step RuO4-catalysed oxidative polycyclization/oxidative spiroketalization of squalene.
Scheme 2.
Synthesis of polycyclic polyethers by one-step RuO4-catalysed oxidative polycyclization/oxidative spiroketalization of squalene.
2. Results and Discussion
2.1. Chemistry
The structure of compound 6 was determined by X-ray diffraction analysis carried out on a single crystal of the substance obtained by slow evaporation of a chloroform solution. The most interesting, and unusual feature of this compound is the double erythro configuration around the C10/C11 and C14/C15 bonds. In fact, previous studies from our group had demonstrated that the oxidation of both linear and isoprenoid polyenes constantly furnishes poly-THF compounds possessing threo inter-THF relationships. This is consistent with the syn addition (a [3+2] cycloaddition) of a O=Ru-O portion across each involved C-C double bond, along the all-trans polyene chain (Scheme 3), that also agrees with mechanistic proposals for related oxidative mono-cyclization of 1,5-dienes catalysed by RuO4 [10,11,12,13,14,15] and related oxo-species OsO4 [16,17,18] and MnO4- [19,20,21,22,23,24,25,26,27], as well as rhenium (VII)-mediated oxidative polycyclization of hydroxypolyenes [28,29,30,31].
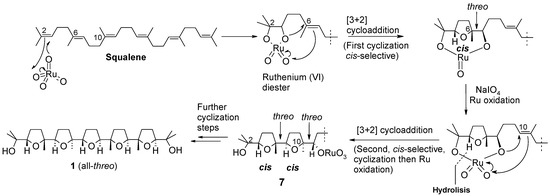
Scheme 3.
Ru-catalysed cascade sequence leading to penta-THF 1.
Scheme 3.
Ru-catalysed cascade sequence leading to penta-THF 1.
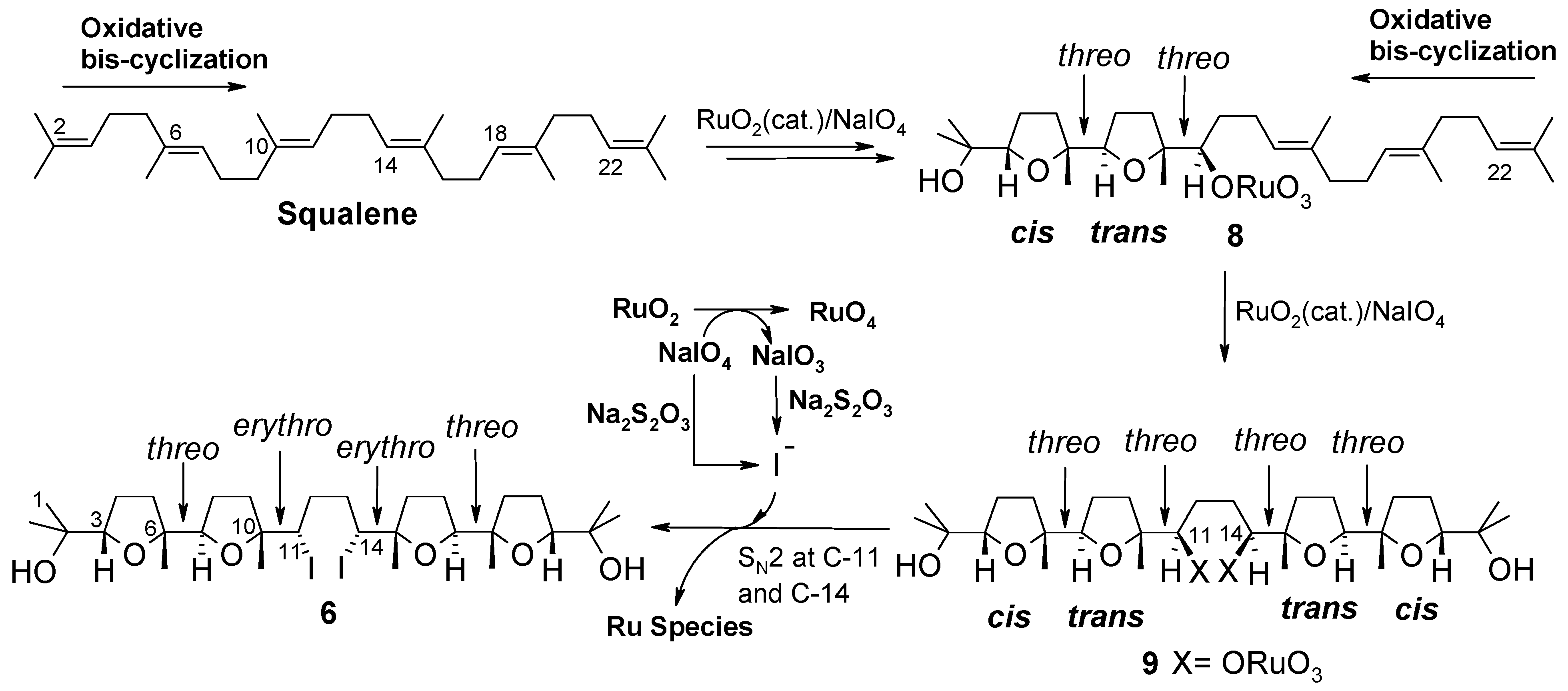
Based on these precedents, formation of 6 was intriguing and could be rationalised through a double cis/trans-selective oxidative bis-cyclization process (Scheme 4). Each bis-cyclization event involves three consecutive double bonds of the polyene chain starting from the terminal ones. In particular, attack of RuO4 to the Δ2 double bond induces two successive cyclization steps giving rise to bis-THF intermediate 8, in the same manner as shown for the synthesis of 1 (see intermediate 7, Scheme 3). A second bis-cyclization would then occur at the other side of the molecule by attack of RuO4 at the terminal, Δ22, double bond to give the all-threo tetra-THF 9 still possessing two oxoruthenate appendages linked to C-11 and C-14. It can be presumed that a double substitution of the ruthenium-containing portions, with inversion of configuration at involved carbon centres, would then occur during the reductive quenching of the process, by iodide ions probably generated in situ by the action of tiosulphate on iodate in turn produced during the oxidation of RuO2 to RuO4. It cannot be excluded that iodide could originate from reduction of periodate itself not completely consumed in the reaction medium. It is probable that such a side-process could be due to the higher concentration of the reaction medium in the new experimented conditions [9].
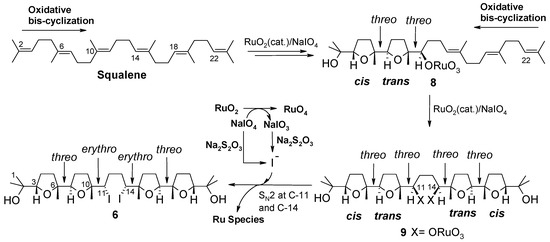
Scheme 4.
A plausible path for the formation of tetra-THF 6.
Scheme 4.
A plausible path for the formation of tetra-THF 6.
In order to enlarge the range of polyether substances accessible through the Ru-mediated polycyclization process we began an exploration of the possible post-synthetic modifications of some of the poly-THF backbones obtained through the above process. We have previously demonstrated that progressive structural simplification of compound 1 to small-sized poly-THF compounds can be achieved via an iterative PCC-mediated oxidative cleavage/reduction [7] sequence. The entire process is possible due to the two alcohol functionalities adjacent to the terminal THF rings that are prone to be intercepted by PCC [32]. In addition, a new type of cytotoxic spyroketal poly-THF compound, strictly related to bis-spiroketals 2-5, could be accessed through a PCC-mediated oxidative spiroketalization process starting from 1 [3]. In a more recent study we have also shown that the same oxidant, or the related system PCC-H5IO6, is able to attack the angular CH position of the THF ring in various mono and poly-THF substrates leading to either the oxidative opening of the THF ring or the oxidative cleavage of suitable inter-THF bonds [33].
As a continuation of this project, we envisaged that compound 6, possessing a central bis-(α-iodo-THF) portion, could be a good model compound to probe a double rearrangement-ring expansion process involving the two internal THF rings as a means to access a new type of mixed THF-THP polyether compounds further functionalised for successive synthetic manipulations providing access to new polyether polycyclic materials. This type of reaction has previously been carried out on substances containing a single α-iodo-THF subunit [34] but has never been attempted on a substrate containing two α-iodo-THF moieties and, in particular, as far as we know, the double rearrangement-ring expansion of a bis-THF substance has never been accomplished. Related chemistry has been successfully employed for example in the synthesis of salynomicin [25] as well as in the synthesis of a bis-oxepane portion of hemibrevetoxin [35]. Pleasingly, when compound 6 was reacted with excess Ag2CO3 (5 equiv.) in acetone/water (8:2, 40 °C, 4 h), compound 10 was obtained in a 65% yield demonstrating the feasibility of the projected transformation (Scheme 5).
Proof for the structure 10 was gained by chemical and high-field 2D-NMR evidence. Attempted acetylation and benzoylation under standard conditions only delivered unreacted 10 indicating, as expected, the presence of tertiary hydroxyl groups in this compound.
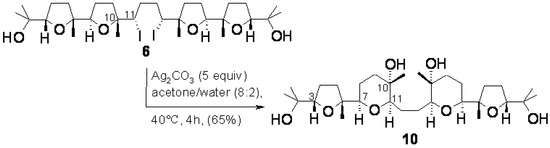
Scheme 5.
Double rearrangement-ring expansion of compound 6.
Scheme 5.
Double rearrangement-ring expansion of compound 6.
A 1H1H-COSY experiment at 700 MHz indicated the presence in 10 of the two five-proton spin systems H-3/H2-4/H2-5 and H-7/H2-8-H2-9 belonging to the two adjacent rings as well as the H-11/H2-12 spin system. Assignment of each of these spin systems to the proper ring came from considerations of spectral data and comparison with strictly related THF- and THP-containing substances. In particular, the signals resonating at δ 3.86 and 2.36 were assigned, respectively, to the angular THF proton (H-3) and to the Hα-5 proton based on the good agreement of their chemical shift values with those typically exhibited by these protons in strictly analogous poly-THF substances including the same cis-THF-containing substructure, previously synthesised in our laboratories [1,2,3,4,5,6,7,8]. This deduction suggested that the two higher field one-proton resonances at δ 3.26 (H-7) and 3.16 (H-11) could be ascribable to the angular hydrogens in the THP ring.
The good proton dispersion of the signals in the 1H-NMR spectrum of 10 allowed us to fully analyse some crucial signals. In particular, the presence of a THP ring in 10 was corroborated by J values (J = 12.5, 3.6 Hz) of the H-9 equatorial proton resonating as a clean double triplet at δ 1.89, as expected for an equatorial proton next to a quaternary centre (C-10) in a six-membered ring possessing a chair conformation. In addition, a W coupling observed between the signal at δ 1.54 for Hax-9 and the singlet methyl resonance at δ 1.19, ascribable to the C-10 methyl group, also pointed to the presence of a THP ring and the axial nature of that methyl. W-type long-range couplings were also observed between the singlet resonances at δ 1.23 and 1.05 allowing assignment of these signals to the two methyls belonging to the terminal 2-hydroxyisopropyl group. Similarly, a long-range coupling between the methyl signal at δ 1.12 and the Ha-5 resonance at δ2.36 allowed to assign the former resonance to the angular methyl of the THF ring (C6-Me).
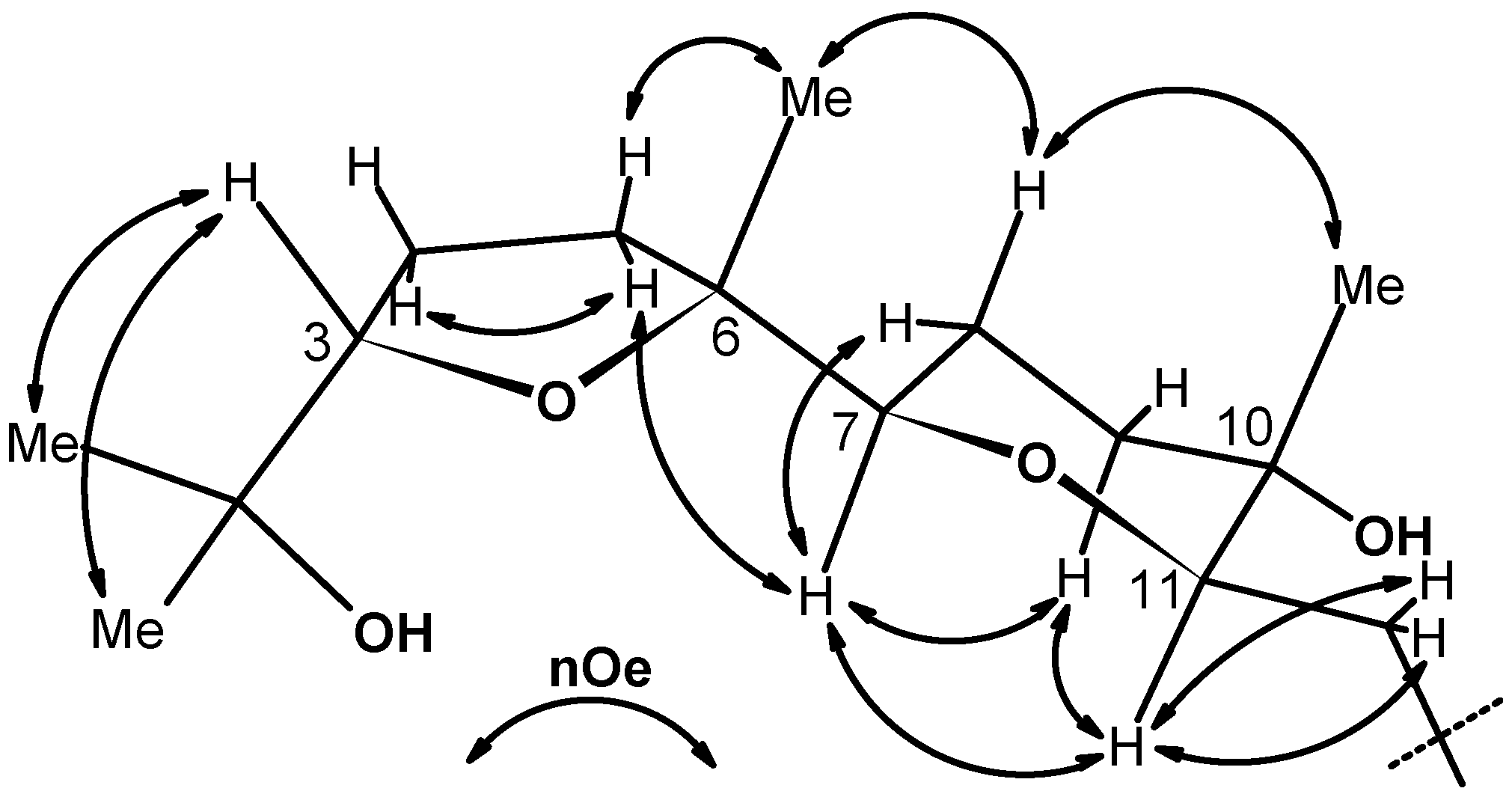
These conclusions were reinforced by data from a very informative 700 MHz NOESY experiment (Figure 1) that also provided conclusive information on the relative configuration of the C-7, C-10 and C-11 centres belonging to the THP ring in 10. In particular, the cis nature of the THP ring was inferred by the presence of a strong correlation peak between signals for the H-7 and H-11 angular protons. Similarly, the axial nature of the C-10 methyl, was further corroborated by a nOe correlation between its resonance at δ 1.19 and that of the Hax-8 proton at δ 1.74. The rest of nOe cross peaks shown in Figure 1 were in full agreement with the given stereostructure.
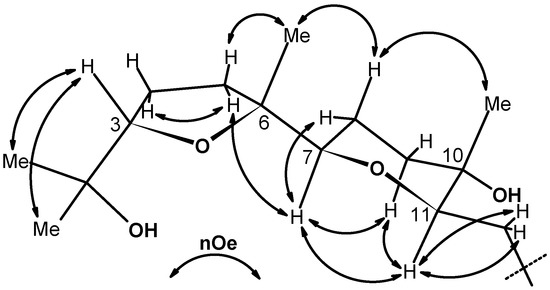
Figure 1.
Summary of some significant 700 MHz NOESY correlations for compound 10 (due to the symmetry, half molecule is shown).
Figure 1.
Summary of some significant 700 MHz NOESY correlations for compound 10 (due to the symmetry, half molecule is shown).
2.2. X-ray crystallography.
Molecules of 6 in the crystals are centrosymmetric (Ci point group) as they lie on crystallographic inversion centres (Figure 2). The molecules have a stretched winding shape, which is due to the double cis-trans sequence of the 2,5-disubstituted THF rings and to the trans-planar conformation of the carbon chain.
The molecular conformation is stabilized by an intramolecular H bonding between O-H donor and the oxygen acceptor of the inner THF ring (O1−H∙∙∙O3 0.983, 2.224, 3.175(9) Å, 163°). Ring puckering coordinates of the inner THF ring are q2 = 0.356(6) Å j2 = 211(1)°, and of the outer are q2 = 0.316(7) Å j2 = 324(1)°. On the basis of the calculated phase angles, it can be argued that both are basically in envelope conformation, with C7 and C4 atoms out of the envelope plane.
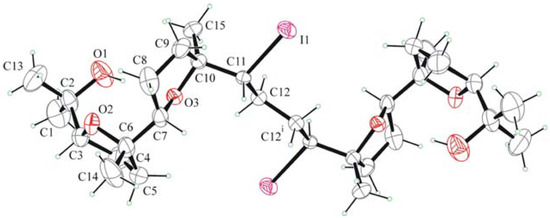
Figure 2.
ORTEP view of 6.
Figure 2.
ORTEP view of 6.
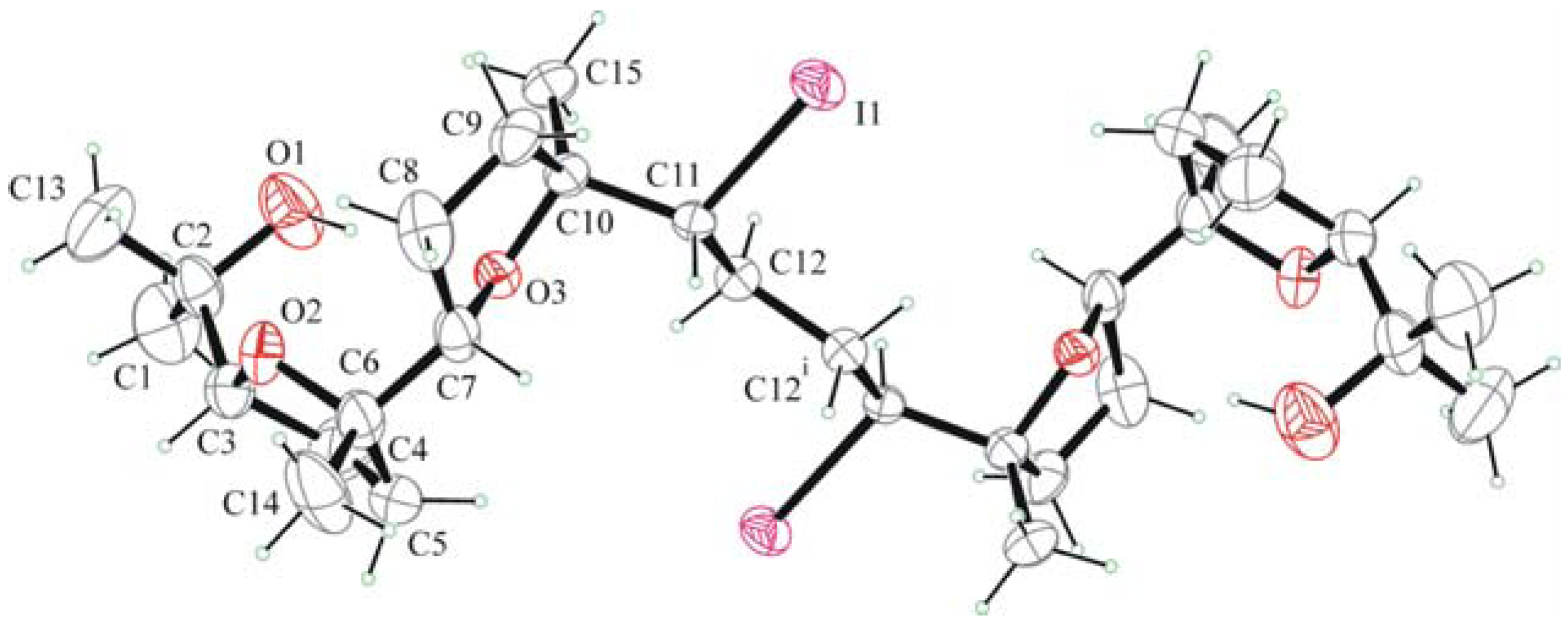
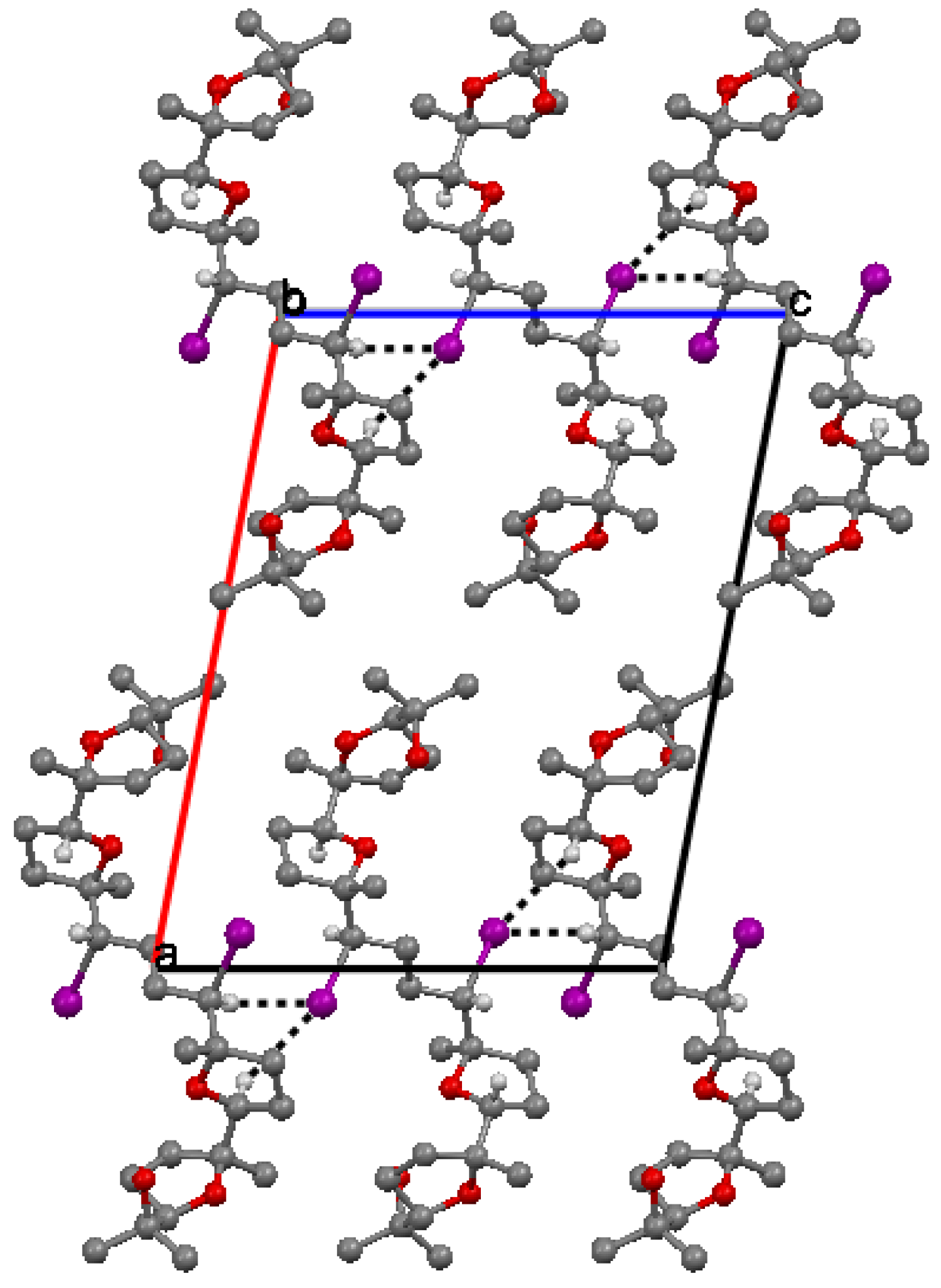
The packing of molecules is accomplished through weak H bonding interactions between iodine atoms as bifurcated acceptors and methyne C-H donors [36]. This is clearly shown in Figure 3. Chains of H-bonded molecules are formed which run along b + c and b – c lattice directions. The weak H-bonding leads to the formation of ring patterns having graph set descriptor 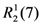 . Along a molecules are stacked in layers through van der Waals contacts.
. Along a molecules are stacked in layers through van der Waals contacts.
 . Along a molecules are stacked in layers through van der Waals contacts.
. Along a molecules are stacked in layers through van der Waals contacts.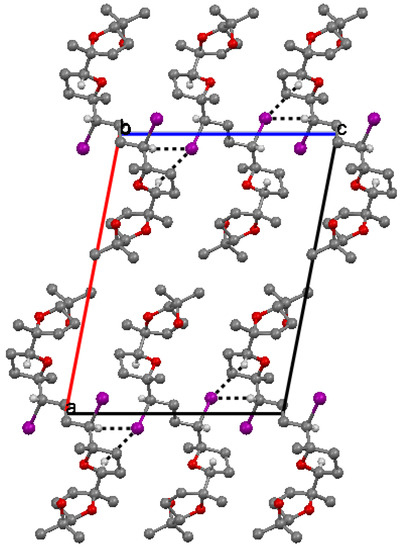
Figure 3.
Crystal packing of 6 viewed down b.
Figure 3.
Crystal packing of 6 viewed down b.
3. Experimental
3.1. General
All reagents were purchased (Aldrich) at the highest commercial quality and used without further purification. Reactions were monitored by thin-layer chromatography carried out on precoated silica gel plates (Merck 60, F254, 0.25 mm thick). Merck silica gel (Kieselgel 40, particle size 0.063-0.200 mm) was used for column chromatography. HPLC separations were carried out on a Varian 2510 apparatus equipped with a Waters R403 dual cell differential refractometer using Phenomenex 250 × 10 mm, Phenomenex 250 × 4.6 mm and Nucleosil 250 × 10 mm columns. NMR experiments were performed on Varian Mercury Plus 400 MHz, Varian Unity Inova 700 MHz and Gemini 200 spectrometers in CDCl3. Proton chemical shifts were referenced to the residual CHCl3 signal (7.26 ppm); 13C-NMR chemical shifts were referenced to the solvent (77.0 ppm). J values are in Hz. Abbreviations for signal coupling are as follows: s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet. IR spectra were collected on a Jasco FT-IR-430 spectrometer. High Resolution MS spectra were recorded on a Bruker APEX II FT-ICR mass spectrometer using the electrospray ionization (ESI) technique in positive mode.
3.2. Synthesis
Squalene (50 g, 122 mmol) was placed into a 5 L round-bottomed flask equipped with a mechanical stirrer and dissolved in the biphasic mixture EtOAc/CH3CN/H2O (3:3:2, 1.6 L). The solution was cooled to 0 °C and NaIO4 (8 equiv., 976 mmol, 209 g) and RuO2•2H2O (20 mol%, 24.4 mmol, 3.25 g) were sequentially added under vigorous stirring. After 30 min excess Na2S2O3•5H2O was added and the mixture was stirred for further 10 min and then filtered through a Buchner funnel. The solid left on the Buchner was thoroughly washed with EtOAc and the resulting biphasic solution was concentrated in vacuo. The aqueous suspension was extracted with EtOAc (3 × 300 mL). The combined organic phase was dried (Na2SO4) and evaporated in vacuo to give an oily product that was chromatographed on silica gel (50 × 8 cm column) eluting with petroleum ether (40-70)/Et2O mixtures (from 7:3 to 100% ether) and then with CHCl3/MeOH mixtures (up to CHCl3/MeOH 8:2) to give three fractions: fraction A (7.40 g) eluted before penta-THF 1; fraction B (4.75 g) containing penta–THF 1 and fraction C (35.18 g) eluted after penta-THF 1. A sample (500 mg) of the less polar fraction A was separated by HPLC (250×10 mm column, eluent: hexane-EtOAc, 8:2, flow 2.5 mL/min) to give previously isolated bis-spiroketals 2-5. The fraction eluted in the range 20-30 min was subjected to a further reversed-phase HPLC separation (250×10 mm column; flow: 1.0 mL/min, eluent: MeOH/H2O, 8:2, tR = 14.5 min) to give pure 2,2'-(5',5''-(1,4-diiodobutane-1,4-diyl)bis(2,5'-dimethyl-octahydro-2,2'-bifuran-5',5-diyl))dipropan-2-ol (6, 2.5 mg, 0.03%). IR (neat): υmax 3706, 3780, 1054, 1013 cm-1; 1H-NMR: (400 MHz, CDCl3) d 4.00 (1H, bd, J = 9.7), 3.91 (1H, m), 3.85 (1H, dd, J = 7.7, 5.2), 2.32 (1H, m), 2.23 (1H, ddd, J = 12.1, 8.7, 8.7), 1.45, 1.25, 1.13, 1.09 (3H each, s’s, 4xMe); 13C-NMR (50 MHz, CDCl3): d 85.6, 85.0, 84.4, 83.0, 71.9, 47.9, 39.9, 36.4, 34.7, 27.9, 27.2, 25.8, 24.9, 24.3, 22.9; HRMS (ESI) m/z calcd for C30H52I2NaO6 [M+Na]+ 785.1751, found 785.1748.
3.3. Ring expansion of 6 to 10
To compound 6 (1.5 mg, 0.02 mmol) dissolved in acetone-water (4:1, 500 μL) was added silver carbonate (16.8 mg, 0.1 mmol) and the mixture stirred at 40 °C. After 4h, the mixture was filtered and the solid thoroughly washed with acetone. The organic phase was taken to dryness to give an oily product. HPLC purification (250 × 4.6 mm column; flow: 1.0 mL/min; CHCl3MeOH, 98:2) gave pure 2,2'-(butane-1,4-diyl)bis(6-(5-(2-hydroxypropan-2-yl)-2-methyl-tetrahydrofuran-2-yl)-3-methyl-
tetrahydro-2H-pyran-3-ol) (10, 0.7 mg, 65%, tR =16.5 min). Oil; IR (neat): υmax 3440 cm-1; 1H-NMR: (700 MHz, CDCl3) d 3.86 (1H, dd, J = 8.4, 3.7), 3.26 (1H, bdd, J = 7.0, 7.0), 3.16 (1H, bd, J = 6.3), 2.36 (1H, ddd, J = 10.0, 10.0, 10.0), 2.05 (1H, dddd, J = 12.7, 9.6, 3.7, 3.7), 2.03-1.94 (2H, m), 1.89 (1H, ddd, J = 12.5, 3.6, 3.6), 1.75 (2H, m), 1.54 (2H, m), 1.31 (1H, m), 1.23, 1.19, 1.12, 1.05 (3H each, s’s, 4 × Me); HRMS (ESI) m/z calcd for C30H54NaO8 [M+Na]+ 565.3716, found 565.3710.
3.4. X-Ray Crystallography
Crystals of 6 suitable for X-ray analysis were obtained from CHCl3 by slow evaporation of the solvent. Data were collected at 298 K on a Bruker-Nonius Kappa-CCD diffractometer using graphite monochromated MoKα radiation (λ = 0.71073 Å). Data reduction and multi-scan absorption correction were done using SADABS program [37]. The structure was solved by direct methods (SIR97 program [38]) and refined by the full matrix least-squares method on F2 using SHELXL-97 program [39] with the aid of the program WinGX [40]. Non-hydrogen atoms were refined anisotropically. H atoms of the hydroxy group was located in difference Fourier maps and refined with Uiso = 1.2∙Ueq of the carrier atom. The positions of the other H atoms were determined stereochemically and refined by the riding model with Uiso = 1.2∙Ueq of the carrier atom (1.5∙Ueq for H atoms of methyl groups). Ring puckering coordinates [41] were determined using the program PARST [42]. The analysis of the crystal packing and the drawing of the molecule were performed using the programs Mercury [43] and ORTEP [44]. Crystal and refinement data are summarized in Table 1. CCDC reference number 821334 contains the supplementary crystallographic data for 6.
4. Conclusion
In conclusion, the isolation of bis-iodocompound 6 was interesting from both a mechanistic and a synthetic point of view. Its existence among the oxidation products of squalene with RuO4 was indicative of the existence of an intermediate species (see 9, Scheme 4), carrying oxoruthenium substituents, likely ORuO3 groups, adjacent to the two internal THF rings, able to undergo a facile nucleophilic displacement, that enforces the mechanistic hypothesis previously put forward to explain the formation of penta-THF 1 from the same substrate (Scheme 3). In addition, the postulated mechanism for the formation of 6 also suggests a new possible use of the RuO4-catalysed polycyclization process where suitable polyenes can be induced to undergo bidirectional poly-THF-forming oxidative sequences. The facile access to a novel type of bis-THF-bis-THP compound (10) has been demonstrated via a double ring-enlargement process. Studies are in progress to further develop the chemistry presented here toward the synthesis of new THP-containing polyether compounds.
Acknowledgments
We are grateful to the “Centro di Metodologie Chimico-Fisiche” and the “Centro di Servizio Interdipartimentale di Analisi Strumentale” (CSIAS) of the University of Napoli “Federico II” for NMR facilities.
References
- Bifulco, G.; Caserta, T.; Gomez-Paloma, L.; Piccialli, V. RuO4-promoted syn-oxidative polycyclization of isoprenoid polyenes: a new stereoselective cascade process. Tetrahedron Lett. 2002, 43, 9265–9269, corrigendum Tetrahedron Lett. 2003, 44, 3429. [Google Scholar] [CrossRef]
- Centore, R.; Tuzi, A.; Zaccaria, S.; Piccialli, V. Synthesis, stereostructure and H-bonding patterns of a tris-THF compound. J. Chem. Crystallogr. [CrossRef]
- Piccialli, V.; Oliviero, G.; Borbone, N.; Tuzi, A.; Centore, R.; Hemminki, A.; Ugolini, M.; Cerullo, V. Discovery of a new PCC-mediated stereoselective oxidative spiroketalization process. An access to a new type of poly-THF spiroketal compound displaying anticancer activity. Org. Biomol. Chem. 2009, 7, 3036–3039. [Google Scholar] [CrossRef]
- Piccialli, V.; Borbone, N.; Oliviero, G. RuO4-catalyzed oxidative polycyclization of the CS-symmetric isoprenoid polyene digeranyl. An unexpected stereochemical outcome. Tetrahedron 2008, 64, 11185–11192. [Google Scholar] [CrossRef]
- Piccialli, V. Oxidative cyclization of dienes and polyenes mediated by transition-metal-oxo species. Synthesis 2007, 17, 2585–2607. [Google Scholar] [CrossRef]
- Piccialli, V.; Caserta, T.; Caruso, L.; Gomez-Paloma, L.; Bifulco, G. RuO4-mediated oxidative polycyclization of linear polyenes. A new approach to the synthesis of the bis-THF diol core of antitumour cis-cis adjacent bis-THF annonaceous acetogenins. Tetrahedron 2006, 62, 10989–11007. [Google Scholar] [CrossRef]
- Caserta, T.; Piccialli, V.; Gomez-Paloma, L.; Bifulco, G. RuO4-catalyzed oxidative polycyclization of squalene. Determination of the configuration of the penta-tetrahydrofuranyl diol product. Tetrahedron 2005, 61, 927–939. [Google Scholar] [CrossRef]
- Bifulco, G.; Caserta, T.; Gomez-Paloma, L.; Piccialli, V. RuO4-promoted oxidative polycyclization of isoprenoid polyenes. A further insight into the stereochemistry of the process. Tetrahedron Lett. 2003, 44, 5499–5503. [Google Scholar]
- Piccialli, V.; Zaccaria, S.; Borbone, N.; Oliviero, G.; D'Errico, S.; Hemminki, A.; Cerullo, V.; Romano, V.; Tuzi, A.; Centore, R. Discovery of a novel one-step RuO4-catalysed tandem oxidative polycyclization/double spiroketalization process. Access to a new type of polyether bis-spiroketal compound displaying antitumour activity. Tetrahedron 2010, 66, 9370–9378. [Google Scholar]
- Carlsen, P.H.J.; Katsuki, T.; Martin, V.S.; Sharpless, K.B. A greatly improved procedure for ruthenium tetroxide catalyzed oxidations of organic compounds. J. Org. Chem. 1981, 46, 3936–3938. [Google Scholar] [CrossRef]
- Piccialli, V.; Cavallo, N. Improved RuO4-catalysed oxidative cyclisation of geraniol-type 1,5-dienes to cis-2,5-bis(hydroxymethyl)tetrahydrofuranyldiols. Tetrahedron Lett. 2001, 42, 4695–4699. [Google Scholar] [CrossRef]
- Albarella, L.; Musumeci, D.; Sica, D. Reactions of 1,5-dienes with ruthenium tetraoxide: Stereoselective synthesis of tetrahydrofurandiols. Eur. J. Org. Chem. 2001, 5, 997–1003. [Google Scholar]
- Roth, S.; Göhler, S.; Cheng, H.; Stark, C.B.W. A highly efficient procedure for ruthenium tetroxide catalyzed oxidative cyclizations of 1,5-dienes. Eur. J. Org. Chem. 2005, 19, 4109–4118. [Google Scholar]
- Göhler, S.; Cheng, H.; Stark, C.B.W. Catalytic diastereo- and positionselective oxidative mono-cyclization of 1,5,9-trienes and polyenes. Org. Biomol. Chem. 2007, 5, 1605–1614. [Google Scholar] [CrossRef]
- Göhler, S.; Roth, S.; Cheng, H.; Göksel, H.; Rupp, A.; Haustedt, L.O.; Stark, C.B.W. Multigram synthesis of diastereomerically pure tetrahydrofuran-diols. Synthesis 2007, 17, 2751–2754. [Google Scholar]
- de Champdoré, M.; Lasalvia, M.; Piccialli, V. OsO4-catalyzed oxidative cyclization of geranyl and neryl acetate to cis-2,5-bis(hydroxymethyl)tetrahydrofurans. Tetrahedron Lett. 1998, 39, 9781–9784. [Google Scholar]
- Donohoe, T.J.; Winter, J.J.G.; Helliwell, M.; Stemp, G. Hydrogen bonding control in the oxidative cyclisation of 1,5-dienes. Tetrahedron Lett. 2001, 42, 971–974. [Google Scholar] [CrossRef]
- Donohoe, T.J; Butterworth, S. A general oxidative cyclization of 1,5-dienes using catalytic osmium tetroxide. Angew. Chem. Int. Ed. 2003, 42, 948–951. [Google Scholar] [CrossRef]
- Klein, E.; Rojahn, W. Oxidation of olefins by potassium permanganate. Oxygen-labeling experiments and mechanism of the oxidation of 1,5-hexadiene. Evidence for a manganese intermediate with coordination number greater than four. Tetrahedron 1965, 21, 2353–2358. [Google Scholar] [CrossRef]
- Baldwin, J.E.; Crossley, M.J.; Lehtonen, E.-M.M. Stereospecificity of oxidative cycloaddition reactions of 1,5-dienes. J. Chem. Soc., Chem. Commun. 1979, 918–920. [Google Scholar]
- Walba, D.M.; Wand, M.D.; Wilkes, M.C. Stereochemistry of the permanganate oxidation of 1,5-dienes. J. Am. Chem. Soc. 1979, 101, 4396–4397. [Google Scholar] [CrossRef]
- Walba, D.M.; Edwards, P.D. Total synthesis of ionophores the monensin BC-rings via permanganate promoted stereospecific oxidative cyclization. Tetrahedron Lett. 1980, 21, 3531–3534. [Google Scholar] [CrossRef]
- Spino, C.; Weiler, L. A stereoselective synthesis of the tetrahydrofuran unit in ionomycin. Tetrahedron Lett. 1987, 28, 731–734. [Google Scholar] [CrossRef]
- Walba, D.M.; Przybyla, C.A.; Walker, C.B. J. Total synthesis of ionophores. 6. Asymmetric induction in the permanganate-promoted oxidative cyclization of 1,5-dienes. J. Am. Chem. Soc. 1990, 112, 5624–5625. [Google Scholar]
- Kocienskyi, P.J.; Brown, R.C.D.; Pommier, A.; Procter, M.; Schmidt, B. Synthesis of salinomycin. J. Chem. Soc., Perkin Trans.1 1998, 9–40. [Google Scholar]
- Brown, R.C.D.; Hughes, R.M.; Keily, J.; Kenney, A. Diastereoselective synthesis of tetrahydrofuran-containing fragments by the permanganate oxidation of 1,5,9-trienes. Chem. Commun. 2000, 1735–1736. [Google Scholar]
- Brown, R.C.D.; Keily, J. F. Asymmetric permanganate-promoted oxidative cyclization of 1,5-dienes by using chiral phase-transfer catalysis. Angew. Chem. Int. Ed. 2001, 40, 4496–4498. [Google Scholar] [CrossRef]
- Towne, T.B.; McDonald, F.E. Syn-oxidative polycyclizations of hydroxypolyenes: highly stereoselective and potentially biomimetic syntheses of all-trans-polytetrahydrofurans. J. Am. Chem. Soc. 1997, 119, 6022–6028. [Google Scholar] [CrossRef]
- Morimoto, Y.; Iwai, T. Highly diastereoselective cyclizations of bishomoallylic tertiary alcohols promoted by rhenium(VII) oxide. Critical steric versus chelation effects in alkoxyrhenium intermediates. J. Am. Chem. Soc. 1998, 120, 1633–1634. [Google Scholar] [CrossRef]
- Sinha, S.C.; Keinan, E.; Sinha, S.C. Rules of Stereoselectivity in Tandem Oxidative Polycyclization Reaction with Rhenium(VII) Oxides. J. Am. Chem. Soc. 1998, 120, 9076–9077. [Google Scholar] [CrossRef]
- Keinan, E.; Sinha, S.C. Oxidative polycyclizations with rhenium(VII) oxides. Pure Appl. Chem. 2002, 74, 93–105. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; D’Auria, M. Pyridinium chlorochromate: a versatile oxidant in organic synthesis. Synthesis 1982, 245–258. [Google Scholar]
- Piccialli, V.; Zaccaria, S.; Oliviero, G.; D’Errico, S.; D’Atri, V.; Borbone, N. Pyridinium chlorochromate-mediated oxidation of mono- and poly-tetrahydrofurans. Disclosure of novel oxidative pathways. Tetrahedron 2011. submitted. [Google Scholar]
- Brimble, M.A.; Edmonds, M.K. Synthesis of bis-2,5-linked tetrahydrofurans via iodoetherification. Tetrahedron 1995, 51, 9995–10012. [Google Scholar] [CrossRef]
- Nakata, T.; Nomura, S.; Matsukura, H.; Masamichi, M. Stereoselective synthesis of the C- and CD-ring systems of hemibrevetoxin B. Tetrahedron Lett. 1996, 37, 217–220. [Google Scholar] [CrossRef]
- Dickie, D.A.; Abeysekera, D.; McKenzie, I.D.; Jenkins, H.A.; Clyburne, J.A.C. Crystallographic studies on substituted m-terphenyls: identification of weak [CH3··I] interactions. Cryst. Eng. 2003, 6, 79–86. [Google Scholar] [CrossRef]
- Blessing, R.H. An empirical correction for absorption anisotropy. Acta Crystallogr. 1995, A51, 33–38. [Google Scholar]
- Altomare, A.; Burla, M.C.; Cavalli, M.; Cascarano, G.L.; Giacovazzo, C.; Guagliardi, A.; Moliterni, G.G.; Polidori, G.; Spagna, R. SIR97: a new tool for crystal structure determination and refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar]
- Farrugia, L.J. WinGX suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Cremer, D.; Pople, J.A. General definition of ring puckering coordinates. J. Am. Chem. Soc. 1975, 97, 1354–1358. [Google Scholar] [CrossRef]
- Nardelli, M. PARST95 – an update to PARST: a system of Fortran routines for calculating molecular structure parameters from the results of crystal structure analyses. J. Appl. Crystallogr. 1995, 28, 659–659. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0 – new features for the visualization and investigation of crystal structures. J. Appl. Cryst. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Farrugia, L.J. ORTEP-3 for Windows- a version of ORTEP-III with a graphical user interface (GUI). J. Appl. Cryst. 1997, 30, 565. [Google Scholar]
- Sample Availability: Samples of the compounds 6 and 10 are available from the authors
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
